Introduction
In Alabama, Georgia, and Florida, the most important diseases of peanut are early and late leaf spots (caused by Cercospora arachidicola and Cercosporidium personatum, respectively) and southern stem rot (SSR) (caused by Sclerotium rolfsii) (Kokalis-Burelle, et al., 1997). In recent years, the predominant leaf spot disease on peanut in the southeast Alabama production region has been early leaf spot (Hagan, 1998). Without fungicidal control, leaf spot diseases of peanut can defoliate plants, which can result in 50% or greater loss of pod yield (Shokes and Culbreath, 1997). Recommendations for controlling leaf spot diseases are to initiate a preventative fungicide program 30 to 40 days after planting and repeat applications at 10 to 14 day intervals until approximately 14 days prior to crop inversion (Kemerait et al., 2004; Weeks et al., 2005). This schedule generally results in 7 or 8 fungicide applications in a growing season in the southeastern U.S. Average losses due to SSR are estimated at 5% annually, and in order to minimize this disease, the fungicide program should include a product with efficacy against S. rolfsii (Kemerait et al., 2004).
Fungicide applications can be reduced during a growing season by adhering to an advisory program such as AU-Pnuts (Jacobi et al., 1995). In simplest terms, fungicide applications are triggered by the AU-Pnuts advisory according to the accumulation of rain events, defined as ≥ 2.5 mm (0.10 inch) of rain or equivalent irrigation amount in a 24-hr period. Starting from ‘ground cracking,’ i.e., when the peanut seedlings first emerges from the soil, rain events are counted. When 6 rain events have occurred, an initial fungicide application is recommended by this advisory. Starting 10 days after the first fungicide application, subsequent applications are triggered after 3 events have occurred, if the 5-day probability of rain exceeds 50%, or a combination of rain events and high 5-day rainfall probability. Over four years of evaluation, use of AU-Pnuts reduced the average number of fungicide applications by 1.25 per year when tested on Florunner peanut (Jacobi et al., 1995).
AU-Pnuts was developed as a fungicide advisory for leaf spot diseases only, and was based on the use of the fungicide chlorothalonil on the peanut cultivar Florunner. More recently, fungicidal products have become available that have efficacy against soil borne pathogens, particularly S. rolfsii, as well as leaf spot diseases (Culbreath et al., 1995; Hagan et al., 2004). In addition, newer peanut cultivars that are now widely grown have a greater tolerance or partial resistance to leaf spot diseases and/or SSR (Branch and Brenneman, 1996; Cantonwine et al., 2002; Hagan et al., 2004; Hagan et al., 2005; Monfort et al., 2004). These newer fungicides and cultivars may necessitate modification of AU-Pnuts rules. The objective of this work was to compare the standard AU-Pnut disease advisory rules to the advisory with modified rules and to differing calendar schedules of fungicide applications. The trials include the fungicide azoxystrobin for soil-borne pathogen management, and are conducted on currently available cultivars.
Materials and Methods
Production Methods
This study was conducted at the Wiregrass Research and Extension Center in Headland, Alabama. Peanuts were planted on 20 May 2002, 14 May 2003, and 25 May 2004 at a rate of approximately 20 seed per row meter (6 seed per row foot) in a Dothan fine sandy loam with less than 1% organic material. In 2002 and 2004, the late-maturing (maturity group 5) Florida C-99R peanut was planted, while late-maturing (maturity group 5) DP-1 peanut was planted in 2003. Both of these peanut lines have some level of resistance to late leaf spot and SSR (Carter, 2005; Gorbet and Shokes, 2002; Hagan et al., 2000; Hagan et al., 2005). Florida C-99R is also partially resistant to early leaf spot (Hagan et al., 2000). In late March, the plot area, which was maintained in a peanut–cotton–peanut rotation, was sub-soiled, turned with a moldboard plow, and then prepared for planting with a disk harrow. Approximately one month before planting, a preemergence application of 0.85 kg ai/ha ethalfluralin (Sonalan HFP®, Dow AgroSciences, Indianapolis, IN) + 0.027 kg ai/ha diclosulam (Strongarm®, Dow AgroSciences, Indianapolis, IN) was broadcast and lightly incorporated. Aldicarb at 1.27 kg ai/ha (Temik 15G®, Bayer CropScience Kansas City, MO) was applied in-furrow at planting to control thrips. Postemergence grass control was provided by a broadcast application of 0.14 kg ai/ha clethodim (Select®, Valent Agricultural Products, Walnut Creek, CA) + 2.7 L/ha of a crop oil concentrate. Escape weeds were pulled by hand or killed by cultivating row middles with flat sweeps. The test area received approximately 6.3 cm/ha of water through irrigation on 7 Aug., 19 Aug., and 7 Sept. 2002. Due to frequent summer rains in 2003, the test area was not irrigated. In 2004, the equivalent of 6.3 cm/ha of irrigation water was applied on 30 July and 17 Aug.
Fungicide Programs
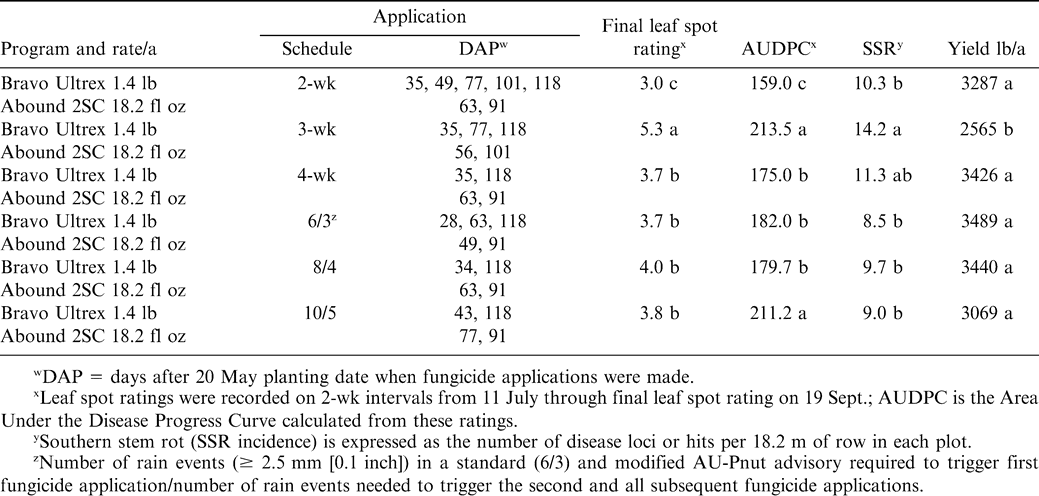
A randomized complete block design with four replications per treatment schedule was used. Plots consisted of four 9.1 m rows spaced 1 m apart and were irrigated as needed (see above). Fungicide treatments were 2-, 3-, and 4-wk intervals between applications (“calendar schedules”), the standard 6/3 AU-Pnut advisory, and modified 8/4 and 10/5 AU-Pnut leaf spot advisories. These AU-Pnut advisories differ by the numerical designation where x/y are: x = the number of rain events (≥ 2.5 mm [0.10 inch]) to trigger the first fungicide application, and y = the number of rain events to trigger each subsequent fungicide application (Jacobi et al., 1995; Weeks et al., 2005). The 2-wk application interval is considered an industry standard and serves as a control in these tests. In all calendar and advisory programs, the first fungicide application was chlorothalonil at 1.26 kg ai/ha (Bravo Ultrex®, Syngenta Crop Protection, Greensboro, NC). Two applications of azoxystrobin at 0.34 kg ai/ha (Abound 2SC®, Syngenta Crop Protection, Greensboro, NC) were made mid-season. Chlorothalonil was applied at all other treatment times. A tractor-mounted boom sprayer with three TX-3 hollow cone nozzles per row, calibrated to deliver 140 L/ha of spray volume, was used to apply all fungicides.
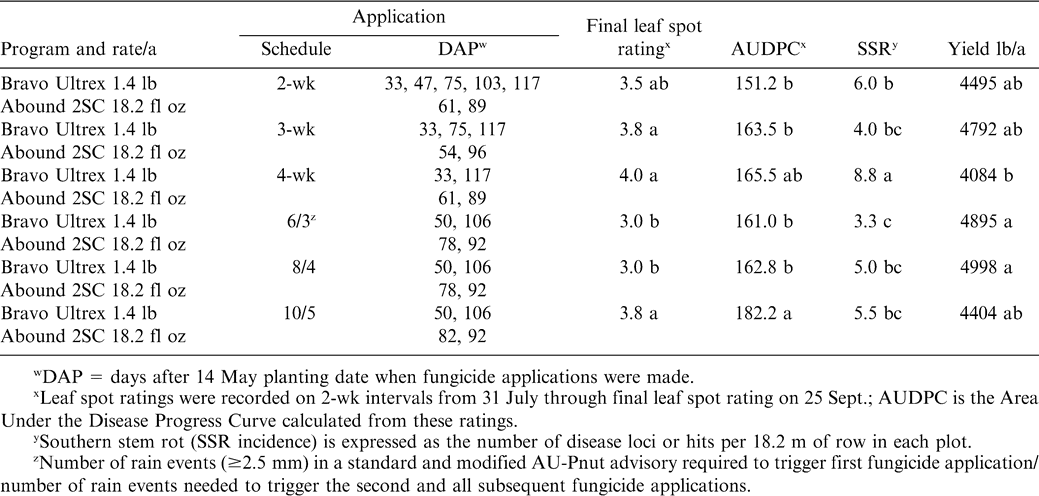
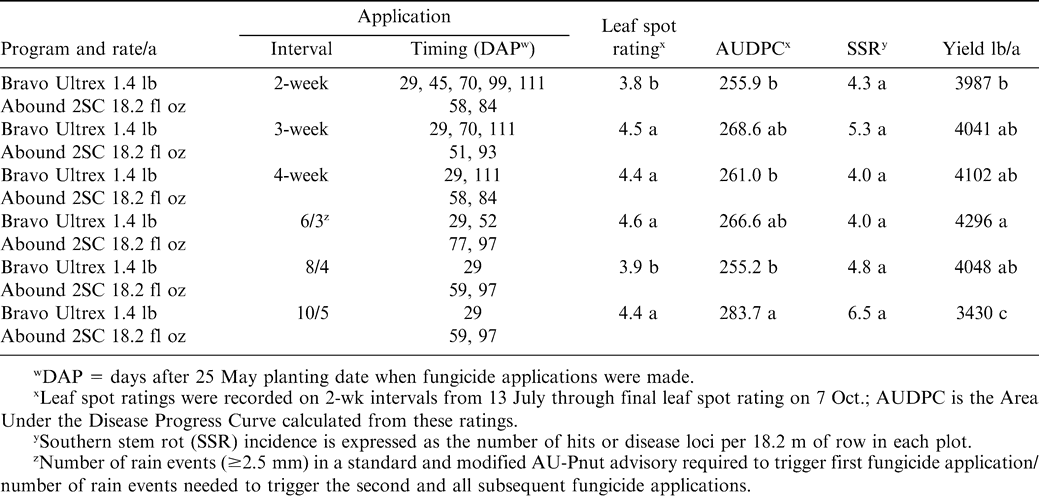
In 2002, chlorothalonil applications were initiated on 24 June for the 2-, 3-, and 4-wk calendar schedules (Table 1). Fungicide was applied according to the 6/3 AU-Pnut advisory on 17 June, 8 July, 22 July, 19 Aug., and 15 Sept, while applications for the 8/4 AU-Pnut advisory programs occurred on 23 June, 22 July, 19 Aug., and 15 Sept. 2002 (Table 1). According to the 10/5 AU-Pnut advisory, fungicide applications were triggered on 2 July, 5 Aug., 19 Aug., and 15 Sept. 2002 (Table 1). In 2003, applications began on 16 June for the calendar schedules (Table 2). Fungicide applications were triggered by the 6/3 and 8/4 AU-Pnut advisories on 3 July, 31 July, 14 Aug., and 28 Aug. 2003; and on 3 July, 4 Aug., 14 Aug., and 28 Aug. 2003 for the 10/5 advisory. For 2004, fungicide applications for all programs (the 2-, 3-, and 4-wk schedules as well as the AU-Pnut advisories) began 23 June (Table 3). Fungicide applications were triggered by the 6/3 AU-Pnut advisory on 23 June, 16 July, 10 Aug., and 30 Aug. 2004; and on 23 June, 23 July and 30 Aug. for the 8/4 and 10/5 advisories (Table 3).
Disease Assessment
Early and late leaf spot were rated together using the Florida peanut leaf spot scoring system where 1 = no disease, 2 = very few lesions in lower canopy, 3 = few lesions in lower and upper canopy, 4 = some lesions in lower and upper canopy with light defoliation (<10%), 5 = lesions noticeable in upper canopy with some defoliation (<25%), 6 = lesions numerous with significant defoliation (<50%), 7 = lesions numerous with heavy defoliation (<75%), 8 = numerous lesions on leaves with severe defoliation (<90%), 9 = few remaining leaves covered with lesions and severe defoliation (<95%), and 10 = plants defoliated or dead (Chiteka et al., 1988). Leaf spot (LS) ratings were recorded every two weeks starting 11 July through 19 Sept. 2002, 31 July through 25 Sept. 2003, and 13 July through 7 Oct. 2004. Incidence of SSR was determined as the number of ‘hits’, or loci counts where 1 hit (locus) is defined as ≤ 30 cm of consecutively damaged plants per row (Rodríguez-Kábana et al., 1975). SSR incidence was determined immediately after plot inversion on 18 Oct. 2002, 13 Oct. 2003, and 28 Oct. 2004. Yields were adjusted to 10% moisture.
Analysis of Data
Areas under disease progress curves (AUDPC's) were calculated (Shaner and Finney, 1977) for each year from leaf spot ratings. AUDPC's were adjusted for the interval of days over which data were collected by dividing by days for analysis across all years. For example, AUDPC's were calculated over 70 days in 2002, so the AUDPC value for 2002 was divided by 70. Year and treatment effects were tested by analysis of variance using Fisher's protected least significant difference test (P = 0.05). Correlation coefficients were also calculated to determine the relative influence of diseases on yield.
Results
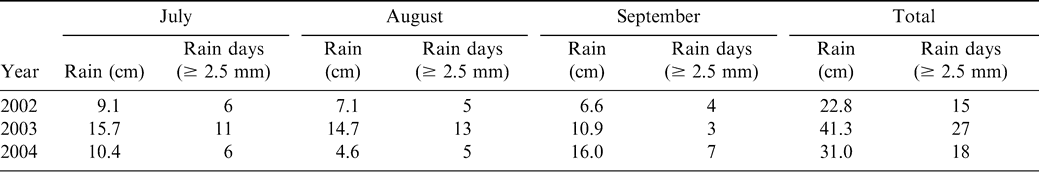
In 2002, through most of the growing season (July through September), rainfall was generally low with few rain days (Table 4). Rain fell more frequently in 2003 and total rain amounts were greater than in the other years of this study. August 2004 was a relative dry month, with only 4.8 cm (1.8 inches) rain falling on five days (Table 4).
Analysis of variance on adjusted AUDPC's for leaf spot, incidence of SSR, and yield indicated significant differences among years. Therefore, further analyses were done on data for individual years using actual AUDPC values. Overall, leaf spot AUDPC's were greatest in 2004, incidence of SSR was highest in 2002, and yields were greatest in 2003 than in other years.
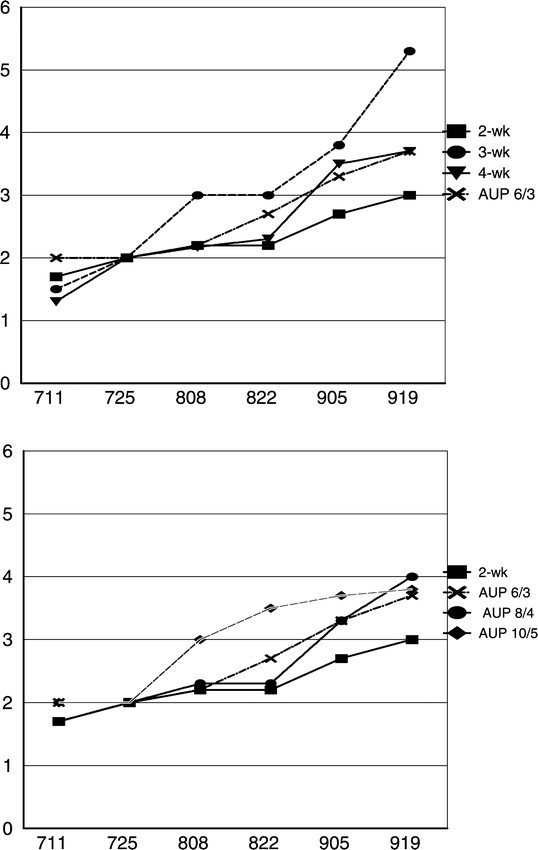
The lowest final rating and AUDPC for leaf spot in 2002 were recorded for the 2-wk calendar fungicide program with 7 fungicide applications (Table 1, Fig. 1). With a final disease rating of 5.3, the poorest leaf spot control was seen with the 3-wk fungicide application program in which defoliation levels exceeded 25%. In all other treatments in 2002, defoliation on 19 Sept. was limited to about 10% of the canopy. There were no significant differences in final leaf spot ratings between the 4-wk calendar schedule and any of the AU-Pnut advisory schedules. AUDPC values reflected the final disease ratings except that the 10/5 AU-Pnut program had an AUDPC value that was similar to the 3-wk calendar program, and these were significantly greater than for other fungicide programs (Table 1).
None of the calendar or AU-Pnut advisory programs with Abound 2SC prevented significant SSR development in 2002, with 8 or more hits observed in 18.2 m row of all plots (Table 1). SSR incidence was greatest with the 3-wk calendar program, which was greater than for all other programs except the 4-wk calendar program (Table 1). While the lowest incidence of SSR was observed for the standard 6/3 AU-Pnut program, SSR incidence did not differ significantly among any AU-Pnut programs, or from the 2- and 4-wk calendar programs in 2002 (Table 1).
Pod yield in 2002 with the 2- and 4-wk calendar and all three AU-Pnut advisory programs were similar, even though up to 3 applications were saved with some of these fungicide programs (Table 1). The high leaf spot and SSR ratings for the 3-wk calendar program were reflected in lower yields in plots treated on 3-wk intervals. Correlation analysis indicated a negative relationship between yield and leaf spots ratings or AUDPCs (r = −0.59 and r = −.45, respectively, P 0.006), but not for SSR incidence (r = −0.27, P = 0.11).
In 2003, defoliation levels in all plots were generally low ( 10%) and each of the AU-Pnut leaf spot advisory programs, with only four fungicide applications, provided as good or better disease control than any of the calendar programs according to final leaf spot ratings (Table 2). The final leaf spot rating for the 2-wk calendar program (with 7 applications) was intermediate between those of the 6/3 and 8/4 AU-Pnut advisory programs and the 3-wk and 4-wk calendar programs. AUDPC's for leaf spots were generally similar for all fungicide application programs, except for the 10/5 AU-Pnut advisory program which resulted in the highest AUDPC.
Highest SSR incidence in 2003 was noted in plots treated according to the 4-wk calendar program in 2003 (Table 2). SSR incidence was similar with fungicides applied according to any of the tested AU-Pnut advisory programs and the 3-wk calendar schedule, and was lower with the 6/3 AU-Pnut program than the 2-wk calendar schedule (Table 2).
Highest yields were obtained from plots treated according to the 6/3 and 8/4 AU-Pnut advisories in 2003, yet these yields were similar to those from plots treated according to the 2- and 3-wk calendar schedules and the 10/5 AU-Pnut advisory. Lowest yields were observed from plots treated on the 4-wk calendar schedule (Table 2). Negative correlation coefficients were calculated for final leaf spot rating (r = −0.50, P = 0.012) and SSR incidence (r = −0.65, P = 0.0006) with yield.
On the basis of the final leaf spot ratings in 2004, the 2-wk calendar program provided better control of these diseases than the 3- or 4-wk calendar schedules, or the 6/3 and 10/5 AU-Pnut advisory programs (Table 3). The 8/4 AU-Pnut advisory program was similar in effectiveness to the 2-wk calendar program in protecting peanut from early leaf spot and provided better control than the 6/3 and 10/5 AU-Pnut programs. AUDPC was greatest for the 10/5 AU-Pnut program, and differed significantly from the 2-and 4-wk calendar and the 8/4 AU-Pnut programs in 2004.
No differences in SSR incidence were noted between any of the calendar and advisory programs in 2004 (Table 3), although the 10/5 AU-Pnut program tended to have higher SSR incidence than did other programs. Higher yield was recorded from plots treated according to the 6/3 AU-Pnut advisory program than for the 2-wk calendar program despite 3 fewer fungicide applications with this advisory. Pod yield from plots treated according to the 10/5 AU-Pnut was lower than for any other tested programs. No significant correlations were observed between disease variables and yield (P 0.10).
Discussion
In the southeastern U.S., it is standard practice when producing peanuts to apply fungicides on 10 to 14 day intervals (Kemerait et al., 2004). Longer intervals between applications may provide sufficient disease control when a resistant cultivar is used or if the weather is dry and a spray advisory is being used (Kemerait et al., 2004). Longer intervals between fungicide applications can reduce the number of fungicide applications, which does decrease peanut production costs. The current study evaluated two general strategies for reducing fungicide applications when leaf spot resistant cultivars are grown. The strategies were: extended application intervals that are calendar-based and use of three modifications of the AU-Pnut advisory (Jacobi et al., 1995).
Previous work has shown that extending the application interval generally decreases leaf spot control, with a corresponding loss in yield, compared to a 2-wk fungicide schedule (Brenneman and Culbreath, 1994; Monfort et al., 2004). These previous studies have even included cultivars such as Southern Runner and Florida C-99R which have partial resistance to leaf spot diseases. Results of the current study are similar to previously published observations in that final ratings and AUDPC's for leaf spot diseases tended to be greater in all three study years when fungicide applications were made every 3- or 4-wks instead of on 2-wk intervals. The differences in final ratings and AUDPC's for leaf spots, as well as pod yields, between the 2-, 3- and 4-wk application schedules did not differ in 2 of the 3 yrs in the current study, even though up to 3 fungicide applications were saved with the longer intervals of application. However, in 2002, AUDPC for leaf spots was 34% greater, incidence of SSR was 38% greater, and yield was 22% lower, with the 3-wk application interval than with the 2-wk interval. To a producer, this indicates a 1 in 3 chance of inadequate disease control.
Another strategy for reducing numbers of fungicide applications is to apply fungicides according to an advisory such as AU-Pnut (Brenneman and Culbreath, 1994; Jacobi et al., 1995). In the current study, in two of three years, AUDPC's for leaf spots were similar in plots treated every 2-wk or treated according to the original AU-Pnut advisory, despite 2 (in 2003) or 3 (in 2004) fewer fungicide applications using the advisory system. Further reductions in fungicide applications are possible when a leaf spot resistant cultivar is grown and AU-Pnut thresholds are raised (Jacobi and Backman, 1995). Two modifications of the original AU-Pnut rules were also evaluated in the current study. These modifications allow additional rainfall events before a fungicide application is triggered. In each of the three study years, AUDPC's were similar when fungicides were applied according to the original AU-Pnut (6/3 thresholds) (Jacobi et al., 1995) or according to the 8/4 modification. The 8/4 modification of AU-Pnut saved one fungicide application in two years (2002 and 2004) compared to the original advisory, and saved up to four applications compared to the 2-wk calendar-based application schedule. Incidence of SSR and yields were similar among all treatments in all years of this study, except for yield from plots treated according to the 10/5 AU-Pnut advisory in 2004 when pod yields were significantly lower than in all other treatments. These results are similar to those of Jacobi and Backman (1995) when modifications to AU-Pnut were tested on the partially resistant Southern Runner peanut cultivar. Over two years, 2 to 4 fungicide applications were saved without yield decreases with 9/4 or 12/4 thresholds for AU-Pnuts in this previous study.
The AU-Pnut advisory was originally developed and validated using only the fungicide chlorothalonil. Over the past decade, fungicidal products have become available that provide control of leaf spots of peanuts as well as soil-borne pathogens. These newer products, such as tebuconazole, flutolanil, azoxystrobin and others, have some systemic activity. Previous studies have demonstrated that these systemic products can be used successfully in conjunction with the AU-Pnut advisory (Brenneman and Culbreath, 1994; Jaks et al., 2002). Since the inclusion of a systemic fungicide in the season-long regime for leaf spot control is currently common practice, this study did not include any regime with only chlorothalonil.
Scrutiny of disease progress curves in 2002 indicates that leaf spot infection increased more after 25 July with the 3-wk calendar and 10/5 AU-Pnut fungicide regimes than with other regimes in that year. Prior to the 25 July 2002 assessment date, fungicide applications had been made on 15 July (azoxystrobin) and 2 July (chlorothalonil) for the 3-wk calendar treatment and the AU-Pnut 10/5 treatment, respectively. Unprotected new growth may have developed or the fungicides had lost efficacy in both of these treatments by 28 July and/or 3 August when substantial rainfall occurred that allowed infection. With all other programs in 2002, fungicide had been applied on 22 July. This indicates that 3-wk intervals or 5 rain events between fungicide applications are likely to be inadequate even with cultivars that have some leaf spot resistance. Distinct differences in disease progress curves among treatments were not observed in 2003 or 2004, so possible differences in disease development due to rain could not be scrutinized in those years.
Grower use of the AU-Pnut advisory for scheduling fungicide applications on peanuts can contribute to lower fungicide and labor costs through a reduction in the numbers of applications. This advisory was developed with the Florunner peanut and the protectant fungicide, chlorothalonil (Jacobi et al., 1995). Over the past decade, new fungicides have become available that have efficacy against soil-borne pathogens and peanut cultivars now grown in the southeastern U.S. have multiple disease resistance. Results of the current study demonstrate that modifications of the AU-Pnut advisory such that thresholds between fungicide applications are higher, can provide adequate disease control without yield loss when a leaf spot resistant cultivar is grown.
Literature Cited
Brenneman T. B. and Culbreath A. K. 1994 Utilizing a sterol demethylation inhibiting fungicide in an advisory program to manage foliar and soilborne pathogens of peanut. Plant Dis 78 : 866 – 872 .
Branch W. D. and Brenneman T. B. 1996 Pod yield and stem rot evaluation of peanut cultivars treated with tebuconazole. Agron. J 88 : 933 – 936 .
Cantonwine E. G. , Culbreath A. K. , Holbrook C. C. , and Gorbet D. W. 2002 Response of moderately resistant peanut breeding lines and cultivars to chlorothalonil for management of early leaf spot. Proc. Amer. Peanut Res. Educ. Soc. 34: 92- 93 (abstr.)
Carter J. 2005 Variety Guidebook; A comparison of the varieties available for farmers today. Southeastern Peanut Farmer Jan/Feb 2005 : 13 – 16 .
Chiteka Z. A. , Gorbet D. W. , Shokes F. M. , Kucharek T. A. , and Knauft D. A. 1988 Components of resistance to late leaf spot in peanut I. Levels of variability-implications for selection. Peanut Sci 15 : 25 – 30 .
Culbreath A. K. , Brenneman T. B. , Bondari K. , Reynolds K. L. , and McLean H. S. 1995 Late leaf spot, southern stem rot, and peanut yield responses to rates of cyproconazole and chlorothalonil applied alone and in combination. Plant Dis 79 : 1121 – 1124 .
Gorbet D. W. and Shokes F. M. 2002 Registration of ‘C-99R’ Peanut. Crop Sci 42 : 2207 .
Hagan A. K. 1998 Foliar Diseases of Peanuts. Ala. Coop. Ext. Sys. ANR-369
Hagan A. K. , Bowen K. L. , Campbell H. L. , Rivas-Davila M. E. , Wells L. , and Pegues M. 2000 Begin the regime… fungicide regimes for foliar and soil disease control in peanuts compared. Alabama Agri. Exp. Sta, Auburn University Highlights of Agric. Res 47 / 4 : 20 – 22 .
Hagan A. K. , Campbell H. L. , and Bowen K. L. 2005 Resistance of commercial runner peanut lines to early leaf spot, tomato spotted wilt, and southern stem rot. Phytopathology 95 : S39 .
Hagan A. K. , Rivas-Davila M. E. , Bowen K. L. , and Wells L. 2004 Comparison of fungicide programs for the control of early leaf spot and southern stem rot on selected peanut cultivars. Peanut Sci 31 : 22 – 27 .
Jacobi J. C. , Backman P. A. , Davis D. P. , and Branneman P. M. 1995 AU-Pnuts advisory I: Development of a rule-based system for scheduling peanut leaf spot fungicide applications. Plant Dis 79 : 666 – 671 .
Jacobi J. C. and Backman P. A. 1995 AU-Pnuts advisory II: Modification of the rule-based leaf spot advisory system for a partially resistant peanut cultivar. Plant Dis 79 : 672 – 676 .
Jaks A. J. , Grichar W. J. , and Beslar B. A. 2002 Use of AUPNUT advisory and fungicides to control peanut diseases, 2001. Fungicide and Nematicide Tests 58 : FC061 .
Kemerait R. , Brenneman T. B. , and Culbreath A. K. 2004 2005 Peanut disease update. University of Georgia College of Agricultural and Environmental Sciences, Online.
Kokalis-Burelle N. , Porter D. M. , Rodriguez-Kabana R. , Smith D. H. , and Subrahmanyam P. 1997 Compendium of Peanut Diseases APS Press St. Paul, MN .
Monfort , Culbreath W. S. A. K. , Stevenson K. L. , Brenneman T. B. , Gorbet D. W. , and Phatak S. C. 2004 Effects of reduced tillage, resistant cultivars, and reduced fungicide inputs on progress of early leaf spot of peanut (Arachis hypogaea). Plant Dis 88 : 858 – 864 .
Rodríguez-Kábana R. , Backman P. A. , and Williams J. C. 1975 Determination of yield losses to Sclerotium rolfsii in peanut fields. Plant Dis. Rep 59 : 855 – 858 .
Shaner G. and Finney R. E. 1977 The effect of nitrogen fertilization on the expression of slow-mildewing resistance in wheat. Phytopathology 67 : 1051 – 1056 .
Shokes F. M. and Culbreath A. K. 1997 Early and late leaf spots. 17- 20 In Kokalis-Burelle N. Porter D. M. Rodriguez-Kabana R. Smith D. H. and Subrahmanyam P. eds. Compendium of Peanut Diseases, 2nd ed APS Press St. Paul, MN.
Weeks J. R. , Hagan A. K. , Hartzog D. , Everest J. W. , and Wehtje G. 2005 Peanut insect, disease, nematode, and weed control recommendations. Ala. Coop. Ext. Ser. Cir 2005IPM-360.
Notes
- This research was supported in part by grants from the Alabama Peanut Producers Association and the National Peanut Board. [^]
- Professor, Alumni Professor, and Research Associate, Department of Entomology and Plant Pathology, Auburn University, Auburn, AL 36849-5624. [^]
- Superintendent, Wiregrass Research and Extension Center, Alabama Agricultural Experiment Station, Headland, AL 36345. [^] *Corresponding author: (email: bowenkl@auburn.edu).
Author Affiliations