Introduction
Tomato spotted wilt virus, a Tospovirus, has recently become one of the most devastating pathogens of peanut (Arachis hypogaea L.) in North Carolina (Cochran et al., 2003) causing a disease known as tomato spotted wilt (TSW). In peanut-producing areas of North Carolina, Frankliniella fusca Hinds, the tobacco thrips, is the most prevalent thrips species, accounting for about 95% of the total thrips (Barbour and Brandenburg, 1994; Eckel et al., 1996). Although the economic threshold for leaf injury by thrips is 25% and is frequently exceeded (Brandenburg, 2003), the concern for feeding injury to foliage has become secondary to the greater economic risk of thrips-transmitted disease. When a peanut plant is infected with TSW, oil content of seeds can be reduced in infected plants (Ali and Rao, 1982) and yield loss may occur (Pappu et al., 1999). Yield loss is due to both decreased weight of seeds and overall lower seed production in infected plants (Culbreath et al., 1992).
Management practices that reduce incidence of TSW include increasing the plant population, planting cultivars with field resistance to TSW, seeding peanut in twin row planting patterns (rows spaced approximately 18 cm apart on centers spaced 91 to 100 cm apart), altering planting dates, and applying the insecticide phorate in-furrow rather than aldicarb or no in-furrow insecticide (Brown et al., 2003; Hurt et al., 2003). Decreased TSW and increased pod yield are often associated with increased plant populations (Hurt et al., 2005; Johnson et al., 2001). Higher plant populations may result in a greater number of healthy plants, which can compensate for diseased and dead plants within rows (Brown et al., 2003). Gorbet and Shokes (1993) found a positive relationship between within-row plant spacing and TSW incidence. The higher seeding rate needed to establish elevated plant population increases production costs; these added costs must be offset by increases in yield and quality in order to justify the use of higher plant populations. In North Carolina, in-row seeding rates of 17 seeds/m are currently recommended for TSW suppression (Hurt et al., 2003; Jordan, 2005).
Cultivar selection is extremely important in managing TSW in peanut (Black et al., 2001; Brown et al., 2003; Culbreath et al., 2000; Hurt et al., 2003). The cultivar Gregory has the best field resistance to TSW of the commercially available Virginia market type cultivars (Hurt et al., 2003). The cultivar NC-V 11 has moderate field resistance to TSW (Shew, 2005), although results are sometimes inconsistent (Culbreath et al., 2000). The cultivar Perry is considered to be susceptible to TSW (Hurt et al., 2003).
Peanut planted in twin rows spaced 17 cm apart with 91 cm centers frequently has higher yield and improved market grade characteristics in many instances when compared with peanut planted in single rows (Culbreath et al., 1999; Lanier et al., 2004). Levels of TSW in fields planted to twin rows are also reduced (Brown et al., 1996), which may be due in part to the earlier canopy cover that may impact the migrating thrips (Culbreath et al., 1999).
Planting peanut in reduced tillage systems often reduces incidence of TSW (Baldwin and Hook, 1998; Brown et al., 2003; Hurt et al., 2003). However, yield of Virginia market types can be lower when planted in reduced tillage systems compared with conventional tillage systems (Brandenburg et al., 1998; Jordan et al., 2003; Jordan et al., 2004). Therefore, employing tillage as a component to manage TSW of peanut needs to be considered carefully.
No single production or pest management practice provides maximum control of TSW in peanut. Several methods must be incorporated into a management program to reasonably suppress this virus (Brown et al., 1996; Culbreath et al., 1999). Preventative methods are necessary to minimize the impact of TSW on peanut because no “rescue” or curative control options are available (Brown et al., 2003; Hurt et al., 2003). Additionally, management of TSW in peanut, as well as other crops, is extremely complex due to the extensive host ranges of the virus and the thrips vectors.
Peanut farmers must consider the impact of management practices designed specifically for TSW on development of other disease and on agronomic performance. This is particularly the case for CBR, a soil-borne disease that has no rescue or curative control measure (Shew, 2005). Additionally, management strategies for TSW often do not affect CBR, and cultivars that are tolerant of TSW are susceptible to CBR (Shew, 2005). Likewise, the CBR-tolerant cultivars NC 12C and Perry are very susceptible to TSW (Shew, 2005). While a combination of production and pest management practices has been successful in reducing the impact of TSW on peanut in the southeastern and southwestern United States, a combination of practices that is effective in the Virginia-Carolina peanut producing region has not been clearly identified, especially with respect to incorporating reduced tillage practices. Therefore, the objectives of this research were to determine the impact of plant population/row pattern, cultivar, in-furrow insecticide, and tillage on TSW and CBR incidence and yield of virginia market-type peanut grown in North Carolina.
Materials and Methods
The experiment was conducted during 2002 in North Carolina at the Peanut Belt Research Station located near Lewiston-Woodville and during 2003 and 2004 at this location and at the Upper Coastal Plain Research Station located near Rocky Mount. In 2003 the experiment was also conducted in a commercial field located near Tyner, NC. Soil at Lewiston-Woodville in all years was a Norfolk loamy sand soil (fine-loamy, siliceous, thermic, Typic Paleudults). At Rocky Mount soil was a Goldsboro sandy loam (fine-loamy, siliceous, thermic Aquic Paleudults) while at Tyner soil was a Wanda fine sand (Siliceous, thermic, Typic Udipsamments). Peanut in either conventional raised beds or strip-tilled beds were planted 5 May 2002, 6 May 2003, and 8 May 2004 at Lewiston-Woodville; 12 May 2003 and 30 April 2004 at Rocky Mount; and 15 May 2003 at Tyner. Peanut at Lewiston-Woodville during 2002 and 2003 was irrigated regularly; however, peanut was not irrigated in 2004 at this location or at the other locations during any year. Plot size was two rows spaced 91 cm apart by 12 m.
Treatments in all experiments consisted of two tillage systems (conventional or reduced tillage), plant population/pattern (in-row densities of 13 plants/m or 17 plants/m), two cultivars (Gregory or Perry), and two in-furrow insecticide treatments (aldicarb or phorate). The conventional tillage treatment was prepared by disking twice and field cultivating followed by establishing raised beds using a row bedder that included in-row subsoiler set to a depth of 30 cm. Reduced tillage was prepared by strip tilling a 45-cm section of each 91-cm row with a strip till implement that included two coulters, in-row subsoiler shank penetrating to a depth of 30 cm, and two rolling basket attachments. A wheat (Triticum aestivum L.) cover crop was established at Lewiston-Woodville during 2002 and 2003, Rocky Mount during 2004, and Tyner. Approximately 2 weeks prior to planting glyphosate at 1.1 kg ai/ha (Roundup Ultra MAX, Monsanto Co., St. Louis, MO) was applied to kill wheat or native vegetation. The low plant population (13 plants/m) was established in single rows spaced 91 cm apart. The high plant population was established in a twin-row planting pattern (two rows spaced 18 cm apart on 91 cm centers) for an in-row population of 17 plants/row m (sum of both twin rows) at Lewiston-Woodville during 2002 and 2003. The high plant population was established in single rows during 2004 at Lewiston-Woodville and at the other locations during all years. Seeds were placed 5 to 8 cm deep depending on soil moisture. Aldicarb (Bayer Crop Science, Research Triangle Park, NC) or phorate (BASF Corporation, Research Triangle Park, NC) was applied at 1.1 kg ai/ha or 1.1 kg ai/ha, respectively, in the seed furrow at planting as a granular formulation prior to seed drop. The cultivars Gregory and Perry offer various levels of field resistance to TSW (Shew, 2005) and pod characteristics ( Jordan, 2005). All other production and pest management practices were held constant across the experiment and were based on Cooperative Extension Service recommendations (Brandenburg, 2005; Jordan, 2005a, 2005b; Shew, 2005). The experimental design was a split plot with tillage serving as whole plots and combinations of cultivar, plant population/pattern, and in-furrow insecticide serving as sub-plot units. Subplots were replicated four times.
Thrips injury was recorded 4 wks after planting by evaluating the most recently emerged leaves for signs of thrips feeding on 25 plants per plot. Leaflets were scored as injured or non-injured based on the presence or absence of scarring. The percentage of leaflets with feeding injury was then calculated. Within 1 wk prior to plant inversion, plant condition ratings were recorded. The ratings were comprised of visual estimates of percent diseased plants in the peanut canopy were recorded using a scale where 0 = no symptoms and 100 = all plants in a plot exhibiting symptoms with increments of 1. ImmunoStrip assays (Agdia STX 89300, ACC 00936, Elkhart, IN) were used to document the dominant disease at each location. Peanut plants were inverted in late September or early October based on pod mesocarp color to optimize pod yield. Pods were allowed to air dry for a period of 4 to 7 days prior to combining.
Data for thrips injury, visual estimates of plant condition, and pod yield were subjected to analyses of variance for a split block yield design with mixed factors (SAS Institute, Cary, NC). Means of significant main effects and interactions were separated using Fisher's Protected LSD Test at P ≤ 0.05 (Steel et al., 1997).
Results and Discussion
Thrips Injury
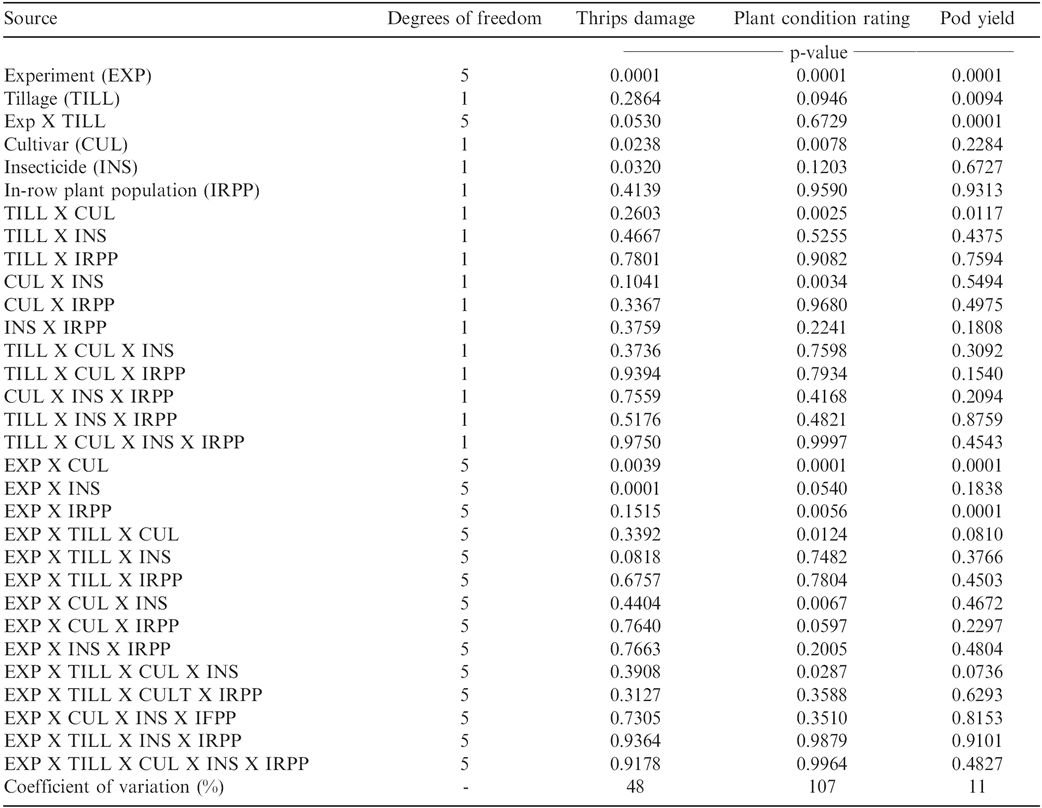
Main effects of cultivar and insecticide were significant for thrips injury (Table 1). Also, interactions of experiment (location and year combinations) with tillage, cultivar, and in-furrow insecticide were significant. All other main effects and interactions were not significant. When pooled over cultivars, in-furrow insecticides, and in-row plant populations, thrips damage was lower in reduced tillage than in conventional tillage at Lewiston-Woodville during 2002 (Table 1). However, there was no difference in thrips damage when comparing tillage systems at the other locations or during the other years at Lewiston-Woodville. Brandenburg et al. (1998) reported less thrips damage in reduced tillage systems compared with conventional tillage systems, although the response was inconsistent. Differences in thrips damage were also noted when comparing cultivars at the various locations. Thrips damage was lower for the cultivar Gregory than for the cultivar Perry during 2004 at Lewiston-Woodville and Rocky Mount (Table 2). Hurt et al. (2005) reported greater thrips damage for the cultivar Perry than for the cultivar NC-V 11. Additionally, the percentage of thrips damage was lower at one location (Lewiston-Woodville in 2003) when the in-row peanut population was 17 plants/m2 rather than 13 plants/m2 (Table 2), and this response is consistent with previous research demonstrating less thrips damage when the in-row plant population is higher (Hurt et al., 2005).
Plant Health Condition
Interactions of experiment X tillage system X cultivar X in-furrow insecticide and experiment by in-row plant population were significant for plant health condition (Table 1). Plant health condition was a measure of plants expressing CBR, TSW, or a combination of both diseases. Other foliar and soil borne diseases were controlled in these experiments based on Cooperative Extension recommendations.
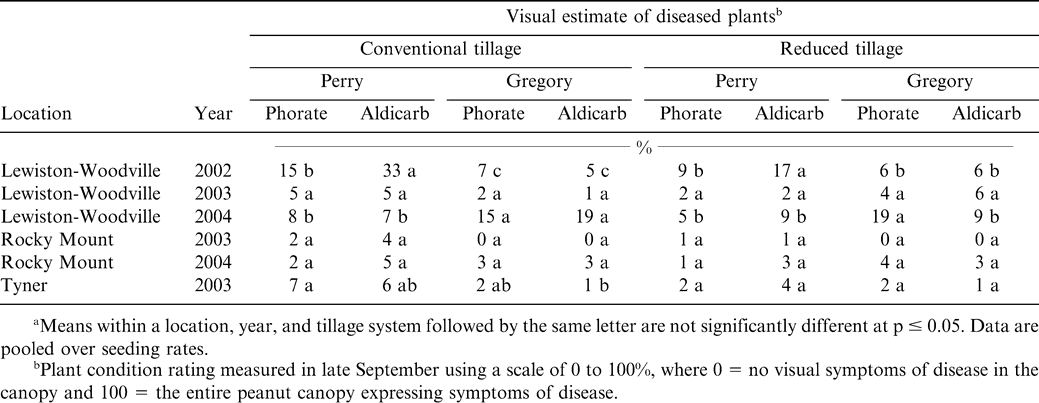
The percentage of plants expressing visible symptoms of CBR or TSW was 6% or less at Lewiston-Woodville in 2003 and at Rocky Mount during both years (Table 3). Field history at Rocky Mount suggested that CBR was not present. Also, TSW was not abundant at these locations during 2003 and 2004 in these experiments and trials conducted by other researchers (data not presented). In 2002 at Lewiston-Woodville, TSW was the dominant disease in the field, and differences were noted when comparing among cultivars and in-furrow insecticides within tillage systems. The highest level of TSW was noted when the cultivar Perry was planted in a conventional tillage system and when aldicarb was applied in the seed furrow (Table 3). Similarly, the highest level of TSW was noted when aldicarb was applied in the seed furrow with the culitvar Perry in the strip tillage system. However, the amount of TSW was 17% for this cultivar/in-furrow insecticide combination in strip tillage compared to 33% in conventional tillage. Planting the cultivar Gregory resulted in 7% or less TSW regardless of tillage system or in-furrow insecticide. Applying phorate with Perry resulted in less disease than when aldicab was applied regardless of tillage system. At Tyner in 2003, no differences in TSW were noted when comparing cultivar and in-furrow insecticide treatments in the reduced tillage system (Table 3). Although disease was 7% or less under conventional tillage systems for all treatments, less disease was noted with the combination of Gregory and aldicarb in-furrow than Perry and phorate applied in-furrow (Table 3). Tomato spotted wilt incidence is often lower when peanut is planted in reduced tillage systems compared with conventional tillage systems (Johnson et al., 2001; Jordan et al., 2003) or when Gregory is planted rather than Perry (Hurt et al., 2005).
The primary disease in 2004 at Lewiston-Woodville was CBR, and based on ImmunoStrip assay results, very little TSW was found in the experiment (data not presented). The highest level of diseased plants was noted for the cultivar Gregory compared with Perry in conventional tillage regardless of the in-furrow insecticide (Table 3). A similar trend was noted in strip tillage when comparing culitvars with phorate applied in the seed furrow, however, less disease was noted when aldicarb was applied with the cultivar Gregory compared to application with phorate. A higher percentage of plants with CBR for the cultivar Gregory is consistent with other research comparing Perry with Gregory (Shew, 2005). However, the difference between disease for Gregory when comparing in-furrow insecticides could not be explained.
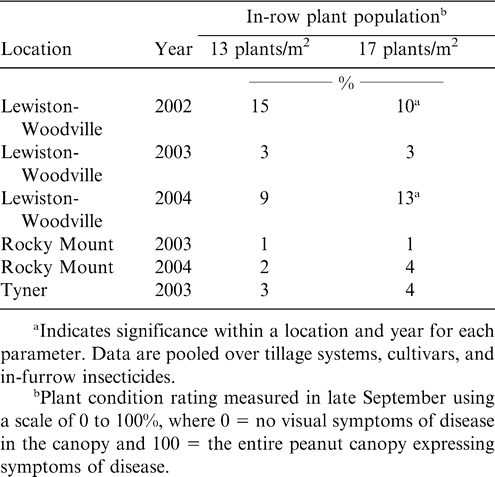
Less disease was noted in 2002 at Lewiston-Woodville when peanut was established at a higher in-row plant population (Table 4). Tomato spotted wilt was the dominant disease in this experiment, and less TSW is often associated with higher plant populations or twin row planting patterns (Brown et al., 2003; Hurt et al., 2003, 2005). In contrast, there was no difference in disease at this location in 2003 at this location or at Rocky Mount during either year or at Tyner. A higher level of CBR was noted for the higher in-row plant population in 2004 at Lewiston-Woodville. The differential response to plant population and in-furrow insecticide noted for CBR in this experiment has not been documented in the literature, and additional research is needed to substantiate or refute these results.
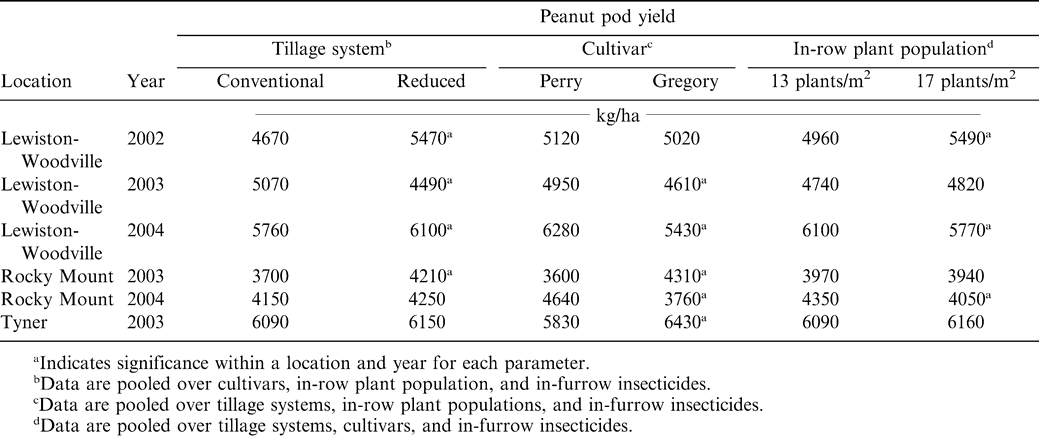
Interactions of experiment X tillage system, experiment X cultivar, experiment X in-row plant population, and tillage system X cultivar was noted for peanut pod yield (Table 1). No other interactions were significant for this parameter. In three experiments pod yield was higher when peanut was planted in reduced tillage systems compared to planting in conventional tillage (Table 5). In one experiment yield in conventional tillage exceeded that in reduced tillage and in the remaining two experiments yield did not differ when comparing tillage systems. Peanut response to tillage can be inconsistent (Baldwin and Hook, 1998; Brandenburg et al., 1998; Johnson et al., 2001; Jordan et al., 2003).
Yield differed between cultivars in five of the six experiments (Table 5). While yield at Lewiston-Woodville did not differ between cultivars, yield of Gregory exceeded that of Perry in three experiments while in two experiments the opposite result was observed. Lower yield for the cultivar Gregory in 2003 at Lewiston-Woodville or in 2004 at Rocky Mount could not be explained by differences in thrips damage or plant health condition (Tables 2 and 3). Likewise, higher yields for Gregory compared to Perry at Rocky Mount in 2003 or at Tyner could not be explained by differences in thrips damage or plant health condition. Lower yield at Lewiston-Woodville in 2003 for Gregory compared to Perry was most likely associated with greater levels of CBR noted for Gregory (Shew, 2005).
Peanut yield was similar during 2003 when comparing plant populations at Lewiston-Woodville, Rocky Mount, and Tyner (Table 5). Yield was higher in 2002 at Lewiston-Woodville when the plant population was higher, and this most likely was associated with reduction in TSW for a higher plant population. However, there was no difference in yield when comparing Gregory and Perry at this location, and there was considerable difference in TSW when comparing these cultivars. One possible explanation for the discrepancy between yield response to similar levels of TSW for the two treatment factors is the positive yield response often observed when peanut is established at higher plant populations and/or twin row planting patterns in absence of TSW (Lanier et al., 2004). Lower yield at a higher plant population was consistent with higher levels of disease for this treatment comparison (Tables 4 and 5). Lower yield for the higher plant population at Rocky Mount in 2003 could not be explained by thrips damage or plant health condition.
Although the interaction of tillage system X cultivar was significant of pod yield (Table 1), there were no differences in yield when comparing yield of cultivars within a tillage system (Table 6). A numerical increase in yield was noted when Gregory was planted in conventional tillage compared to Perry. However, the opposite response was noted in reduced tillage. Previous research (Jordan et al. 2003) reported no interaction among tillage systems and cultivars for Virginia market types.
These data indicate that considerable variation in response to tillage, cultivars, plant population, and in-furrow insecticide can be observed when comparing across years and locations. While data from these experiments generally support current recommendations designed to manage TSW, they also demonstrate variable and unpredictable responses. Distinguishing between CBR and TSW in previous years is critical when incorporating appropriate cultural and pest management practices for control of these diseases. Collectively, these results underscore the challenge of managing TSW in peanut, especially given that management practices for this disease must be incorporated preventatively with no assurance that TSW will emerge as a significant problem in a given field.
Acknowledgements
The authors wish to thank Brenda Penny, Michael Williams, Carl Murphy, Brenda Watson, Sarah Thompson, James Lanier, and Sarah Lancaster for their assistance. Appreciation is also expressed to staff at the Peanut Belt Research and the Upper Coastal Plain Research Stations, Dan Ward, and Gene Roberson for assistance with these experiments. This research was supported by the North Carolina Peanut Growers Association Inc. and USAID Peanut CRSP (LAG-G-00-96-90013-00).
References Cited
Ali M. I. M. and Rao R. D. V. J. P. 1982 Effect of Tomato spotted wilt virus on the oil content of groundnut seeds. Madras Agric. J 69 : 269 – 270 .
Baldwin J. A. and Hook J. 1998 Reduced tillage systems for peanut production in Georgia. Proc. Am. Peanut Res. Ed. Soc 30 : 48 .
Barbour J. D. and Brandenburg R. L. 1994 Vernal infusion of thrips into North Carolina peanut fields. J. Econ. Entomol 87 : 446 – 451 .
Black M. C. , Tewolde H. , Fernandez C. J. , and Schubert A. M. 2001 Seeding rate, irrigation and cultivar effects on tomato spotted wilt, rust, and southern blight diseases of peanut. Peanut Sci 28 : 1 – 4 .
Brandenburg R. L. 2005 Peanut insect and mite management. 61 – 78 In 2005 Peanut Information North Carolina Coop. Ext. Ser. Series AG-331 114 .
Brandenburg R. L. , Herbert D. A. , Sullivan G. A. , Naderman G. C. , and Wright S. F. 1998 The impact of tillage practices on thrips injury of peanut in North Carolina and Virginia. Peanut Sci 25 : 27 – 31 .
Brown S. L. , Todd J. W. , and Culbreath A. K. 1996 Effect of selected cultural practices on incidence of tomato spotted wilt virus and populations of thrips in peanuts. Acta Hort 431 : 491 – 498 .
Brown S. , Tood J. , Culbreath A. , Baldwin J. , Beasley J. , Kemerait B. , and Prostko E. 2003 Minimizing spotted wilt of peanut. Univ. of Georgia, Coop. Ext. Serv. Bull 1165 .
Culbreath A. K. , Todd J. W. , and Demski J. W. 1992 Productivity of Florunner peanut infected with tomato spotted wilt virus. Peanut Sci 19 : 11 – 14 .
Culbreath A. K. , Todd J. W. , Brown S. L. , Baldwin J. A. , and Pappu H. 1999 A genetic and cultural “package” for management of tomato spotted wilt virus in peanut. Biological and Cultural Tests 14 : 1 – 8 .
Culbreath A. K. , Todd J. W. , Gorbet D. W. , Brown S. L. , Baldwin J. , Pappu H. R. , and Shokes F. M. 2000 Reaction of peanut cultivars to spotted wilt. Peanut Sci 27 : 35 – 39 .
Eckel C. S. , Cho K. , Walgenbach J. F. , Kennedy G. G. , and Moyer J. W. 1996 Variation in thrips species composition in field crops and implications for tomato spotted wilt epidemiology in North Carolina. Entomol. Exp. App 78 : 19 – 29 .
Gorbet D. W. and Shokes F. M. 1993 Plant spacing and tomato spotted wilt virus. Proc. Amer. Peanut Res. Ed. Soc 25 : 50 (abstr.).
Hurt C. , Brandenburg R. , Jordan D. , Shew B. , Isleib T. , Linker M. , Herbert A. , Phipps P. , Swann C. , and Mozingo W. 2003 Managing Tomato spotted wilt virus in peanuts in North Carolina and Virginia North Carolina Coop. Ext. Ser. Series AG-638 4 .
Hurt C. A. , Brandenburg R. L. , Jordan D. L. , Kenneday G. G. , and Bailey J. E. 2005 Management of spotted wilt vectored by Frankliniella fusca (Thysanoptera: Thripidae) in Virginia market-type peanut. J. Econ. Entomol 98 : 1435 – 1440 .
Johnson W. C. , Brenneman T. B. , Baker S. H. , Gaschco G. J. , Johnson A. W. , Sumner D. R. , and Mullinix B. G. 2001 Tillage and pest management considerations in a peanut-cotton rotation in the southeastern Coastal Plain. Agron. J 93 : 570 – 576 .
Jordan D. L. 2005 Peanut production practices. 17 – 34 In 2005 Peanut Information North Carolina Coop. Ext. Ser. Series AG-331 114 .
Jordan D. L. 2005 Weed management in peanuts. 35 – 60 In 2005 Peanut Information North Carolina Coop. Ext. Series AG-331 114 .
Jordan D. L. , Barnes J. S. , Bogle C. R. , Brandenburg R. L. , Bailey J. E. , Johnson P. D. , and Culpepper A. S. 2003 Peanut response to cultivar selection, digging date, and tillage intensity. Agron. J 95 : 380 – 385 .
Jordan D. , Brandenburg R. , Shew B. , Naderman G. , Barnes S. , and Bogle C. 2004 Advisory index for transitioning from conventional to reduced tillage peanut production in North Carolina North Carolina Coop. Ext. Ser. AG-644 4 .
Lanier J. E. , Jordan D. L. , Spears J. J. , Wells R. , Johnson P. D. , Barnes J. S. , Hurt C. A. , and Brandenburg R. L. 2004 Peanut response to planting pattern, row spacing, and irrigation. Agron. J 96 : 1066 – 1072 .
Pappu S. S. , Pappu H. R. , Culbreath A. K. , and Todd J. W. 1999 Localization of tomato spotted wilt virus (Genus Tospovirus, Family Bunyaviridae) in peanut pods. Peanut Sci 26 : 98 – 100 .
Shew B. 2005 Peanut disease management. 79 – 104 In 2005 Peanut Information North Carolina Coop. Ext. Ser. Series AG-331 114 .
Steel R. G. D. , Torrie J. H. , and Dickey D. A. 1997 Principles and procedures of statistics: a biometrical approach. 3rd Ed WCB McGraw-Hill New York, NY .
Notes
- Former Graduate Student, Dept. of Entomology, North Carolina State Univ., Current address: USDA-APHIS, 1730 Varsity Dr., Suite 300, Raleigh, NC 27606. [^]
- Prof. and Ag. Tech., Dept. of Entomology, Box 7613, North Carolina State Univ., Raleigh, NC 27695-7613. [^]
- Assoc. Prof. and Ag. Tech., Dept. of Crop Science, Box 7620, North Carolina State Univ., Raleigh, NC 27695-7620. [^] *Corresponding author: david_jordan@ncsu.edu
Author Affiliations