Introduction
Protein accessibility remains a global issue, particularly in the developing world, requiring solutions from all fields, including that of food sciences, in order to investigate and design alternative protein sources. Proteins can be integrated in a diet as animal proteins (meat, eggs, poultry, milk or fish) and/or vegetal proteins such as legumes (lupine, bean, lentils, chickpeas, soy or peanut) and cereals. An equally important need is to decrease the carbon footprint of agriculture, hopefully leading to more responsible and sustainable agro-industries (Report of the EAT-Lancet Commission, 2019). This latter goal can be aided by bringing value to local raw materials and shortening the production to consumption cycle.
Interestingly, several African countries belong to the top 10 list of countries producing groundnuts with shell in the world. Indeed, Nigeria (rank 3) produced over 2.8 million tons of peanuts in 2018 and the Food and Agriculture Organization registered a slightly lower production volume for Sudan (rank 4) for the same year. The United Republic of Tanzania (rank 7), Chad (rank 9) and Senegal (rank 10) exhibited production volumes of 940 thousand tons, 893 thousand tons and 846 thousand tons in 2018, respectively (FAO Statistical Database, 2018). Arya, Salve and Chauhan (2016) have compared peanut nutritional potential to other grains and legumes and have shown that the protein content of peanuts (Arachis hypogaea) is comparable to soybean (Glycine max) and mung beans (Vigna radiata), essentially representing 31% of its dry mass. Thus, peanut products provide a potential key to addressing some of the need for producing protein sources, particularly in the developing world.
The transformation of peanuts can be achieved by means of many methods, including toasting to make salty snacks, crushing to obtain peanut butter or crushing with water to produce peanut milk. We present here an investigation of making an analogue cheese from peanuts. It should be noted that for a cheese product to be accepted by a consuming population, it needs to have certain characteristics. The most important property is the texture, which is the focus of this study.
Yu et al. (2007) have shown that peanut protein concentrate has the potential to add value to the peanut industry and provide food processors with an affordable source of plant proteins with unique flavor and functional characteristics, such as thickening and emulsifying capacities. For example, Alcalase® (Subtilisin) is a serine protease produced by Bacillus licheniformis. It has broad specificity but is mainly used to cleave the carboxyl groups of hydrophobic amino acids (Adler-Nissen, 1986). Alcalase hydrolysis has been employed to increase solubility of the soy protein in combination with transglutaminase in acidic conditions (Walsh et al., 2003). Transglutaminase (TG) is a widely available enzyme in nature that can modify protein covalent bonds by catalyzing acyl transfer between a λ-carboxyamide of a peptide/protein bound glutamine and the lysine residue forming an ℇ-(λ-glutamyl) lysine [ℇ-(λ-Glu)Lys]cross-link (Kuraishi, Yamazaki, & Susa, 2001). This cross-linking results in modified proteins presenting an increased molecular mass. TG is used in the processing of dairy, seafood (surimi), meat, noodle, soybean (tofu, kamaboko) and bakery products to make gels or increase viscosity (Kuraishi, Yamazaki, & Susa, 2001). Studied by Renkema & Jacoba (2002), heat-induced gel formation by soybean proteins involves denaturation, aggregation (in which disulfide bridges play a role), network formation, and gel stiffening. Gel stiffening during prolonged heating is caused by rearrangements in the network structure and probably to some extent by further incorporation of proteins into the network. Gel stiffening during cooling is a thermoreversible process and does, therefore, not involve disulfide bond formation or rearrangements in the network structure. Also, according to Ramel and Marangoni (2017), animal processed cheese microstructure was found to greatly affect the rheological properties of the final product. Results of their study suggest that the embedding of milk fat globules within the protein matrix and/or presence of other ingredients increases the ratio of the 𝛃 crystalline polymorph to the '𝛃' form. In addition to that, Masotti et al. (2018) have shown that the inclusion of starch in analogue cheese formulation modifies the physico-chemical and functional properties of the final product to an extent related to starch type and starch properties, such as shape, size, swelling ability and the amylose/amylopectin ratio. Some of these processes were explored by Guo et al. (2018) who have presented a novel method of preparing peanut tofu by combining transglutaminase (TG) and high-temperature pressure cooking (HTPC, 115 C, 0.17 MPa) treatment, and its gel formation.
The following focuses on how the properties of a peanut-based analogue of cheese can be modified by shearing modulation designed via analysis and characterization of the rheological behavior.
Materials and Methods
The ingredients used to prepare the samples were tap water, peanuts (runner high oleic from Argentina, cultivar unknown, Bredabest B.V., Netherland), palm oil (Elaeis guineensis, Ets Ch Daudruy Van Cauwenberghe Et Fils, France), cassava flour (Manihot esculenta, PhilAfrica Foods Ldt, Mozambic), hydrochloric acid 37% v/v (AppliChem GmbH, USA), table salt (Salinor, Compagnie des Salins du Midi et Salines De L'Est SASU, France) and sugar (sucrose, Saint Louis Sucre SAS, France), Transglutaminase Activa® WM EC 2.3.2.13 (Ajinomoto Co., Inc., Japan), and Alcalase® 2.4 L FG EC 3.4.21.62 (Novozymes, Nasdaq Copenhagen, Denmark). The selection of high oleic peanuts has been made for increasing health benefits and shelf life (Chamberlin et al. 2011).
Samples Preparation
The process of sample preparation is published in patent number US20170150734A1 and includes crushing, mixing, shearing, enzymatic activation, sterilization, cooling and shearing. Raw peanuts were crushed in a crusher (MC15, 200 μm, Stephan Machinery GmbH, Germany), then combined with most of the ingredients: water, crushed peanuts (35% m/m), palm oil (10% m/m), cassava flour (7% m/m), salt and sugar. This was followed by the first shearing (for either 0, 1, 2, 4, 6, 8, or 10 min) that was done with a double knife at 3000 rpm in a mixer (UMSK24E, 18L, Stephan Machinery GmbH, Germany). Then enzymatic reaction was performed by adding the two enzymes while maintaining 40 C for 20 min during slow mixing. An acidification was added to reach a pH of 4.50 by adding hydrochloric acid 37% v/v. The 10 kg batch was subjected to sterilization (118 C for 4 min) in the same mixer before being cooled to 70 C. A second shearing step (for either 0, 1, 2, 4, 6, 8, or 10 min) was performed in order to obtain the final consistency. All measurements were made on the final product after the second shearing step.
Rheological Properties
Indentation measurements were performed with a TA.XT plus Texture Analyzer (Stable MicroSystems Ltd., United Kingdom). A conic probe (angle 45°) was used, the minimum force detection was 0.049 N, and the displacement of the probe in the sample was 5 mm in depth at a constant speed of 2 mm/s. The reported values are averaged from three indentation measurements at room temperature. Viscoelastic measurements of peanut gels have been recorded on a Thermo Scientific™ HAAKE™ MARS™ (ThermoFisher Scientific Inc., United States) rheometer by using parallel plates, the sample thickness was 1 mm and flow curves were obtained by scanning shear strain rates from 0 to 40 s-1 with a constant angular acceleration of 0.12 s-2. These measurements were repeated three times at room temperature and the average of the force at maximum indentation depth is reported.
Color Measurements
Clarity of the peanut gels were measured by using the CIELAB color space method with a Chromameter CR-410 (Konica Minolta Co. Ltd., Japan). The samples analyzed were spread on the glass cell. All results were taken in triplicate at room temperature.
Particle Size Distribution
A first dispersion was performed mixing 1 mL of the peanut gel to 30 mL of Lauryl-Sodium Dodecyl Sulfate (SDS, AppliChem GmbH, USA) at 4 g/L. SDS was added as a tensioactive and the sample was then mixed for 3 h under magnetic stirring. Particle size distribution measurements were performed by using a Mastersizer 3000 (Malvern Panalytical Ltd, Netherlands). Refractive indices of the dispersed and continuous phases (water) were taken at 1.449 and 1.333, respectively. All results were replicated at least 3 times.
Oil Separation Capacity (OSC)
The percentage of OSC of the peanut gels were determined by measurements performed at room temperature. Samples were incubated at 50 C for 2 h. After centrifugation (10 min, 1300 g), the OSC was calculated with the following equation:
Before centrifugation, the sample was fully melted at 50 C for 2 hours (Tfusion(palm oil) = 36-40 C ; Tfusion(peanut oil) = 3 C) (Juliano et al., 2012) which allows the mobility of the fatty molecules that are not trapped inside the cheese.
Statistical Analysis
Data analysis was performed using XLSTAT (Addinsoft, Paris, France, 2009). Principal Component Analysis (PCA) was performed on all data.
Results
The present study is for different peanut gels prepared as described above. All samples showed the same general visual appearance as in Fig. 1. The final product has the appearance of a light-colored spread (i.e., a light paste).
Impact of Shearing on the Rheological Properties of Peanut Gels
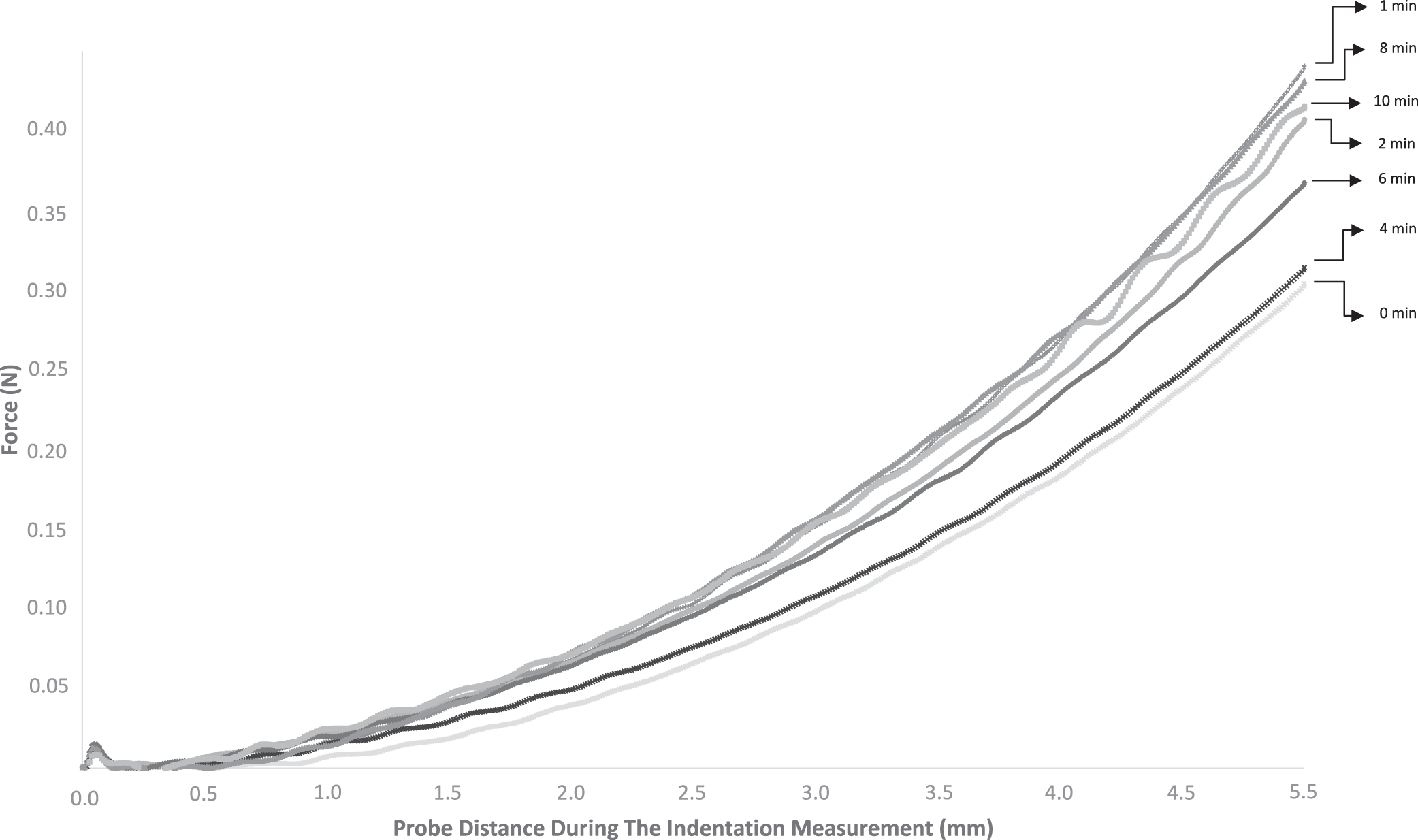
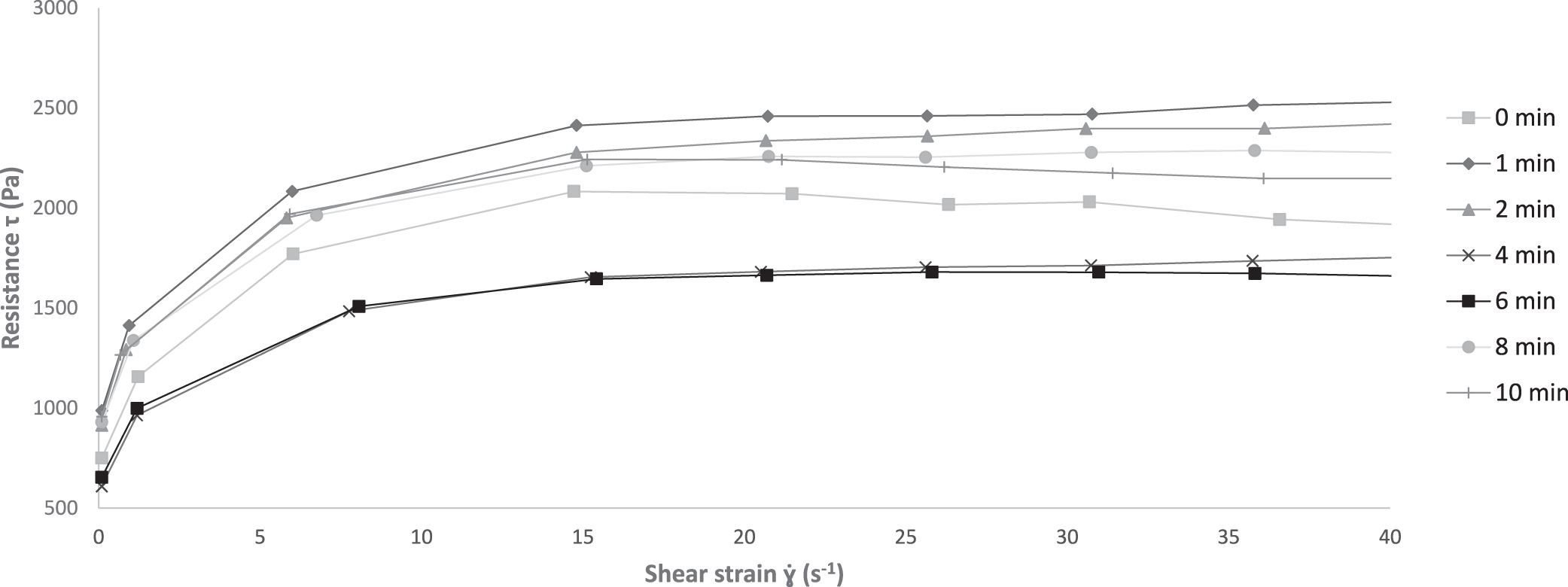
The results of the indentation are reported in Fig. 2, where the resistance (N) of the product is recorded as a function of the probe's depth (in mm). Probe displacement speed was constant and set at 2 mm/s. While the indentation probe penetrated into the sample, it was observed that the force increased non-linearly from 0 mm to the maximum value at 5 mm depth irrespective of shearing duration. For no shearing (i.e., 0 min), the measured maximum force was the lowest with a value of 0.304 N. This maximum increased to 140% of this initial value for 1 or 2 min of shearing (i.e., about 0.730 N). This maximum decreases for 4 min and 6 min of shearing with respective values of maximum resistance of 0.314 N and 0.367 N. After 8 min of shearing, the value stabilized around 0.420 N. The flow characteristics are shown in Fig. 3. These curves were constructed by plotting the shear stress (Pa) against the shear strain rate (s-1), which, respectively, correlate with the load required to sustain a given shearing rate, and the associated shearing rate of the sample. The same tendencies were observed for all the cases. The initial zero-strain-rate shear stress resisting the flow (𝛕0) first increases as the shear strain rate increases and then reaches a plateau (𝛕plateau) as the shear strain rate became large (Fig. 3). The initial resistance 𝛕0 is 880 Pa for 0, 1 and 2 min of shearing, it decreases to a minimum of 664 Pa for 4-6 min of shearing and increases to 985 Pa for 6 min of shearing. Although the values are different, the ratio 𝛕plateau/ 𝛕0 remains the same for all the shearing durations.
Impact of Shearing on the Clarity of Peanut Gels
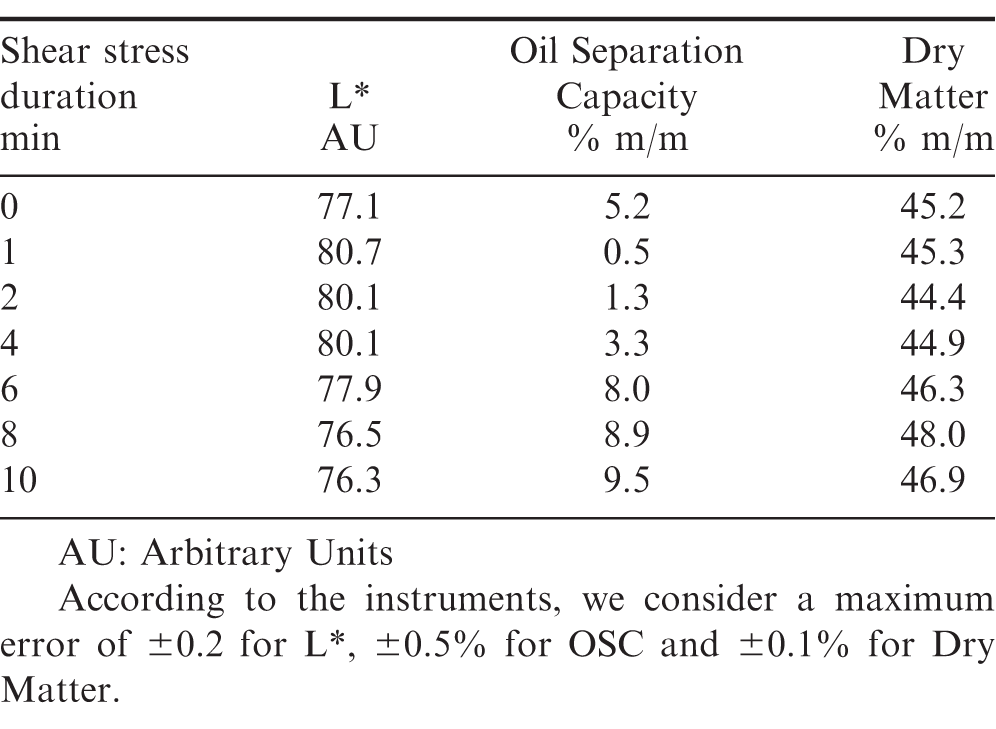
The values of the clarity (L*) obtained from color measurements are reported in Table 1. For all the samples, the values are between 76 and 81, which corresponds to off-white. The initial value of L* = 77.1 changes as soon as the shearing process is initiated, and reaches a maximum value of 80.7 for shearing durations lower than 2 min. For longer shearing durations, L* decreases suddenly, coming back to the initial value and remains at this value for all longer shearing durations. The values of the OSC and the dry matter fraction determined for each experimental condition are reported in Table 1. The values lay between 0.5% and 10.0%, which constitute a significant variation. OSC, dry matter and clarity exhibit the same behavior.
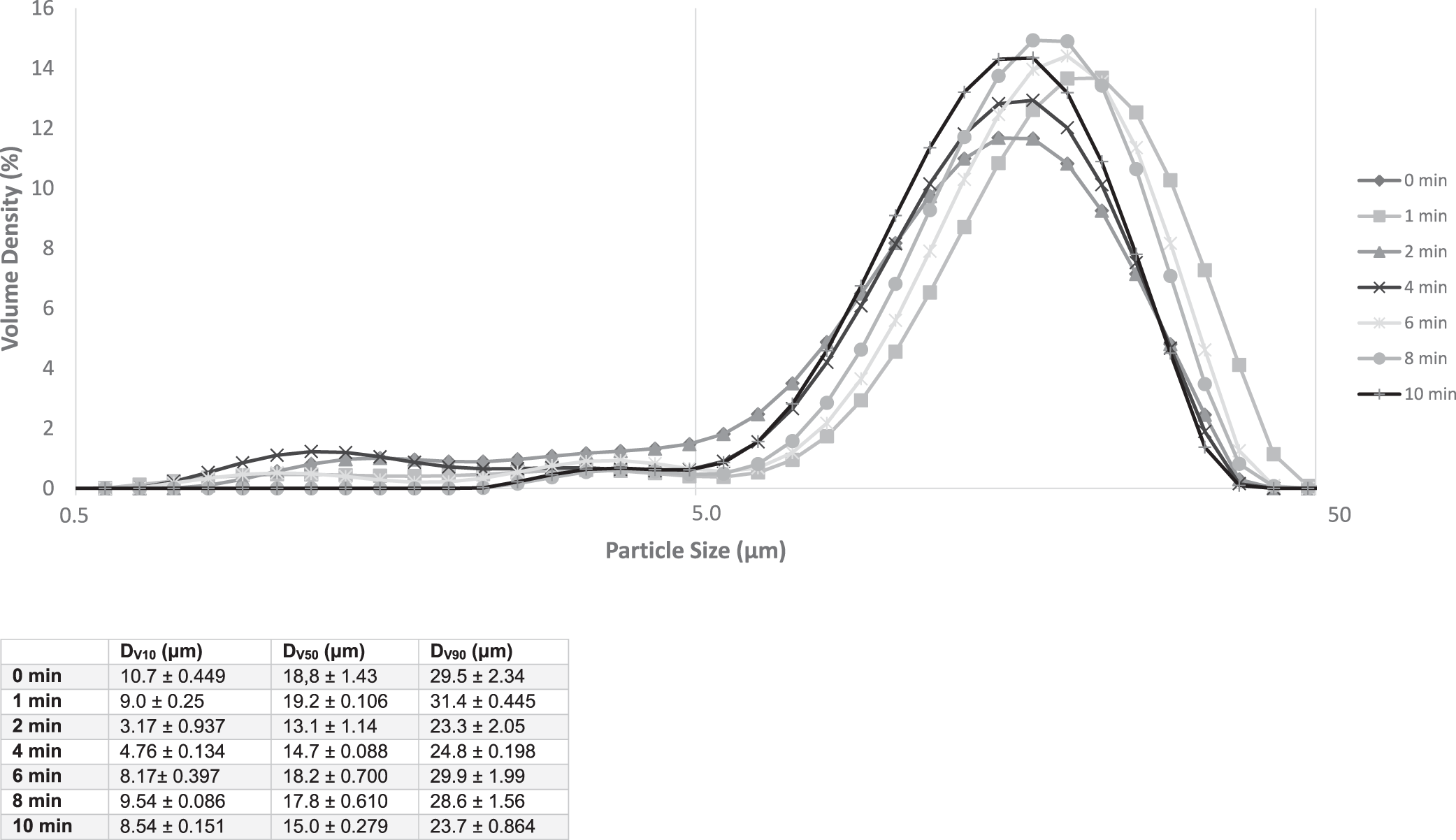
Impact of Shearing on the Particle Size Distribution
The measurement of particle distribution is given in Fig. 4. From these curves it is possible to define domains of particle size. The first domain of particle size is between 0.5 and 5 μm leading to what will be call the initiation of the measurement, which is not considered a distribution. A distribution is found for sizes between 5 to 50 μm. Considering spherical particles, because most of the particles are aggregates of lipids and proteins, the D[4;3] repartition is considered as the corrected diameter. The measurements of D[4;3] give an initial average size of 19.5 μm, which is reduced to 13.4 μm after 2 min of shearing, and then seems to come back to the initial values for higher shearing durations.
Discussion
Enzymatic gelation due to transglutaminase and subtilisin activities contributes to the cheese texture, such as does thermal treatment, by creating a protein network. In addition to this, the thermal treatment will induce a starch gelatinization that creates a second network.
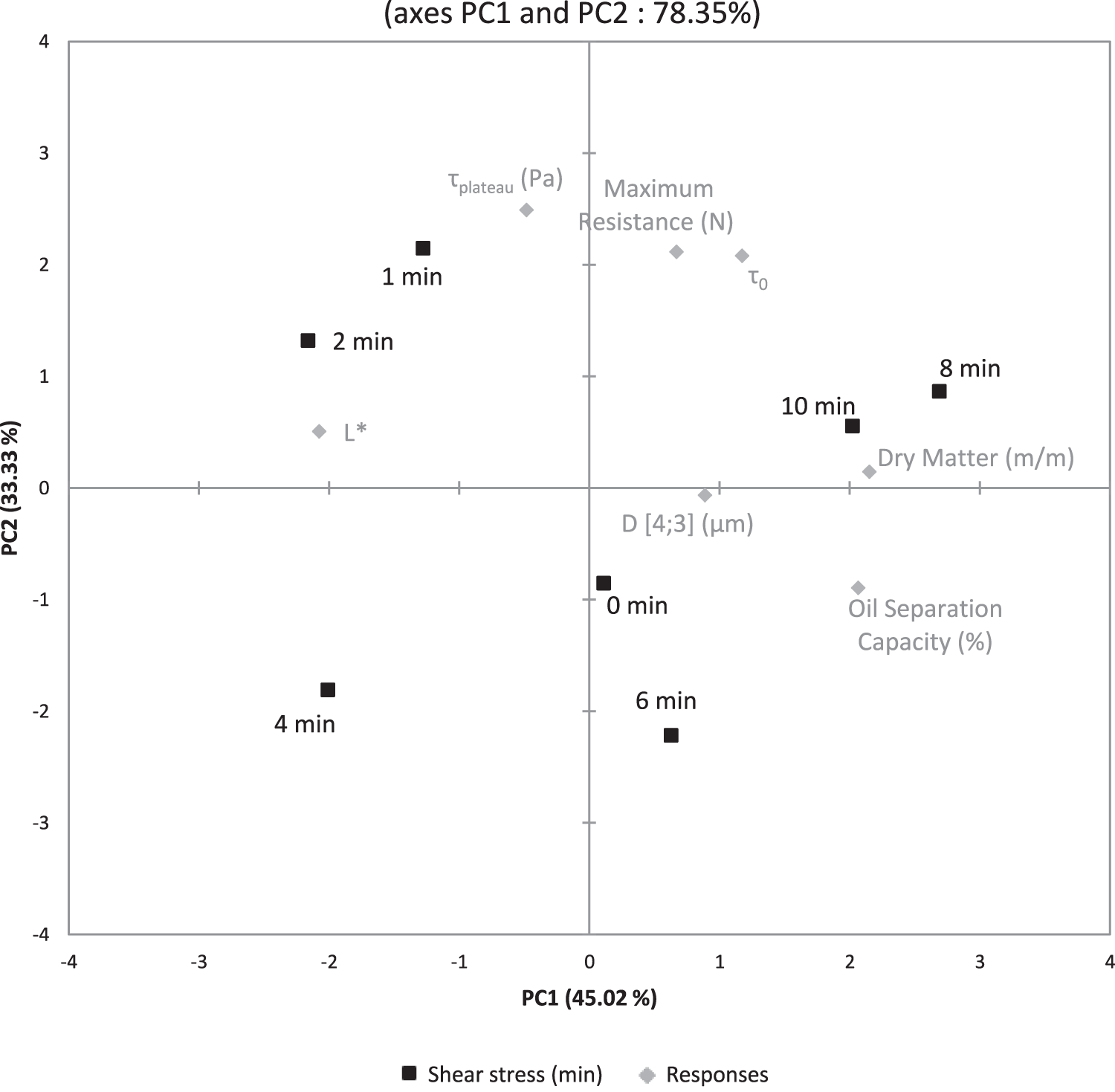
Principal Component Analysis (PCA) was applied to all data. That is, to the different peanut gels obtained by changing the time of shearing. The first two principal components (PCs), as shown in Fig. 5, accounted for 78.35% of the total variance with a predominance of the PC1 (45.02% of the variance).
According to the PC1, a clear discrimination between two groups was observed since products obtained by low shearing (1, 2 and 4min) presented negative scores, while those obtained by strong shearing (6, 8 and 10min) showed positive values.
The first grouping is seen on the left quadrant and includes clarity L*. The second grouping of terms is seen on the right quadrant and includes dry matter, OSC and average particle size. All these terms relate to the physical stability of the material, which is clarity negatively correlated to dry matter content and OSC. In this case, negative values on the left quadrant describe a hydrophobic matrix which traps oil and release water during dry matter analysis and, on the contrary, values in the right quadrant describe a hydrophilic matrix which trap water and release oil during centrifugation. Pereira et al. (2001) have shown that increasing moisture tends to produce softer cheese, which has not been evidenced here with this particular peanut matrix. Finally, the rheology and indentation responses are represented more by the PC2 axis which tends to be the firmness and spreadability of the material.
It is noted that no shearing (i.e., 0 min), which represents the initial state of the matrix, is the closest point to the center, indicating that the responses are well chosen to describe variations. For 1 and 2 min of shearing, the matrix gets firmer compared to the other shearing time. The shapes obtained for the indentations are those expected for peanut based food (Basse et al., 2018) and also similar to processed cheddar cheese (Goh et al., 2005). We also observe the same behavior for a processed cheese (Kaminarides and Stachtiaris, 2000). In the first reference, the peanut gel was obtained by cross-linking peanut protein by TG, the range of indentation force for 5 mm penetration was between 0.05 and 0.25 N. In the second reference, the cheddar gel was obtained by agglomeration of milk protein with lactic acid. The observed indentation values were between 2.0 and 3.0 N. In the latter reference, the control processed cheese is mainly made of kasseri cheese (60%) without whey protein concentrate or soybean oil, and three other cheeses containing increasing amounts of whey protein concentrate and soybean oil. Indentation values was measured between 0.36 and 1.56 N. Considering flow curves presented here, similar behavior has been reported by Carré & Eustache (2000) on a model system of polydiméthylsiloxane (PDMS). The 𝛕plateau of this system was 150 Pa and has a 𝛕0 that is not zero. When the value of the shear stress at zero shear strain rate (𝛕0) is not zero, the behavior of our materials can be classified as a pseudoplastic material with yield stress as proposed by Souza et al., (2004). Lee et al. (2004) described a pseudoplastic behavior but without yield stress for processed spreadable cow cheese. This characteristics in the peanut cheese can be explained by considering the existence of starch in our peanut cheese, which behave as a thickener inducing a threshold for the matrix flow. For 1 and 2 min of shearing, the matrix gets whiter and more hydrophobic. For the clarity L*, it is of interest to note that depending on the shearing duration, the value of the clarity changes. The same range of L* (i.e., around 80) was also observed by El-Nimr et al. (2010) during ripening of Egyptian gouda cheese. As shown by Chanamai and McClements (2001), the change of clarity L* can be explained by smaller oil droplets that create a thinner emulsion and a higher clarity for monodisperse oil-in-water emulsions. This occurs with our results of particle distribution and color evolution. For the particle distribution, Floury et al. (2000) and Kiokas et al. (2004) have observed similar distributions respectively on a model lipoproteic matrix and on whey protein-stabilized emulsions. The peanut cheese can be considered as a lipoproteic matrix emulsion with peanut protein as tensioactive. The associated curves can be fitted by a sum of two normal distributions.
At 4 min of shearing, the matrix gets less firm. After 4 min, the matrix changes and became more hydrophilic and darker and closer to what the matrix was at tshearing = 0 min. For 8 and 10 min, the firmness is increased by the shearing. It is observed that 1, 2, 8 and 10 min of shearing are firmer and less spreadable than 4 and 6 min of shearing. This concurs with the results of Chen & Tao (2005), which have shown that oil-water model emulsions have a maximum of stability of stirring time (15 min) due to the drop-out of the emulsifier from the oil-water interface.
Summary and Conclusions
This product was similar to a processed cheese and can be flavored with various spices and enhanced with aromas. This vegetal analogue cheese was produced using an optimization process, considering multiple response characteristics. The rheological behavior obtained is like a milk-based cheese. To get the rheological impact of the shearing step on the peanut-based analogue cheese, it was shown that a shearing time larger than 4 min tends to destabilize the matrix by decreasing its firmness. It probably destroys the protein and starch networks. To get a maximum firmness and physical stability, the optimal shearing time is 1 min. It should be noted that organoleptic changes must also be studied to confirm the impact of shearing, particularly on the product taste. Further characterization is also needed to study the water/oil holding capacity resulting from the different processing conditions.
Literature Cited
J., Adler-Nissen, (1986). Enzymic hydrolysis of food proteins. Elsevier Applied Science Publishers; Sole distributor in the USA and Canada: Elsevier Science Pub. Co, London; New York: New York, NY, USA..
S.S., Arya, A.R., Salve, S., Chauhan, (2016). Peanuts as functional food: a review. Journal of Food Sci and Tech 53, 31- 41.
Basse, B., d'Huart, J.-B., Lorand, J.-P., Dupas, J.-P., Saiter, J.-M., 2018 Peanut Suspension Crosslinking by Means of Microbial Transglutaminase Studied with the Indentation Technique Macromolecular Symposia 382, 1800067.
A., Carré, F., Eustache, (2000). Spreading Kinetics of Shear-Thinning Fluids in Wetting and Dewetting Modes. Langmuir 16, 2936- 2941.
K. D., Chamberlin, H. A., Melouk, R., Madden, J. W., Dillwith, Y., Bannore, & Z. E Rassi, ((2011)). Determining the oleic/linoleic acid ratio in a single peanut seed: a comparison of two methods. Peanut Sci 38: 78- 84.
R., Chanamai, D.J., McClements, (2001). Prediction of emulsion color from droplet characteristics: dilute monodisperse oil-in-water emulsions. Food Hydrocolloids 15, 83- 91.
G., Chen, D., Tao, (2005). An experimental study of stability of oil-water emulsion. Fuel Processing Technology 86, 499- 508.
EAT-Lancet Commission Summary Report, 2019 Food Planet Health EAT.
A.A., El-Nimr, H.A., Eissa, M.M., El-Abd, A.A., Mehriz, H.M., Abbas, H.M., Bayoumi, (2010). Water Activity, Color Characteristics and Sensory Properties of Egyptian Gouda Cheese during Ripening 8..
J., Floury, A., Desrumaux, J., Lardières, (2000). Effect of high-pressure homogenization on droplet size distributions and rheological properties of model oil-in-water emulsions. Innovative Food Sci & Emerging Tech 1, 127- 134.
Food and Agriculture Organization of the United Nations. FAOSTAT Statistical Database. [Rome] :FAO, 2018.
S.M., Goh, M.N., Charalambides, J.G., Williams, (2005). Characterization of the nonlinear viscoelastic constitutive properties of mild cheddar cheese from indentation tests. Journal of Texture Studies 36, 459- 477.
Y., Guo, H., Hu, Q., Wang, H., Liu, (2018). A novel process for peanut tofu gel: Its texture, microstructure and protein behavioral changes affected by processing conditions. LWT 96, 140- 146.
P., Juliano, P., Swiergon, R., Mawson, K., Knoerzer, M.A., Augustin, (2013). Application of Ultrasound for Oil Separation and Recovery of Palm Oil. Journal of the American Oil Chemists' Society 90, 579- 588.
S., Kaminarides, & S Stachtiaris, ((2000)). Production of processed cheese using kasseri cheese and processed cheese analogues incorporating whey protein concentrate and soybean oil. International Journal of Dairy Technology , 53 ((2)), 69- 74.
S., Kiokias, A.A., Reszka, A., Bot, (2004). The use of static light scattering and pulsed-field gradient NMR to measure droplet sizes in heat-treated acidified protein-stabilised oil-in-water emulsion gels. International Dairy Journal 14, 287- 295.
C., Kuraishi, K., Yamazaki, Y., Susa, (2001). Transglutaminase: its utilization in the food industry. Food Reviews International 17, 221- 246.
S.K., Lee, S., Anema, H., Klostermeyer, (2004). The influence of moisture content on the rheological properties of processed cheese spreads. International Journal of Food Science and Technology 39, 763- 771.
Lorand, J.P., Kandil, L., D'Huart, J.B., 2017 Food comprising proteins mainly of plant origin and preparation method thereof US20170150734A1.
F., Masotti, S., Cattaneo, M., Stuknytė, I., De Noni, (2018). Status and developments in analogue cheese formulations and functionalities. Trends in Food Science & Technology 74, 158- 169.
R.B., Pereira, R.J., Bennett, Y., Hemar, O.H., Campanella, (2001). Rheological and microstructural characteristics of model processed cheese analogues. journal of texture studies 32, 349- 373.
P.R., Ramel, A.G., Marangoni, (2017). Characterization of the polymorphism of milk fat within processed cheese products. Food Structure 12, 15- 25.
J.M.S., Renkema, T., van Vliet, (2002). Heat-Induced Gel Formation by Soy Proteins at Neutral pH. Journal of Agricultural and Food Chemistry 50, 1569- 1573.
P.R., Souza Mendes, E.S.S., Dutra, (2004). Viscosity Function for Yield-Stress Liquids. Applied Rheology 14, 296- 302.
D.J., Walsh, D., Cleary, E., McCarthy, S., Murphy, R.J., FitzGerald, (2003). Modification of the nitrogen solubility properties of soy protein isolate following proteolysis and transglutaminase cross-linking. Food Research International 36, 677- 683.
J., Yu, M., Ahmedna, I., Goktepe, (2007). Peanut protein concentrate: Production and functional properties as affected by processing. Food Chemistry 103, 121- 129.
Notes
- First, second, third, fourth and seventh authors: Engineer, Doctor, Engineer, Technician and Doctor, Tweed Research Center, Onyx Développement SAS, Nutriset Group, 640 Chemin du Chateau, 76770 Malaunay, France; Fifth author: Professor, Mechanical & Materials Engineering, University of Nebraska-Lincoln, Lincoln, NE 68588, USA; Sixth author: Professor Emeritus, Laboratoire SMS, Faculté des Sciences, Université de Rouen, Normandie, France. [^] *Corresponding author's E-mail: heudier@onyx-dev.fr
Author Affiliations