Introduction
Aflatoxin contamination of peanuts has been recognized as a serious food safety problem since the discovery that aflatoxins in Brazilian peanut meal were the causative agents of an intoxication (turkey X disease) that killed thousands of turkeys and other animals in England in 1960 (Lancaster et al., 1961; Sargeant et al., 1961). Aflatoxins occur naturally as four related chemicals, designated B1, B2, G1, and G2, that are not only acutely toxic, but are also highly carcinogenic (Eaton and Groopman, 1994). Contamination of peanuts results from invasion and growth of the fungi, Aspergillus flavus (Link) and A. parasiticus (Speare), which can occur both prior to and after harvest (Diener et al., 1987). Preharvest aflatoxin contamination of peanuts is associated with plant stress that occurs under late-season drought conditions. Most of this contamination can be prevented if adequate irrigation is provided during the crop maturation period, but this is not an option for the majority of peanut producers. Good agronomic practices to maintain optimum plant health can help reduce the degree of contamination when drought conditions are not severe, but in years with significant late-season water stress aflatoxin contamination can be widespread. Because of the toxicity and carcinogenicity of the aflatoxins, their concentration in both human food and animal feeds is closely monitored and regulated. In the USA, the Food and Drug Administration has established an action level of 20 µg/kg for total aflatoxins in human food. Levels for animals are variable and depend on their sensitivity to the toxins. Many other countries have established even lower tolerances. For example, the European Union allowance is only 2 µg/kg for aflatoxin B1 and 4 µg/kg for total aflatoxins. While these regulations ensure a much safer food supply, they place an economic burden on various segments of the peanut industry to guarantee that they are met.
During research studies designed to determine the primary route of invasion of peanuts by A. flavus and A. parasiticus, we observed that inoculation of peanut plots with a toxigenic, color mutant strain of A. parasiticus (NRRL 6111) resulted in a dominance of that strain both in the soil and in peanuts (Cole et al., 1986). It appeared that the A. parasiticus color mutant, commonly referred to as the NOR mutant because it produces norsolorinic acid, competitively excluded other strains of A. flavus and A. parasiticus in the colonization of peanuts. Thus was born the idea for an aflatoxin biocontrol strategy using nontoxigenic strains of A. flavus or A. parasiticus. The purpose of this paper is to review the development of this biocontrol strategy and describe the current state of the technology.
Initial proof of concept
After observing the dominance of the NOR mutant both in soil and in subsequently harvested peanuts (Cole et al., 1986), the hypothesis was made that inoculation of soil with a nontoxigenic strain would result in reduced aflatoxin in peanuts if the nontoxigenic strain was similarly dominant and competitive. To test the hypothesis, a naturally-occurring, non-aflatoxigenic, O-methylsterigmatocystin (OMST)-producing strain of A. parasiticus (NRRL 13539) was grown on liquid media, homogenized in water and Tween 20, and applied over peanut plants at 30 and 100 d after planting using a garden sprinkler (Dorner et al., 1992). Peanuts were subjected to late-season drought in environmental control plots (Blankenship et al., 1983) at the National Peanut Research Laboratory, in Dawson, Georgia harvested, and analyzed for aflatoxin and OMST. Inconclusive results indicated that aflatoxin concentrations were reduced and OMST concentrations were increased in peanuts from inoculated soil. When the experiment was repeated the following year, treated edible grade peanuts contained 1 µg/kg of aflatoxin compared with 96 µg/kg in controls. Treated peanuts also contained 172 µg/kg of OMST, providing strong evidence that the applied strain of A. parasiticus essentially displaced toxigenic strains of A. flavus and A. parasiticus in peanuts. In the 3rd yr of this study, another non-aflatoxigenic strain of A. parasiticus also was tested. This was a UV-induced mutant of the OMST-producing strain that was subsequently shown to produce versicolorin A, an early precursor in the aflatoxin biosynthetic pathway. This strain also was effective in reducing aflatoxin concentrations. Results of these experiments demonstrated the potential for biological control of aflatoxin contamination in peanuts using the concept of competitive exclusion. However, several areas still needed to be investigated before a commercial biocontrol product could be fully developed and released, including: (a) strain selection, (b) inoculum rate, (c) timing of field application of inoculum, (d) effect on storage contamination, (e) delivery method, (f) safety, and (g) a large scale demonstration of efficacy.
Strain selection
The strains of A. parasiticus used in the initial experiments demonstrating proof of concept were considered unacceptable for a commercial biopesticide because they accumulated compounds (OMST and versicolorin A) that were toxic, mutagenic, or carcinogenic. Any fungus used as a commercial biocontrol agent should not produce potentially harmful metabolites because if the agent is successful, the commodity will likely be contaminated with those metabolites, as was seen with OMST. Therefore, many isolates of A. flavus and A. parasiticus were screened for their ability to produce aflatoxins, cyclopiazonic acid, OMST, versicolorins, and other aflatoxin biosynthetic precursors. In addition, efforts were continued to produce mutant strains through UV irradiation. Eventually, an isolate of A. flavus (NRRL 21882) was found as a natural contaminant of a peanut, and an orange-colored mutant of A. parasiticus (NRRL 21369) was produced by UV irradiation of the NOR mutant. A UV-induced orange color mutant (NRRL 21368) of the natural isolate of A. flavus (NRRL 21882) also was produced for use in plot and field studies to simplify identification of the competitive strain in soil and peanut dilution platings. Studies to test these various isolates began in 1992 in the environmental control plots. Individual plots (12.2 m × 5.5 m; 6 rows) were partitioned so that half of each plot could be inoculated while the other half served as an uninoculated control. For inoculation of soil, isolates were grown in shake or tumbling culture (to minimize sporulation) on sterilized, long-grain rice. After colonized rice was applied to soil, isolates would resume growth and sporulate until rice grains were consumed.
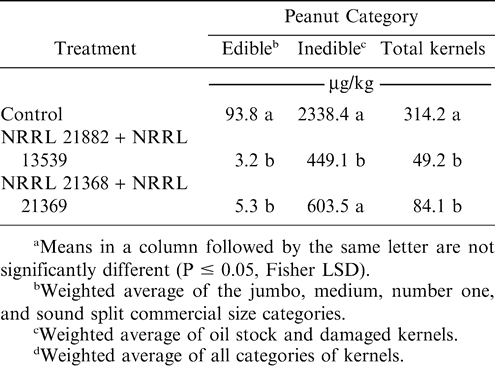
In 1992, plots were inoculated with the rice inocula of the naturally-occurring, nontoxigenic A. flavus (NRRL 21882) alone, the OMST-producing strain of A. parasiticus (NRRL 13539) alone, a combination of those two strains, and the A. parasiticus color mutant (NRRL 21369) alone. Treatments were not replicated because of space limitations. Results of aflatoxin analyses of harvested peanuts (Table 1) indicated that all applications reduced aflatoxin concentrations although the color mutant of A. parasiticus (62.9% reduction in total kernels) did not appear to be as effective as the naturally occurring isolates. The mixture of nontoxigenic strains of A. flavus and A. parasiticus provided the greatest reduction, reaching 98.9% in edible category kernels.
By the 1993 crop year, a color mutant (designated NRRL 21368) had been produced from A. flavus NRRL 21882. It was applied in combination with the A. parasiticus color mutant (NRRL 21369) as one treatment with the other treatment being a combination of the two naturally-occurring isolates of A. flavus and A. parasiticus (NRRL 21882 and NRRL 13539). Results from the replicated test showed that application of the naturally-occurring isolates produced significant reductions in aflatoxin in all kernel categories. Application of the color mutants produced significant reductions in the edible and total kernel categories (Table 2). Although differences between the naturally occurring and color mutant isolates were not significant in the total kernels category, it appeared that the naturally-occurring isolates might provide a slightly greater reduction. Nevertheless, because the color mutants provided the advantage of visual differentiation between the applied strains and wild types in mycofloral analyses of soil and peanuts, it was decided to continue using those isolates in further studies.
Before making a final decision on strain selection for a potential commercial biocontrol product, a 2-yr study was carried out to determine the efficacy of NRRL 21882 (A. flavus) and NRRL 21369 (A. parasiticus color mutant) applied separately and in combination (Dorner and Horn, 2007). Since the initial study using individual isolates of A. parasiticus (Dorner et al., 1992), most of the succeeding work had utilized a combination of nontoxigenic strains of A. flavus and A. parasiticus based on findings by Horn et al. (1994) that A. flavus was more aggressive in invading peanut seeds than A. parasiticus. However, a single strain biocontrol product would be preferable for registration purposes if it were as effective. Results of the study showed that application of nontoxigenic A. flavus alone was as effective as the combination and more effective than nontoxigenic A. parasiticus alone. Therefore, A. flavus NRRL 21882 was selected as the active ingredient of the biocontrol product.
Inoculum rate
A 2-yr study was conducted to determine the effect of inoculum rate on preharvest aflatoxin contamination (Dorner et al., 1998). Nontoxigenic color mutants of A. flavus (NRRL 21368) and A. parasiticus (NRRL 21369) were used as biocontrol agents and inocula were prepared by culturing the fungi on long-grain rice. An equal mixture of the two inocula were applied to replicated plots at rates of 22.4, 112, and 560 kg/ha in each of the 2 yr. Plots that were inoculated in year 1 were re-inoculated with the same rate in year 2. Regression analysis of the aflatoxin data showed a significant trend toward lower aflatoxin concentrations with increasing rates of inoculum in each year. In the 2nd yr of the study, mean aflatoxin concentrations (compared with controls) were reduced by 74.3, 95.0, and 99.9% for the 22.4, 112, and 560 kg/ha rates, respectively. The virtual elimination of aflatoxin after 2 yr of application at the highest rate demonstrated the potential of the technology if overwhelming numbers of nontoxigenic fungal propagules were established in the soil. However, it would not be affordable to use such a high rate commercially; therefore, most of the additional studies were carried out at the more affordable 22.4 kg/ha rate.
Application timing
Specific studies were not conducted to address the issue of the optimum time to apply the nontoxigenic strain to the field. However, the variety of studies that were conducted over a period of several years made it apparent that the middle of the growing season was the best time to apply inoculum. First, the importance of having the nontoxigenic strain established and in a competitively advantageous position when peanuts are subject to A. flavus invasion was recognized. Therefore, inoculum should not be applied late in the growing season because developing peanuts may already be infected by wild-type, toxigenic strains of A. flavus. In studies where inoculum was applied early in the growing season, poor growth and sporulation often occurred. In these cases, exposure of the granules to direct sunlight during the day tended to dry them out. The fungus grows and sporulates best on the granules when the water activity is high, and high water activity cannot be maintained in direct sun. In other studies in which inoculum was applied after the plant canopy was well established, we observed much better growth and sporulation by the nontoxigenic strains. Therefore, it is recommended that inoculum be applied to the field at between 60 and 80d after planting when the foliage canopy is well developed and at a time when good soil moisture is available. This can be soon after a rain or shortly before (if a high probability of rain exists). Even in the absence of rain, good growth of the fungus can take place in the warm, humid environment under the plant canopy if there is good protection from direct sunlight.
Effect on storage contamination
Having demonstrated that competitive exclusion could reduce preharvest aflatoxin contamination, we conducted studies to determine the potential for reductions in contamination that occur during long-term peanut storage (Dorner and Cole, 2002). Experiments were carried out over 2 yr in which nontoxigenic color mutants of A. flavus and A. parasiticus (NRRL 21368 and 21369) were applied to field plots and also to harvested peanut pods before they were placed in a 1/10 scale miniature warehouse (Smith et al., 1989). Treated and control peanuts from the field were each divided in half after harvest, and each half was then sprayed with an aqueous conidial suspension of the nontoxigenic strains. Therefore, storage treatments consisted of peanuts that were never treated (control-control), peanuts that were not field-treated but were treated prior to storage (control-treated), peanuts that were treated in the field but not treated prior to storage (treated-control), and peanuts that were treated at both times (treated-treated). The warehouse contained wire-mesh partitions to separate the four groups while allowing exposure to the same storage conditions. In the 1st yr of the study (1998), there was no preharvest aflatoxin contamination in either field-treated or control peanuts (Table 3). After a 3-m storage period, the control-control peanuts contained significantly more aflatoxin than the other groups, averaging 78.0 µg/kg. The control-treated group contained an average of 48.8 µg/kg, which was significantly higher than both the treated-control and treated-treated groups. These results were confirmed in 1999 when late-season drought conditions resulted in significant preharvest contamination (Table 3). Again, the spray treatment prior to storage produced a significant reduction in contamination that occurred during storage, but the reduction was not nearly as great as in the field-treated peanuts regardless of the pre-storage treatment. Although spraying the pods prior to storage did reduce storage contamination in peanuts that had not been treated in the field, it did not add to the reduction brought about through field treatment. This is explained by results of dilution plating of homogenized peanut-water slurries, which showed that the fungi that proliferated in storage were predominately those that invaded peanuts in the field. Even in 1998 when there was no preharvest contamination, the control peanuts had been invaded predominately by toxigenic strains, which then proliferated during storage. The opposite was true of the treated peanuts, resulting in reduced aflatoxin after storage. Therefore, the slight benefit from spraying nontoxigenic strains on pods before storage is not warranted, especially compared with the dual benefit of field treatment.
Delivery method
A major question during the development of the biocontrol strategy dealt with how to deliver the competitive, nontoxigenic strain to peanuts by the most economical and effective method. In the early studies, designed simply to show whether the strategy had potential for reducing aflatoxin, the fungi were grown in liquid culture, homogenized, and the homogenate was sprinkled over plants or applied as a soil drench at planting time (Dorner et al., 1992). Whereas this proved effective for aflatoxin reduction, it was obvious that it would not be practical for a commercially-produced formulation. In several subsequent studies, fungal-colonized rice was used to inoculate soil. The advantage of this formulation technique over liquid fermentation was that adequate amounts of inoculum could be made ahead of the time it would be needed. The rice not only served as a carrier, but it was also a substrate for continued production of conidia in the field until the rice grains were totally consumed. Although there were advantages over liquid fermentation, it was evident that this method also had drawbacks that were problematic. Commercial facilities did not exist in the USA for large-scale solid state fermentation of Aspergillus. In addition, it was not deemed economical to produce a biopesticide in this manner because a) the substrate had to be sterilized prior to inoculation, b) the time required for the fermentation, and c) the fermented rice had to be dried at relatively low temperatures that would not kill the fungus. All of these steps introduced excessive costs (both time and money) that would make the biopesticide very expensive to produce commercially. Therefore, two other formulation options were evaluated along with fermented rice in a 2-yr study. First was pesta, which is the product of an adaptation of the extrusion process for making pasta (Connick et al., 1991; Daigle et al., 1997). Conidia of A. flavus (NRRL 21368) and A. parasiticus (NRRL 21369) were extruded with semolina (wheat gluten) and kaolin, cut into small granules, and dried at 50C for 1h. Second, conidia of each strain were encapsulated in pregelatinized corn-flour granules (Dorner et al., 2003), a process that has been used with other microbial biocontrol agents and other matrices (Dunkle and Shasha, 1988; McGuire and Shasha, 1992). These two formulations were tested along with colonized rice in a 2-yr study which showed that all were effective in establishing the nontoxigenic strains and all produced significant aflatoxin reductions in year two (Dorner et al., 2003). Although the pesta and corn-flour granule formulations were effective, economic analysis by potential commercial partners showed that those options were still not commercially viable. Primary problems were the cost of raw materials and time-consuming low-temperature drying.
Therefore, other options were considered, and the key to developing an economical, commercially viable formulation was the availability of pure conidia of the desired nontoxigenic strain from commercial fermentation companies in Japan. These companies produce large quantities of Aspergillus conidia to be used in the fermentation industry to produce a variety of products such as soy sauce, miso, sake, and others. The conidia most often produced are of A. oryzae (Ahlb.) Cohn and A. sojae Sakaguchi & Yamada, which are closely related to A. flavus and A. parasiticus, respectively. Therefore, the availability of an essentially unlimited supply of conidia at very low cost solved one problem. The remaining questions involved choice of substrate and how best to join substrate and conidia. Several possible substrates were investigated including many small grains. Readily available hulled barley was chosen because the hull, which is a barrier to the fungus, is removed in the pearling process and it is relatively inexpensive compared with other equally suitable substrates, such as polished rice. To join substrate and conidia, conidia were suspended in vegetable oil and sprayed onto the barley surface. Diatomaceous earth was then added to absorb the oil leaving the conidia bound to the surface (Cole and Dorner, 2001). A final concentration of 5 × 105 conidia/g of barley ensured rapid growth and sporulation covering the surface of the grain after uptake of moisture. This formulation method offers several advantages over others that were investigated. First, the barley does not need to be sterilized because the pearling process, which removes the hull, also destroys the germ. Also, because of the conidial load on the seed, competing organisms that may be present are overwhelmed by the A. flavus resulting in an abundance of A. flavus conidia produced after field application. Second, there is a tremendous savings in time because the fermentation step is eliminated. Third, the formulated product does not have to be dried because no water is introduced in the process. This not only eliminated drying costs but it also saved time that is required for relatively low-temperature drying. Finally, seed coating equipment is flow-through and greatly increases production capacity over fermentation, which is limited to whatever batch size the fermenter can accommodate. Typical seed coating equipment can produce several tons of product per hour. This formulation technique greatly economized the process of producing biocontrol inoculum and provided the impetus for a private company to license the technology and produce a commercial biocontrol product, which is sold under the trade name afla-guard®.
The conidia-coated hulled barley formulation was compared with colonized rice and conidia-coated rice in a 3-yr field study (Dorner, 2004). Actually, coated wheat was used in the 1st yr before switching that treatment to coated hulled barley in the 2nd and 3rd yrs. The coated wheat was not as effective in establishing the nontoxigenic strains as the coated and colonized rice, apparently because the wheat hull prevented the abundant growth and sporulation by the fungi that was achieved with the other two formulations. Because this was a field study, natural, late-season drought conditions were necessary to produce significant aflatoxin contamination of peanuts. This occurred only in the 2nd yr of the study when all three formulations produced significant reductions in aflatoxin that averaged 81%. In the 3rd yr, when drought conditions were not severe enough to produce aflatoxin contamination, analysis of peanuts for A. flavus colonization showed that all three formulations produced a significant reduction in the incidence of toxigenic A. flavus in peanuts. Also, the treatments did not result in an increase in total A. flavus colonization of peanuts. Because the coated barley formulation was more economical to produce and as effective as the colonized rice formulation both in establishing the nontoxigenic strain in soil and reducing aflatoxin, it was chosen for commercial production.
Safety
The U. S. Environmental Protection Agency (EPA) considers a nontoxigenic strain of A. flavus being used for biological control of aflatoxin contamination a biopesticide. As such, several tests of potential toxicity and pathogenicity were required to ensure the safety of both the active ingredient (conidia of nontoxigenic A. flavus) and the formulated product in the environment. Contracts with independent laboratories were established to conduct the following studies: (a) acute mammalian (rat) oral toxicity with the formulated product; (b) field testing of the formulated product to determine hazards to pollinators (honey bees); (c) acute injection (intraperitoneal) of the active ingredient in rats to determine toxicity/pathogenicity when the skin is bypassed as a barrier; (d) acute mammalian (rat) pulmonary toxicity/pathogenicity of the active ingredient; (e) acute avian (bobwhite quail) pulmonary toxicity/pathogenicity of the active ingredient. In all of these studies, no toxicity, pathogenicity, or adverse effects were found to be associated with either the active ingredient or the formulated product.
Based on the demonstration of the safety of the biopesticide in conjunction with efficacy data submitted to the EPA, a conditional registration was issued in May, 2004 (http://www.epa.gov/oppbppd1/biopesticides/ingredients/tech_docs/brad_006500.pdf), for use of afla-guard® in peanuts. However, a condition of the registration was to provide the EPA with data demonstrating the efficacy of commercially-produced afla-guard® in large-scale field trials.
Large-scale efficacy demonstration
The large-scale field trials were conducted in crop year 2004 immediately after conditional registration was issued. Afla-guard® was applied at a rate of 22.5 kg/ha to approximately 2000 ha in commercial peanut fields at several locations. Samples of farmers' stock peanuts were collected from both treated and control (untreated) fields at seven peanut buying points in an area stretching from southeastern Alabama to middle Georgia. Samples were analyzed for aflatoxin, colonization by A. flavus, and the incidence of toxigenic A. flavus isolates in peanuts. In addition, at two locations the treated and control peanuts were placed in commercial storage warehouses, shelled, sized, and the shelled product was also analyzed for aflatoxin.
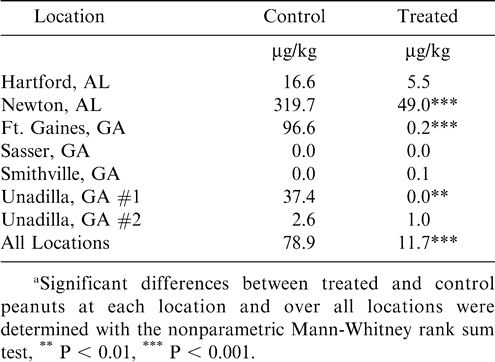
Results of aflatoxin analyses of farmers' stock peanuts at each location are shown in Table 4. In areas where there was little to no late-season drought, including Hartford, AL; Sasser, GA; Smithville, GA; and Unadilla, GA #2, aflatoxin concentrations were very low in both control and treated peanuts. However, in the other three locations that experienced mild to severe drought, aflatoxin was significantly reduced in treated peanuts. Over all locations, the mean aflatoxin concentration was reduced by 85% in treated peanuts.
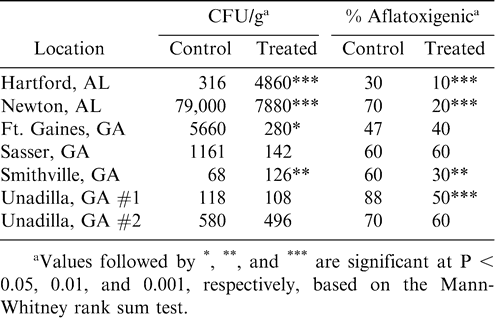
The densities of A. flavus in farmers' stock peanuts (Table 5) were extremely variable, and data indicated that weather conditions had a much greater effect on colonization of peanuts by A. flavus than did treatment with the nontoxigenic strain. The density of A. flavus was highest in control peanuts from Newton, AL, which suffered from the most severe late-season drought conditions among all locations. At Newton and Ft. Gaines, GA, the density of A. flavus was higher in control peanuts than in treated peanuts. However, at Hartford, AL, and Smithville, GA, the density was higher in treated peanuts. In general, the data corroborate results of other studies showing that application of the nontoxigenic strain does not increase the quantity of A. flavus in peanuts. The incidence of aflatoxigenic A. flavus in peanuts was significantly reduced in treated peanuts at four of the locations. This was reflected in significantly reduced aflatoxin concentrations (Table 4) in Newton, AL, and Unadilla, GA #1. Although the aflatoxigenic incidence was not significantly reduced at Ft. Gaines, GA, the significant reduction in A. flavus density probably resulted in the significant aflatoxin reduction.
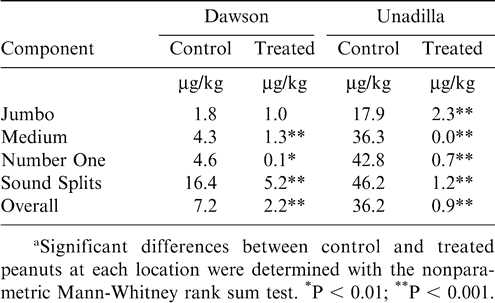
Treated and control peanuts from the Ft. Gaines and Unadilla #2 location were transferred to Dawson, GA, where they were stored in separate bins within the same warehouse. Treated and control peanuts from the Unadilla #1 location were also stored separately in another warehouse. The purpose was to carry these peanuts through a typical storage season and determine the aflatoxin concentrations in subsequently shelled peanuts. Those results showed that highly significant reductions in shelled lots were achieved in treated peanuts at both locations (Table 6). Aflatoxin concentrations were generally lower in peanuts stored in Dawson, but the biocontrol treatment still produced significant reductions in all but the Jumbo category, which also had a relatively low level in the controls. The reduction in all categories at the Unadilla location were highly significant, averaging 98% overall. Official aflatoxin analyses are conducted on all shelled lots of peanuts in the USA destined for sale, and they must contain ≤ 15 µg/kg of total aflatoxins in order to be sold. At the Unadilla location, 48% of shelled lots from control fields tested at > 15 µg/kg, which meant they had to be remilled, blanched, or sold at reduced oil stock prices. This greatly reduced the value of those peanuts. At the Dawson location, 16% of shelled lots from control fields tested at > 15 µg/kg. At both locations, no lots of shelled peanuts from treated fields tested at > 15 µg/kg. An economic analysis of the costs associated with blanching the shelled lots from untreated fields that tested at > 15 µg/kg showed that untreated peanuts at Unadilla and Dawson had reductions in net shelled stock value of 13% and 4%, respectively (Dorner and Lamb, 2006). This converts to increases in net shelled stock value of 16 and 6% for the treated peanuts at Unadilla and Dawson, respectively.
Conclusions
Research conducted over the last 20 yrs has resulted in the development and commercialization of biological control technology to manage aflatoxin contamination in peanuts. Biological control is based on competitive exclusion whereby a large population of a nontoxigenic strain of A. flavus is established in soil before peanuts are exposed to late-season drought conditions under which aflatoxin contamination occurs. The established nontoxigenic strain out-competes toxigenic strains in soil for infection sites resulting in reduced concentrations of aflatoxin in peanuts. Studies were conducted to: establish that this concept of biological control was effective, identify the best nontoxigenic strain to use, determine the application rate of inoculum needed, determine efficacy in reducing storage contamination, develop a cost-effective delivery method, and establish the safety of the biocontrol product. After receiving EPA registration of the commercially-produced biopesticide, afla-guard®, efficacy was demonstrated in a large-scale field trial in commercial peanut fields. Although this biocontrol technology does not completely eliminate aflatoxin in peanuts, it does provide the peanut industry with a cost-effective aflatoxin management tool.
Literature Cited
Blankenship P. D. , Cole R. J. , Sanders T. H. , and Hill R. A. 1983 Environmental control plot facility with manipulable soil temperature. Oleagineux 38 : 615 – 620 .
Cole R. J. and Dorner J. W. 2001 Biological control formulations containing spores of nontoxigenic strains of fungi for toxin control of food crops. U.S. Patent 6, 306, 386 .
Cole R. J. , Hill R. A. , Blankenship P. D. , and Sanders T. H. 1986 Color mutants of Aspergillus flavus and Aspergillus parasiticus in a study of preharvest invasion of peanuts. Appl. Environ. Microbiol 52 : 1128 – 1131 .
Connick W. J. , Boyette C. D. , and McAlpine J. R. 1991 Formulation of mycoherbicides using a pasta-like process. Biol. Control 1 : 281 – 287 .
Daigle D. J. , Connick W. J. , Boyette C. D. , Lovisa M. P. , Williams K. S. , and Watson M. 1997 Twin-screw extrusion of ‘Pesta’-encapsulated biocontrol agents. World J. Microbiol. Biotechnol 13 : 671 – 676 .
Diener U. L. , Cole R. J. , Sanders T. H. , Payne G. A. , Lee L. S. , and Klich M. A. 1987 Epidemiology of aflatoxin formation by Aspergillus flavus. Annu. Rev. Phytopathol 25 : 249 – 270 .
Dorner J. W. 2004 Combined effects of biological control formulations, cultivars, and fungicides on preharvest colonization and aflatoxin contamination of peanuts by Aspergillus species. Peanut Sci 31 : 79 – 86 .
Dorner J. W. and Cole R. J. 2002 Effect of application of nontoxigenic strains of Aspergillus flavus and A. parasiticus on subsequent aflatoxin contamination of peanuts in storage. J. Stored Prod. Res 38 : 329 – 339 .
Dorner J. W. , Cole R. J. , and Blankenship P. D. 1992 Use of a biocompetitive agent to control preharvest aflatoxin in drought stressed peanuts. J. Food Prot 55 : 888 – 892 .
Dorner J. W. , Cole R. J. , and Blankenship P. D. 1998 Effect of inoculum rate of biological control agents on preharvest aflatoxin contamination of peanuts. Biol. Control 12 : 171 – 176 .
Dorner J. W. , Cole R. J. , Connick W. J. , Daigle D. J. , McGuire M. R. , and Shasha B. S. 2003 Evaluation of biological control formulations to reduce aflatoxin contamination in peanuts. Biol. Control 26 : 318 – 324 .
Dorner J. W. and Horn B. W. 2007 Separate and combined applications of nontoxigenic Aspergillus flavus and A. parasiticus for biocontrol of aflatoxin in peanuts. Mycopathologia 163 : 215 – 223 .
Dorner J. W. and Lamb M. C. 2006 Development and commercial use of afla-guard®, an aflatoxin biocontrol agent. Mycotoxin Res 22 : 33 – 38 .
Dunkle R. L. and Shasha B. S. 1988 Starch-encapsulated Bacillus thuringiensis: A potential new method for increasing environmental stability of entomopathogens. Environmen. Entomol 17 : 120 – 126 .
Eaton D. L. and Groopman J. D. 1994 The Toxicology of Aflatoxins Academic Press San Diego, CA .
Horn B. W. , Dorner J. W. , Greene R. L. , Blankenship P. D. , and Cole R. J. 1994 Effect of Aspergillus parasiticus soil inoculum on invasion of peanut seeds. Mycopathologia 125 : 179 – 191 .
Lancaster M. C. , Jenkins G. P. , and Philp J. M. 1961 Toxicity associated with certain samples of groundnuts. Nature 192 : 1095 – 1096 .
McGuire M. R. and Shasha B. S. 1992 Adherent starch granules for encapsulation of insect control agents. J. Econ. Entomol 85 : 1425 – 1433 .
Sargeant K. , Sheridan A. , O'Kelly J. , and Carnaghan R. B. A. 1961 Toxicity associated with certain samples of groundnuts. Nature 192 : 1096 – 1097 .
Smith J. S. , Sanders T. H. , and Crippen K. L. 1989 Storability of farmers stock peanuts at two moisture levels in mechanically and naturally ventilated miniature warehouses. Peanut Sci 16 : 58 – 62 .
Notes
1 Research Microbiologist, USDA, ARS, National Peanut Research Laboratory, P. O. Box 509, 1011 Forrester Dr., SE, Dawson, GA 39842.
*Corresponding author (email: Joe.Dorner@ars.usda.gov).