Introduction
Seed deterioration is inexorable and non-uniform (McDonald, 2004). Many factors contribute to seed deterioration including genetic composition, seed moisture content, mechanical and insect damage, pathogen attack, seed maturity, and relative humidity and temperature of the storage environment. Of the environmental factors affecting seed storage, relative humidity and temperature are the most important (McDonald, 2004). Relative humidity directly influences seed moisture. Increasing temperature increases the amount of moisture air can hold and the rate of cellular metabolism.
Ideal conditions for storage of peanut seed are 10 C and 65% RH (Ketring, 1992). The resulting seed moisture content is approximately 6%. Maintaining these conditions is not possible when peanuts are stored in warehouses without climate control. Navarro et al. (1989) studied the interaction of temperature and moisture concentration on the germination of peanut seeds of different cultivars. Germination of peanut seeds stored in-shell (pods stored intact and later shelled to provide seeds for germination) at 15 C and 79 to 83% RH remained above 80% for over 150 days. Germination of the same peanut cultivars stored at 15 C and 85 to 89% RH decreased dramatically to 30% in 80 days. When the storage temperature increased to 20 C and RH was at 79 to 83%, germination steadily decreased to 80% after 80 days and, if stored at 20 C and 85 to 89% RH, germination dropped to 20% in 80 days. Navarro et al. (1989) found an interaction between the tested cultivars and the temperature and RH. The cultivar Congo (valencia type) tolerated higher storage temperature and RH better than the cultivar Hanoch (virginia type). Ketring (1992) reported peanut seed tolerated temperatures of 44 C when relative humidity was low, but that genotypes varied in tolerance of high temperatures within seasons and across seasons, showing the effect of genetic variation and environmental conditions during seed maturation. Hypocotyl-radicle length was more adversely affected than germination.
Seed deterioration is the result of changes within the seed that decrease the ability of the seed to survive. It is distinct from seed development and germination and it is cumulative (McDonald, 2004). Peanut seed quality is highest just prior to physiological maturity, but can deteriorate rapidly during storage (Perez and Arguello, 1995). McDonald (2004) points out that a seed is a composite of tissues that differ in their chemistry and proximity to the external environment and that deterioration does not occur uniformly throughout the seed. The embryonic axis is more sensitive to aging than the cotyledons. In the axis, the radicle is more sensitive to deterioration than the shoot. McDonald (1998) suggested that water present in the atmosphere may be attracted by the same matric forces as soil water resulting in higher water content in the radicle compared to the storage reserves. Higher moisture content could selectively accelerate deterioration of the seed axis.
The University of Florida Agricultural Experiment Station (FAES) at the North Florida Research and Education Center (NFREC) near Marianna, Florida, has released several late-maturing peanut cultivars, namely Florida MDR-98 (Gorbet and Shokes, 2002), C-99R (Gorbet and Shokes, 2003), DP-1 (Gorbet, 2003), and Hull (Gorbet, 2007a). These cultivars have improved resistance to late leaf spot (Cercosporidium personatum, Berk & Curt.), white mold (Sclerotium rolfsii, Sacc.), and tomato spotted wilt virus (genus Tospovirus; family Bunyaviridae). The improved resistances were derived primarily from lineage to PI (Plant Introduction) 203396 through a common parent or grandparent UF81206-1 (Islieb, et al., 2001). Based on pedigrees, DP-1 has 50% genetics inherited from PI 203396, Florida MDR-98 and Hull have 38%, and C-99R has 25%. The cultivars are high yielding with good grades and acceptable flavor and processing characteristics. They provide the grower an opportunity to reduce fungicide applications and, therefore, to reduce variable costs without reducing yields.
The cultivar Florida MDR-98 was released for commercial production in 1998, but within two years, seed production was terminated because of poor field emergence. Similarly, DP-1 and Hull suffered from poor field emergence and seed is no longer commercially available. Field emergence problems for C-99R have been less frequent and less severe and seed has been commercially produced. Official germination tests usually show acceptable seed quality for these late-maturing cultivars (Tillman, 2004, Per. Comm.). Preliminary research data from Florida shows that reduced field emergence of these cultivars seldom occurs when the seed has been grown, harvested, and stored in research storage facilities (Tillman, 2004, Per. Comm.). The poor field emergence occurs when seed is produced in commercial channels with large volumes of peanut pods being harvested, stored in bulk facilities, shelled, and the seed treated with fungicides.
Seed peanut fields in the southeastern USA are harvested starting in early September by digging the pods and vines, inverting the biomass to air dry. Then, 3 to 5 days after digging, plants are combined by machine to separate pods from stems. With forced heated air, pods and seeds are dried in wagons to approximately 9.5% seed moisture content (Stadsklev, 2004, Per. Comm.). The peanut pods are stored in large bulk bins at ambient atmospheric temperatures that may exceed 32 C during the warmest part of the day and RH that may exceed 95% during the night. Dimensions of the bulk pile may be very large, often exceeding 7,000 m3. In some operations, exhaust fans mounted in the gable end of the warehouse draw ambient air in inlets mounted in the opposing gable and through the headspace between the roof and the top of the peanut pile. As cooler air is drawn into the headspace, it falls and displaces the warmer moist in the peanut pile. In other operations, ventilation is accomplished by circulating surface air through open doors and/or louvers in the wall and exhausting through roof vents. Shelling seed peanuts begins as early as December. Shelled peanuts are stored in a warehouse at ambient temperature and relative humidity in solid cardboard containers or large tote bags on pallets until treated with fungicides, bagged, and ready for delivery to the farmer.
This work was conducted to determine if various storage environments affect the germination and emergence of peanut cultivars with PI 203396 parentage. Based on experience and observation we hypothesized that cultivars with this parental lineage would have poor field emergence and perhaps germination compared to other cultivars when stored in a commercial style facility.
Materials and Methods
To study the interaction of cultivars and storage environment, four cultivars were placed in four storage environments. The cultivars were C-99R, DP-1, Hull, and AP-3 (Gorbet, 2007b). The cultivars C-99R, DP-1, and Hull are late -maturing with parentage tracing to PI 203396, the primary source of their resistance to tomato spotted wilt virus (genus Tospovirus; family Bunyaviridae), late leaf spot (Cercosporidium personatum, Berk & Curt.), and white mold (Sclerotium rolfsii, Sacc.). These cultivars consistently produce high yields and good grades. However, field emergence has been unreliable after storage in commercial bulk peanut bins. AP-3 was chosen as the control, mainly because AP-3 has no lineage of PI 203396 and has demonstrated good field emergence after storage in commercial facilities.
Seed was obtained in October 2004 and 2005 from commercial fields of the Florida Foundation Seed Producers (FFSP) near Marianna, Florida. Pods were dried with forced heated air (≈33 C) to seed moisture contents of 8 to 10%. The pods of each cultivar within a seed source were thoroughly mixed to minimize variation and bagged in burlap sacks for placement in four storage locations. Bags of peanut pods of each cultivar from each seed origin in each year were placed into four storage environments as follows, 1) temperature and RH controlled cool room (COOL), 2) approximately 1.5 meters into the base of the bulk in-shell peanut pile within the FFSP commercial peanut storage warehouse (WHSE), 3) in the center of bulk peanuts being stored in wagons housed in open-sided sheds at FFSP (WAGON), and 4) in burlap bags on pallets in a closed shed (SHED). Pod samples of each cultivar were frozen to preserve seed condition at the time of harvest
Temperature and relative humidity were recorded using a data logger (HOBO® Pro Series, Onset Computer Corp., Bourne, Mass). The device was placed within stored samples in each environment and temperature and relative humidity data were recorded every 15 min. The COOL environment provided uniform air temperature (12 to 15 C) and relative humidity (60 to 70%) from October to December. In the WHSE environment, piles varied in height to 15 meters and had a volume of up to 7,000 m3. Ventilation was accomplished through ridge vents on the roof creating a natural flow of air through louvered vents in the side of the warehouse. No forced air or intra-pile ventilation was provided. The WAGON environment consisted of peanut dryer wagons that have a plenum about 0.3 m deep with peanuts on top of the plenum about 1.5 m deep. Burlap bags containing the samples were buried within the peanut pods in the wagon. Wagons were placed under an open shed. Ambient temperature and relative humidity outside the storage facilities were recorded by the Florida Automated Weather Network (FAWN) substation located at Latitude 30.850 and Longitude −85.165 approximately 50 m from the FFSP storage facilities.
Placement of bagged peanut pod samples from 2004 was delayed and bagged peanuts could not be placed within the center of WHSE or WAGON. For comparison of storage effects deeper within the WHSE and WAGON, a second set of samples designated “Bulk Stored Seed” was collected at the end of the storage period from pods deep within the bulk piles of WHSE and the center of WAGON. This sample set is similar to the bagged peanut samples in all aspects except that the peanuts during storage were located deeper within the bulk piles of the WHSE or the bulk peanuts of the WAGON.
Pods from bagged samples were shelled in small lots using a pod sizer and a standard Federal/State grade sheller. The seeds that rode a 6.3 × 19.1 mm (16/64 × ¾ inch) slotted screen were used for field and germination tests.
Bulk seed were shelled by FFSP as follows: a front-end loader scooped the peanuts out of the WHSE, loaded the peanuts into a wagon, and the contents of the wagon were dumped into an elevator which lifted them into the sheller. After shelling, all seeds were sized over a 6.3 × 19.1 mm (16/64 × ¾ inch) slotted screen. Seed that rode the screen were used for field and germination tests. The shelled and sized seed was stored in large cardboard boxes in a warehouse.
Germination Tests
Germination tests were conducted according to the Rules for Testing Seeds published by the Association of Official Seed Analysts (2004) and included using ethephon (ethylene) to break dormancy. Seeds were treated with Vitavax® PC for control of fungi. Four replications of 50 seeds of each cultivar were evenly spaced on double germination towels, covered with a third towel, the lower edge of the towels was folded to retain seeds, and the towels were rolled into a cylinder shape and set on end in sealed plastic containers. Unless otherwise noted, germination tests were conducted in a germination chamber (Model I-35LVL, Percival Mfg. Co., Boone, Iowa) at 25 C. Seedlings with a radicle longer than 1 cm were counted after 10 days of imbibition. Each year, germination tests were conducted just prior to sowing field emergence tests.
Field Emergence Tests
To determine field emergence, four replications of 50 seeds of each cultivar were sown on successive days during April 2005 and 2006 and October 2005 in a Randomized Block Design (RBD) in field research plots located on the campus of the University of Florida, Gainesville, FL. The soil type was a Millhopper fine sand (Loamy, Siliceous, Semiactive, Hyperthermic Grossarenic Paleudult). Soil temperatures exceeded 15.5 C at the sowing depth of 5 cm. Seeds were placed in rows spaced 15 cm apart with in-row spacing between seeds of 8 cm. The plots were watered by overhead irrigation and emerged plants were counted 14 days after planting (DAP). No fertilizer was applied. To minimize impact of the experiments sown in 2005 on those sown in 2006, all seedlings from the 2005 tests were pulled out within 20 days of sowing and the soil was tilled and lay fallow for about 12 months with periodic tillage to control weeds.
In the October field emergence test, the seed came from the bulk stored seed (FFSP origin) that had been stored from the time of the April sowing until October in conditions similar to the COOL environment at 12 to 13 C and 66 to 68% relative humidity. The additional storage time was 5.5 months. Treatments were sown October 4, 2005 in a polyethylene-covered hoop greenhouse located in Gainesville, FL. The greenhouse was used to insure that soil temperature was similar to the April 2005 planting. Replications were sown at five-day intervals. All seeds were treated with Vitavax® PC.
Seeds from the 2005 production year were sown in a field test on May 5, 2006 in the manner described above. Four replications were sown on successive days in the same field site as the prior year's field emergence test.
Laboratory tests and field emergence trials were set up as RBD. Analysis of variance (ANOVA) was accomplished by the procedures in SAS System Release 9.1 (SAS Institute, Inc., Cary, NC). Least Squares Means and Duncan's Multiple Range Tests were generated using the general linear model (PROC GLM). Unless stated otherwise, differences reported were significant at alpha of less than or equal to 0.05.
Results and Discussion
Pods Stored in Bags- Germination
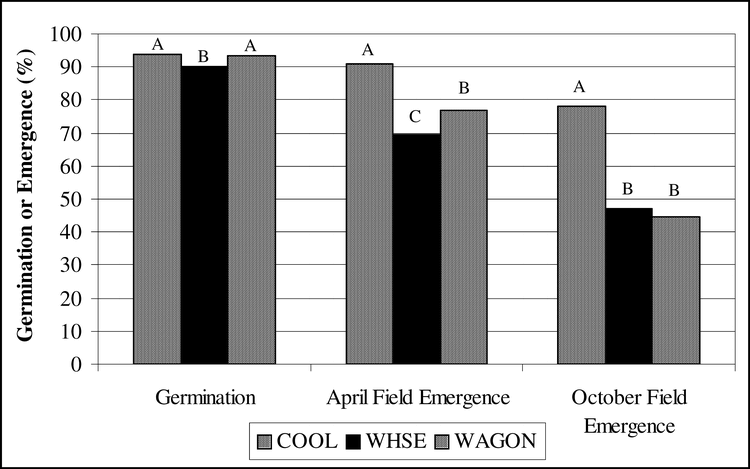
The effect of cultivar and storage environment on standard germination was variable in 2004 and 2005 (Table 1). Neither cultivar nor storage environment affected germination of seeds from 2004, but there was cultivar by environment interaction. Germination of seeds from 2005 was affected by cultivar (P = 0.0442 and storage location (P = 0.0498), but there was no interaction between treatments.
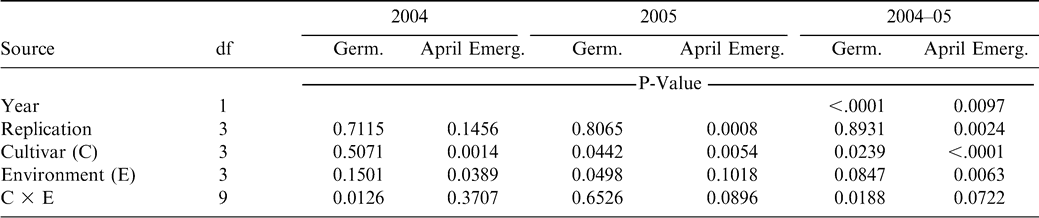
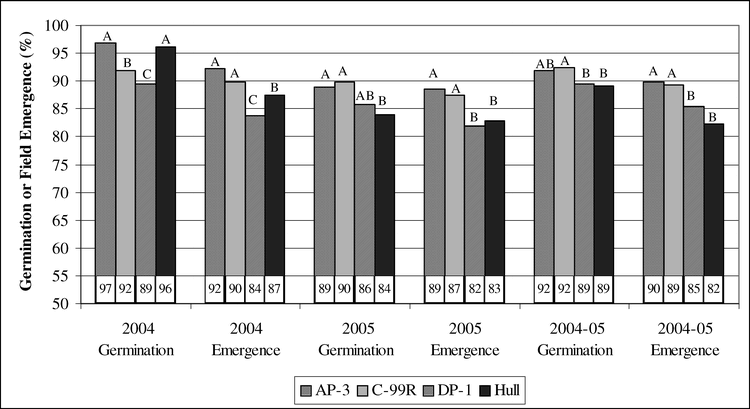
When data from the two years were combined, the main effects of cultivar and storage environment were significant as well as the interaction between the treatments. Germination of C-99R was greater than that of DP-1 and Hull, and germination of AP-3 was intermediate (Fig. 1). Given the fact that the lowest germination value was 89% and the highest was 92%, these differences were negligible. Similarly, the average germination in each storage environment ranged from 89% in COOL to 92% in WAGON. Germination was greatest in WAGON and least in COOL and intermediate in WHSE and SHED (Fig. 2). Evaluation of the interaction between cultivar and storage environment revealed that germination of DP-1 in COOL was 83%, lower than all other storage environments (P>0.002). There was no difference in germination among the storage environments for AP-3 or Hull. Similar to DP-1, germination of C-99R in the WHSE and WAGON was greater than that in the COOL and SHED. Again, the overall germination values ranged from 83% to 95% with and average of 90% and the next lowest germination was 89%. The low germination of DP-1 in COOL could have been related to lingering dormancy although ethephon was used in the germination process. In any case, germination of all cultivars from all storage environments was acceptable based on commercial standards for marketing seed. The minimum germination required for peanut seed is 70 to 75% and varies from state to state.
Field Emergence
For both 2004 and 2005 crop years, cultivar affected field emergence (2004, P = 0.0014; 2005, P = 0.0054; combined P<0.0001) (Table 1). Seeds of AP-3 from the 2004 crop year emerged better than DP-1 and Hull, but similar to C-99R (Fig. 1). Field emergence of DP-1 was less than AP-3, C-99R, and Hull. From the 2005 crop year, field emergence of AP-3 and C-99R was similar and greater than that of DP-1 and Hull. Combined across years, field emergence of AP-3 and C-99R was about 89%, which was greater than both DP-1 and Hull.
Storage environment affected field emergence (2004, P = 0.0389; 2005, P = 0.1018; combined, P = 0.0063). From the 2004 crop, field emergence of seeds stored in the WSHE was lower than that from COOL and WAGON (Fig. 2). Similarly, seeds from the 2005 crop year stored in the WHSE had poorer emergence than those in the SHED (Fig. 2). In the combined analysis, field emergence of seeds stored in the WHSE was less than all other locations.
Germination and field emergence of peanut seeds after storage in four different locations as follows: 1) COOL- cool room, 12 to 15 C and 60 to 70% relative humidity, 2) WHSE, warehouse- a large pile of peanut pods in a closed metal building on concrete floors, 3) WAGON- a peanut drying wagon under an open-sided barn, and 4) SHED- pods in bags stored on a pallet in a large enclosed shed. Within a grouping, means with the same letter are not significantly different by Duncan's Multiple Range Test at the 5% probability level.
Averaged over the two years, interaction between cultivar and storage environment for field emergence was significant (P = 0.0722) (Table 1). This indicates that emergence of the cultivars was not consistent among storage environments. Analysis showed that emergence of DP-1 and Hull was lower than that of AP-3 and C-99R when seeds were stored in the WHSE. Emergence of AP-3 and C-99R seeds was similar across all storage environments and was at least 85% whereas emergence of DP-1 and Hull fell to 80% or less when stored in the WHSE. Emergence of DP-1 and Hull in the other storage locations was at least 85% except for Hull stored in COOL which had an emergence rate of 79%.
Pods Stored in Bulk
Samples used in the results described above were stored in burlap bags and were derived by hand mixing peanuts from several wagons and placing the bags as deep as possible into WHSE and WAGON. This depth was approximately 1.5 m. In contrast to the bagged samples, bulk stored seed samples were collected from the same WHSE and WAGON either by probing into the bulk pile or as the peanuts were being emptied at the conclusion of the storage season. Since these seed samples came from deeper within the pile or wagon than the bagged seed, they should more representative of the storage environment.
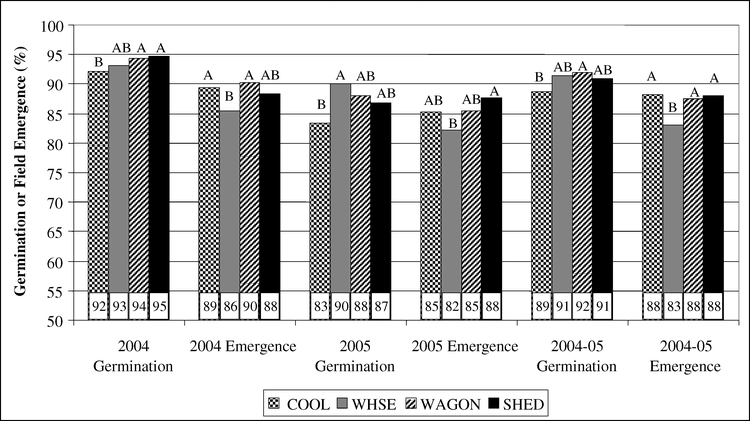
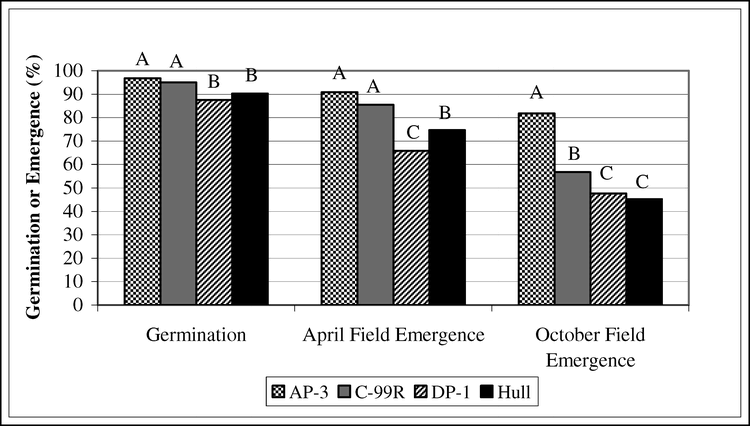
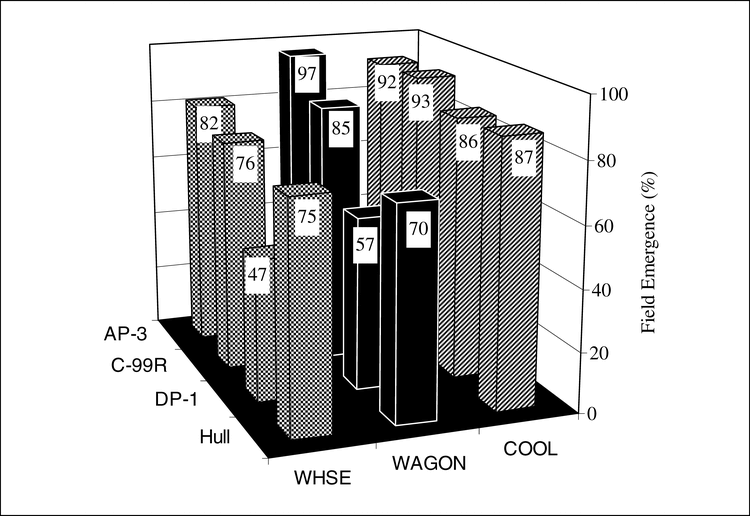
The main effects of cultivar and storage location for germination tests and field emergence of bulk stored peanut pods sampled from various locations for production year 2004 are presented in Fig. 3 and Fig. 4. Cultivar differences were evident in field emergence tests in April and October (April field emergence, P<0.0001; October field emergence P<0.0001). In the April test, field emergence of AP-3 and C-99R was superior to DP-1 and Hull and Hull was superior to DP-1 (Fig. 3). In the October test, emergence of AP-3 was superior to C-99R, DP-1, and Hull and emergence of C-99R was superior to DP-1 and Hull.
Effect of storage environment on germination and field emergence of bulk stored peanut produced in 2004. Storage environments were 1) COOL, cool room, 12 to 15 C and 60 to 70% relative humidity, 2) WHSE- a large pile of peanut pods in a closed metal warehouse on a concrete floor, and 3) WAGON- a peanut drying wagon under an open-sided barn. Seed from COOL is a second sample from bagged seed and not truly bulk seed. Within a grouping, means with the same letter are not significantly different by Duncan's Multiple Range Test at the 5% probability level.
Storage location also affected April and October field emergence (April field emergence, P<0.0001; October field emergence, P<0.0001). In the April test, field emergence of seeds stored in COOL was superior to those stored in WHSE and WAGON, and WAGON was superior to WHSE (Fig. 4). In the October test, emergence of seeds stored in COOL was superior to both WHSE and WAGON.
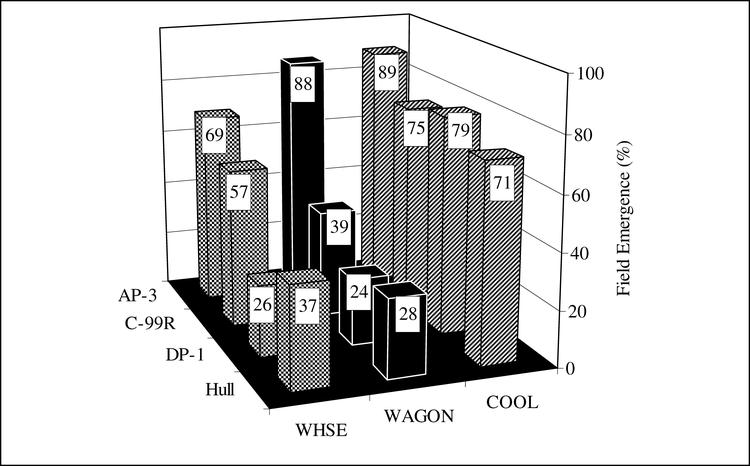
The interaction of cultivar and storage location is presented in Figures 5 and 6. The P-values for interaction of cultivar and storage location for April field emergence and October field emergence were P<0.0001. April field emergence of AP-3, C-99R and DP-1 was similar when stored in the COOL, but when stored in either the WAGON or WHSE, field emergence of DP-1 was inferior to that of AP-3 or C-99R (Fig. 5). The interaction of cultivar and storage location was pronounced when seeds were tested in October (Fig. 6). Field emergence of all cultivars stored in COOL was superior to storage in WHSE. Comparing COOL to WHSE, October field emergence of AP-3 decreased from 89% in COOL to 69%, C-99R from 75% to 57%, DP-1 from 79% to 26%, and Hull from 71% to 37%. AP-3 stored as well in the WAGON as in the COOL, but C-99R, DP-1, and Hull had greatly reduced field emergence when stored in the WAGON as compared to COOL. Note that field emergence of AP-3 in October from seeds stored in WHSE was less than field emergence of AP-3 stored in COOL (P = 0.0003), indicating that seed vigor of AP-3 stored in WHSE also declined. The implication is that WHSE storage past normal planting time (spring after fall harvest) may reduce vigor for all cultivars resulting in stand establishment failures.
Field emergence in April 2005 of bulk stored peanut as affected by cultivar and storage environment of seed peanut produced in 2004. Seed from COOL is a second sample from bagged seed and not truly bulk seed. Storage environments were 1) COOL- cool room, 12 to 15 C and 60 to 70% relative humidity, 2) WHSE- a large pile of peanut pods in a closed metal warehouse on a concrete floor, and 3) WAGON- a peanut drying wagon under an open-sided barn.
Field emergence in October 2005 of bulk stored peanut that was produced in 2004 as affected by cultivar and storage environment. Seed from COOL is a second sample from bagged seed and not truly bulk seed. Storage environments were 1) COOL- cool room, 12 to 15 C and 60 to 70% relative humidity, 2) WHSE- a large pile of peanut pods in a closed metal warehouse on a concrete floor, and 3) WAGON- a peanut drying wagon under an open-sided barn.
Seed deterioration is a result of changes within the seed that decrease the vigor followed by reductions in germination of the seed (McDonald, 2004). Over time, damage from a suite of degrading reactions accumulates and changes occur that begin to weaken the seed from strong viability to a weaker seed to a non-viable seed (Walters, 1998). Seed vigor declines faster than germinability (Delouche and Caldwell, 1960). The storage period for all treatments from the time of the April 2005 field emergence test to the October 2005 field emergence test was 5.5 months in COOL at 12 to13 C and 66 to 68% relative humidity. The additional stress was uniform and minimal for all samples yet, in October field emergence tests, the number of emerged seedlings was dramatically lower. The greatly reduced field emergence in October indicates that vigor of seed not stored in COOL, as influenced by quality and storage conditions, declined as described by Delouche and Caldwell (1960).
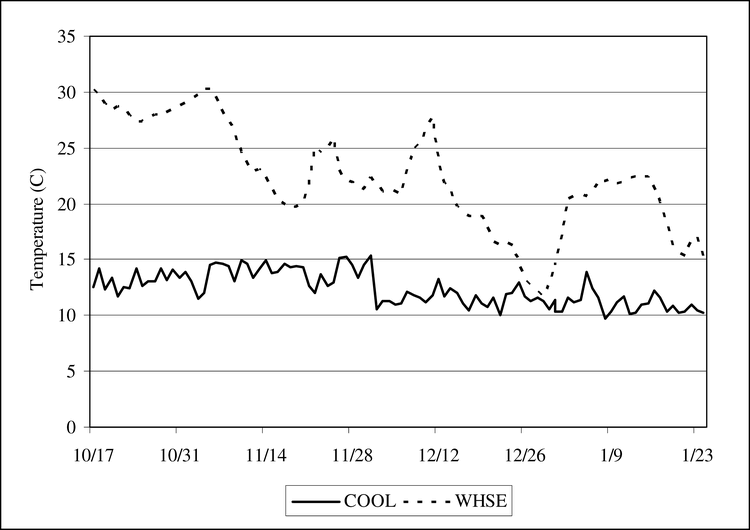
External atmospheric temperature and relative humidity data at 2 m height were recorded at the Florida Automated Weather Network (FAWN) adjacent to the storage facilities. The mean of the daily temperature was 15.8 C in 2004 and 15.7 C in 2005. Although there is no meaningful difference in the mean daily temperature of the external air for ventilation, the temperatures within the center of WHSE October 11 to 24 January for the 2004 crop year averaged 22.8 C compared to 15.8 C for the 2005 crop, a difference of 7 C. Conceivably the timing of warm and cool weather fronts for ventilation will affect the relative rate of cooling of peanuts in WHSE. In 2005 from 23 October to 11 November, external air temperatures averaged 13.4 C, which was 8.6 C cooler than the average of 22 C for the same period in 2004. During that time interval, temperature in WHSE in 2005 decreased 5 C whereas temperature in WHSE in 2004 increased 3 C. However, the 11-day period of higher temperatures in 2004 by itself accounts for only part of the higher temperatures throughout the storage period in 2004. Although external air temperature may change rapidly, sometimes as much as 10 C within 24 hours, temperatures in the center of the peanut pile in the WHSE changed slowly, usually less than 2 C per day. A comparison of temperature in WHSE to the outside temperatures recorded by FAWN and by sensors in WAGON under sheds suggests that the temperature in WHSE in 2004 is unexpectedly higher, possibly associated with heating in WHSE by peanut respiration resulting from insufficient drying or a climate event in 2004 that may have affected maturity of the seed prior to drying.
Temperatures in COOL varied between 10 and 15 C in 2004. Compared to WHSE, in 2004 temperature in the center of WHSE exceeded that in COOL by 5 to 15 C until the end of December (Figure 7). In the COOL environment, the relative humidity fluctuated between 58% and 79% in 2004. In the WHSE the relative humidity decreased from 86% to a range fluctuating between 52% and 62%.
Comparison of mean daily temperature in the seed storage room located at the University of Florida, North Florida Research and Education Center (NFREC) and the mean daily temperature within the bulk pile (WHSE) of peanut cultivar AP-3 stored in a traditional storage bin at the Florida Foundation Seed Producers, Inc., (FFSP) for the period 17 Oct. 2004 to 24 Jan. 2005. Key: COOL- cool room- 12 to 15 C and 60 to 70% relative humidity, WHSE- a large pile of peanut pods in a closed metal warehouse on a concrete floor.
Summary and Conclusions
The data from the crop year 2004 and 2005 field emergence tests is in accord with the anecdotal reports that poor field emergence and stand failures vary from year to year and that failures are more frequent with DP-1 and Hull. The results of these studies show that seed vigor, as measured by field emergence, of some cultivars like DP-1, C-99R, and Hull may decline more than other cultivars when stored in WHSE or WAGON. Loss of vigor can result in unacceptable plant stands even if germination tests meet minimum requirements.
Literature Cited
Delouche J. C. and Caldwell W. P. 1960 Seed vigor tests. Proceedings of the AOSA 50 / 1 : 136 .
Gorbet D. W. 2003 DP-1 - A new late maturity multiple disease-resistant peanut variety. Marianna NFREC Research Report 03-7, University of Florida North Florida Research and Education Center, Marianna, FL.
Gorbet D. W. and Shokes F. M. 2003 Registration of ‘C-99R’ peanut. Crop Sci 42 : 2207 .
Gorbet D. W. and Shokes F. M. 2002 Registration of ‘Florida MDR 98’ peanut. Crop Sci 42 : 2207 – 2208 .
Gorbet D. W. 2007a Registration of ‘Hull’ peanut. J. Plant Reg 1 : 125 – 126 doi: 10.3198/jpr2007.01.0035crc.
Gorbet D. W. 2007b Registration of ‘AP-3’ peanut. J. Plant Reg 1 : 126 – 127 doi: 10.3198/jpr2007.01.0037crc.
Isleib T. G. , Holbrook C. C. , and Gorbet D. W. 2001 Use of plant introductions in peanut cultivar development. Peanut Sci 28 : 96 – 113 .
Ketring D. L. 1992 Physiology of oil seeds. X. Seed quality of peanut genotypes as affected by ambient storage temperature. Peanut Sci 19 : 72 – 77 .
McDonald M. B. 2004 Orthodox seed deterioration and its repair. 273 – 296 In Benech-Arnold R. L. and Sanchez R. J. Handbook of Seed Physiology Food Products Press and Haworth Reference Press New York .
McDonald M. B. 1998 Seed quality assessment. Seed Sci. Res 8 : 265 – 275 .
Perez M. A. and Arguello J. A. 1995 Deterioration in peanut (Arachis hypogaea L. cv. Florman) seeds under natural and accelerated aging. Seed Sci. and Technol 23 : 439 – 445 .
SAS Institute System 9.1 SAS User's Guide: Statistics. SAS Institute Cary, NC .
Stadsklev T. D. 2004 Personal communication. Florida Foundation Seed Producers, P.O. Box 309, Greenwood, FL, 32443.
Tillman B. L. 2004 Personal communication. University of Florida North Florida Research and Education Center, Marianna, FL.
Walters C. 1998 Understanding the mechanisms and kinetics of seed aging. Seed Sci. Res 8 : 223 – 244 .
Notes
- First and second authors: Res. Agron. , USDA-ARS and Agric. Res. Statistician; Coastal Plain Exp. Sta., Tifton, GA 31793-0748. Third author: President and CEO, Hebert Green AgroEcology; Asheville, NC 28801. [^]
- Former Graduate Student, Dept. of Agronomy, University of Florida, Current address: National Association of Wheat Growers, 415 Second St., N.E. Washington D.C. 20002-4993 [^]
- Assistant Professor, Dept. of Agronomy, North Florida Research and Education Center, 3925 Highway 71, Marianna, FL 32446 [^]
- Professor, Dept. of Agronomy, North Florida Research and Education Center, 3925 Highway 71, Marianna, FL 32446 [^]
- Professor, Dept of Agronomy, University of Florida, P.O. Box 110500, Gainesville, FL 32611-0500 [^] *Corresponding author (email: btillman@ufl.edu)
Author Affiliations