Introduction
Jimsonweed possesses many characteristics that make it a competitive summer annual broadleaf weed. It is capable of very high growth rates, prodigious seed production, and rapid seed germination (Scott et al., 2000). Jimsonweed is native in the continental U.S. States except Wyoming (Anonymous, 2006). While jimsonweed is not typically considered to be a problematic peanut weed in all of the peanut-producing states, it can be a problematic weed in the mid-Atlantic peanut production region including North Carolina and Virginia (Oliver et al., 1991). Jimsonweed can reach heights of 67 cm (Scott et al., 2000) and the resulting canopy can intercept light and reduce peanut productivity. According to the 2005 North Carolina Peanut Production manual, jimsonweed has a rank of 5.8 on a competitive index, based on a 10-point scale, with 10 being most competitive (Jordan, 2006).
Jimsonweed interference has been evaluated in cotton (Gossypium hirsutum L.) (Scott et al., 2000) and soybean (Glycine max L.) (Frazee and Stoller, 1974; Hagood et al., 1981; Oliver et al., 1991; Paterson and Flint, 1983; Stoller and Woolley, 1985), and both crops suffered yield loss as a result of jimsonweed infestation. Jimsonweed height was not affected by either cotton or soybean; however crop height was decreased as weed density increased in both cases (Scott et al., 2000; Hagood et al., 1981). Scott et al., (2000) reported decreased seed pods per jimsonweed plant with increasing density; however, the number of seed pods per acre was increased.
Currently registered postemergence herbicides allow growers to successfully manage jimsonweed in peanut; however, questions exist concerning treatment threshold levels. Additionally, weed seed production has been cited as a concern of growers and other agricultural personnel (Czapar et al., 1997). By comparison, common ragweed (Ambrosia artemisiifolia L.) is considered a problematic weed whose interference in peanut had negative impacts including increased disease incidence, peanut size and yield reductions (Clewis et al., 2001). Common ragweed is ranked at 3.8 on a competitive index, 2 points less than jimsonweed (Jordan, 2006).
Not only do weeds cause yield loss in peanut but also peanut inversion can be affected, thus causing reduced harvesting efficiency (Clewis et al., 2001). Jimsonweed has been noted as a problematic weed in North Carolina and Virginia and the effects of interference in peanut have not been evaluated. Therefore, objectives of this study were to determine yield and growth reductions caused by jimsonweed interference in peanut.
Materials and Methods
Experiments were conducted in 2004 at the Upper Coastal Plain Research Station located near Rocky Mount, NC and the Cherry Research Farm near Goldsboro, NC. The soil types were Norfolk loamy sand (fine-loamy siliceous thermic Typic Paleudults) with 2.1% organic matter and pH 6.1 and Wickham loamy sand (fine-loamy, mixed, thermic Typic Hapludults) with 2.1% organic matter and pH 5.8, respectively. The soil was disked and seedbeds were conventionally prepared (Jordan, 2006). The peanut cultivars ‘Perry’ and ‘VA-98R’ were planted 5.0 cm deep at 134 kg/ha on May 10, 2004 in Goldsboro and May 12, 2004 in Rocky Mount.
The experimental design was a randomized complete block with 3 replications. Individual plots consisted of four rows spaced 91 cm apart that were 6.1 m long. Fertilization, insect, and disease management practices were standard for peanut production in North Carolina (Brandenburg, 2005; Jordan, 2005; Shew, 2006).
Greenhouse-grown jimsonweed seedlings at the cotyledon to 2-leaf stage were planted into plots immediately after peanut planting at the following densities: 0, 1, 2, 4, 8, 16, or 32 plants per 6.1 m of row or 0, 0.16, 0.33, 0.66, 1.31, 2.62, or 5.25 plants per m of row. Jimsonweed seedlings were planted into the center two rows of each plot with the two outer rows left as weed-free borders. The transplanting method allowed the establishment of plants with uniform size and distribution along the row. Although jimsonweed seedlings were transplanted at peanut planting, an acclimation period of 1 to 2 wk was required before the jimsonweed plants resumed growth (data not shown). Peanut emerged within 1 wk of planting and had nearly equaled jimsonweed height at the first measuring interval 2 wk after planting. Herbicide applications of clethodim at 0.28 g ai/ha over the top and a hooded application of acifluorfen at 0.14 kg ai/ha plus bentazon at 0.28 g ai/ha (to keep the herbicide treatments off of the jimsonweed) were made six weeks after planting to control weeds other than jimsonweed. Clethodim does not control or injure jimsonweed (Jordan, 2006). In addition to herbicide application, the experimental area was kept weed-free by weekly hand removal.
Height measurements were recorded for up to four jimsonweed plants (one treatment contained only two jimsonweed plants in a plot) and four peanut plants and peanut canopy diameter measurements were taken bi-weekly during the season. At the end of the growing season, up to four jimsonweed plants were harvested in each plot, and the remaining plants were cut at ground level to facilitate peanut inversion and harvest. At jimsonweed harvest, seed pods were hand-removed from plants, kept separate, and seed production was quantified. Plants and pods were dried and dry weights were taken for each sample. Peanut yield was determined by inverting the middle two rows of each plot and allowing the peanuts to air-dry in the field for approximately 1 wk. Finally, peanuts were harvested with a combine modified for small-plot research and weighed.
Statistical Analyses
Data were tested for homogeneity of variance prior to statistical analysis by plotting residuals. Analysis of variance (ANOVA) was performed on peanut yield loss, jimsonweed dry biomass, jimsonweed seed weight and number, and jimsonweed pod weight. Linear, quadratic, and higher-order effects were tested by partitioning sums of squares (Draper and Smith, 1981). Location was considered a random variable, and the weed-density main effects were tested by the error associated with the appropriate location by weed-density interaction (McIntosh, 1983). Significant effects were explained using appropriate regression. Nonlinear models were used if ANOVA indicated that higher-order polynomial effects of jimsonweed density were more significant than linear or quadratic effects. Iterations were performed to determine parameter estimates with least sums of squares for all nonlinear models using the Gauss-Newton method via PRO NLIN in SAS ([SAS] 1998).
Plant height was measured at different time intervals after planting each location. Therefore, the Gompertz equation (Equation 1, Draper and Smith, 1981) was fitted to plant height of each species in each plot:
 1
1
The relationship between jimsonweed density per meter of row and percent peanut yield loss was fitted to the rectangular hyperbola (Equation 2) (Cousens, 1998).
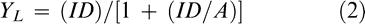 2
2
Coefficients of determination (R 2) were calculated for all regressions. For linear equations, R 2 values were calculated as 100 times the ratio of regression sums of squares to corrected total sums of squares (Askew et al., 2001; Draper and Smith, 1981). Where a nonlinear equation was fitted to the data, an approximate R 2 value, was calculated by other researchers (Askew et al., 2001; Draper and Smith, 1981; Jasieniuk et al., 1999), was obtained by subtracting the ratio of residual sums of squares to corrected total sums of squares from Equation 1. The R 2 and residual mean squares were used to determine goodness of fit to nonlinear models.
Results and Discussion
Plant Diameter and Height
Peanut diameter, measured as canopy row width at four random locations, increased over time in plots with low jimsonweed density. However, in the two highest density jimsonweed plots, peanut diameters were less than 25 cm. Average peanut diameter at the lower jimsonweed densities was near 85 cm (93% of row width) at harvest. Thus, peanut diameter does not seem a reliable predictor of peanut productivity or jimsonweed interference.
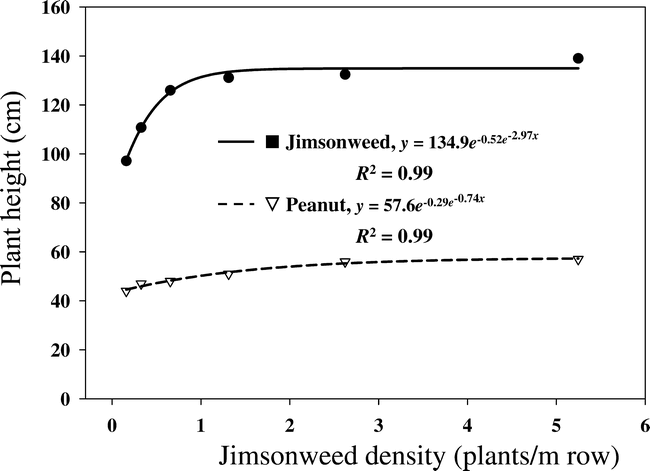
Analysis of variance on estimated parameters of the Gompertz equation (Equation 1) (Draper and Smith, 1981) indicated that jimsonweed and peanut heights were both significantly affected by jimsonweed density while location affects were not significant. Data were therefore pooled to explain the effect of jimsonweed density on jimsonweed and peanut heights (Figure 1). Furthermore, trends in plant height over time indicated that maximum jimsonweed density effects occurred at 11 weeks after planting (WAP). Peanut height increased from 44 to 57 cm as jimsonweed density increased from 0 to 5.25 plants/m of row (Figure 1). Likewise, jimsonweed heights increased from 97 to 139 cm as jimsonweed density increased from 0 to 5.25 plants/m of row.
Jimsonweed Biomass
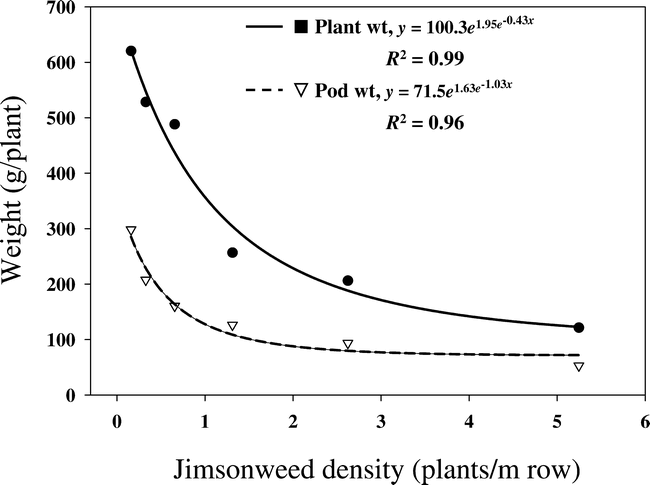
There was no location effect for jimsonweed plant and seedpod weights; therefore, data were combined over locations. Jimsonweed plant and seedpod weights were significantly influenced by jimsonweed density (Figure 2). As jimsonweed density increased, plant and pod weight decreased logarithmically, indicating intra-specific weed interference weed interference at the higher weed densities. Plant weight in Figure 2 consists of dry vegetative matter and totaled 620 g when plants were grown at the lowest jimsonweed density. The 918 g total plant weight was 68% vegetative matter and 32% pods (Figure 2). Thus, reproductive structures contribute a substantial amount of jimsonweed dry biomass at peanut harvest. When jimsonweed were grown at the highest density, vegetative weight was 121 g (70% of total) and pods contributed 52 g (30% of total). Although both plant weight and pod weight decreased as jimsonweed density increased, jimsonweed's competitive index remained more stable at between 30 and 32% based on trends in Figure 2.
Jimsonweed Seed Production
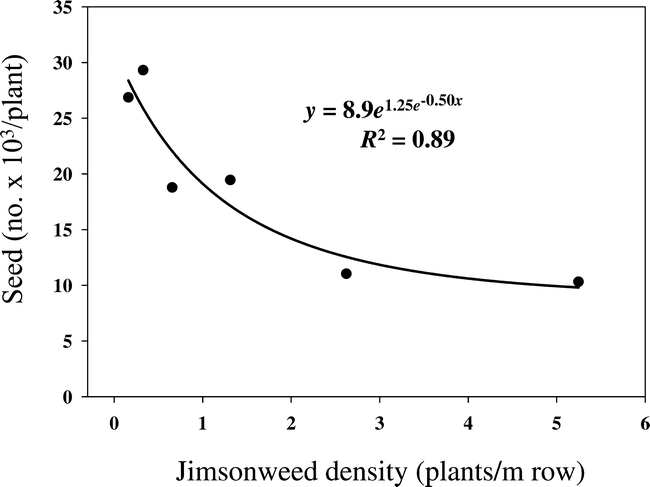
There was no location effect for jimsonweed seed production; therefore, data were combined over locations. Number of seed produced per plant decreased with increasing jimsonweed density (Figure 3). When jimsonweed was grown at lower densities, seed production amounted to nearly 30,000 per plant, which was reduced to 10,000 per plant when densities increased to 5.25 plants per m of row. However, overall seed production increased from 60,0000 seed/plot at the lowest density to 640,000 seed/plot at the highest density (data not shown). Increasing jimsonweed density also decreased seed per plant and increased total seed produced when grown in competition with cotton (Scott et al., 2000).
Peanut Yield Loss
Analysis of variance on estimated parameters of the rectangular hyperbola equation (Equation 2) (Cousens, 1987) indicated that peanut heights were significantly affected by jimsonweed density while location effects were not significant. Data were therefore pooled to explain the effect of jimsonweed density on peanut yield (Figure 4). Percent peanut yield reduction increased with increasing jimsonweed density. Jimsonweed density on percent peanut yield loss resulted in i and a values of 10.7 and 98, respectively (Figure 4). Jimsonweed's competitiveness with peanut at lower densities is similar to competitiveness in cotton (Scott et al., 2000). As additional comparisons in peanut, the i value for common ragweed was 68 (Clewis et al., 2001) and 149.5 for cocklebur (Royal et al., 1997), indicating common ragweed and cocklebur were more competitive in peanut than jimsonweed. Common cocklebur (Royal et al., 1997), common ragweed (Clewis et al., 2001), Florida beggarweed (Cardina and Brecke, 1989), and tropic croton (Thomas et al., 2004) plant biomass were also inversely related to peanut yield. Similarities between locations reflect the similarity of peanut yield and weed dry biomass accumulation at each location, however it should be noted that weeds affect crop yield more when yield potential is higher (Swanton et al., 1999).
Predicted peanut yield loss from season-long interference of one jimsonweed plant per meter of crop row was 40% (Figure 4). Using the hyperbolic function (Cousens, 1987) and asymptotic values constrained to 100% yield loss, maximum peanut yield loss when grown with one wild poinsettia (Bridges et al., 1992), tropic croton (Thomas et al., 2004), horsenettle (Hacket et al., 1987), and bristly starbur (Walker et al., 1989) plant per meter of crop was 17, 17, 14, and 13%, respectively. Jimsonweed is more competitive than these weeds; however, its interference is similar to common ragweed and less than common cocklebur. Common ragweed reduced peanut pod yield 40% at one weed per meter of crop row (Clewis et al., 2001), while common cocklebur reduced peanut pod yield 70% at the same density (Royal et al., 1997).
Conclusions
Peanut diameter did not accurately predict peanut productivity or jimsonweed interference. Weed biomass response to plant density indicated that intra-specific weed interference occurred at higher weed densities. Weed biomass was also inversely related to peanut yield. The high growth rate of jimsonweed coupled with the height of the jimsonweed observed in this study makes jimsonweed one of the most competitive weeds in peanut. When jimsonweed is present and emerges at or before peanut emergence, treatment should be eminent as population densities of jimsonweed rarely occur below economic thresholds. Future studies need to focus on yield loss caused by infestations emerging during the growing season.
Acknowledgements
We thank Scott Clewis, Caitlyn Wilcut, and Jared Wilcut for technical support and Cavell Brownie for reviewing statistical analyses. Appreciation is also extended to the North Carolina Peanut Growers Association for partial funding of this research.
Literature Cited
Anonymous 2006 USDA Plants Database. http://plants.usda.gov/java/profile?symbol = DAST Accessed 3/2/2006.
Brandenburg R. L. 2005 Weed management in peanuts. 61 – 78 in Peanut Information 2005 North Carolina Cooperative Extension Service Raleigh, NC .
Bridges D. C. , Brecke B. J. , and Barbour J. C. 1992 Wild poinsettia (Euphorbia heterophylla) interference with peanut (Arachis hypogaea). Weed Sci 40 : 37 – 42 .
Cardina J. and Brecke B. J. 1989 Growth and development of Florida beggarweed (Desmodium tortuosum) selections. Weed Sci 37 : 207 – 210 .
Clewis S. B. , Askew S. D. , and Wilcut J. W. 2001 Common ragweed interference in peanut. Weed Sci 49 : 768 – 772 .
Cousens R. 1987 Theory and reality of weed control thresholds. Plant Prot. Q 2 : 13 – 20.
Czapar G. F. , Curry M. P. , and Wax L. M. 1997 Grower acceptance of economic thresholds for weed management in Illinois. Weed Technol 11 : 828 – 831 .
Draper N. R. and Smith H. 1981 Applied Regression Analysis. J. Wiley New York 33 – 42, 511 .
Frazee R. W. and Stoller E. W. 1974 Differential growth of corn, soybean, and seven dicotyledonous weed seedlings. Weed Sci 22 : 336 – 339 .
Hackett N. M. , Murray D. S. , and Weeks D. L. 1987 Interference of horsenettle (Solanum carolinense) with peanuts (Arachis hypogaea). Weed Sci 35 : 780 – 784 .
Hagood E. S. , Bauman T. T. , Williams J. L. , and Schreiber M. 1981 Growth analysis of soybean (Glycine max) in competition with jimsonweed (Datura stramonium). Weed Sci 29 : 500 – 504 .
Jordan D. L. 2006 Weed management in peanuts. 17 – 60 in Peanut Information 2006 North Carolina Cooperative Extension Service Raleigh, NC http://www.peanut.ncsu.edu/peanutinfo2006.htm Accessed 03/1/2006.
Oliver L. R. , Chandler J. M. , and Buchanan G. A. 1991 Influence of geographic region on jimsonweed (Datura stramonium) interference in soybean (Glycine max) and cotton (Gossypium hirsutum). Weed Sci 39 : 585 – 589 .
Patterson D. T. and Flint E. P. 1983 Comparative water relations, photosynthesis, and growth of soybean (Glycine max) and seven associated weeds. Weed Sci 31 : 318 – 323 .
Royal S. S. , Brecke B. J. , and Colvin D. L. 1997 Common cocklebur (Xanthium strumarium) interference with peanut (Arachis hypogaea). Weed Science 45 : 38 – 43 .
Scott G. H. , Askew S. D. , Wilcut J. W. , and Brownie C. 2000 Datura stramonium interference and seed rain in Gossypium hirsutum. Weed Technol 48 : 613 – 617 .
Shew B. 2006 Weed management in peanuts. 89 – 102 in Peanut Information 2006 North Carolina Cooperative Extension Service Raleigh, NC http://www.peanut.ncsu.edu/peanutinfo2006.htm Accessed 03/1/2006.
Stoller E. W. and Woolley J. T. 1985 Competition for light by broadleaf weeds in soybean (Glycine max). Weed Sci 33 : 199 – 202 .
Swanton C. J. , Weaver S. , Cowan P. , Van Acker R. , Deen W. , and Shreshta A. 1999 Weed Thresholds: Theory and Applicability. 9 – 29 in Buhler D. D. eds. Expanding the Context of Weed Management Food Products Press, an imprint of The Haworth Press, Inc Binghamton, NY .
Thomas W. E. , Askew S. D. , and Wilcut J. W. 2004 Tropic croton interference in peanut. Weed Technol 18 : 119 – 123 .
Walker R. H. , Wells L. W. , and McGuire J. A. 1989 Bristly starbur (Acanthospermum hispidium) interference in peanuts (Arachis hypogaea). Weed Sci 37 : 196 – 200 .
Notes
- Plant Physiol., National Soil Dynamics Laboratory, Agriculture Research Service, United States Department of Agriculture, 411 South Donahue Drive, Auburn, AL 36832. Email address of corresponding author: john_wilcut@ncsu.edu. [^]
- Grad. Res. Assist., Grad. Res. Assist., Assist. Prof., Grad. Res. Assist. and Prof., Crop Science Department, Box 7620, North Carolina State University, Raleigh, NC 27695-7620. [^] *Corresponding author (email: aprice@ars.usda.gov).
Author Affiliations