Introduction
Peanut (Arachis hypogaea L.) is an allotetraploid species, 2n = 4x = 40, having an AABB genome derived from its putative progenitor species, Arachis duranensis (AA) and Arachis ipaensis (BB) (Burow et al., 2009; Kochert et al., 1996; Seijo et al., 2004), though other wild progenitor species have been proposed (Raina and Mukai, 1999; Smartt et al., 1978). Thus, at least two genes, one from each sub-genome, are likely to encode each protein in peanut. The seed storage proteins of peanut, which are largely 2S albumin (Ara h 2, 6, and 7), 7S globulin (Ara h 1), and 11S globulin (Ara h 3, 4), are allergenic (Ozias-Akins et al., 2006). Of these seed storage proteins, 11S globulins are the most abundant comprising on average 30% of total protein (Kang et al., 2007a), whereas Ara h 2 is the most potent allergen (Flinterman et al., 2007). In the course of our study to assign expressed allergen genes to their sub-genomes (Ramos et al., 2006), we cloned the regulatory and coding regions of Ara h 2 genes. This study revealed that Ara h 2 has two copies, one from the A genome designated Ara h 2.01 and the other from B genome, Ara h 2.02. The two protein isoforms mainly differ by a 12-amino acid insertion in Ara h 2.02. Ara h 2 expression was shown to begin in immature seeds (stages 1 and 2) and peak as seeds matured (stages 3–4) (Kang et al., 2007b). Also, the expression of Ara h 2 was reduced in embryonic axes compared to cotyledons in the post germination (seedling) growth stages. The Ara h 2 transcript was absent from the vegetative parts of the plant as confirmed by Northern blots and RT-PCR analysis (Kang et al., 2007b).
Accurate prediction of promoter function in eukaryotic systems is difficult, since promoters may contain many different motifs, some present in multiple copies, that can interact with transcription factors (Priest et al., 2009). Core promoter regions typically extend 40 bp upstream of a start codon, although upstream promoter regions may encompass more than 200 bp farther upstream (Potenza et al., 2004). Studies on promoters controlling seed specific expression have shown that essential motifs are present within 500 bp upstream of the ATG start codon (Priest et al., 2009).
The primary objective of this study was to identify the promoter region responsible for tissue-specific expression patterns resulting in accumulation of Ara h 2 allergen protein in peanut seeds. Seed-specific storage protein accumulation is regulated by hormonal and metabolic regimes (Priest et al., 2009; Verdier and Thompson, 2008); therefore, in silico analysis of the sequence upstream from the start codon of Ara h 2 was carried out to identify motifs known to play a role in seed-specific expression. Since upstream regulatory regions of arbitrary length often are used to test for promoter function, an ∼ 1 kb (989 bp) region was selected for fusion with reporter genes, green fluorescent protein (Gfp) and ß-glucuronidase (Gus), and expression analysis in peanut, whereas 989 bp, 1927 bp, and 2517 bp regions were tested in Arabidopsis.
Materials and Methods
Motif analysis
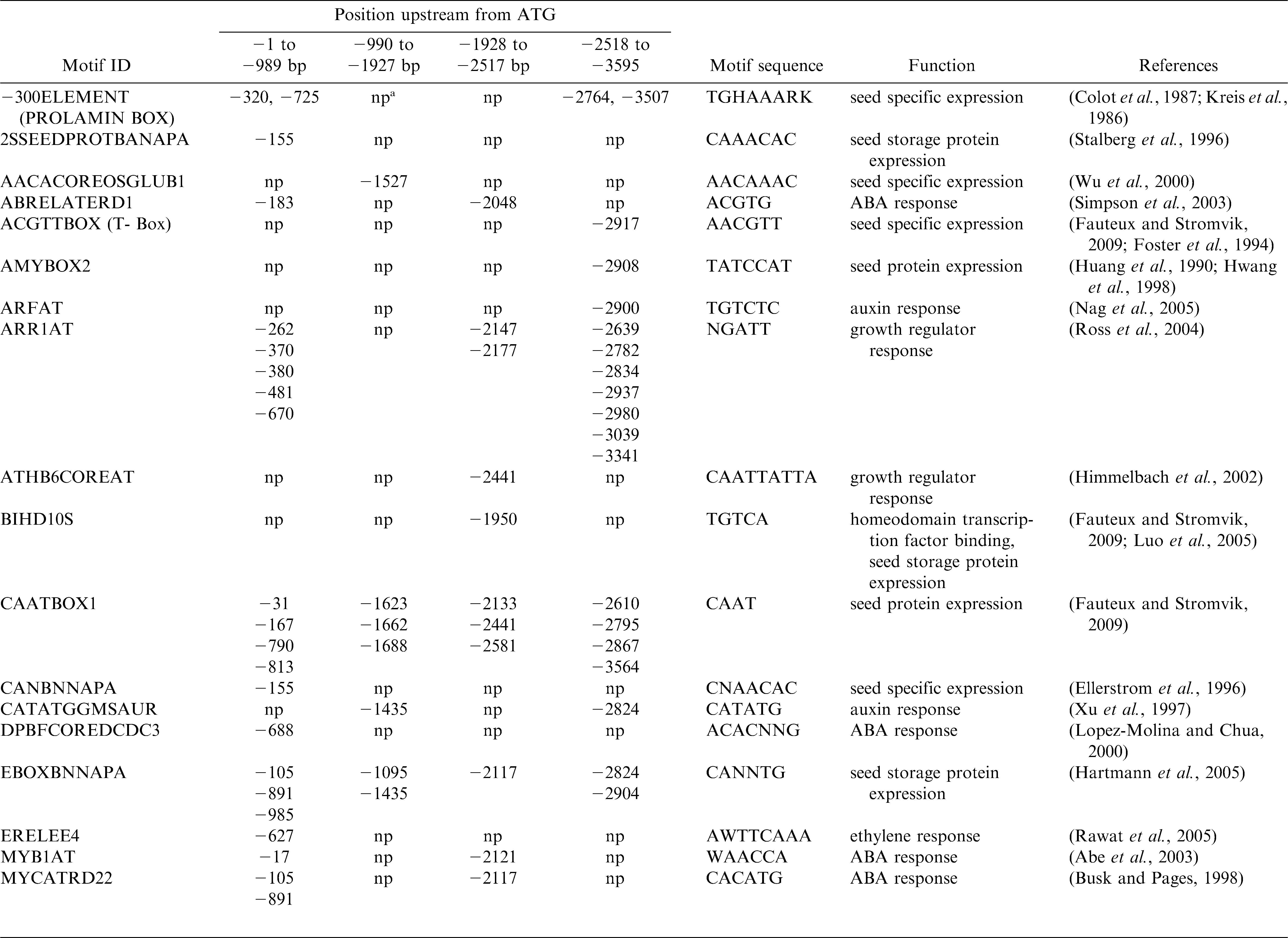
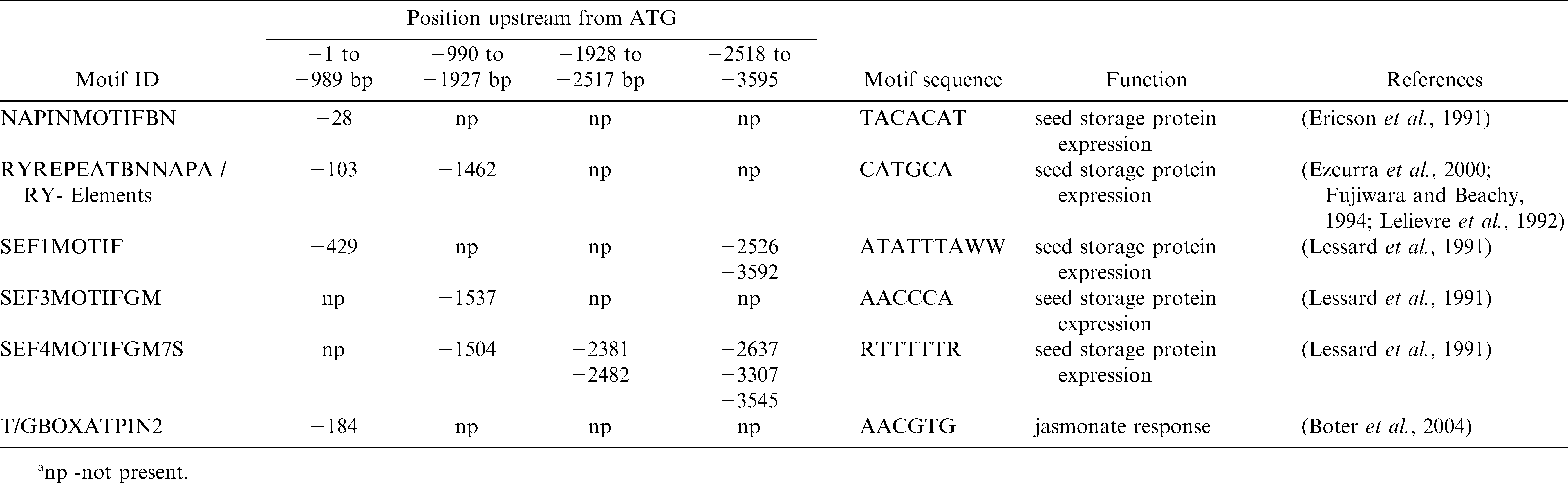
The 3595 bp region upstream of the start codon in Ara h 2.02 was analyzed for different motifs, particularly those known to confer seed specificity, using PLACE (Higo et al., 1999) and PLANTCARE (Lescot et al., 2002) databases and search tools.
Plasmid construct
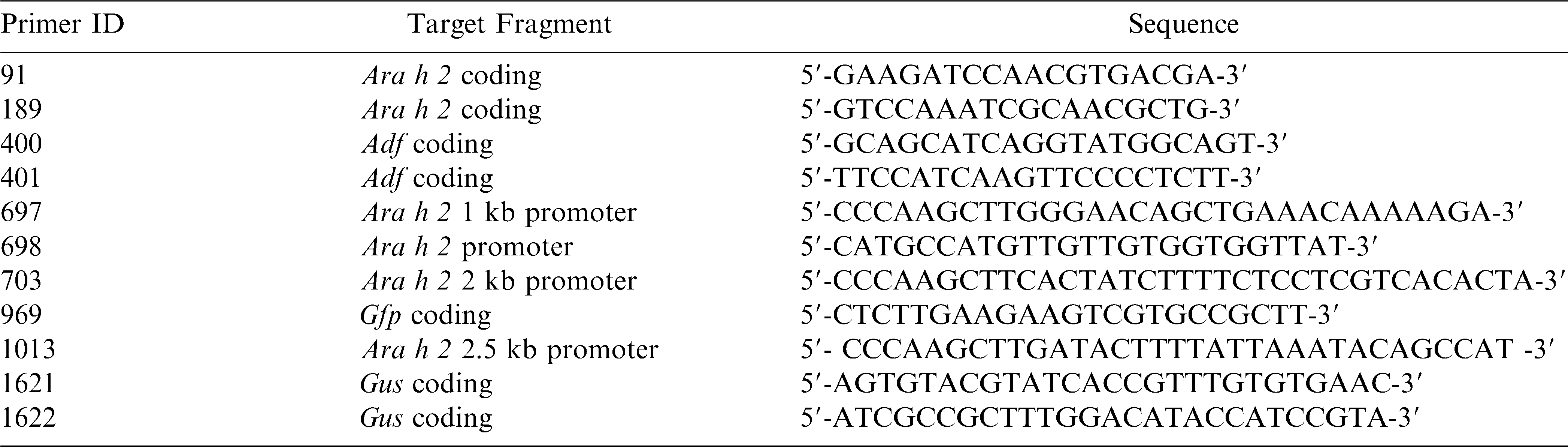
The Ara h 2.02pro∶Gfp∶Gus fusion was constructed from pCambia1304 where the CaMV 35S promoter driving the Gfp∶Gus cassette had been excised by digestion with NcoI and HindIII. The promoter region of Ara h 2.02 was amplified from the plasmid clone 8A2a (GI 148613180, EF 609644) using specific primer combinations, 697 sense / 698 anti-sense (989 bp), 703 sense / 698 anti-sense (1927 bp), or 1013 sense / 698 anti-sense (2517 bp) that included appropriate restriction sites (Table 1). PCR amplification was carried out according to the following conditions: initial denaturation at 94 C for 5 min followed by 35 cycles of 94 C for 30 s, variable annealing temperatures and extension times at 72 C (primers 697 / 698: 48 C for 30 sec, 1-min extension; 703/698: 50 C for 30 sec, 2-min extension; 1013/698: 50 C for 30 sec and 3.5-min extension) with a final extension of 7 min at 72 C and a 4 C hold. The PCR products were digested with NcoI and HindIII and gel purified using a Qiagen kit as per the manufacturer's protocol (Qiagen). After ligation of the digested vector and PCR products, the plasmids were transformed into E. coli DH5α (NEB) competent cells, extracted using the Qiagen Miniprep Kit (Qiagen), and inserts were sequenced. Plasmids were transformed into Agrobacterium tumefaciens strain GV 3101 by electroporation.
Plant tissue culture
Somatic embryos (SE) were obtained by initiating cultures from mature peanut seeds of ‘Georgia Green’ in the SE induction medium which consisted of FN lite macro salts (Samoylov et al., 1998), MS micronutrients (Murashige and Skoog, 1962), B5 vitamins (Gamborg et al., 1968), 1 g/L L-asparagine (Fisher Scientific), 3 mg/L picloram (Dow Agrosciences), 30 g/L sucrose (J. T. Baker, Inc.), and 8 g/L agar (Sigma). The pH was adjusted to 5.7–5.8 prior to autoclaving at 121 C for 20 min. The medium was cooled below 65 C and then filter sterilized L-glutamine solution, 1 g/L (Acros Organics), was added as a supplement prior to dispensing to plates. Seeds were surface sterilized in 50% solution of commercial bleach (Clorox) for 15 min on a shaker, followed by three rinses with sterile deionized water. The plumule region of the embryo axis was carefully excised under a stereomicroscope. Embryogenic cultures were grown in the dark at 26 ± 2 C for up to 9 mo with periodic subculture every 4 wk. Bombardment was conducted 12–14 d after subculture.
Microprojectile bombardment, selection, and regeneration of peanut plants
The Qiagen Plasmid Maxi/Midi Kit was used to extract plasmid DNA from an overnight culture of E. coli cells. DNA quantification was carried out with the Quant-iT PicoGreen dsDNA Assay Kit (Invitrogen). Microprojectile bombardment and selection were conducted as described previously (Chu et al., 2008). Hygromycin-resistant embryogenic tissues were regenerated on MS medium supplemented with 1 mg/L thidiazuron (NOR-AM Chemical Co.) along with 20 mg/L hygromycin (Sigma), 30 g/L sucrose, and 8 g/L agar under a daily photoperiod of 16 h light (50 µmol/m2/s) followed by 8 h of darkness for at least 7–8 wk. Somatic embryos undergoing conversion were transferred to fresh MS medium containing 20 mg/L hygromycin. Embryos displaying well-developed shoot-root axes were subsequently transferred to ventilated vessels containing MS medium with 0.5 mg/L kinetin (Sigma) and 0.25 mg/L 6-benzylaminopurine (Sigma). Well-developed shoots were selected for rooting in Magenta vessels on MS medium supplemented with 0.1 mg/L 1-naphthaleneacetic acid (Sigma). Plants with well-developed roots were transferred to soil, acclimatized, and grown in the greenhouse.
Agrobacterium-mediated Arabidopsis transformation and screening
Arabidopsis transformation was carried out by the standard floral dip procedure (Clough and Bent, 1998). Seeds (T1) from treated inflorescences and subsequent generations (T2–T3) were sterilized in 70% alcohol for 30 sec, 50% Clorox/0.02% Triton X-100 for 6 min, and washed three times with sterile deionized water. The putative transgenic seeds were selected in ½-strength MS medium containing hygromycin (50 mg/L final concentration) and 8 g/L agar. Seeds were plated and stratified at 4 C for 4–7 d before being transferred to a growth chamber at 22 ± 2 C under a 16 h light: 8 h dark photoperiod. Hygromycin-resistant seedlings were transferred to soil and analyzed after 2 wk by PCR and GUS histochemistry to confirm positive transformants. From each T2 line showing 3∶1 Mendelian segregation for hygromycin resistance, at least 8 transgenic plants were chosen for bulk harvest and T3 seeds were screened for hygromycin resistance. Plates showing 100% survival and growth were considered to contain homozygous lines.
PCR analysis
Genomic DNA was extracted as described previously (Singsit et al., 1997). Putative transgenic Arabidopsis plants were screened by amplifying the 989 bp, 1927 bp, or 2517 bp promoter region by the same set of primers used initially for cloning (Table 1). The Gus gene was detected by using primers 1621 sense/1622 antisense, which generated a product of 1061 bp. Actin depolymerizing factor, a housekeeping gene, was detected by primers 400 sense / 401 antisense producing a 450 bp product, and Ara h 2 by primers 91 sense / 189 antisense, for a PCR product size of 337 bp. Putative transgenic peanut plants were screened by using the 1 kb promoter forward primer 697 and Gfp reverse primer 969, product size of 1270 bp. PCR amplification was carried out according to the following conditions: initial denaturation at 94 C for 5 min followed by 35 cycles of 94 C for 30 s, variable annealing temperatures and extension times (annealing temperatures: 697 / 969, 59.3 C; 400 / 401, 53 C; 91 / 189, 54 C; 1621 / 1622, 71 C for 60 s; extension times: 697 / 969, 120 s; 400 / 401, 60 s; 91 / 189, 60 s; 1621 / 1622, 75 s at 72 C) with a final extension of 7 min at 72 C and a 4 C hold. PCR products (8 µl) were separated in 1% (w/v) agarose gels (70V for 1 h) containing ethidium bromide (0.1 µg/ml) with a 1 kb Hi-Low ladder (Minnesota Molecular) as marker. Gels were visualized using a UV transilluminator.
Southern blot hybridization
Genomic DNA was extracted with the Qiagen Plant DNA extraction kit according to the manufacturer's protocol and quantified by a Nanodrop1000 (Nanodrop). Approximately 20 µg DNA was digested overnight at 37°C with 50 units of Hind III restriction enzyme in a 20 µl final reaction volume. Digested DNA fragments were separated overnight by electrophoresis on a 1% agarose gel in 1× TBE buffer at 25V. DNA was blotted onto Genescreen Plus nylon membrane (NEN Life Sciences) by capillary transfer. The membrane was pre-hybridized in 30 ml hybridization buffer (6×SSC, 1% SDS, 100 µg/ml salmon sperm DNA) at 65 C overnight. Probes were labeled with α-32P-dCTP following the protocol described in the PCR labeling kit (Sigma) and cleaned with Sephadex® G-50 (Amersham). Labeled probe was denatured at 95 C for 10 min and placed immediately on ice before adding into the hybridization buffer. Hybridization was conducted at 65 C overnight followed by four washes at the same temperature for 15 min each with the following buffers: 1) 2×SSC, 0.1% SDS; 2) 1× SSC, 0.1% SDS; 3) 0.5× SSC, 0.1% SDS; and 4) 0.1 ×SSC, 0.1% SDS. Signal was detected with the Storm Phosphorimager system (Amersham Biosciences).
Semi-quantitative reverse transcriptase PCR (RT-PCR)
RNA was extracted from 100 mg leaf samples / seed cotyledons (stage 3) (Paik-Ro et al., 2002) of transformed and non-transformed control plants using an RNeasy® Plant Mini Kit (Qiagen). Two rounds of DNase treatment were employed, one at the column purification stage (Qiagen) and another post purification (DNase from Invitrogen) according to the manufacturers' protocols. RT-PCR employed a One Step RT-PCR Kit (Qiagen) according to the manufacturer's instructions. Subsequent conditions were as above for PCR analysis. A set of negative RT controls was also included to test for genomic DNA contamination. Actin depolymerizing factor (Adf) served as a control housekeeping gene and was amplified for 24 cycles. Gus was amplified for 30 cycles and Ara h 2 for 28 cycles for semi-quantitative RT-PCR purposes. Image quantification (ImageQuant TL, Amersham Biosciences) was used to estimate amounts of RT-PCR products by densitometry.
GUS expression analysis
Four seeds were selected from the T1 peanut lines and each seed was divided into two cotyledons, one containing the embryo axis. The half containing the embryo axis was transferred to soil. The other half was further divided into two pieces, one of which was used for GUS assay and the other for GFP analysis. Leaf tissues (2 and 8 wk post planting) were tested for GUS activity. Tissues were immersed in a GUS reagent solution (Jefferson et al., 1987) and incubated overnight at 37 C in the dark with appropriate controls, i.e. non-transformed or 35S transformed plants. An indigo blue color confirmed GUS expression.
GUS quantification
GUS quantification was carried out by grinding 100 mg of sample (seeds/leaf tissues) in 200 µl of extraction buffer (100 µl 0.1 M NaH2PO4, pH 7, 0.014 µl ß-mercaptoethanol, 0.4 µl 0.5 M Na2EDTA, 0.2 mg Sarkosyl, 0.2 µl Triton X-100 and 99.38 µl of dH2O). Cell debris was removed by centrifugation at 12,000 rpm for 15 min at 4 C. The supernatant was transferred to a new tube and protein was estimated by the Bradford assay (Bio-Rad Laboratories). Homogenate (20 µl) containing 5 µg protein was mixed with 80 µl of GUS assay buffer prepared by adding 0.88 mg MUG (4-methylumbelliferyl ß-D-glucuronide; Sigma) in each ml of extraction buffer. A 1 mM MU (sodium methylumbelliferone; Sigma) stock solution was used to prepare dilutions for generation of a standard curve. After 30 min, 475 µl of 200 mM Na2CO3, pH 11.2, stop-buffer was added to 25 µl of each reaction mixture or standard dilution. Duplicate samples of 200 µl for each of three plants per line were loaded in a microtiter plate and fluorescence was determined at excitation and emission wavelengths of 365 nm and 444 nm, respectively. To determine endogenous background GUS activity, plant extract from non-transgenic plants was taken as a control. GUS activity was calculated as pmoles MU/mg protein/min.
GFP signal detection
GFP was detected by placing the plant material (seeds or leaf tissues, as appropriate) directly under blue light and observing with a SV11 epifluorescence stereomicroscope (Zeiss) equipped with a 100W mercury lamp as the light source with a 480 ± 30 nm excitation filter and a 515 nm long-pass emission filter (Chroma Technology). Images were captured with an Axiocam digital camera (Zeiss).
Statistical analysis
Data were subjected to Student's t-test using standard MINITAB version 15, statistical software (Minitab, Inc.).
Results
Motif analysis of Ara h 2.02 upstream sequence
A detailed analysis for short conserved putative cis-regulatory sequences was undertaken for 3595 bp upstream of the translational start site for Ara h 2.02 using PLACE and PLANTCARE databases and search tools with the sequence information of EF 609644, GI148613180. The analysis revealed cis-regulatory elements known to confer seed-specific expression and response to growth regulators (Table 2). Since numerous cis-regulatory elements responsible for seed-specific expression were present in the 1 kb promoter region (Table 2), both peanut and Arabidopsis transformation were conducted with this promoter-reporter construct.
Transgenic peanut
Copy number determination by Southern blot hybridization
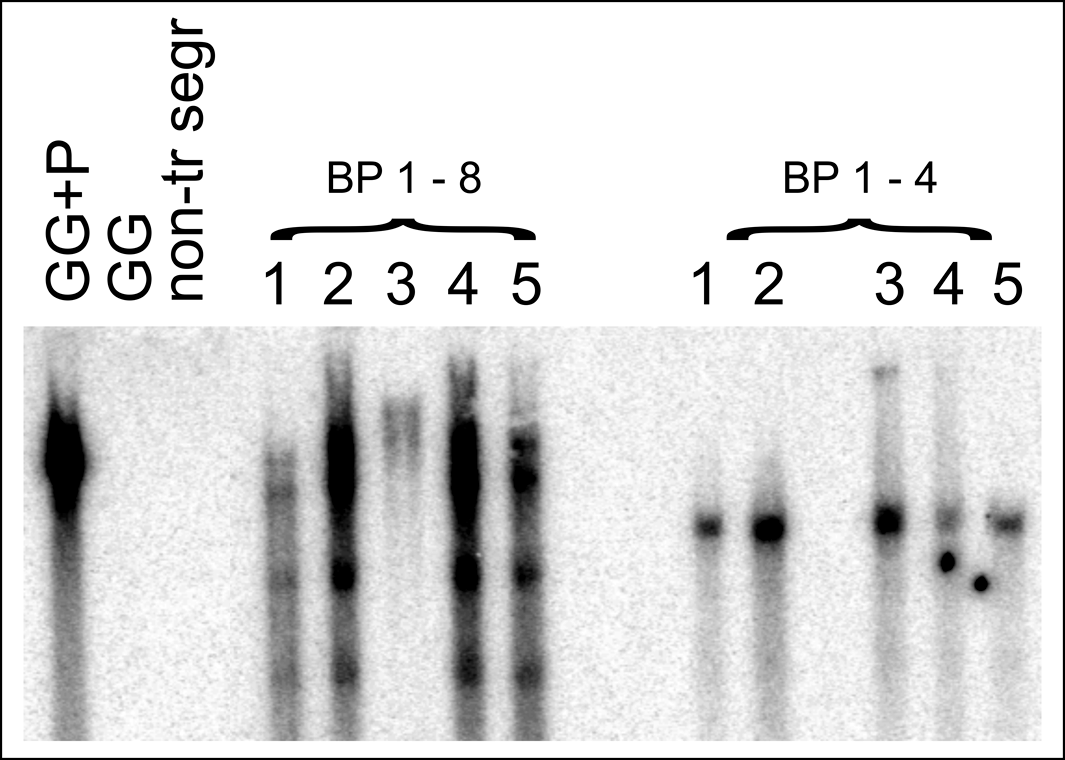
Two fertile transgenic lines, designated BP 1.4 and BP 1.8, were obtained via hygromycin selection of peanut embryogenic cultures bombarded with a plasmid carrying the Ara h 2.02pro∶Gfp∶Gus fusion. Southern blot hybridization results showed that both the lines were homozygous in the T3 generation although BP 1.4 showed one band and BP 1.8 showed four bands hybridizing with the Gus gene (Fig. 1). Transgene integration at a single locus seems likely given the similar hybridization pattern among all T3 progeny from each parental line.
Southern blot hybridization analysis from two Ara h 2.02pro∶Gfp∶Gus transgenic lines, BP 1.4 and BP 1.8, in the T3 generation (5 plants from each individual line numbered 1 through to 5). Upon digestion with HindIII, BP 1.4 showed one band whereas BP 1.8 showed four bands (except for lane 3 which was incompletely digested) when probed with Gus. Controls include a non-transgenic segregant from BP 1.8 (non-tr segr), non-transformed ‘Georgia Green’ (GG), and non-transformed ‘Georgia Green’ DNA mixed with HindIII-digested plasmid DNA (GG+P).
GUS and GFP expression in seeds
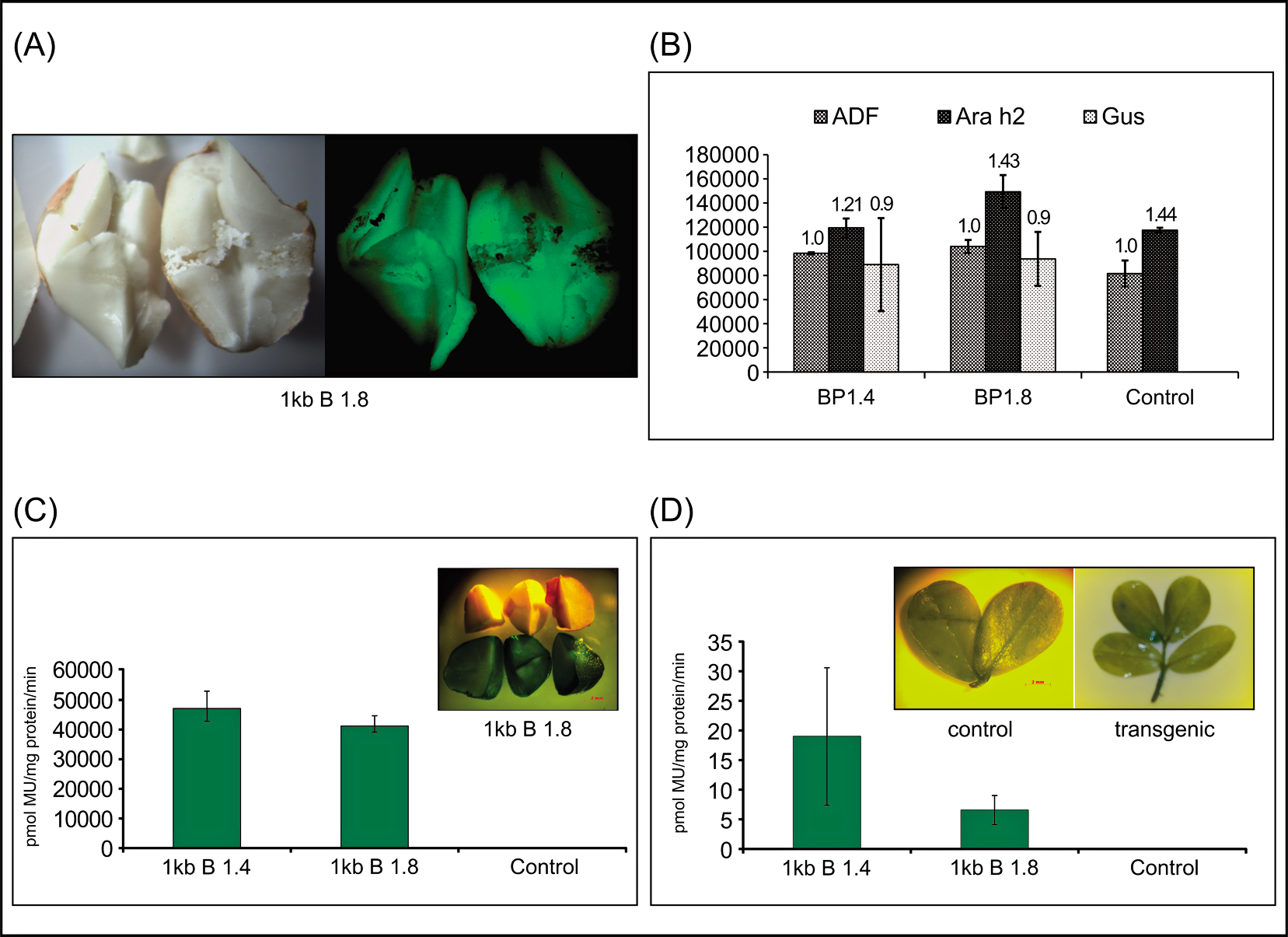
GUS and GFP expression was restricted to the cotyledons of seeds and essentially absent from vegetative tissues (Fig. 2). Semi-quantitative RT-PCR results showed that Ara h 2 gene expression in transgenic stage 3 peanut seeds was similar to the control (Fig. 2B), particularly when expressed as a fraction of Adf (Adf : Ara h 2 ratio of 1.32 in transgenic vs. 1.44 in control). Between the two transgenic lines BP 1.4 and BP 1.8, Gus transcripts were not significantly different at P<0.01 (Fig. 2B). Even though BP 1.8 carried a higher copy number of Ara h 2.02pro∶Gfp∶Gus than BP 1.4, GUS expression levels as measured in pmol MU/mg protein/min were not significantly different between the two lines for both tissues tested, although GUS expression was shown to be several orders of magnitude higher in stage 3 seeds compared with 2-wk-old vegetative tissues of transgenic peanut lines (Fig. 2C, D).
Transgenic 1 kb Ara h 2.02pro∶Gfp∶Gus peanut lines (T2 generation) (A) GFP expression (right) in transgenic cotyledons. Left panel shows half-seeds under white light. (B) Densitometric quantification of Adf (Actin depolymerizing factor), Ara h 2 and Gus transcripts. Error bars indicate standard error of mean. Numbers above bars are ratio of respective gene transcript level to Adf transcript level. (C) Quantitative GUS assay with transgenic (B1.4 and B1.8) and non-transgenic control seeds. Inset - GUS expression in peanut cotyledons (top – non-transgenic; bottom – transgenic). (D) Quantitative GUS assay with 2-wk-old leaves from transgenic (B1.4 and B1.8) and non-transgenic control plants. Inset - leaflets stained for GUS 2 wk post-planting; non-transgenic (left panel), transgenic (right panel).
GUS expression in vegetative tissues declines post germination
Faint GUS expression was observed in vegetative parts of young seedlings 2 wk after germination and GUS expression was undetectable in 8-wk-old leaves. The presence of transcript (Gfp∶Gus cassette) was confirmed by RT-PCR in 2-wk-old tissues from T2 individuals (Fig. 3A) and also by semi-quantitative RT-PCR in the T3 generation (Gus transcript only, data not shown). There was no significant difference between the two transgenic lines (BP 1.4 and BP 1.8) in the level of Gus transcripts from 2-wk-old vegetative tissues (data not shown). Quantitative GUS analysis showed that expression in line BP 1.4 was similar to BP 1.8 after a 30-min assay (Fig. 2D). Negative controls consisted of two lines, which were transgene nulls segregating from the transformed lines, i.e., lacking the transgene cassette (confirmed by PCR and Southern blot). Neither GUS enzyme activity nor Gus transcripts was detectable in 8-wk-old plants from either transgenic line at any of the three generations.
(A) Top panel - RT-PCR with primers for detecting Gfp∶Gus transcripts in 2-wk-old leaves of transgenic BP 1.8 (1 kb Ah) and non-transgenic (non-tr Ah) peanut, and leaves from transgenic Arabidopsis plants carrying 1 kb (1 kb At) or 2 kb (2 kb At) promoter regions. Each sample included a corresponding negative control (no RT) with all reaction components except reverse transcriptase enzyme. Bottom panel - actin depolymerizing factor (Adf) primers were also used to confirm integrity of the cDNA. (B) GUS expression in Arabidopsis vegetative tissue transformed with the 1 kb promoter (line no. 58). (C) GUS expression in Arabidopsis seeds from plants transformed with the 2 kb promoter (line no. 67); top row of seeds is non-transgenic control. (D) GUS expression in Arabidopsis vegetative tissue transformed with the 2.5 kb promoter (line no. 1).
Transgenic Arabidopsis
Six transgenic Arabidopsis T1 lines containing the 1 kb Ara h 2.02pro∶Gfp∶Gus cassette, five with the 2 kb promoter, and six with the 2.5 kb promoter were recovered. Five transgenic lines containing the reporter genes controlled by the CaMV 35S promoter also were obtained. Single locus lines for each construct were identified, namely line numbers 58 (1 kb); 67, 68 (2 kb) and 37, 38 (CaMV 35S). Transgenic lines containing the 2.5 kb promoter showed poor seed set. Similar constitutive GUS expression patterns from all three Ara h 2.02 promoter regions, and the CaMV 35S promoter, were observed in transgenic Arabidopsis plants compared to no expression in the non-transformed controls (Fig. 3).
Discussion
The present study investigates the potential of using promoter sequence from Ara h 2.02 as a seed specific promoter (SSP). Transcriptional regulation of any gene in plants is controlled by a complex interaction of transcription factors and cis-regulatory elements (Dare et al., 2008). Several important seed specific regulatory motifs found in the Ara h 2.02 promoter (PROLAMINBOX, 2SSEEDPROTBANAPA) were identified for high level of activity of the napA promoter from Brassica napus. Deletion of the region containing these motifs from the napA promoter decreased GUS expression in transgenic tobacco plants (Stalberg et al., 1993). A RY motif (CATGCA / CACGT) is commonly found in seed storage protein genes (Fauteux and Stromvik, 2009), including Ara h 1 (Viquez et al., 2003) and Ara h 2 (this study), and acts by enhancing seed specific gene expression (Dickinson et al., 1988). Certain regulatory elements are fundamental to growth regulator signaling pathways which are important in seed development. The ARR1AT element found in the Ara h 2.02 promoter has been shown in other studies to bind ARR1 proteins which are response regulators, act as transcription factors (Kuriakose et al., 2009), and are involved in cytokinin signaling. Abscisic acid (ABA) also plays an important role in expression of seed-storage protein genes and embryogenesis related genes (Luo et al., 2008). Plant et al. (1994) showed that ABA positively regulated expression of GUS in transgenic Brassica containing the oleosin promoter from Arabidopsis fused to the reporter gene. Rowley and Herman (Rowley and Herman, 1997) found that soybean oleosin genes contain regulatory elements in the upstream region which also were found in vacuolar storage protein genes. These included the −300 element (also present in the Ara h 2.02 promoter) that moreover quantitatively regulates prolamin expression, an endosperm protein in cereals (Colot et al., 1987). Qu et al. (2008) found prolamin and endosperm-specific motifs in all but one rice glutelin gene promoter which controlled seed specific expression.
Our results for reporter gene expression in peanut under the control of the Ara h 2 promoter largely conform with the results obtained by Kang et al. (2007b) who showed that Ara h 2 polypeptides were abundant in mature seeds and diminished after germination during seedling growth. An unexpected result was the lack of reporter gene expression in embryo axes, although expression in cotyledons was strong. Furthermore, we did not detect Ara h 2 transcripts in either 2-wk- or 8–wk-old leaf tissues even though reporter gene transcripts could be observed as faint RT-PCR products in the 2-wk-old samples, data which also are consistent with Kang et al. (2007b) who could not detect Ara h 2 transcripts in leaves by Northern blot analysis. The present study also shows that a higher copy number of the transgene did not result in a difference in the steady state level of transcripts. Reporter gene expression pattern largely paralleled Ara h 2 expression pattern with the minor exceptions described above.
Therefore, the 1 kb promoter region reported in this study is sufficient for seed specific reporter gene expression, although it only confers seed-specificity in peanut and not in Arabidopsis. In addition to cis-regulatory elements, transcription factors that bind to a particular element and interact with other recruited transcription factors ultimately control transcriptional activation or suppression (Verdier and Thompson, 2008). The lack of seed-specificity in Arabidopsis could be due to the absence, divergence, or temporal difference in expression of trans-acting factors, which are present and functioning in peanut. There is precedent for altered expression patterns from promoters introduced into a heterologous system. For example, a −765 bp alfalfa isoflavone reductase promoter fused with Gus conferred expression in alfalfa root meristems, cortex and nodules, following the pattern of endogenous isoflavone reductase expression, but in transgenic tobacco, GUS expression was observed in additional vegetative tissues, including leaf and stem, as well as in reproductive tissues (Oommen et al., 1994). While multiple motifs with evidence for seed-specific regulation and growth regulator response may contribute to the expression pattern for Ara h 2 in peanut seeds, further promoter dissection with transgenic peanut would be laborious because of its low transformation efficiency. However, from a comparative perspective, the set of seed-specific motifs present in the Ara h 2.02 promoter reported during this work are very attractive for future in vitro or in vivo analyses in other legume systems to understand evolutionary differences.
Summary and Conclusions
The present study describes a novel promoter from a peanut Ara h 2.02 protein with a seed specific expression pattern. However, the promoter-regulated reporter gene expression patterns differed in homologous versus heterologous (Arabidopsis) systems. Although an approximately 1 kb fragment upstream from the translational start codon in peanut was sufficient to confer seed-specific expression of GUS and GFP in peanut, even up to ∼2.5 kb of upstream sequence was not sufficient in Arabidopsis. Thus, this promoter is a valuable molecular tool accessible for seed-specific expression of transgenes for genetic improvement of peanut and testing in heterologous legumes.
Acknowledgements
This work was supported by the Georgia Seed Development Commission/University of Georgia Research Foundation, the Peanut Foundation, and USDA Specific Cooperative Agreement 58-6435-6-050.
Literature Cited
Abe H Urao T Ito T Seki M Shinozaki K and Yamaguchi-Shinozaki K 2003 Arabidopsis AtMYC2 (bHLH) and AtMYB2 (MYB) function as transcriptional activators in abscisic acid signaling Plant Cell 15 : 63 – 78 .
Boter M Ruiz-Rivero O Abdeen A and Prat S 2004 Conserved MYC transcription factors play a key role in jasmonate signaling both in tomato and Arabidopsis Genes & Development 18 : 1577 – 1591 .
Burow M. D Simpson C.E Faries M.W Starr J.L and Paterson A.H 2009 Molecular biogeographic study of recently described B- and A-genome Arachis species, also providing new insights into the origins of cultivated peanut Genome 52 : 107 – 119 .
Busk P.K and Pages M 1998 Regulation of abscisic acid-induced transcription Plant Mol. Biol. 37 : 425 – 435 .
Chu Y Faustinelli P Ramos M.L Hajduch M Stevenson S Thelen J.J Maleki S.J Cheng H.P and Ozias-Akins P 2008 Reduction of IgE binding and nonpromotion of Aspergillus flavus fungal growth by simultaneously silencing Ara h 2 and Ara h 6 in peanut J. Agric. Food Chem. 56 : 11225 – 11233 .
Clough S.J and Bent A.F 1998 Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana Plant J 16 : 735 – 743 .
Colot V Robert L.S Kavanagh T.A Bevan M.W and Thompson R.D 1987 Localization of sequences in wheat endosperm protein genes which confer tissue-specific expression in tobacco EMBO J. 6 : 3559 – 3564 .
Dare A.P Schaffer R.J Lin-Wang K Allan A.C and Hellens R.P 2008 Identification of a cis-regulatory element by transient analysis of co-ordinately regulated genes Plant Methods 4 : 17 .
Dickinson C.D Evans R.P and Nielsen N.C 1988 RY repeats are conserved in the 5′-flanking regions of legume seed-protein genes Nucl. Acids Res. 16 : 371 – 371 .
Ellerstrom M Stalberg K Ezcurra I and Rask L 1996 Functional dissection of a napin gene promoter: Identification of promoter elements required for embryo and endosperm-specific transcription Plant Mol. Biol. 32 : 1019 – 1027 .
Ericson M.L Muren E Gustavsson H-O Josefsson L-G and Rask L 1991 Analysis of the promoter region of napin genes from Brassica napus demonstrates binding of nuclear protein invitro to a conserved sequence motif Eur. J. Bioch. 197 : 741 – 746 .
Ezcurra I Wycliffe P Nehlin L Ellerstrom M and Rask L 2000 Transactivation of the Brassica napus napin promoter by ABI3 requires interaction of the conserved B2 and B3 domains of ABI3 with different cis-elements: B2 mediates activation through an ABRE, whereas B3 interacts with an RY/G-box Plant Journal 24 : 57 – 66 .
Fauteux F and Stromvik M.V 2009 Seed storage protein gene promoters contain conserved DNA motifs in Brassicaceae, Fabaceae and Poaceae BMC Plant Biol. 9 : 126 .
Flinterman A.E Van Hoffen E Den Hartog Jager C.F Koppelman S Pasmans S.G Hoekstra M.O Bruijnzeel-Koomen C.A Knulst A.C and Knol E.F 2007 Children with peanut allergy recognize predominantly Ara h 2 and Ara h 6, which remains stable over time Clin. Exp. Allergy 37 : 1221 – 1228 .
Foster R Izawa T and Chua N.H 1994 Plant bZIP proteins gather at ACGT elements FASEB J. 8 : 192 – 200 .
Fujiwara T and Beachy R.N 1994 Tissue-specific and temporal regulation of a beta-conglycinin gene - roles of the RY repeat and other cis-acting elements Plant Mol. Biol. 24 : 261 – 272 .
Gamborg O.L Miller R.A and Ojima K 1968 Nutrient requirements of suspension cultures of soybean root cells Exp. Cell Rev. 50 : 151 – 158 .
Hartmann U Sagasser M Mehrtens F Stracke R and Weisshaar B 2005 Differential combinatorial interactions of cis-acting elements recognized by R2R3-MYB, bZIP, and bHLH factors control light-responsive and tissue-specific activation of phenylpropanoid biosynthesis genes Plant Mol. Biol. 57 : 155 – 171 .
Higo K Ugawa Y Iwamoto M and Korenaga T 1999 Plant cis-acting regulatory DNA elements (PLACE) database Nucl.Acids Res. 27 : 297 – 300 .
Himmelbach A Hoffmann T Leube M Hohener B and Grill E 2002 Homeodomain protein AthB6 is a target of the protein phosphatase ABI1 and regulates hormone responses in Arabidopsis EMBO J. 21 : 3029 – 3038 .
Huang N Sutliffe T.D Litts J.C and Rodriguez R.L 1990 Classification and characterization of the rice alpha-amylase multigene family Plant Molec. Biol. 14 : 655 – 668 .
Hwang Y.S Karrer E.E Thomas B.R Chen L and Rodriguez R.L 1998 Three cis-elements required for rice alpha-amylase Amy3D expression during sugar starvation Plant Mol. Biol. 36 : 331 – 341 .
Jefferson R.A Kavanagh T.A and Bevan M.W 1987 Gus fusions: β-glucusonidase as a sensitive and versatile gene fusion marker in higher plants EMBO J. 6 : 3901 – 3907 .
Kang I.H Gallo M and Tillman B.L 2007a Distribution of allergen composition in peanut (Arachis hypogaea L.) and wild progenitor (Arachis) species Crop Sci. 47 : 997 – 1003 .
Kang I.H Srivastava P Ozias-Akins P and Gallo M 2007b Temporal and spatial expression of the major allergens in developing and germinating peanut seed Plant Physiol. 144 : 836 – 845 .
Kochert G Stalker H.T Gimenes M Galgaro L Lopes C.R and Moore K 1996 RFLP and cytogenetic evidence on the origin and evolution of allotetraploid domesticated peanut, Arachis hypogaea (Leguminosae) Am. J. Bot. 83 : 1282 – 1291 .
Kreis M Williamson M.S Forde J Schmutz D Clark J Buxton B Pywell J Marris C Henderson J Harris N Shewry P.R Forde B.G and Miflin B.J 1986 Differential gene-expression in the developing barley endosperm Phil. Trans. Royal Soc. London Ser. B-Biol. Sci. 314 : 355 – 365 .
Kuriakose B Arun V Gnanamanickam S.S and Thomas G 2009 Tissue-specific expression in transgenic rice and Arabidopsis thaliana plants of GUS gene driven by the 5′ regulatory sequences of an anther specific rice gene YY2 Plant Sci. 177 : 390 – 397 .
Lelievre J.M Oliveira L.O and Nielsen N.C 1992 5′-CATGCAT-3′ elements modulate the expression of glycinin genes Plant Physiol. 98 : 387 – 391 .
Lescot M Dehais P Thijs G Marchal K Moreau Y Van De Peer Y Rouze P and Rombauts S 2002 PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences Nucl. Acids Res. 30 : 325 – 327 .
Lessard P.A Allen R.D Bernier F Crispino J.D Fujiwara T and Beachy R.N 1991 Multiple nuclear factors interact with upstream sequences of differentially regulated beta-conglycinin genes Plant Mol. Biol. 16 : 397 – 413 .
Lopez-Molina L and Chua N.H 2000 A null mutation in a bZIP factor confers ABA-insensitivity in Arabidopsis thaliana Plant Cell Physiol. 41 : 541 – 547 .
Luo H Song F Goodman R.M and Zheng Z 2005 Up-regulation of OsBIHD1, a rice gene encoding BELL homeodomain transcriptional factor, in disease resistance responses Plant Biol. 7 : 10 .
Luo K.M Zhang G.F Deng W Luo F.T Qiu K and Pei Y 2008 Functional characterization of a cotton late embryogenesis-abundant d113 gene promoter in transgenic tobacco Plant Cell Rep. 27 : 707 – 717 .
Murashige T and Skoog F 1962 A revised medium for rapid growth and bioassays with tobacco cultures Physiol. Plant. 15 : 473 – 497 .
Nag R Maity M.K and Dasgupta M 2005 Dual DNA binding property of ABA insensitive 3 like factors targeted to promoters responsive to ABA and auxin Plant Mol. Biol. 59 : 821 – 838 .
Oommen A Dixon R.A and Paiva N.L 1994 The elicitor-inducible alfalfa isoflavone reductase promoter confers different patterns of developmental expression in homologous and heterologous transgenic plants Plant Cell 6 : 1789 – 1803 .
Ozias-Akins P Ramos M.L and Chu Y 2006 Hypoallergenic foods beyond infant formulas, In: Maleki S et al (Eds.) , Food allergy ASM Press , Washington, D.C . pp. 287 – 306 .
Paik-Ro O.G Seib J.C and Smith R.L 2002 Seed-specific, developmentally regulated genes of peanut Theor. Appl. Genet. 104 : 236 – 240 .
Plant A.L van Rooijen G.J.H Anderson C.P and Moloney M.M 1994 Regulation of an Arabidopsis oleosin gene promoter in transgenic Brassica napus Plant Mol. Biol. 25 : 193 – 205 .
Potenza C Aleman L and Sengupta-Gopalan C 2004 Targeting transgene expression in research, agricultural, and environmental applications: Promoters used in plant transformation In Vitro Cell. Devel. Biol.-Plant 40 : 1 – 22 .
Priest H.D Filichkin S.A and Mockler T.C 2009 Cis-regulatory elements in plant cell signaling Curr. Opin. Plant Biol. 12 : 643 – 649 .
Qu L.Q Xing Y.P Liu W.X Xu X.P and Song Y.R 2008 Expression pattern and activity of six glutelin gene promoters in transgenic rice J. Exp. Bot. 59 : 2417 – 2424 .
Raina S.N and Mukai Y 1999 Genomic in situ hybridization in Arachis (Fabaceae) identifies the diploid wild progenitors of cultivated (A. hypogaea) and related wild (A. monticola) peanut species Plant Syst. Evol. 214 : 251 – 262 .
Ramos M Fleming G Chu Y Akiyama Y Gallo M and Ozias-Akins P 2006 Chromosomal and phylogenetic context for conglutin genes in Arachis based on genomic sequence Mol. Genet. Genomics 275 : 578 – 592 .
Rawat R Xu Z.F Yao K.M and Chye M.L 2005 Identification of cis-elements for ethylene and circadian regulation of the Solanum melongena gene encoding cysteine proteinase Plant Mol. Biol. 57 : 629 – 643 .
Ross E.J.H Stone J.M Elowsky C.G Arredondo-Peter R Klucas R.V and Sarath G 2004 Activation of the Oryza sativa non-symbiotic haemoglobin-2 promoter by the cytokinin-regulated transcription factor, ARR1 J. Exp. Bot. 55 : 1721 – 1731 .
Rowley D.L and Herman E.M 1997 The upstream domain of soybean oleosin genes contains regulatory elements similar to those of legume storage proteins Biochim. Biophys. Acta -Lipids and Lipid Metabolism 1345 : 1 – 4 .
Samoylov V.M Tucker D.M and Parrott W.A 1998 Soybean [Glycine max (L.) Merill] embryogenic cultures: The role of sucrose and total nitrogen content on proliferation In Vitro Cell. Dev. Biol. 34 : 8 – 13 .
Seijo J.G Lavia G.I Fernandez A Krapovickas A Ducasse D and Moscone E.A 2004 Physical mapping of the 5s and 18s–25s rRNA genes by FISH as evidence that Arachis duranensis and A. ipaensis are the wild diploid progenitors of A. hypogaea (Leguminosae) Am. J. Bot. 91 : 1294 – 1303 .
Simpson S.D Nakashima K Narusaka Y Seki M Shinozaki K and Yamaguchi-Shinozaki K 2003 Two different novel cis-acting elements of erd1, a clpa homologous Arabidopsis gene function in induction by dehydration stress and dark-induced senescence Plant J. 33 : 259 – 270 .
Singsit C Adang M.J Lynch R.E Anderson W.F Wang A Cardineau G and Ozias-Akins P 1997 Expression of a Bacillus thuringiensis CryIA(c) gene in transgenic peanut plants and its efficacy against lesser cornstalk borer Transgen. Res. 6 : 169 – 176 .
Smartt J Gregory W.C and Gregory M.P 1978 The genomes of Arachis hypogaea. 2. The implications in interspecific breeding Euphytica 27 : 677 – 680 .
Stalberg K Ellerstrom M Josefsson L.G and Rask L 1993 Deletion analysis of a 2S seed storage protein promoter of Brassica napus in transgenic tobacco Plant Mol. Biol. 23 : 671 – 683 .
Stalberg K Ellerstom M Ezcurra I Ablov S and Rask L 1996 Disruption of an overlapping E-box/ABRE motif abolished high transcription of the napA storage-protein promoter in transgenic Brassica napus seeds Planta 199 : 515 – 519 .
Verdier J and Thompson R.D 2008 Transcriptional regulation of storage protein synthesis during dicotyledon seed filling Plant Cell Physiol. 49 : 1263 – 1271 .
Viquez O.M Konan K.N and Dodo H.W 2003 Structure and organization of the genomic clone of a major peanut allergen gene, Ara h 1 Mol. Immunol. 40 : 565 – 571 .
Wu C.Y Washida H Onodera Y Harada K and Takaiwa F 2000 Quantitative nature of the prolamin-box, ACGT and AACA motifs in a rice glutelin gene promoter: Minimal cis-element requirements for endosperm-specific gene expression Plant J. 23 : 415 – 421 .
Xu N.F Hagen G and Guilfoyle T 1997 Multiple auxin response modules in the soybean SAUR 15A promoter Plant Sci. 126 : 193 – 201 .