Introduction
The factors that affect the degree of interference between weeds and agricultural crops were described in a scheme proposed by Bleasdale (1960). According to this scheme, the degree of interference depends on factors of the crop (species, row spacing, and planting density), of the weed community (species composition, density, and distribution), the edaphic and climatic conditions, and crop management. Time period where weed interference with crop growth is also important (Pitelli, 1985). Previous research indicates that there is great variation in peanut yield loss due to weed interference, reflecting the influences of the many factors that affect the interference relationship between weeds and crops (Hill and Santelmann, 1969; Feakin, 1973; Hauser et al, 1975; Buchanan et al., 1976; Drennan and Jennings, 1977; York and Coble, 1977; Martins and Pitelli, 1994).
The critical period of weed control is defined as an interval in the life cycle of the crop when it must be kept weed-free to prevent yield loss. This period consisted of two discrete periods: (i) the critical time of weed removal, or the length of time that weeds which emerge with the crop can remain uncontrolled before they begin to compete with the crop and cause yield loss, and (ii) the critical weed-free period, or the length of time that the crop must be free of weeds after sowing, in order to prevent yield losses (Nieto et al., 1968; Kropff et al., 1993).
The critical time of weed removal has varied from three to ten weeks (Hill and Santelmann, 1969; Drennan and Jennings, 1977; Pitelli et al., 1981; Pitelli et al., 1984; Barbosa and Pitelli, 1990; Kasai et al., 1997), and the critical weed-free period from one to ten weeks (Hill and Santelmann, 1969; Drennan and Jennings, 1977; Pitelli et al., 1981; Gavioli, 1985; Barbosa and Pitelli, 1990; Kasai et al., 1997).
Feakin (1973) stated that erect peanut cultivars are more tolerant to competition with weeds than the runner cultivars, probably due to the formation of an above ground compact mass, with greater shading between the lines. In Brazil, critical period studies have been conducted mostly for erect cultivars, mainly the cultivar Tatu (Pacheco, 1980; Pitelli et al., 1981; Pitelli et al., 1984; Gavioli, 1985; Barbosa and Pitelli, 1990; Martins and Pitelli, 1994; Kasai et al., 1997). Currently, in the peanut producing areas in Brazil, runner cultivars are being introduced with wider row spacing are being adopted (45 cm changed to 90 cm). Wider row spacings have been adopted to allow for mechanical harvest. For this new production practice, there are no data on critical periods imposed by weeds common in Brazil. Thus, it is possible that the current control practices are inadequate for this emerging planting style. Therefore, the objective of this study was to determine the critical period of weed control in peanuts conducted in a planting system for mechanical harvest.
Materials and Methods
Five experiments were conducted in a same commercial area located in the county of Morro Agudo, SP, Brazil, on a Red Yellow Latosol with a visual uniform weed infestation. The soil analysis, from a sample collected after soil preparation and before setting the experiments, presented the following characteristics: 31% sand; 20% silt; 49% clay; pH in CaCl2 5.5; organic matter 26 g/dm3; P in resin 24 mg/dm3; contents of K, Ca, Mg, H+Al, and S, 0.6, 32, 14 and 28 mmolc/dm3, respectively.
A different cultivar was planted for each experiment: runner cultivars “Caiapó” and ‘Runner ‘Tégua’’ and erect cultivars “ST-Tatu”, “IAC-22”, and “IAC-1075”. Eight rows were mechanically seeded for each cultivar on 20 November 2000, with a row spacing of 90 cm. Peanut was planted at a rate of 32 seed/row meter for runner cultivar and 22 seed/row meter for erect cultivar. Planting fertilization consisted of an application of 48 kg/ha of phosphorus and 24 kg/ha of potassium. Seed germination occurred approximately five days after planting. During the growing season eight insecticide (four cypermethrin + thiamethoxam and four methamidophos) and five fungicide (three chlorothalonil + hexaconazol and two epoxiconazol + pyraclostrobin) sprays were performed for the control of insects and diseases.
The treatments were separated in two groups. In the first group, the crop remained under competition with the natural weed community from planting until removed at 15, 30, 45, 60, 75, 90, and 105 days after planting. A season-long weedy control check was included. Weeds were removed from the plots at the appropriate time by manual weeding, and maintained weed free for the remainder of the growing season. In the second group of treatments, the crop was kept free of weed competition from planting until 15, 30, 45, 60, 75, 90, and 105 days after planting. A season-long weed-free control check was included. After these periods the weeds that germinated were left to grow freely. The terms “weed-infested” for initial periods under competition and “weed-free” for initial periods free from competition with the infesting community were adopted.
For each cultivar the treatments were arranged in a randomized complete block design, with four replications. The experimental units consisted of four 5-m planting rows (18 m2). Data measurements were made in the two center peanut rows (9.0 m2).
The evaluations of the weed community were conducted at the end of the each association periods in the treatments where the crop remained weed-infested. Two sampling squares of 0.25 m2 per experimental unit were used to measure weed species composition, weed density, and weed biomass. The weed community at the different experiments showed the same behavior. Thus, data were combined across cultivars and the total number of plants and accumulated dry matter by the infesting community were subjected to regression analysis as function of time (days after planting).
Peanut yields were obtained by mechanical digging, air-drying in the field for seven days, and harvesting individual plots manually. Weights were recorded after soil and trash were removed from the samples and moisture adjustment to 8%. Total weight of the samples and weight of 100 seeds were determined.
Peanut yield data were processed separately within each group (weed-infested or weed-free), and subjected to regression analysis (Cousens, 1988; van Acker et al., 1993; Mulugeta and Boerboom, 2000). Linear equation and Boltzman sigmoidal equation, adapted from Kuva et al. (2000), were fit to the yield data. The limits of the critical periods of weed control were determined allowing for a maximum yield loss of 5% in relation to that obtained in the experimental units kept weed-free during the whole growing season.
Results and Discussion
Weed Community
The weed community was composed by the follow family and species: Amaranthaceae: Alternantera tenella Colla, Amaranthus deflexus L.; Asteraceae: Acanthospermum hispidum DC., Acanthospermum australe (Lofl.) Kuntze., Bidens pilosa L., Blainvillea rhomboidea Cass., Emilia sonchifolia DC.; Commelinaceae: Commelina benhalensis L.; Convolvulaceae: Ipomoea acuminata Roem. et Schult; Cyperaceae: Cyperus rotundus L.; Euphorbiaceae: Chamaesyce hirta (L.) Millsp., Chamaesyce hissopifolia (L.) Small., Plyllanthus tenellus Roxb.; Malvaceae: Sida rhombifolia L.; Mimosaceae: Aeschynomense rudis Benth.; Poaceae: Brachiaria decumbens Stapf., B. plantaginea (Link.) Hitch; Cenchrus echinatus L., Cynodon dactylon (L.) Pers., Digitaria horizontalis Willd., Eleusine indica (L.) Gaertn., Panicum maximum Jacq.; Portulacalaceae: Portulaca oleraceae L.; Rubiaceae: Richardia brasiliensis Gómez, Spermacoce latifola Aubl.; Família Solanaceae: Physalis angulata L., Solanum americanum Will.
The dicots predominated, corresponding to 75% of all species found. The most numerous dicot families were Asteraceae, with five species, and Euphorbiaceae, with three species. Among the monocots, the major family was Poaceae, with seven species, which among all weed families present in the experimental area, was the most numerous. The species from Poaceae corresponded to 26% of all species found.
According to Clark (1971), the different species composing an infesting community can affect, in part, the degree of interference, since the competitive ability varies among the species. It is hypothesized that the more physiologically similar two species are, and often these are related to the taxa classification, the closer their needs will be and the more intense the competition for the limited factors of the common ecosystem.
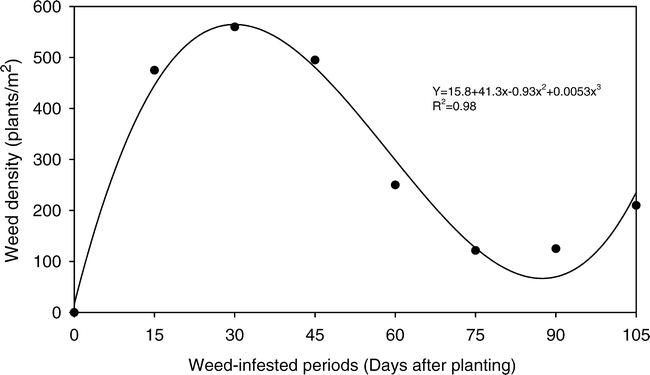
It was observed that in terms of number of individuals (Fig. 1), after the 15th day of planting the infesting community was established, reaching its maximum estimated density on day 30, with 565 plants/m2. Later the population density decreased until day 87, when it reached 67 plants/m2. This reduction in density was probably due to the interference between the individuals, which caused plant mortality in the infesting community. Dekker and Meggit (1983), in studies of population dynamics, observed that the increase in population density of Datura stramonium L. (jimsonweed), associated to plant growth, induced an increase in the mortality rate. In the present study, after the stabilization of the interference effect, an increase in the weed density was observed (Fig. 1), probably resulting from a new emergence outbreak, reaching 235 individuals/m2, on day 105 after peanut planting.
The increase in weed density, until 30 days after peanut seeding, could have affected the crop. Direct competition for the medium resources may have affected the crop. Otherwise, the return of a population increase, at the end of the growing season, also could have affected the crop, prevailing the indirect interference, obstructing the harvest process, in operational terms as well as in maturation time for the peanut branches.
Bhan et al. (1971) observed, in a study done in India, that 75% of the components of an infesting community of the peanut crop could emerge in the first 30 days of the growing season. In Brazil, Pitelli (1980) observed that in a “rainy season” peanut crop the maximum number of individuals was established at 20 days. In a “dry season”, Pitelli et al. (1984) observed that this establishment occurred after 14 days.
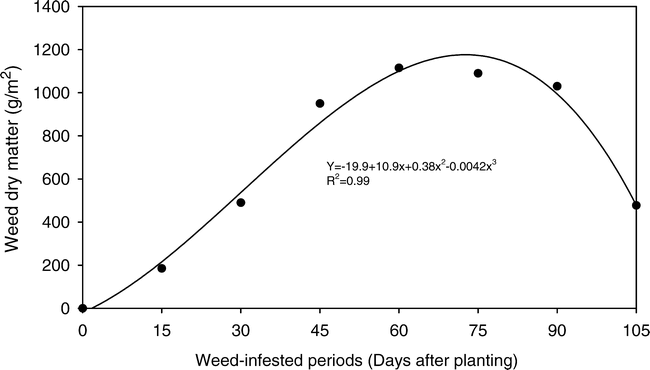
The weed community accumulated dry biomass linearly, until 66 days after peanut seeding, when a maximum accumulation of dry matter (1049 g/m2) was observed (Fig. 2). Dry matter accumulated decreased at 105 days, reaching a value of 630 g/m2, due probability to natural senescence. Making a correlation between the data on infesting community density and dry matter accumulation, one can observe that, although there was plant mortality as a consequence of interference among individuals of the infesting community, the plants remaining in the area developed and accumulated dry biomass.
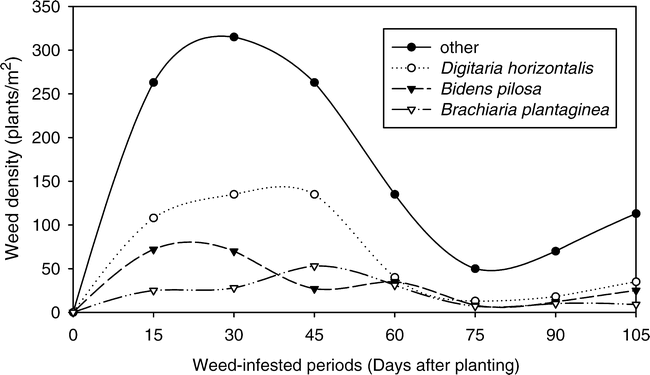
Among the species of the infesting community the three most important were Brachiaria plantaginea (Link) Hitchc. (alexander grass), Digitaria horizontalis Willd. (crabgrass) and Bidens pilosa L. (hairy beggarticks) (Fig. 2). In terms of population density, D. horizontalis was the most numerous species in most evaluations, followed by B. pilosa and B. plantaginea (Fig. 3). At 30 days after peanut seeding, when the infesting community reached its maximum density, D. horizontalis contributed with 22% (123 plants/m2) of this infestation, B. pilosa with 11% (62 plants/m2) and B. plantaginea with 5% (28 plants/m2). At 105 days, the major weed responsible for the new population density increase was B. pilosa with 17% (36 plants/m2), followed by D. horizontalis with 12% (26 plants/m2) and by B. plantaginea with 6% (12 plants/m2).
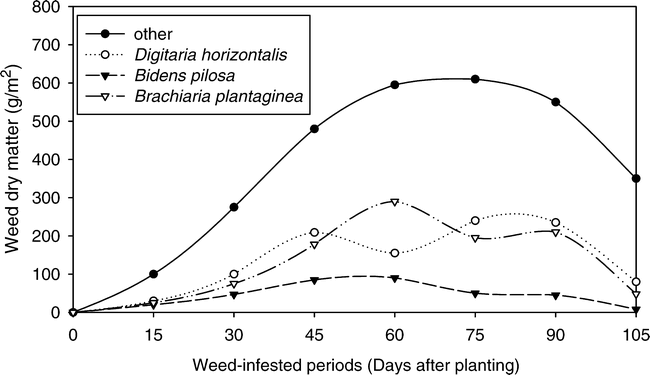
Digitaria horizontalis was also the weed that presented the greatest dry biomass accumulated in most evaluations, reaching, at 75 days, the maximum accumulation of 240.0 g/m2 (Fig. 4). Only when the dry biomass accumulation by the infesting community was maximum, at 60 days after planting, B. plantaginea reached greater value of dry biomass accumulated than D. horizontalis, accumulating 287.2 g/m2. At 60 days after planting, the population of B. plantaginea represented 26% of the total accumulated by the infesting community and the populations of D. horizontalis and B. pilosa represented 15% (161.4 g/m2) and 8% (87.9 g/m2), respectively.
Peanut Yield
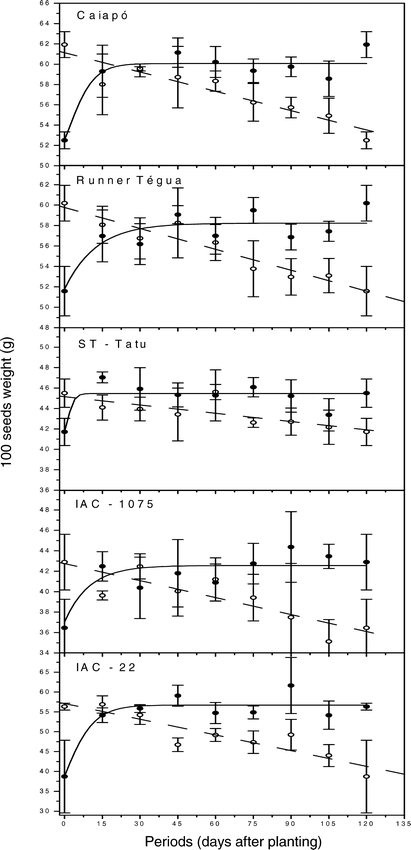
When the weight of 100 seeds is compared between peanut cultivars that had grown free of weed interference (weed-free) (Fig. 5), one observes that the cultivar that presented the greatest weight was ‘Caiapó’ (61.9 g), followed by Runner ‘Tégua’ (60.2 g), ‘IAC-22’ (56.3 g), ‘ST-Tatu’ (45.5 g) and ‘IAC-1075’ (42.9 g). Comparing the weights of 100 seeds from the treatments maintained weed-free with those maintained weed-infested during the whole growing season, the weed interference caused a 15% reduction on the weight of 100 seeds of cultivar ‘Caiapó’, 14% of Runner ‘Tégua’, 31% of ‘IAC-22’, 8% of ‘ST-Tatu’ and 15% of ‘IAC-1075’. Thus, the cultivar most sensitive to weed interference, in relation to the weight of 100 seeds, was ‘IAC-22’ and the least sensitive ‘ST-Tatu’.
Weight of 100 seeds response to increasing length of weed-free (• observed values with standard error, ___ estimated values) or weed-infested (○ observed values with standard error, --- estimated values) periods as calculated from data in table 1.
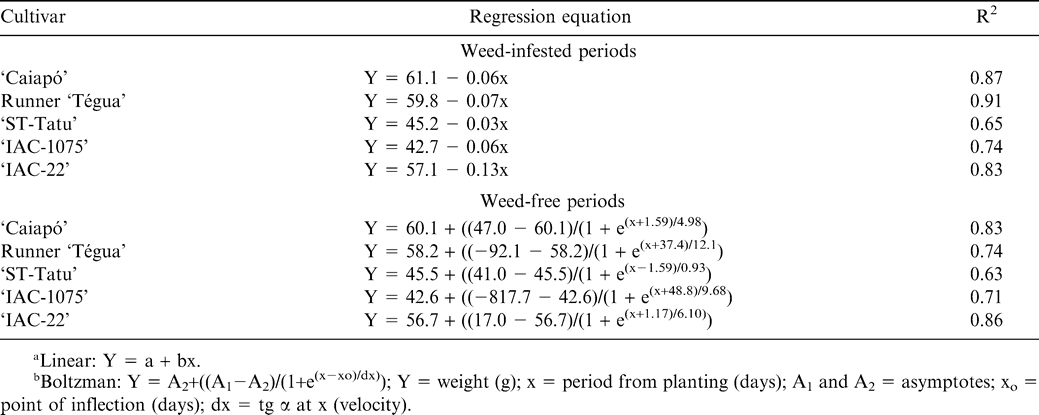
Considering the weed-infested periods, the weight of 100 seeds for all cultivars adjusted well to the linear regression (Fig. 5 and Table 1), where, according to the equations obtained, the reductions on the weight of 100 seeds were of 0.06 g/day for cultivar ‘Caiapó’, 0.07 g/day for Runner ‘Tégua’, 0.13 g/day for ‘IAC-22’, 0.03 g/day for ‘ST-Tatu’ and 0.06 g/day for ‘IAC-1075’. The increase on the period that the crop was kept weed-free resulted in a positive effect on the weight of 100 seeds for all cultivars, with the data adjusted to the Boltzman sigmoidal equation.
Pitelli (1980), Bianco (1978) and Ishag (1971) also observed negative effects of the infesting community presence on the average weight of 100 peanut seeds. Pitelli (1980) mentioned that, due to the great non-uniformity of the infesting community in the crop areas, peanut plants are subjected to the most diverse conditions of shading, competition, of soil thermal and hydric variations, of allelopathic processes and of other factors that could direct or indirectly, change flowering. Thus, with the non-uniformity of flowering and, fruit set, by harvest there would be mature seeds side by side with growing seeds. This would make the average size of the seeds smaller and reflect on the weight of 100 seeds. However, Dajos (1983) stated that, generally, the competition between plants leads to a reduction on the individual weight of a plant and on the number of seeds, but the weight of each seed tends to be constant. Gavioli (1985) observed that in the commercial classification of the seeds as well as in the average size, the seeds did not differ as a consequence of interference by an infesting community, in which D. horizontalis predominated. That author attributed this result to the specific condition of that study, where the infestation was uniform, differently from what was reported by Pitelli (1980).
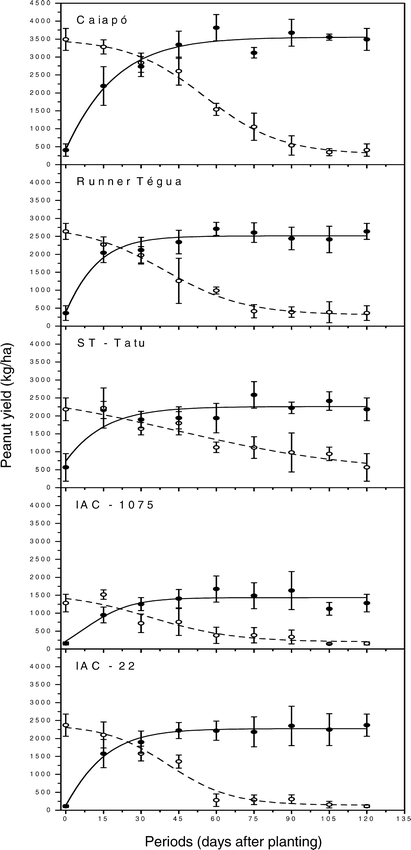
In the absence of weed interference, the most productive cultivar was ‘Caiapó’ (3490 kg/ha), followed by Runner ‘Tégua’ (2640 kg/ha), ‘IAC-22’ (2370 kg/ha), ‘ST-Tatu’ (2180 kg/ha) and ‘IAC-1075’ (1280 kg/ha) (Fig. 6). Weed interference during the whole growing season caused a 88% yield reduction in cultivar ‘Caiapó’, 86% in Runner ‘Tégua’, 92% in ‘IAC-22’, 74% in ‘ST-Tatu’ and 88% in ‘IAC-1075’. Thus, similarly to what was observed for the weight of 100 seeds, the most sensitive cultivar to interference, in relation to yield, was ‘IAC-22’ and the least sensitive ‘ST-Tatu’, both of them with an erect growth habit.
Peanut yield response to increasing length of weed-free (• observed values with standard error, ___ estimated values) or weed-infested (○ observed values with standard error, --- estimated values) periods as calculated from data in table 2.
Previous studies indicated that maximum peanut loss was 63% in Brazil (Bianco, 1978; Pacheco, 1980; Pitelli et al., 1981; Pitelli et al., 1984; Gavioli, 1985; Barbosa and Pitelli, 1990; Martins and Pitelli, 1994; Kasai et al., 1997). For the time of weed removal, the critical periods in the current study were smaller than those previously published. Therefore, in the present study, weeds became to interfere with crop growth earlier in the season, perhaps due to elevated weed population density observed in the beginning of the growing season (Fig. 1). Critical weed-free period in this study were longer than those previously published in Brazil, which could may be attributed to wider row spacing (90 cm), relative to previous studies (<70 cm). The broader spacing impedes the soil shading by the crop, allowing the weeds to germinate for a longer period and, in turn, longer control periods could be needed.
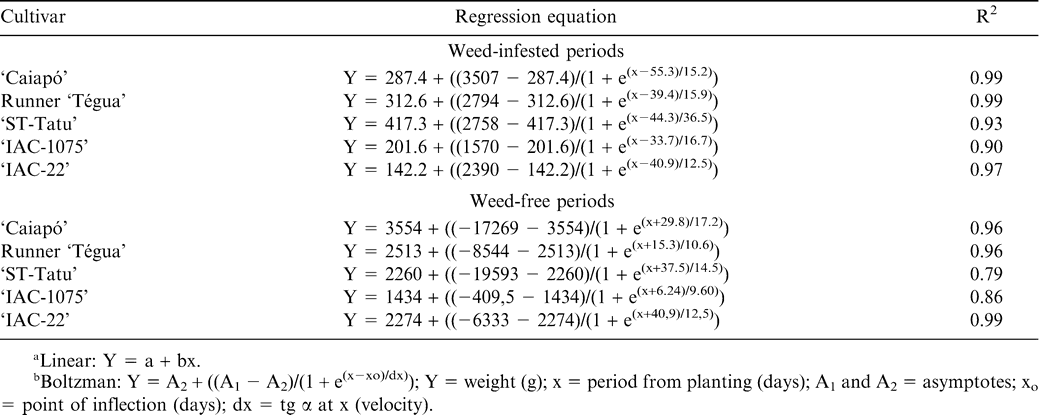
Analyzing the yield of the cultivars as a function of weed-infested or weed-free periods indicated that the Boltzman sigmoidal equation best fit all data (Fig. 6 and Table 2). When the cultivars were maintained weed-infested for increasing periods, there was a more intense yield reduction between 15 and 75 days after planting. As a function of the length of weed-free period with, more intense increase on yield occurred especially when the weed control was done in the first 30 days. Longer control periods did not provide great increases in peanut yield.
Accepting a maximum yield loss of 5% the critical time of weed removal was 13, 7, 12, 16, and 7 days and the critical weed-free period was 47, 65, 31, 26 and 61 days for the cultivars ‘Caiapó’, Runner ‘Tégua’, ‘ST-Tatu’, ‘IAC-1075’ and ‘IAC-22’, respectively (Fig. 6 and Table 3). Consequently, the critical periods of weed control were 13 to 47, 7 to 65, 12 to 31, 16 to 26, and 7 to 61 days for these cultivars, respectively, with no observations of behavior patterns for either growth habit, runner or erect. For the present results the critical period for weed control for all cultivars should begin at 7 DAP and continue until a maximum of 65 DAP.
In the literature review from Brazilian experiments until 2005, no long critical periods of weed control were found. When this period was evaluated, it was determined that only one weed control practice would be enough to ensure high crop yield (Bianco, 1978; Pacheco, 1980; Pitelli et al., 1981; Pitelli et al., 1984; Gavioli, 1985; Barbosa and Pitelli, 1990; Martins and Pitelli, 1994; Kasai et al., 1997). In a comercial area of peanut with Cyperus rotundus, Commelina benghalensis, and Euphorbia heterophylla the productivity was reduced in 76%, with a critical period of weed control of 34 to 90 days after peanut emergency, with a great difficult for mechanical harvest (Nepomuceno et al., 2005). In a weed community composed by Xanthium strumarium, Cenchrus echinatus, Indigofera hirsuta, Portulaca oleraceae, and Digitaria horizontalis, Dias et al. (2005) founded critical periods of weed control of 30 to 88 days after peanut emergency for row spacing of 80 cm and 38 to 101 days for row spacing of 90 cm, with a yield loss more than 80%. All long critical periods of weed control were found outside Brazil. Rodrigues Marquina et al. (1974) determined a critical period of weed control of 30 to 60 days after planting. Drennan and Jennings (1977) determined such a period between six and ten weeks after peanut emergence and Hill and Santelmann (1969) between three and six weeks.
Literature Cited
Barbosa L. M. and Pitelli R. A. 1990 Estudos sobre períodos de interferência de plantas daninhas na produtividade do amendoinzeiro (Arachis hypogaea L.). Hoehnea 17 : 33 – 41 .
Bhan V. M. , Singh M. , and Maurya R. E. 1971 Crop weed competition studies in groundnuts. Indian J. Weed Sci 3 : 32 – 36 .
Bianco S. 1978 Matocompetição em amendoim “das secas” sob diferentes condições de adubação. Agronomy Monograph Jaboticabal, Brasil Faculdade de Ciências Agrárias e Veterinárias, Universidade Estadual Paulista 66 .
Bleasdale J. K. A. 1960 Studies on plant competition. 133 – 142 In Harper J. L. ed. The Biology of Weeds Blackwell Scientific Oxford.
Buchanam G. A. , Hauser E. W. , Ethredge W. J. , and Cecil S. R. 1976 Competition of Florida beggarweed and sicklepod with peanut. II. Effects of cultivation weeds and SADH. Weed Sci 24 : 29 – 39 .
Clark G. L. 1971 Elementos de Ecologia Omega Barcelona 534 .
Cousens R. 1988 Misinterpretations of results in weed research through inappropriate use of statistics. Weed Res 28 : 281 – 289 .
Dajoz R. 1983 Ecologia Geral Vozes, Petrópolis Rio de Janeiro 472 .
Dekker J. and Meggit W. F. 1983 Interference between velvetleaf (Abutilon theophast Medic.) and soybean (Glycine max (L.) Merr.) II. Population dynamics. Weed Res 23 : 103 – 107 .
Dias T. C. S. , Alves P. L. C. A. , Nepomuceno M. , and Souza N. L. 2005 Períodos de interferência das plantas daninhas na cultura do amendoim (IAC 886). In: II Encontro sobre a cultura do amendoim. Jaboticabal, São Paulo, Brasil, 2005. CD
Drennan D. S. H. and Jennings E. A. 1977 Weed competition in irrigated cotton (Gossypium barbadense L.) and groundnut (Arachis hypogaea L.) in the Sudan Gezira. Weed Res 17 : 3 – 9.
Feakin S. D. 1973 Pest Control in Groundnuts. 3th Ed Center for Overseas Pest Research London 197 .
Gavioli V. O. 1985 Efeitos da época e extensão do período de controle das plantas daninhas sobre a cultura do amendoim (Arachis hypogaea L.) em duas épocas de semeadura. Agronomy Monograph Faculdade de Ciências Agrárias e Veterinárias, Universidade Estadual Paulista Jaboticabal, Brasil 62 .
Hauser E. , Buchanan G. A. , and Ethredge W. J. 1975 Competition of Florida beggarweed and sicklepod with peanuts. I. Effects of periods of weed-free maintenance or weed competition. Weed Sci 23 : 368 – 372 .
Hill L. V. and Santelmann P. W. 1969 Competitive effects of annual weeds on Spanish peanuts. Weed Sci 17 : 01 – 02 .
Ishag H. M. 1971 Weed control in irrigated groundnuts (Arachis hypodaea L.) in the Sudan Gezira. J. Agr. Sci 77 : 237 – 242 .
Kasai F. S. , Paulo E. M. , Cavichioli J. C. , Peressin V. A. , and Igue T. 1997 Efeitos dos períodos de competição do mato na cultura do amendoim: I. Safra da seca de 1988. Bragantia 56 : 323 – 331 .
Kropff M. J. , Lotz L. A. , and Weaver S. E. Kropff M. J. and van Laar H. H. 1993 Practical applications. 149- 169 In Modelling Crop-Weed Interactions, CAB International in association with the The International Rice Research Institute.
Kuva M. A. , Pitelli R. A. , Christoffoleti P. J. , and Alves P. L. C. A. 2000 Períodos de interferência das plantas daninhas na cultura da cana-de-açúcar. I. Tiririca. Planta Daninha 18 : 241 – 252 .
Martins D. and Pitelli R. A. 1994 Interferência das plantas daninhas na cultura do amendoim das águas: efeitos de espaçamentos, variedades e períodos de convivência. Planta Daninha 12 : 87 – 92 .
Mulugeta D. and Boerboom C. M. 2000 Critical time of weed removal in glyphosate-resistent Glycine max. Weed Sci 48 : 35 – 42 .
Nepomuceno M. , Dias T. C. S. , A Alves P. L. C. , and Luvezuti R. A. 2005 Interferência das plantas daninhas na cultura do amendoim rasteiro. In: II Encontro sobre a cultura do amendoim Jaboticabal São Paulo, Brasil 2005. CD rom.
Nieto J. H. , Brondo M. A. , and Gonzalez J. T. 1968 Critical periods of the crop grwth cycle for competition from weeds. Pans (c) 14 : 159 – 166 .
Pacheco R. P. B. 1980 Duração do período de competição de plantas invasoras com a cultura do amendoim-da-seca (Arachis hypogaea L.). Vegetalia 3 : 1 – 11 .
Pitelli R. A. 1980 Efeitos do período de competição das plantas daninhas sobre a produtividade do amendoim (Arachis hypogaea L.) e o teor de macronutrientes em suas sementes. M. Sc. Thesis Escola Superior de Agricultura “Luiz de Queiroz”, Universidade de São Paulo Piracicaba, Brasil 89 .
Pitelli R. A. 1985 Interferência das plantas daninhas em culturas agrícolas. Informe Agropecuário 11 : 16 – 27 .
Pitelli R. A. , Ferraz E. C. , and de Marinis G. 1981 Efeito do período de matocompetição sobre a produtividade do amendoim (Arachis hypogaea L.). Planta Daninha 4 : 110 – 119 .
Pitelli R. A. , Peressim V. A. , Pansani L. C. , and Perecin D. 1984 Efeitos de períodos de convivência das plantas daninhas sobre a produtividade da cultura do amendoim das secas. Planta Daninha 7 : 58 – 64 .
Rodrigues Marquina E. L. , Hemsy V. , Andaliaris J. S. , Muños H. L. L. , and Cajaly J. A. 1974 Período crítico de competencia entre las malezas y el cultivo del mani (Arachis hypogeae L.). Rev. Agron. Noroeste Arg 11 : 137 – 143 .
van Acker R. C. , Swanton C. J. , and Weise S. F. 1993 The critical period of weed control in soybean [Glycine max (L.) Merr.]. Weed Sci 41 : 194 – 200 .
York A. C. and Coble H. D. 1977 Fall panicum interference in peanut. Weed Sci 25 : 43 – 47 .
Notes
- 1 Dept. of Agriculture Applied Biology, College of Agronomy and Veterinary Sciences, University of São Paulo State, Access road Prof. Paulo D. Castellane, s/n, 14884-900, Jaboticabal, SP, Brazil [^] *Corresponding Author (email: renangravena@asbyte.com.br)