Introduction
Due to its indeterminate growth habit, identifying the optimum harvest date for peanut (Arachis hypogaea L.) is difficult. During the late reproductive growth stages, there will be pods with a range of maturities present on the plant. At harvest, the combine collects all pods that have reached a given size and weight, regardless of maturity, so that a premature harvest increases the risk of having a higher percentage of immature pods. After harvest and drying, pods are delivered to a commercial sheller and the hull is removed. This mix of seed maturities has critical implications for overall seed quality. Germination is often the trait that is commonly utilized as a surrogate for seed quality; but this presents significant limitations because there is often a clear separation between germination and eventual seed vigor in many crop species. Germination capacity is a trait that occurs in the seed relatively early in development, while desiccation tolerance, genomic repair mechanisms, and the overall physiological ability to emerge and grow vigorously are traits acquired by the seed later in development (Finch-Savage and Bassel, 2016). Therefore in most instances, the assessment of seed maturity quantifies important impacts not only on yield and grade of the crop, but also the physiological performance and biochemical composition of seed peanut, ultimately determining its vigor and germination potential (TeKrony and Egli, 1991; Hampton, 2000). For peanut, Spears and Sullivan (1995) clearly demonstrated that seed maturity impacted not only germination capacity, but overall seed vigor. This fact makes the achievement of optimal maturity for seed peanuts a very critical issue.
It has been demonstrated that proper seed drying is important for all crop and plant production, not just peanut (Ellis et al., 1991; Rao et al., 1991; Elias and Copeland, 2001; Samarah et al., 2004). Drying is an important process during the physiological development of seed as it reflects the displacement of water by insoluble storage reserves (Bewley et al., 2013); so that the preparation of commercial seed often involves some artificial drying scenario prior to storage. Similar to peanut, coffee (Coffea arabica L.) is dried to 10-12%; moisture concentration but further desiccation often damages seed viability (Ellis et al., 1991). This can be the case when seed is dried too quickly and has not acquired desiccation tolerance, a mechanism that improves as a seed develops (Bewley et al., 2013). Another important milestone in seed development is physiological maturity, which is defined as the point at which seed reaches maximum dry weight (TeKrony et al., 1987). Common vetch (Vicia ervilia L.) reaches physiological maturity when the pod color changes to yellow and pre-chilling treatments reportedly improved seed germination in 80-99%; of the later maturing stages (Samarah et al., 2004). It has been observed that vetch harvested in later stages positively responded to air-drying and dry-prechilling, which decreased dormancy and improved germination (Samarah et al., 2003). Optimizing harvest by insuring physiological maturity is also possible for canola (Brassica napus L.). Elias and Copeland (2001) reported that harvesting at full maturity is preferable for the processing (threshing and storage) of canola. Farmers can harvest canola several weeks before full maturity without reductions in dry weight or yield; but seed quality was maximized by harvest at full maturity (Elias and Copeland, 2001). Similar to canola, pearl millet (Pennisetum glaucum) should be harvested one week after physiological maturity to maximize the storage potential of seed used for planting (Rao et al., 1991).
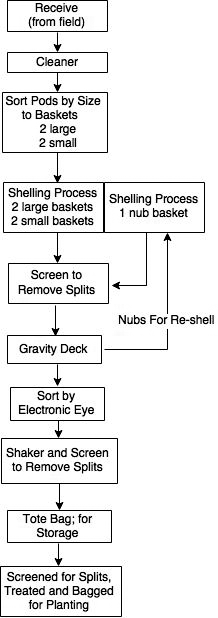
These examples indicate that seed maturity is important for optimal seed quality. However, for most crops, there is little in the seed processing procedures that can be utilized to improve seed maturity prior to storage other than through the process of drying. Peanut is unique because the pods are harvested and require shelling to liberate the seeds for planting. Although peanut was planted in-shell over a hundred years ago, and shown to have adequate germination and emergence (Sturkie and Buchanan, 1973), all seed for commercial production in the U.S. is shelled prior to planting. Therefore, it is possible that the maturity of the harvested crop could be improved in the shelling process during the various preparation stages involved. These stages include: 1) cleaning, where shriveled immature pods, stems, leaves, and other debris are removed mechanically; 2) size sorting after cleaning where intact pods are sorted by size and diverted to different sheller inputs to allow for more optimized shelling; and 3) a final stage that occurs after separation of shelled kernels on a gravity deck, which allows for extra small pods that were not shelled during the first shelling operation (often known as nubs), to be passed back into the smallest sheller (Davidson et al., 1982). This last step also removes small, shriveled, and/or damaged seeds.
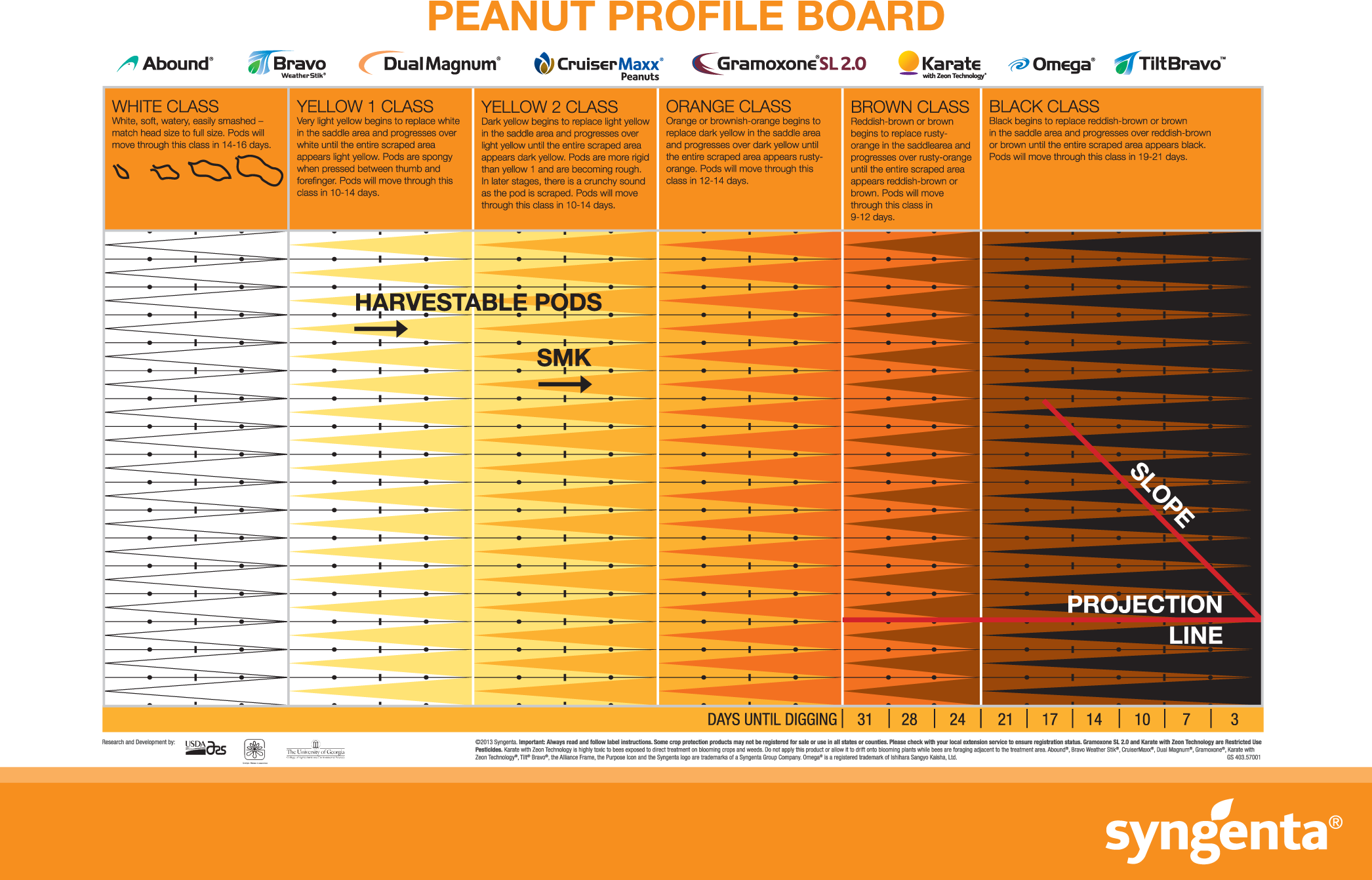
Given the impact maturity has on seed vigor and possibly subsequent crop performance in other species (Samarah et al., 2003; Elias and Copeland, 2001), it is important to identify all areas during the production and seed handling process where maturity can be optimized. The typical maturity determination process utilized by growers, consultants, and extension personnel involves the classification of pods by mesocarp color. A maturity profile board (MPB) for harvest determination developed by Williams and Drexler (1981), involves the separation of individual pods into distinct and multiple color classifications of white, yellow 1, yellow 2, orange, brown, and black (Figure 1). Additional classification within each of the larger classes is also often accomplished by separating pods within a given color class depending on the development of that color along some subjective scale. It has been shown that having 70 to 80%; black/brown pods in a field optimizes yield and grade (Boote, 1982; Rowland et al., 2006); therefore, determining harvest date through this color assessment provides growers with a tool to help ensure maximal yield and grade. However, the subjective nature of using visual assessment of mesocarp color to classify pods into very minute maturity classes is often widely variable and dependent entirely on the skill of the visual observer (Colvin et al., 2014). Because of the difficulties and error involved in the maturity determination process, oftentimes the maturity level of the crop is not evaluated and is based solely on planting date. Therefore, it appears that the impact of peanut maturity on seed production may not be fully understood by producers, resulting in more frequent premature harvests. To combat increasing levels of immaturity in seed peanut, it is important to concentrate efforts on two priorities: 1) continued extension efforts aimed at educating and communicating to producers about the importance of optimal peanut maturity; and 2) determining if there are steps within the shelling process for seed that could be utilized to cull out immature seed if harvest maturity is not optimal.
Maturity Profile Board developed from the work of Williams and Drexler (1981). Illustration used with permission from Syngenta.
To evaluate the particular steps in the shelling process where immature seed could be removed, a study was initiated that quantified pod maturity at five steps in the shelling process for four peanut cultivars. The specific objectives of the study were: 1) to quantify the percentage of mature seed at each stage and compare to the initial maturity level from the field at harvest (or receipt at the shelling plant); and 2) because grade is taken routinely at the buying point and has often been assumed to be indicative of maturity, grade was evaluated for its relationship with maturity.
Materials and Methods
All samples evaluated in this study were collected from peanut seed loads received at the Florida Foundation Seed Producers (FFSP; N 30°; 50'; 26.1816, W 85°; 9'; 57.6288) in Marianna, Florida. All fields were grown under certification conditions for seed, at least two years out of peanut and gypsum was applied. The experiment was conducted as a completely randomized design. The maturity of the seed was chosen (as opposed to germination) because it is the most closely linked trait to overall seed vigor and quality. To track the maturity ratio from harvest to shelling, samples were collected from multiple stages of the process.
The shelling process at the FFSP involves five distinct stages where pods are cleaned, separated by size in-shell, and sent back for repeated attempts at shelling if they were not shelled in the first pass. This process is illustrated by Figure 2 adapted from Davidson et al. 1982. Each of these stages was targeted for sampling in this study. The first set of samples collected were from the FFSP storage bay where seed coming directly from the field were placed after drying to 10%; moisture (FIELD). Samples within the FFSP shelling process itself included: collection of pods after passing through the cleaner (CLEANER); collection of pods after being screen separated into two in-shell sizes prior to shelling and categorized as either large (LARGE) or small (SMALL); and collection of pods after passing over the gravity deck that were not shelled during the first pass through the sheller (NUBS) (Figure 2).
The FIELD samples were collected for four peanut cultivars: FloRun '107' (Tillman and Gorbet, 2015), TUFRunner '727' (Tillman et al., 2015), TUFRunner '297' (Tillman et al., 2015), and TUFRunner '511' (Tillman et al., 2015). Samples were collected at each of the four stages within the FFSP shelling process for all except FloRun 107, which had completed the shelling process prior to the implementation of the study. In-shell sizing during the shelling process was accomplished through various screens and shelling baskets that varied due to inherent pod size differences by cultivar. For 2014-2015, the pod size screens were 10.3 mm (often labeled 26/64 inches in the industry), 12.7 mm (32/64 inch), 14.2 mm (36/64 inch), and 12.7 mm (32/64 inch) for FloRun 107, TUFRunner 727, TUFRunner 297, and TUFRunner 511, respectively. The sheller baskets included two for the LARGE, two for the SMALL, and one for the nub baskets: for FloRun 107 they were 10.3 mm (26/64 inch), 10.3 mm (26/64 inch), 8.3 mm (21/64 inch), 7.9 mm (20/64 inch), 6.7 mm (17/64 inch); for TUFRunner 727, they were 11.1 mm (28/64 inch), 10.7 mm (27/64 inch), 10.3 mm (26/64 inch), 9.9 mm (25/64 inch), 7.5 mm (19/64 inch); for TUFRunner 297, they were 12.7 mm (32/64 inch), 12.3 mm (31/64 inch), 11.1 mm (28/64 inch), 11.1 mm (28/64 inch), and 7.5 mm (19/64 inch); and for TUFRunner 511, they were 12.3 mm (31/64 inch), 11.9 mm (30/64 inch), 11.1 mm (28/64 inch), 11.1 mm (28/64 inch), and 7.5 mm (19/64 inch).
All samples collected from the FIELD, CLEANER, LARGE, SMALL, and NUBS stages were standardized by weight totaling approximately 22.7 kg. Subsamples of 2.3 kg were then taken from the original sample. Due to variability in the samples available, the number of samples evaluated at each stage per cultivar varied slightly: four samples were obtained at the FIELD and CLEANER stages, with six samples from the LARGE, SMALL, and NUBS stages.
Maturity Separation by Color Class
The dried samples of each cultivar and stage were rehydrated to decrease risk of the pods being destroyed during exocarp removal when placed in a wire basket and blasted with water using a pressure washer and turbo nozzle (Williams, 2003). After blasting, pods were sorted by mesocarp color into two color classes representing relative maturity level. To minimize the error introduced by visual determination of minute differences in mesocarp color, it was deemed necessary to group pods into only two color classes as opposed to the multiple categories listed on the MPB. The mature group was considered the most physiologically developed seeds and represented the visual separation of pods into brown and black classes following the convention of the MPB (Williams and Drexler, 1981); while the immature group was considered as the least developed seeds and represented the visual separation of pods into the Yellow 1 and 2 classes based on the MPB (Figure 3). To ensure that samples were clearly separated between mature and immature classes and immaturity was not overestimated, pods with a conflicted status of orange or slightly developed brown colors were categorized as mature (Figure 3). After sorting into these two color classes, the total number of pods in each group was counted, and a maturity proportion was calculated:
Illustration of how peanut pods were separated into the mature brown/black class (top row), orange or brown pods classified as mature (middle row) and the immature yellow (bottom row) class for this study. The middle row was categorized as mature for data processing purposes. Photograph by Ethan Carter.
Grade
After calculating the maturity ratio from the field stage, the group of mature and immature pods from each sample were shelled and graded. Grade was calculated by summing the total sound mature kernels (SMK) and sound splits (SS), dividing that number by the sample weight, and then multiplying by 100. Grade was determined for the mature and immature groups of seed from all samples collected at each stage and for each cultivar, with the exception of FloRun 107 that had samples limited to the FIELD.
Statistical Analysis
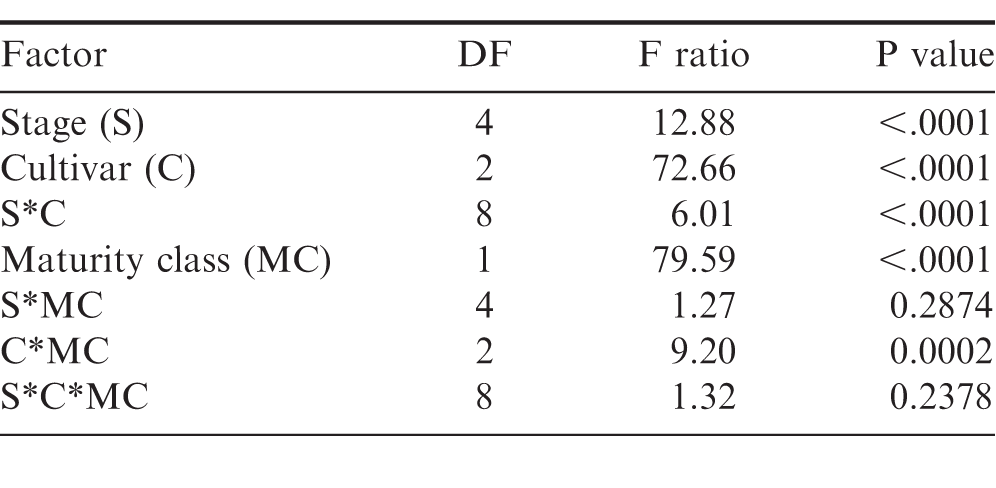
All data were evaluated in JMP 10 (SAS, Cary, North Carolina) through univariate analysis using a mixed model ANOVA consisting of a cross factorial design. The model was designed for a data set consisting of non-repeated measures. The restricted maximum likelihood method was used with standard least squares and an emphasis on effect leverage. The model was comprised of three fixed factors (maturity class, cultivar, and stage) and one random factor (rep). This model was run for three cultivars, TUFRunner 727, TUFRunner 297, and TUFRunner 511, for all five shelling stages (FIELD, CLEANER, LARGE BASKET, SMALL BASKET, NUBS). Because FloRun 107 was available only at the field stage, a separate ANOVA was run for FIELD samples that included all four cultivars. A Tukey's HSD multiple comparison test was run to separate means when significant differences in factors were noted. All graphical representations of data from this experiment are displayed with one standard error from the mean.
Results
Maturity Separation by Stage
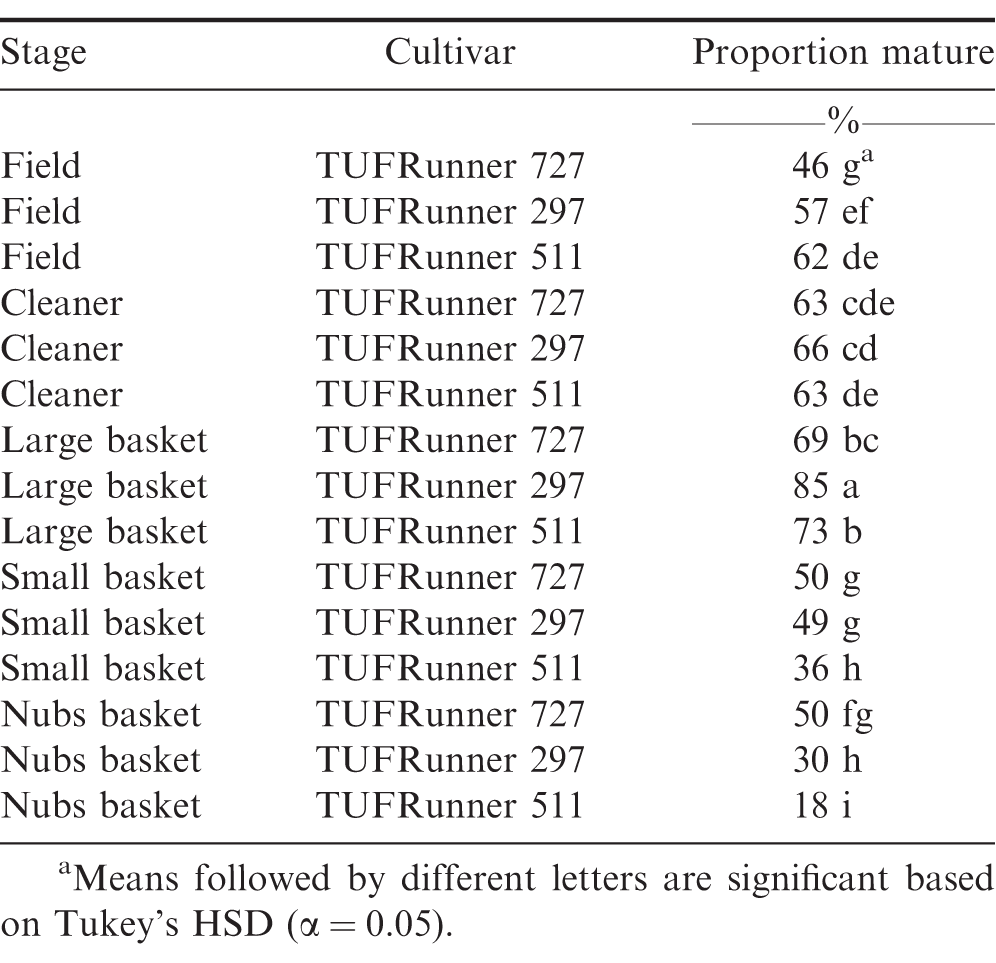
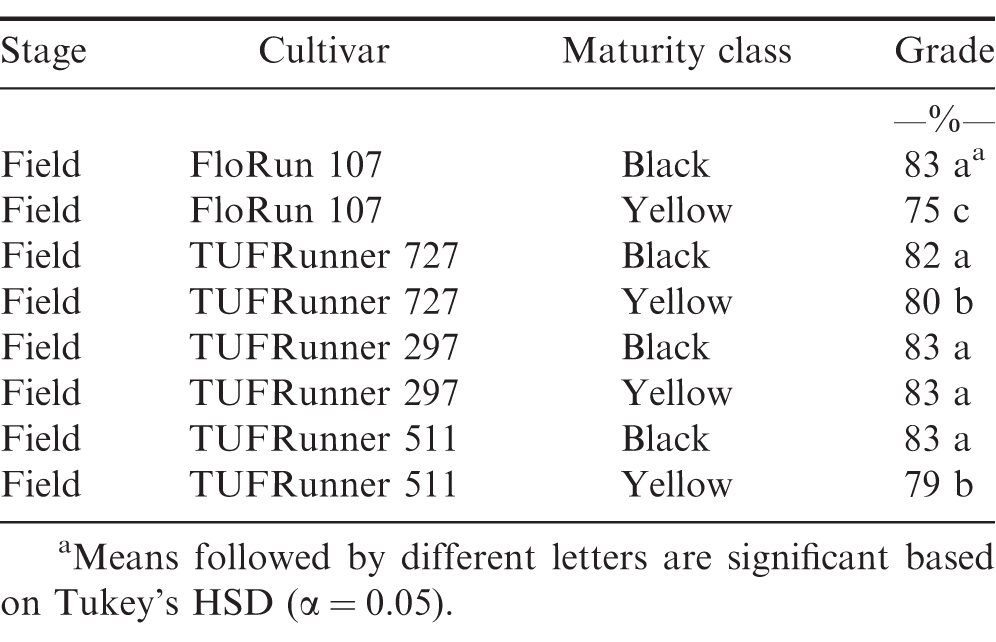
At the FIELD stage with all four cultivars represented, there were differences among cultivars (P Value 0.0001), with TUFRunner 727 having a lower percentage of mature pods than the other three cultivars (Table 1). On average, samples from the FIELD showed that the percentage of mature pods of TUFRunner 297, TUFRunner 511, and FloRun 107 was above 50%;, whereas TUFRunner 727 was composed primarily of immature pods (Table 1). Despite samples from three of the four cultivars being predominantly mature (>50%;), none of them reached the desired harvest level of 70-80%; mature pods shown to be representative of optimal crop maturity.
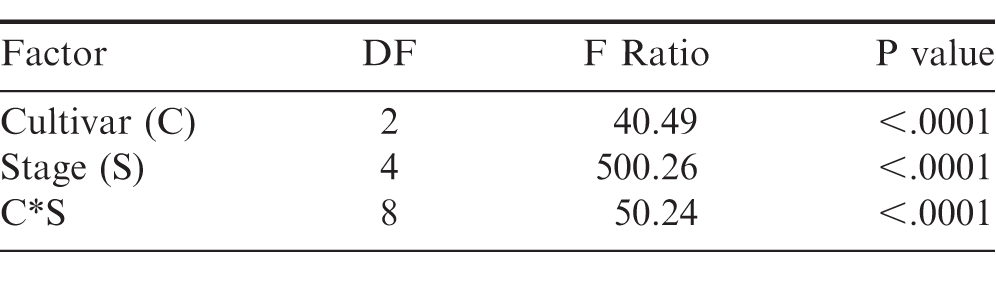
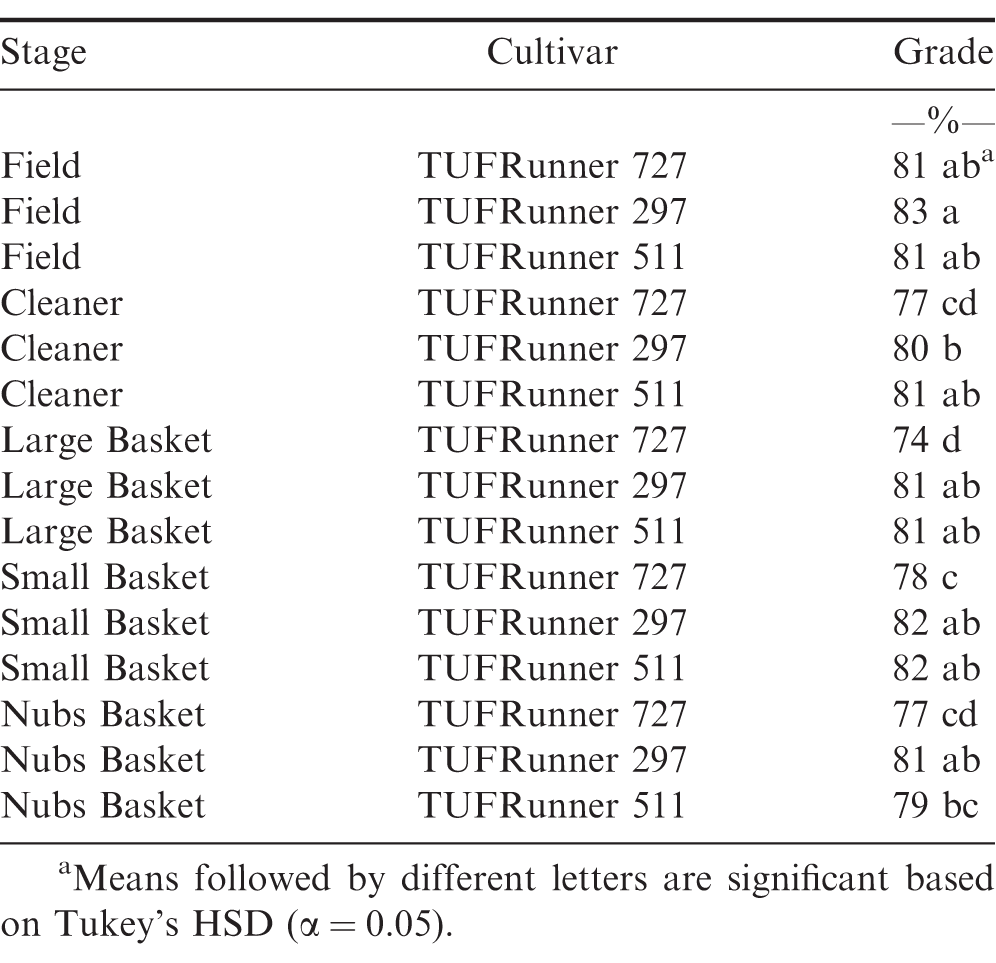
When examining the FSPP stages within the shelling process, cultivar, stage, and the interaction between cultivar and stage affected maturity (Table 2). The percentage of mature pods increased in all three cultivars after passing through the CLEANER, but the interaction between cultivar and stage was evident by the impact the cleaner had on the maturity of TUFRunner 727 and TUFRunner 297 compared to TUFRunner 511 (Table 3). Mature pods of TUFRunner 727 increased from 46 to 63%;, whereas the mature pods of TUFRunner 297 rose from 57 to 66%; and there was no change in TUFRunner 511 at the cleaner stage. Improvements in percentage of mature pods was also evident between the cleaner and the LARGE basket for all of the cultivars except TUFRunner 727, but the interaction with cultivar at this stage showed a greater impact for TUFRunner 297 than for the other two cultivars (Table 3). The brown/black classes of TUFRunner 297 and TUFRunner 511 had reached the 70%; mature mark by this stage showing an improvement in maturity that had not been reached for either cultivar at the previous stages. For TUFRunner 727, the improvement was quite remarkable, showing an improvement from 46%; mature in the field to 69%; mature at the LARGE stage. At the SMALL and NUBS stages, the impacts to maturity were relatively minor in comparison to the other stages and were relatively similar across cultivars (Table 3). There was a large drop in percentage mature pods from the LARGE to the SMALL basket for each cultivar, respectively: TUFRunner 727 dropped from 69%; to 50%; mature; TUFRunner 297 dropped from 85%; to 49%;; and TUFRunner 511 displayed the greatest decline of 37 percentage points. The pod maturity from the NUBS basket was 50%; for TUFRunner 727, a similar percentage to the SMALL basket. The other two cultivars TUFRunner 297, and TUFRunner 511 showed very low levels of maturity at the NUBS stage with 30%; and 18%; mature, respectively.
Grade
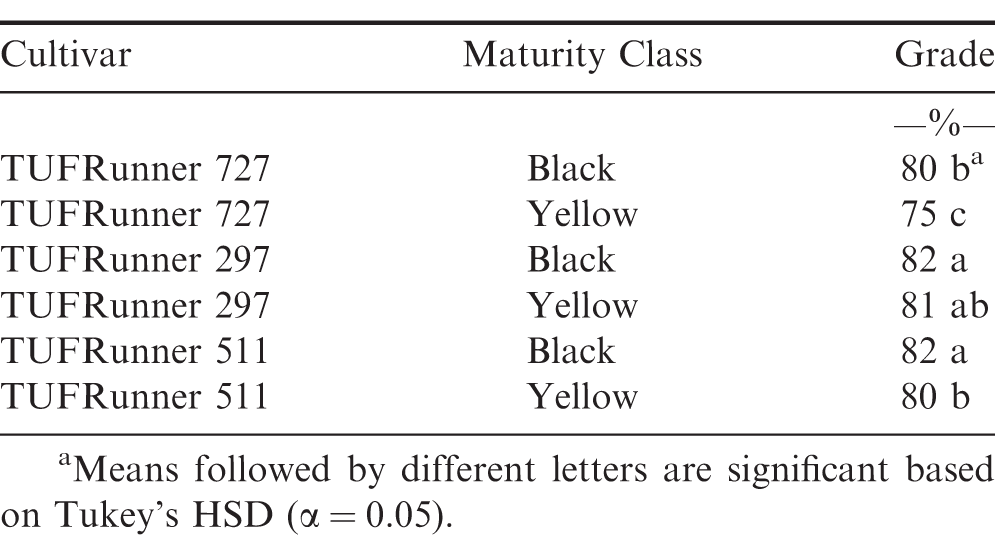
The evaluation of grade at the FIELD stage showed a difference among cultivars (P value 0.0001), maturity classes (P value 0.0001) and the interaction between cultivar and maturity class (P value 0.0001). Every cultivar graded 70%; or above regardless of maturity class (Table 4). At the FIELD stage, the mature and immature samples of TUFRunner 297 both had a grade of 83%;, whereas the mature class of the other three cultivars graded higher than their respective immature class. When evaluating grade for TUFRunner 727, TUFRunner 297, and TUFRunner 511 at the other shelling stages, all stages, cultivars and maturity classes graded 70%; or above, but there were interactions of cultivar by maturity class and cultivar by stage (Table 5). Both the stage by cultivar and cultivar by maturity class interactions were driven primarily by effects in TUFRunner 727, with grades being relatively uniform for the other two cultivars. For TUFRunner 727 the stages CLEANER, SMALL, and LARGE had lower grades than the other two cultivars and lower grade in the nubs stage than TUFRunner 297 (Table 6). The cultivar by maturity class interaction was driven mainly by lower grades for yellow pods in TUFRunner 727 and TUFRunner 511 (Table 7).
Discussion
This main objective of this experiment was to census multiple points within the shelling process to determine the level of pod maturity and whether certain stages within the process could increase the percentage of mature pods from the initial field level. It was clear from the FIELD samples, that maturity had not reached the optimal 70%; level recommended for harvest, with the actual maturity level across cultivars averaging near 50%;. These findings at the FIELD stage suggest that none of the cultivars were harvested at an optimum date that would insure maximum yield and grade, with the harvest of TUFRunner 727 being the most premature. This indicates a need to identify stages within the seed shelling process that could improve maturity, likely by culling and removing immature pods. Ultimately, it is most important to harvest the seed crop at optimal maturity. For example, Elias and Copeland (2001) reported that canola should be left in the field until reaching its full harvest maturity if the primary purpose is for seed as opposed to consumption, because this will ensure high seed quality and optimal germination and vigor. In the current study, the results indicate that the shelling process may present an opportunity for improving seed maturity if field harvest is not performed at optimal crop maturity.
The CLEANER stage increased the proportion of mature pods for each cultivar by removing the lightest and most immature pods. These small pods usually contain shriveled seeds that also have higher moisture content and are known to mold during storage (Davidson et al., 1982). Achieving low moisture concentration for seed storage is important in reducing and preventing microbial growth, fermentation, premature germination and other quality issues (Venkatachalam and Sathe, 2006).
For the three shelling baskets (LARGE, SMALL, NUBS), the LARGE stage consistently accumulated a significantly higher percentage of mature pods for every cultivar throughout the shelling process. This indicates that the in-shell sizing and subsequent sorting to the different baskets is an excellent way of pinpointing the majority of mature pods when collecting seed for planting. It can be assumed for some plant species that pod/seed size is reflective of greater subsequent seedling vigor (Marshall, 1986). Therefore, by selecting large seed sizes in the LARGE basket, this process is likely selecting an optimal level of maturity. However, seed size may not always be linked to field emergence and vigor. TeKrony et al. (1987) reported for soybean (Glycine max L.) that emergence was indeed linked to vigor but not with seed size. Further, large seeds from soybean and those of low density performed poorest during germination testing as well as had high levels of seed leachate conductivity, an indication of low vigor (Hoy and Gamble, 1985). While the LARGE basket clearly showed the potential for improving maturity, the SMALL and NUBS stages showed no improvement and may actually represent an input of immature pods into seed peanuts by repeatedly attempting to shell from these stages.
Another objective of this study was to determine if grading could be a reliable and repeatable method for determining maturity, thus allowing maturity assessment at the buying point itself when field samples are received from growers. Currently, samples are collected from farmer lots at buying points, graded, and then this grade value is used along with net lot weight to determine the total economic return to the grower (Lamb and Blankenship, 2005). At the FIELD level, the mature group of pods graded higher than their respective yellow group for three of the four cultivars, FloRun 107, TUFRunner 727, and TUFRunner 511. When assessing the other stages for the three TUFRunner cultivars, the grade really only reflected the differences in maturity groups for TUFRunner 727 and TUFRunner 511. Therefore, across different shelling stages, the immature class of most of the cultivars was found to grade similar to the mature class. The findings of our study suggest that grade may not always accurately represent maturity when taken at the field level. Therefore, when a grade sample is taken at the buying point (FIELD) a high grade does not necessarily correlate with a mature crop.
Conclusion
As illustrated by the FIELD maturity levels of all four cultivars (Table 2), growers do not always optimize the harvest maturity of peanut. The CLEANER was shown to increase the percentage of mature pods by removing immature pods, improving some cultivars more than others. This is best shown by comparing the significant rise in pod maturity for TUFRunner 727 after the CLEANER to the consistent maturity for TUFRunner 511. This study revealed that seed maturity is indeed linked to some extent with in-shell pod size. The biggest pods associated with the LARGE stage after in-shell sizing had the highest proportion of mature pods of all three stages, and was near or over 70%; for each cultivar, despite the much lower maturity proportion associated with each at the FIELD stage. The SMALL basket was found to be relatively immature, and NUBS extremely immature for certain cultivars. Grade was not a consistent representation of maturity, as both the mature and immature seed graded similarly across stages for most cultivars.
Based on the results of this study, it is recommended that peanut used for seed be sized in-shell and seed collected from the LARGE stage. This is an alternative to the current process for most shellers whereby seed is taken from a collection of jumbo or medium sized seed which is likely to include seed from more immature developmental stages. Because the in-shell sizing increases the level of pod maturity viewed at the FIELD stage, it can be used to offset lots harvested prematurely by growers such as TUFRunner 727 when selecting for seed. Continued research into the link between seed size and grade needs to be pursued, as this study found grade to not always be an accurate representation of maturity.
Acknowledgements
We are thankful for the research funding support from the Florida Peanut Producers Association and the National Peanut Board. We are also grateful to Florida Foundation Seed Producers, Inc. (FFSP) for their help with procuring the needed samples. Special thanks to Cody Smith, Carter Ullman and Thomas Burley for their help with pod processing, as well as Mihai Giurcanu of the University of Florida's Institute of Food and Agricultural Sciences Statistics department for statistical consulting.
Literature Cited
J.D., Bewley K.J Bradford, H.W.M Hilhorst, and H Nonogaki (2013). Development and maturation. In. Seeds: Physiology of Development, Germination and Dormancy, 3rd Edition, Springer, New York,. pp. 27- 83.
K.J Boote (1982). Growth stages of peanut (Arachis hypogaea L.). Peanut Science 9: 35- 40.
B.C., Colvin D.L Rowland, J.A Ferrell, and W.H Faircloth (2014). Development of digital analysis system to evaluate peanut maturity. Peanut Science 41: 8- 16.
Davidson J., T Whitaker, and J Dickens 1982 Grading, cleaning, storage, shelling, and marketing of peanuts in the United States In Peanut Science and Technology (eds. Harold, P and Young, C ), pp 571- 596 American Peanut Research and Education Society, Inc., Yokam, Texas.
S.G., Eliasand L.O Copeland (2001). Physiological and harvest maturity of canola in relation to seed quality. Agronomy Journal 93: 1054- 1058.
R.H., Ellis T.D Hong, and E.H Roberts (1991). An intermediate category of seed storage behaviour? II. Effects of provenance, immaturity, and imbibition on desiccation-tolerance in coffee. Journal of Experimental Botany 42: 653- 657.
D.J., Hoy, and E.E Gamble (1985). The effects of seed size and seed density on germination and vigor in soybean (Glycine max (L.) Merr.). Canadian Journal of Plant Science 65: 1- 8.
M.C., Lamb, and P.D Blankenship (2005). The capacity and efficiency of official grade shellers. Peanut Science 32: 132- 135.
D.L Marshall, (1986). Effect of seed size on seedling success in three species of sesbania (Fabaceae). American Journal of Botany 73: 457- 464.
N.K., Rao, S.A Rao, M.H Mengesha, and R.H Ellis (1991). Longevity of pearl millet (Pennisetum glaucum) seeds harvested at different stages of maturity. Annals of Applied Biology 119: 97- 103.
D.L., Rowland, R.B Sorensen, C.L Butts, and W.H Faircloth (2006). Determination of maturity and degree day indices and their success in predicting peanut maturity. Peanut Science 33: 125- 136.
N.H., Samarah, N Allataifeh, M Turk, and A.R Tawaha (2003). Effect of maturity stage on germination and dormancy of fresh and air-dried seeds of bitter vetch (Vicia ervilia L.). New Zealand Journal of Agricultural Research 46: 347- 354.
N.H., Samarah, N Allataifeh, M.A Turk, and A.M Tawaha (2004). Seed germination and dormancy of fresh air-dried seeds of common vetch (Vicia ervilia L.) harvested at different stages of maturity. Seed Science and Technology 32: 11- 19.
J.F., Spears, G.A Sullivan, (1995). Relationship of hull mesocarp color to seed germination and vigor in large-seeded Virginia-type peanuts. Peanut Science 22: 22- 26.
D.G., Sturkie, and G.A Buchanan (1973). Cultural Practices. In. Peanuts - Culture and Uses, American Peanut Research and Education Society, , Stillwater, OK..
D.M., TeKrony, T Bustamam, D.B Egli, and T.W Pfeiffer (1987). Effects of soybean seed size, vigor, and maturity on crop performance in row and hill plots. Crop Science 27: 1040- 1045.
D.M., TeKrony, and D.B Egli, (1991). Relationship of seed vigor to crop yield: a review. Crop Science 31: 816- 822.
Tillman, B., M Gomillion, J McKinney, and G Person 2015 Peanut Variety Performance in Florida, 2011-2014 University of Florida. Accessed on January 10, 2015. http://wfrec.ifas.ufl.edu/media/wfrecifasufledu/docs/pdf/EDIS-Tillman-Peanut-Variety-Performance-in-FL-2011-2014-for-EDIS.pdf
B.L., Tillman, and D.W Gorbet (2015). Registration of FloRun '107' peanut. Journal of Plant Registrations 9: 162- 167.
M., Venkatachalam, and S.K Sathe (2006). Chemical composition of selected edible nut seeds. Journal of Agricultural and Food Chemistry 54: 4705- 4714.
Williams, E.J (2003) A simple, quick, inexpensive peanut blaster University of Georgia http://santarosa.ifas.ufl.edu/documents/ag_podblaster.pdf. Retrieved 30 June 2015.
E.J., Williams, and J.S Drexler (1981). A non-destructive method for determining peanut pod maturity. Peanut Science 8: 134- 141.
Notes
- First, second, third, and fourth authors: Graduate Student, Professor, Associate Professor, and Professor, Agronomy Department, University of Florida, Gainesville, FL 32601; Fifth author: Professor, Crop and Soil Sciences Department, University of Georgia, Tifton, GA 31793. Sixth author: Associate Extension Scientist, Entomology and Nematology Department, University of Florida, Gainesville, FL 32601; Seventh author: Associate Professor, Soil and Water Sciences Department, University of Florida, Gainesville, FL 32601. [^] *Corresponding author's E-mail: ethancarter@ufl.edu
Author Affiliations