Introduction
Flavor is among the most important quality attributes for commercial acceptance of roasted peanuts (Arachis hypogaea L.) as snacks or peanut butter. Relatively little research has been published on flavor and other sensory attributes of peanut cultivars under development in the USA, mainly because breeding efforts have been directed at developing cultivars with increased yield, grade and disease resistance, the traits with the most economic impact on peanut production. Traits such as peanut flavor, an important attribute for consumers, but one with little direct and measurable economic value, have received little weight in selecting lines for release to commercial producers. Isleib et al. (2001) suggested that there is greater variation in peanut flavor among cultivars and breeding lines developed and/or released during the past 10 years than there was in cultivars released earlier, in part due to the incorporation of exotic parents into breeding programs in order to introduce genes for disease resistance. Recently manufacturers of products such as snack foods or peanut butter have demanded increased emphasis on maintenance or improvement of peanut flavor in new releases.
To date, only two cultivars have been released that trace their immediate ancestry to diploid wild species of genus Arachis: COAN and NemaTAM (Simpson and Starr, 2001; Simpson et al., 2003). Because of changes in the U.S. peanut price support program, growers are demanding greater levels of host plant resistance in new cultivar releases, and use of wild species-derived resistances is likely to become more common in the future. The peanut genetics program at N.C. State Univ. has been actively developing peanut lines with multiple disease resistance using diploid Arachis species as sources of resistance. Several germplasm lines derived from crosses between A. hypogaea (2n = 4x = 40) and A. cardenasii Krap. & Greg. (2n = 2x = 20), a species with high levels of resistance to several pathogens and insect pests of peanut, have been released (Stalker and Beute, 1993; Stalker and Lynch, 2002; Stalker et al., 2002a,b). These germplasm lines are being used as parents in several U.S. peanut breeding programs. At N.C. State and Texas A&M Univ., additional populations have been derived from the diploid species A. diogoi Hoehne, A. correntina (Burk.) Krap. & Greg., A. stenosperma Greg. & Greg., and A. batizocoi Krap. & Greg. (Tallury and Stalker, unpublished data; Simpson et al., 1993). These new germplasm lines and other interspecific hybrid selections have been hybridized with commercial cultivars. Several advanced populations are being evaluated in field tests for yield, grade, and resistance to the diseases tomato spotted wilt virus (TSWV), leaf spots [caused by Cercospora arachidicola Hori and Cercosporidium personatum (Berk. & Curt.) Deighton], Cylindrocladium black rot (CBR) (caused by Cylindrocladium parasiticum Crous, Wingfield, & Alfenas), and Sclerotinia blight (caused by Sclerotinia minor Jagger). Preliminary yield and disease resistance data suggest that several lines are promising candidates for commercial cultivation in the Virginia-Carolina production area (Tallury et al., unpublished). However, no sensory evaluations have been made on these materials. The overall objective of this research was to evaluate sensory attributes of interspecific hybrid-derived breeding lines for peanut flavor to determine if transfer of disease resistance from wild species was associated with concomitant changes in flavor.
Materials and Methods
Plant Material
An array of 16 wild species-derived breeding lines (Table 1) were grown as entries in the Leafspot Advanced Test during 2005 (Sprayed) and 2006 (Sprayed and Unsprayed) at the Peanut Belt Research Station at Lewiston, NC. The lines traced to five Arachis species: A. batizocoi, A. cardenasii, A. correntina, A. diogoi, and A. stenosperma. Lines designated “HTS” were developed by H.T. Stalker; those designated “SPT” were developed by S.P. Tallury. Parentage of the wild species-derived lines was complex and included U.S. virginia-type parents Florigiant (Carver, 1969), NC 5 (Emery and Gregory, 1970), NC 6 (Campbell et al., 1977), NC 7 (Wynne et al., 1979), NC 12C (Isleib et al., 1997), Perry (Isleib et al., 2003), runner-type parents Southern Runner (Gorbet et al., 1987) and NC 3033 (Beute et al., 1976), and A. hypogaea plant introductions PI 261942, PI 270806, and PI 337396 (USDA-ARS, Nat. Genetic Resources Program. Germplasm Resources Information Network). The virginia-type cultivar NC 7 was included as a performance and flavor standard. The tests were conducted in a randomized complete block design with 2 replications. Each plot was comprised of two rows 7.3 m in length spaced 91 cm apart. Seed spacing at planting was 25 cm. Plots were planted on 11 May 2005 and 11 May 2006, dug 30 Sept. 2005 and 2 Oct. 2006, and harvested by combine on 5 Oct. 2005 and 5 Oct. 2006. Standard cultural practices, including irrigation and a full program of chemical control of leaf spots, were applied to the plots except that leaf spot fungicides were not applied to the “unsprayed” test in 2006.
Wild species-derived lines for sensory evaluation using the notation Purdy et al., (1968).
Sample Handling, Roasting and Preparation
Pods were sized on a rolling grader and shelled with a standard reciprocating grading sheller (Georgia Federal-State Inspection Service, Albany, GA). Seeds were screened over a 6.0 × 25.4 mm (15/64 × 1 in) grading screen. Samples from the 2005 sprayed test and from both 2006 tests were retained for sensory analysis. The shelled peanuts were stored at 5 C and 60% RH. Samples were roasted in March, 2006 and June, 2007 using a Blue M “Power-O-Matic 60” laboratory oven, ground into a paste, and stored in glass jars at -20 C until evaluated. The roasting, grinding, and color measurement protocols were as described by Pattee and Giesbrecht (1990). Roast color of the peanut paste was evaluated as CIELAB L*. Six samples failed to produce paste within two color units of the target value (58 CIELAB L*), so duplicate samples were processed to produce paste within the desired color range.
Sensory Evaluation
An eight-member, trained, roasted peanut profile panel at the Food Science Dept. at N.C. State Univ., Raleigh, NC, conducted a descriptive sensory analysis of all peanut-paste samples using a 14-point intensity scale. Panel orientation and reference control were as described by Pattee and Giesbrecht (1990) and Pattee et al., (1993). Sensory evaluation commenced on 20 March 2006 and 3 July 2007 and continued until all samples were evaluated. The averages of individual panelists' scores on sensory attributes were used in all analyses.
Statistical Analysis
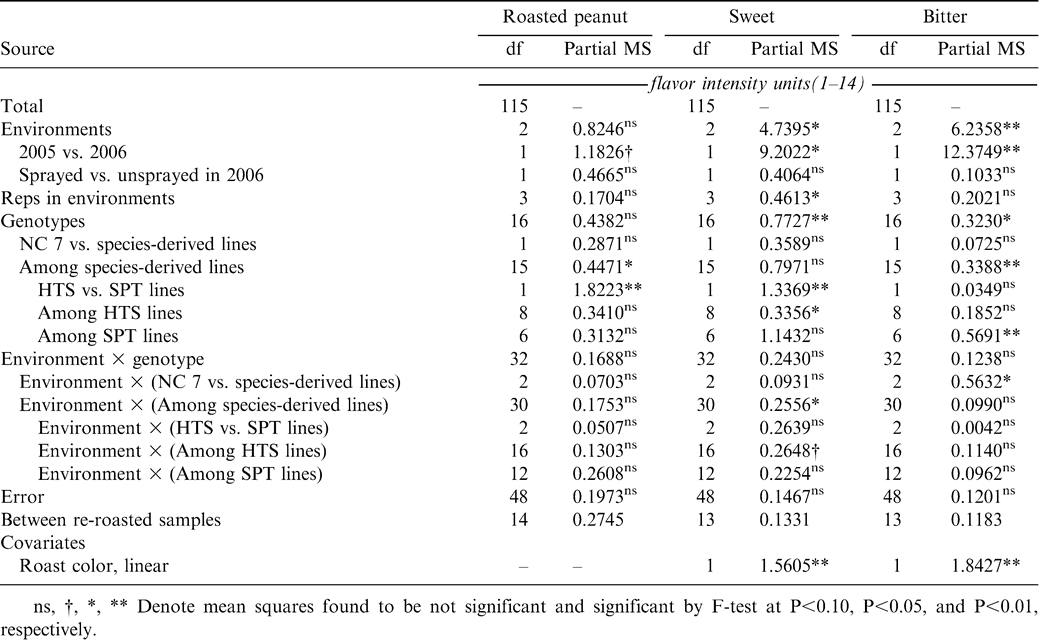
Flavor data were analyzed using a linear model for a randomized complete block design (RCBD) repeated across three environments with each test (the 2005 sprayed and the 2006 sprayed and unsprayed tests) being considered a separate environment using the general linear models procedure (PROC GLM) of SAS statistical software (SAS Institute, Cary, NC). Data from all samples (including re-roasted samples) were included in the analysis. The sum of squares for variation among environments was partitioned into two orthogonal contrasts each with a single degree of freedom, one between the 2 test years and the other between the sprayed and unsprayed tests grown in 2006. The sum of squares for variation among entries was partitioned into two parts, one comparing NC 7 to all wild species-derived lines, the other reflecting variation among wild species-derived lines. The latter sum of squares was further partitioned into a contrast between HTS and SPT lines, and two contrasts of means reflecting the variation within the two groups of wild species-derived lines. An analogous partition was made of the sum of squares for variation due to environment-by-entry interaction. Variation between initial and re-roasted samples was partitioned from the experimental error. Fruity attribute intensity and linear and quadratic effects of roast color were applied as covariates in the initial analysis. Covariates that did not significantly reduce the experimental error were dropped from the model. Means were adjusted to a common environmental effect and to the mean level for any covariates applied in ANOVA. Because the standard errors and covariances of the genotypic means were not constant, mean separation was by individual t-tests.
Results and Discussion
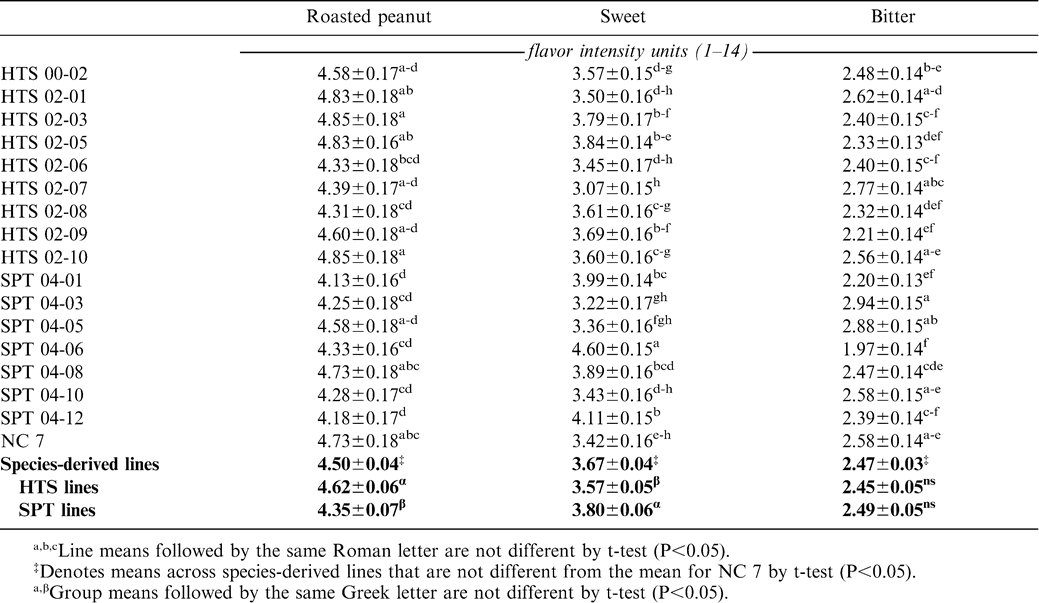
Variation among environments was primarily attributable to the difference between the 2005 and 2006 seasons (Table 2). This observation is consistent with earlier studies where year-to-year variation was shown to be a major part of the total variation in peanut sensory quality (Pattee et al., 1994, 1997, 1998, 2004). Although significant variation among entries was found for the roasted peanut, sweet, and bitter sensory attributes, in no case was the contrast between NC 7 and the mean of wild species-derived lines significant (Tables 2, 3). There was a difference between the means of the HTS and SPT lines for roasted peanut [4.62 vs. 4.35 flavor intensity units (fiu), P = 0.0038] and sweet attributes (3.57 vs. 3.80 fiu, P = 0.0041), but not for bitter (2.45 vs. 2.49 fiu, P = 0.5924). Significant variation was also observed among the HTS lines for sweet and among the SPT lines for bitter (Table 2). For roasted peanut, five of the 16 species-derived lines scored as great as or numerically greater than NC 7 for sensory intensity, although none was significantly greater (Table 3). Two lines, SPT 04-01 and SPT 04-12, had significantly lower roasted peanut intensity scores than NC 7. For sweet attribute intensity, the mean of SPT lines was significantly greater than that for NC 7 (3.80 vs. 3.42 fiu, P = 0.0264), and four individual SPT lines scored significantly greater than NC 7. One line, SPT 04-06, also scored significantly less in the bitter attribute. There was very little environment-by-genotype interaction (Table 2).
These results are positive in the sense that, except in two cases, introduction of disease and pest resistance traits from the Arachis species did not result in degradation of the flavor profile relative to NC 7, the long-time flavor standard for the virginia market-type. This has not always been the case in breeding for disease resistance because large-seeded virginia-type cultivars developed for resistance to CBR (NC 8C, NC 10C, NC 12C, and Perry) had inferior flavor profiles (Isleib et al., 2001). In the case of those cultivars, their resistance to CBR was derived from sources within A. hypogaea. As a result of these observations, it appears that flavor of wild species-derived germplasm will not prevent its use either as parents in peanut breeding programs or as cultivars. Variation observed among wild species-derived lines for sensory quality is consonant with the variation observed among lines derived exclusively from A. hypogaea ancestry. However, with these lines and as additional Arachis species are utilized for germplasm enhancement, it will be necessary to monitor flavor profiles in lines deriving resistance from them.
Literature Cited
Beute M. K. , Wynne J. C. , and Emery D. A. 1976 Registration of NC 3033 peanut germplasm (Reg. No. GP 9). Crop Sci 16 : 887 .
Campbell W. V. , Wynne J. C. , Emery D. A. , and Mozingo R. W. 1977 Registration of NC 6 peanuts (Reg. No. 20). Crop Sci 17 : 346 .
Carver W. A. 1969 Registration of Florigiant peanuts (Reg. No. 1). Crop Sci 9 : 849 – 850 .
Emery D. A. and Gregory W. C. 1970 Registration of NC5 peanuts (Reg. No. 6). Crop Sci 10 : 460 .
Gorbet D. W. , Norden A. J. , Shokes F. M. , and Knauft D. A. 1987 Registration of ‘Southern Runner’ peanut. Crop Sci 27 : 817 .
Isleib T. G. , Pattee H. E. , Gorbet D. W. , and Giesbrecht F. G. 2001 Genotypic variation in roasted peanut flavor quality across 60 years of breeding. Peanut Sci 29 : 92 – 98 .
Isleib T. G. , Rice P. W. , Bailey J. E. , Mozingo R. W. , and Pattee H. E. 1997 Registration of ‘NC 12C’ peanut. Crop Sci 37 : 1976 .
Isleib T. G. , Rice P. W. , Mozingo R. W. , Bailey J. E. , Mozingo R. W. , and Pattee H. E. 2003 Registration of ‘Perry’ peanut. Crop Sci 43 : 739 – 740 .
Pattee H. E. and Giesbrecht F. G. 1990 Roasted peanut flavor variation across germplasm sources. Peanut Sci 17 : 109 – 112 .
Pattee H. E. , Giesbrecht F. G. , and Mozingo R. W. 1993 A note on broad-sense heritability of selected sensory descriptors in virginia-type Arachis hypogaea L. Peanut Sci 20 : 24 – 26 .
Pattee H. E. , Isleib T. G. , and Giesbrecht F. G. 1994 Genotype-by-environment interaction in roasted peanut attribute. Peanut Sci 21 : 94 – 99 .
Pattee H. E. , Isleib T. G. , and Giesbrecht F. G. 1997 Genotype-by-environment interaction in the sweet and bitter attributes of peanut. Peanut Sci 24 : 117 – 123 .
Pattee H. E. , Isleib T. G. , and Giesbrecht F. G. 1998 Variation in intensity of sweet and bitter sensory attributes across peanut genotypes. Peanut Sci 25 : 63 – 69 .
Pattee H. E. , Isleib T. G. , and Gorbet D. W. 2004 Trends in sensory quality of roasted peanuts across 15 years (1986–2000). Peanut Sci 31 : 45 – 50 .
Purdy L. H. , Loegering W. Q. , Konzak C. F. , Peterson C. J. , and Allan R. E. 1968 A proposed standard method for illustrating pedigrees of small grain varieties. Crop Sci 8 : 405 – 406 .
Simpson C. E. and Starr J. L. 2001 Registration of COAN peanut. Crop Sci 41 : 918 .
Simpson C. E. , Nelson S. C. , Starr J. L. , Woodard K. E. , and Smith O. D. 1993 Registration of TxAG-6 and TxAG-7 peanut germplasm lines. Crop Sci 33 : 1418 .
Simpson C. E. , Starr J. L. , Church G. T. , Burow M. D. , and Paterson A. H. 2003 Registration of NemaTAM peanut. Crop Sci 43 : 1561 .
Stalker H. T. and Beute M. K. 1993 Registration of four leafspot-resistant peanut germplasm lines. Crop Sci 33 : 1117 .
Stalker H. T. and Lynch R. E. 2002 Registration of four insect-resistant peanut germplasm lines. Crop Sci 42 : 313 – 314 .
Stalker H. T. , Beute M. K. , Shew B. B. , and Barker K. R. 2002a Registration of two root-knot nematode-resistant peanut germplasm lines. Crop Sci 42 : 312 – 313 .
Stalker H. T. , Beute M. K. , Shew B. B. , and Isleib T. G. 2002b Registration of five leafspot-resistant peanut germplasm lines. Crop Sci 42 : 314 – 316 .
Wynne J. C. , Mozingo R. W. , and Emery D. A. 1979 Registration of NC 7 peanut (Reg. No. 22). Crop Sci 19 : 563 .
Notes
- First and second authors: Res. Agron. , USDA-ARS and Agric. Res. Statistician; Coastal Plain Exp. Sta., Tifton, GA 31793-0748. Third author: President and CEO, Hebert Green AgroEcology; Asheville, NC 28801. [^]
- Dep. of Crop Science, Box 7629, N.C. State Univ., Raleigh, NC 27695-7629 [^]
- Dep. of Biological and Agricultural Engineering, Box 7625, N.C. State Univ., Raleigh, NC 27695-7625 [^] *Corresponding author (tallury@ncsu.edu)
Author Affiliations