Introduction
Simple sequence repeat (SSR) markers are a valuable tool in genetic mapping, genotyping, and marker-assisted selection in breeding due to their characterization of co-dominant loci, high allelic variation, even distribution, and be easily used by PCR. In peanut, SSR markers have been developed by several groups (Hopkins et al., 1999; He et al., 2003; Ferguson et al., 2004; Luo et al., 2005a), however, they are still not fully exploited and developed compared to other crops. Peanut genome research has made less progress than other legumes because of insufficient genomic tools available (Luo et al., 2005a, 2005b). Thus, one of the pressing needs in peanut genomic research is to take advantage of progress made in the well-characterized legumes. Searching for transferable SSR marker developed from model legumes, such as soybean, Medicago truncatula, and Lotus japonicus is a cost-effective way to increase DNA markers for peanut genomic studies and genetic linkage map development.
Many studies have shown that RFLP and SSR markers were transferable among cereal crops (Saghai Maroof et al., 1996; Devos et al., 1998; Zhang et al., 1998; Hernandez et al., 2001; Babula et al., 2003; Kuleung et al., 2004; La Rota and Sorrells 2004; Spielmeyer et al., 2004), and legume crops (Boutin et al., 1995; Humphry et al., 2002; Isobe et al., 2003). The transferable markers are developed from the conserved genomic regions among related species. Transferability of DNA markers between the genomes of different species not only provides researchers with large pools of available markers, but also allows us to better understand the evolution and speciation of crops through comparative mapping. However, the transferability of DNA markers from other crops to peanut is unknown. Therefore, the objective of this study is to examine the transferability of soybean SSR markers to cultivated peanut.
Materials and Methods
Plant Material
Two peanut cultivars (GK7 and COAN) and two breeding lines (C11-2-39 and 448A) were used in this study to test the transferability of soybean SSR markers in peanut. These four peanut lines were selected because they are used as parental lines in current peanut breeding programs and mapping population development. The cultivar, COAN, was developed by introgression from A. cardenasii, A. duranensis and A. batizocoi to cultivated peanut (Simpson and Starr, 2001).
Dna Extraction
DNA was extracted from young leaf tissue using the CTAB method of Murray and Thompson (1980) with some modifications. Leaves (3–5 g) were ground in liquid nitrogen and suspended in a buffer containing 2% CTAB, 30 mM Tris-HCl (pH 8.0), 10 mM EDTA, and 0.1 M NaCl. One-third volume of 5 M potassium acetate was added, and the supernatant was extracted twice with phenol and once with chloroform. Nucleic acid was precipitated with two volumes of ethanol and suspended in 0.5 ml TE (10 mM Tris-HCl, pH 8.0, and 1 mM EDTA) containing 5 µg/ml RNaseA (Sigma). This mixture was then extracted twice with phenol:chloroform (1:1) and precipitated with two volume of ethanol, dissolved in 0.2 ml of TE, and diluted to 50 ng/µl for PCR amplification.
Pcr Amplification
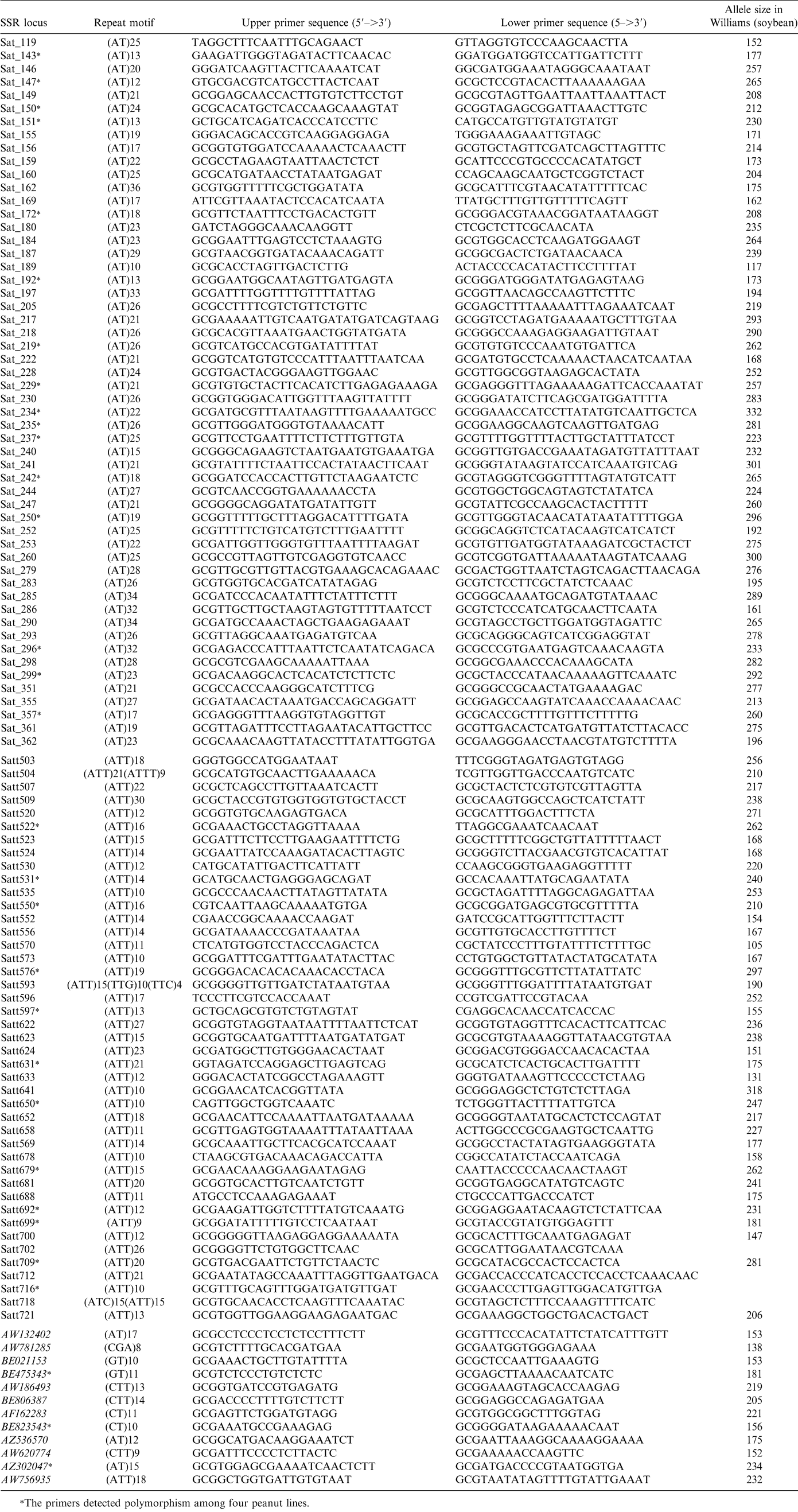
Soybean SSR primer pairs were provided by Drs. Cregan and Song at the USDA-ARS, Beltsville, MD. A total of 432 soybean SSR primer pairs including 200 (AT)n motifs (named as Sat #), 197 (ATT)n motifs (named as Satt #), and 35 EST-SSR (named as GenBank accession #) primer pairs were used. PCR amplification was carried out in 10 µl solution containing 0.25 units of AmpliTaq polymerase (Applied Biosystems, CA), 1.5 µM of each primer, 5 µl of FailSafe PCR 2X PreMix-B (Epicentre, WI), and 50 ng peanut genomic DNA. Amplification was carried out under the following conditions: 3 min at 94 C for initial denaturation; two cycles of 30 sec at 94 C, 30 sec at 65 C, and 1 min at 72 C; two cycles of 30 sec at 94 C, 30 sec at 56 C, and 1 min at 72 C; 30 cycles of 15 sec at 94 C, 30 sec at 55 C, and 1 min at 72 C; and 10 min at 72 C for final extension (Mellersh and Sampson, 1993). PCR products were analyzed on a 6% denaturing polyacrylamide gel and visualized by silver staining (He and Prakash, 1997).
Results and Discussion
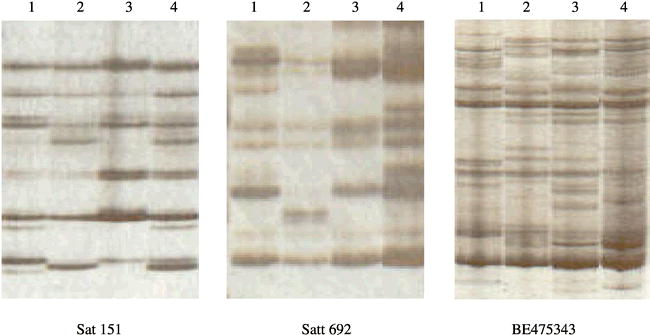
Out of 432 soybean SSR primer pairs tested, 109 (25%) were amplifiable in peanut (Table 1). Among 109 soybean SSR markers, 28% detected polymorphism among 4 cultivated peanut lines (Fig. 1). The polymorphism detection rate in this study is higher than the previous report (He et al. 2003). This difference may be due to the fact that cultivar ‘COAN’ contains wild species segments of chromosomes, from which additional genetic variation could be detected. For 54 (AT)n markers, 30% detected genetic variation, while 28% of 43(ATT)n markers detected a polymorphism. The transferability of EST-SSR in peanut was higher (34%) than genomic SSR markers, but only 25% of EST-SSR markers could detect polymorphism (Table 2). The result of more transferable markers from the coding regions (EST-SSR) was consistent with the previous study (Liewlaksaneeyanawin et al., 2003) because coding regions are more conserved between related species.
These amplifiable markers implied that 25% of primer-binding sites were conserved between soybean and peanut. However, most banding patterns amplified by these transferable markers were similar to multiple-band patterns produced by random amplified polymorphic DNAs (RAPDs) rather than typical SSR banding patterns (Fig. 1). Choumane et al. (2004) reported that 54.4% of chickpea SSR primer-binding sites were conserved among the three genera, chickpea, dry pea, and lentil. They also found that SSR motifs were present in chickpea, but absent either in dry pea or lentil, after sequencing the amplicons produced by the same chickpea SSR primer. In this study, we speculate that amplicons from soybean SSR primers that produced complex patterns may not contain SSR motifs. This speculation needs to be confirmed by sequencing these amplicons. Nevertheless, these transferable SSR markers derived from soybean could be used to detect genetic variation in peanut.
Among legume crops, there is a high level of conservation between cowpea and mungbean, mungbean and common bean, and mungbean and lablab genomes (Menancio-Hautea et al., 1993; Boutin et al., 1995; Humphry et al. 2002). In contrast, the homology between soybean and common bean was retained only in dispersed blocks throughout their genomes (Boutin et al., 1995). Although the comparative mapping has been progressed in legume crops, the comparison of peanut genome with other legumes could not be carried out due to the lack of a set of common DNA markers. The level of transferability observed in this study would provide genome tools for comparative mapping between peanut and soybean because there are a large number of soybean SSR markers available. The comparative mapping between peanut and soybean will allow us to gain deeper insight into the degree of chromosome colinearity between them, and to elucidate the biological relationship among legume crops.
In our previous study, we have searched for SSR markers in 1350 peanut ESTs. Three hundred fifty-three ESTs were found to contain SSRs. Primers were designed for 44 EST-SSRs and 9 of them detected polymorphism, for a polymorphism rate of 20% in coding regions (Lou et al. 2005a). This study shows that 25% of transferable soybean EST-SSR markers detected a polymorphism in peanut genome. The results from both studies may suggest that there might be an abundance of mutations in coding regions in peanut. Using EST-SSR markers in comparative mapping between peanut and soybean will allow us to predict the location of genes of interest from soybean which has a well developed genetic map compared to peanut for which such information is scarce.
Peanut and most legume crops belong to the same subfamily Papilionoideae of the Leguminosae family. However, peanut is isolated in a different clade (dalbergioid) from most legume crops in the legume phylogenetic tree (Doyle and Luckow, 2003). There is a relatively under-developed infrastructure of genomic tools in peanut. Therefore, in peanut, there is a great need to integrate the knowledge gained from the study of model legume genomes with the important biological and agronomic questions of peanut. Identification of transferable DNA markers from model legumes in peanut would be useful for peanut improvement through comparative genome research because the information of these transferable markers from other legumes is known. The common transferable DNA markers among legumes would also provide us tools to compare legume genomes, gain insight into relationship of legumes, and elucidate legume evolution.
Acknowledgements
The authors thank the support from the George Washington Carver Agricultural Experiment Station in Tuskegee University. The authors also express gratitude to Drs. Cregan and Song for providing soybean SSR markers. This work has been partially funded by a grant from USDA/CSREES/CBG (No. 00-38814-9541) and funds provided by National Peanut Foundation and Georgia Peanut Commission. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply approval to the exclusion of other products that may be suitable.
Literature Cited
Babula D. , Kaczmarek M. , Barakat A. , Delseny M. , Quiros C. F. , and Sadowski J. 2003 Chromosomal mapping of Brassica oleracea based on ESTs from Arabidopsis thaliana: complexity of the comparative map. Mol Gen Genomics 268 : 656 – 665 .
Boutin S. R. , Young N. D. , Olson T. C. , Yu Z. H. , Shoemaker R. C. , and Vallejos C. E. 1995 Genome conservation among three legume genera detected with DNA markers. Genome 38 : 928 – 937 .
Choumane W. , Winter P. , Baum M. , and Kahl G. 2004 Conservation of microsatellite flanking sequences in different taxa of Leguminosae. Euphytica 138 : 239 – 245 .
Devos K. M. , Wang Z. M. , Beales J. , Sasaki T. , and Gale M. D. 1998 Comparative genetic maps of foxtail millet (Setaia italica) and (Oryza sativa). Theor Appl Genet 96 : 63 – 68 .
Doyle J. J. and Luckow M. A. 2003 The rest of the iceberg. Legume diversity and evolution in a phylogenetic context. Plant Physiology 131 : 900 – 910 .
Ferguson M. E. , Burow M. D. , Schulze S. R. , Bramel P. J. , Paterson A. H. , Kresovich S. , and Mitchell S. 2004 Microsatellite identification and characterization in peanut (A. hypogaea L.). Theor Appl Genet 108 : 1064 – 1070 .
He G. H. and Prakash C. S. 1997 Identification of polymorphic DNA markers in cultivated peanut (Arachis hypogaea L.). Euphytica 97 : 143 – 149 .
He G. H. , Meng R. H. , Newman M. , Gao G. Q. , Pittman R. N. , and Prakash C. S. 2003 Microsatellites as DNA markers in cultivated peanut (Arachis hypogaea L.). BMC Plant Biology 3 : 3 .
Hernandez P. , Dorado G. , Prieto P. , Gimenez M. J. , Ramirez M. C. , Laurie D. A. , Snape J. W. , and Martin A. 2001 A core genetic map of Hordeum chilense and comparisons with maps of barley (Hordeum vulgare) and wheat (Triticum aestivum). Theor Appl Genet 102 : 1259 – 1264 .
Hopkins M. , Casa A. , Wang T. , Mitchell S. , Dean R. , Kochert G. , and Kresovich S. 1999 Discovery and characterization of polymorphic simple sequence repeats (SSRs) in peanut. Crop Sci 39 : 1243 – 1247 .
Humphry M. E. , Konduri V. , Lambrides C. J. , Magner T. , McIntyre C. L. , Aitken E. A. B. , and Liu C. J. 2002 Development of a mungbean (Vigna radiata) RFLP linkage map and its comparison with lablab (Lablab purpireus) reveals a high level of colinearity between the two genomes. Theor Appl Genet 105 : 160 – 166 .
Isobe S. , Klimenko I. , Ivashuta S. , Gau M. , and Kozlov N. N. 2003 First RFLP linkage map of red clover (Trifolium pratense L.) based on cDNA probes and its transferability to other red clover germplasm. Theor Appl Genet 108 : 105 – 112 .
Kuleung C. , Baenziger P. S. , and Dweikat I. 2004 Transferability of SSR markers among wheat, rye, and triticale. Theor Appl Genet 108 : 1147 – 1150 .
La Rota , Mauricio , and Sorrells M. E. 2004 Comparative DNA sequence analysis of mapped wheat ESTs reveals the complexity of genome relationships between rice and wheat. Funct Integr Genomics 4 : 34 – 46 .
Liewlaksaneeyanawin , Cherdsak , Ritland C. E. , El-Kassaby Y. A. , and Ritland K. 2003 Single-copy, species-transferable microsatellite markers developed from loblolly pine ESTs. Theor Appl Genet 109 : 361 – 369 .
Luo M. , Dang P. , Guo B. Z. , He G. , Holbrook C. C. , Bausher M. G. , and Lee R. D. 2005a Generation of expressed sequence tags (ESTs) for gene discovery and marker development in cultivated peanut. Crop Sci 45 : 346 – 353 .
Luo M. , Dang P. , Holbrook C. C. , Bausher M. G. , Lee R. D. , Lynch R. E. , and Guo B. Z. 2005b Identification of transcripts involved in resistance responses to leaf spot disease Caused by Cercosporidium personatum in peanut (Arachis hypogaea). Phytopath 95 : 381 – 387 .
Mellersh C. and Sampson J. 1993 Simplifying detection of microsatellite length polymorphisms. BioTechniques 15 : 582 – 584 .
Menancio-Hautea D. , Fatokun C. A. , Kumar L. , Danush D. , and Young N. D. 1993 Comparative genome analysis of mungbean (Vigna radiata L. Wilczek) and cowpea (Vigna unguiculata L. Walpers) using RFLP mapping data. Theor Appl Genet 86 : 797 – 810 .
Murray M. G. and Thompson W. F. 1980 Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8 : 4321 – 4326 .
Saghai Maroof M. A. , Yang G. P. , Biyashev R. M. , Maughan P. J. , and Zhang Q. 1996 Analysis of the barley and rice genomes by comparative RFLP linkage mapping. Theor Appl Genet 92 : 541 – 551 .
Simpson C. E. and Starr J. L. 2001 Registration of ‘COAN’. Crop Sci 41 : 118 .
Spielmeyer W. , Ellis M. , Robertson M. , Ali S. , Lenton J. R. , and Chandle P. M. 2004 Isolation of gibberellin metabolic pathway genes from barley and comparative mapping in barley, wheat and rice. Theor Appl Genet 109 / 4 : 847 – 855 .
Zhang H. , Jia J. , Gale M. D. , and Devos K. M. 1998 Relationships between the chromosomes of Aegilops umbellulata and wheat. Theor Appl Genet 96 : 69 – 75 .