Introduction
Peanut (Arachis hypogea L.) is one of the main cash crops in Ghana, West Africa and is grown primarily by resource-poor, smallholder farmers. Confectionary nuts make up the bulk of production and consumption in Ghana, similar to their use in Zambia (Sandhu et al., 1986). Peanut productivity in West Africa is limited by a number of biotic and abiotic constraints including soil arthropods, foliar and soil borne diseases, peanut rosette virus (Peanut rosette umbravirus), aflatoxin (Aspergilus flavus) contamination, interference from weeds, and drought (Bowen and Mack, 1993; Johnson and Gumel, 1981; Johnson et al., 1981; Lynch et al., 1989; Lynch and Douce, 1992; Wightman and Ranga Rao, 1993; Wightman and Wightman, 1994; Wightman et al., 1990; Umeh et al., 2001).
Arthropods infesting peanut and consistently reducing yield in sub-Saharan Africa include termites, white grubs, millipedes, and nematodes (Rhabditida: Steinermatidae and Heterorhabditidae) (Feakin, 1973; Johnson et al., 1981; Raheja, 1975; Sohati and Sithanathan, 1990; Tsigbey et al., 2003). Other pests such as wireworms (Elateridae), doryline ants (Hymenoptera: Formicidae), earwigs (Dermaptera: Forficulidae), and false wireworms (Tenebrionidae) are reported to be occasionally damaging in peanut (Wightman and Ranga Rao, 1993). Termites include Macrotermes spp., Odontotermes spp. and Microtermes spp. The above pests damage peanut pods and seeds. White grubs, symphilids (Myriapoda: Symphyla), termites, and wireworms destroy the roots, while termites cause pod scarification and eat the haulm (Wightman et al., 1990; Wightman and Ranga Rao, 1993). Johnson et al. (1981) reported a yield loss of 40% caused by Microtermes spp. to peanut in Nigeria. Damage to the pods can also result in invasion of the seeds by soil fungi. Infection by fungi can lead to reduction in quality, caused by internal decay, deterioration of the seed and the production of mycotoxins, especially aflatoxins (Johnson and Gumel, 1981; Martin and Gilman; 1976; McDonald, 1970; Lynch et al., 1989; Perry, 1967; Porter et al., 1972; Wilson, 1945).
Rosette and Cercospora leaf spot (Cercospora arachidicola and Cercospora personatum) and interference from weeds are major limitations to optimum peanut production in Ghana. Cultural practices including improper application of manure, leaving crop residues on farms, delaying planting and harvesting, and abiotic factors such as rainfall and soil texture also influence soil pest occurrence and damage. Despite pest concerns, peanut farmers in most areas of sub-Saharan Africa seldom apply effective control measures against soil pests (Johnson et al., 1981).
Research indicates that carbofuran and chlorpyrifos control a wide range of soil arthropods without damaging peanut (Wightman et al., 1990; Wightman and Ranga Rao, 1993). Numerous studies have documented yield increases following application of insecticides in peanut in Africa (Wightman et al., 1990; Wightman and Ranga Rao, 1993). Although synthetic insecticides are used routinely in production systems for peanut in developed countries, they are not routinely applied in countries like Ghana where the availability of commercial products is limited and financial constraints prevent purchasing these materials. However, the cost effectiveness of synthetic insecticides has not been adequately addressed in Ghana for peanut and empirical data on the actual losses caused by soil arthropods are limited in Ghana. The objectives of this study were to identify soil arthropods associated with peanut production in southern Ghana, to quantify yield loss due to these pests, and to determine the interaction of application timing of insecticides and cultivar in order to develop strategies for more effective peanut production in West Africa.
Materials and Methods
Survey of Farmer Fields for Soil Arthropods
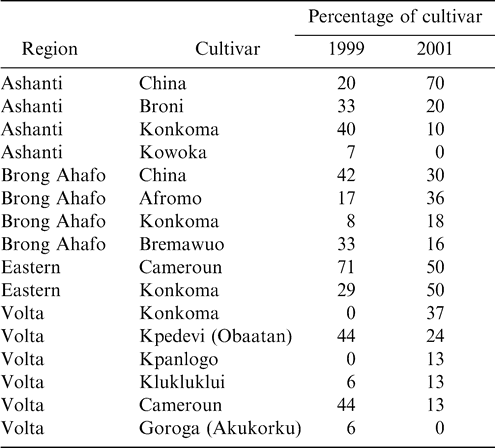
Fields in the peanut production districts of Ashanti, Brong Ahafo, Eastern, and Volta regions in Ghana were visited during the major growing seasons during 1999 and 2001. Ten (1999) and eight (2001) villages in each region were visited and two fields per village were sampled (Table 1). The percentage of each cultivar grown in that area was determined. In each field, a 5 m × 5 m section was marked and five plants were randomly selected from two, 5 m by 5 m sections selected at random to assess presence of insect-feeding holes on pods and seeds. Sampling during 1999 occurred in late July in Ashanti and Brong Ahafo regions and in late August in Eastern and Volta regions. During 2001, fields were sampled in late August in Ashanti and Grong Ahafo regions and in mid-September in Eastern and Volta regions. Additionally, within each section five soil cores (15 cm3) were removed to quantify soil arthropods. The percentage of unfilled pods was also determined from the five plants sampled.
Arthropod density was transformed to log10 prior to statistical analysis (Sokal and Rohlf, 1981). Data for counts of each arthropod, percentage unfilled pods, and damaged pods and kernels were analyzed using a general linear model procedure (SAS Institute, 1999–2000). When ANOVAs (analysis of variance) were significant (P ≤ 0.05) treatment means were compared against control by Tukey's (HSD) test.
Efficacy of Carbofuran and Chlorpyrifos Against Soil Arthropods
Experiments were conducted at Kwadaso, Kumasi (Lat, 6° 42 N; Long, 1° 40 W; 262 m above sea level) and at Hiawoannwu/Ejura (Lat, 7° 40 N; Long, 1° 39 W; 221.9 m above sea level) in the Ashanti region of Ghana to determine the effect of soil pesticides for arthropod control and peanut yield. Seedbeds were prepared by hand tillage and peanut was seeded in rows spaced 50 cm apart (Kwadaso) and 15 cm apart (Ejura). Plot length was 6 m and consisted of eight rows. Air temperature ranged from 29–32 C and 31–34 C with relative humidity of 70–80% and 50–65% for Kwadaso and Ejura, respectively. One improved peanut cultivar, ICG FDRS-20 × F-MIX-38 and a local cultivar, Konkoma, were planted in these experiments. Chlorpyrifos (Dursban 4E, Dow AgroSciences, Indianapolis, IN) was applied at planting as side dressing at 0.68 kg ai/ha. Similarly, carbofuran (Furadan 3G, FMC Corp., Philadelphia, PA) was applied in furrows at 1.0 kg ai/ha. The side dressing application was done in shallow furrows 20 mm deep and 0.10 m from the plant row. The furrows were covered with soil after application. A non-treated control for each cultivar was included. Treatments consisted of carbofuran or chlorpyrifos applied at planting or at planting followed by a repeat application 60 days after planting (DAP) at the same rate. Peanut was planted at Kwadaso on June 3, 2003 and on May 21, 2004 at Hiawoannwu and Ejura.
The experimental design was a randomized complete block with a 2 × 2 × 2 factorial treatment arrangement with three replications. The day before harvest, soil from a 15 cm × 15 cm × 15 cm section each was removed from two spots in each plot to determine arthropod density for each species. Damaged pods and kernels from two peanut plants removed from each plot were recorded. Pod weight from plants within one m of the two middle rows of each plot was recorded.
Data for total arthropod density, density of each arthropod group, damaged pods, damaged kernels, unfilled pods, and pod yield were subjected to analyses of variance (SAS Institute, 1999–2000). Means were separated using Tukey's (HSD) test at p ≤ 0.05. Arthropod density was transformed (log10) before analyses (Sokal and Rohlf, 1981).
Results and Discussion
Survey of Farmer Fields for Soil Arthropods
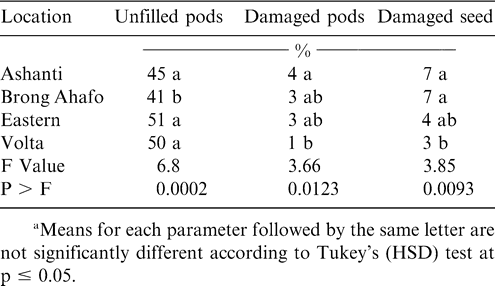
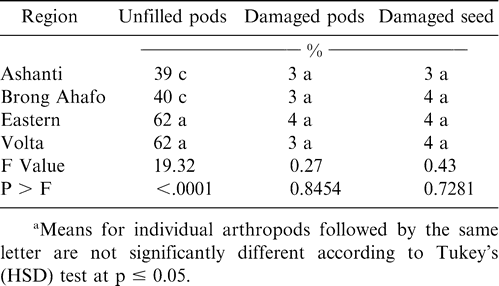
The peanut cultivar Konkoma was planted in all regions during 2001 and in three regions during 1999 (Table 1). Although the percentage of Konkoma decreased from 1999 to 2001 in Ashanti region, the percentage of this cultivar increased in Brong Ahafo, Eastern, and Volta regions. Konkoma was cultivated across the regions because it has been a standard cultivar for decades.
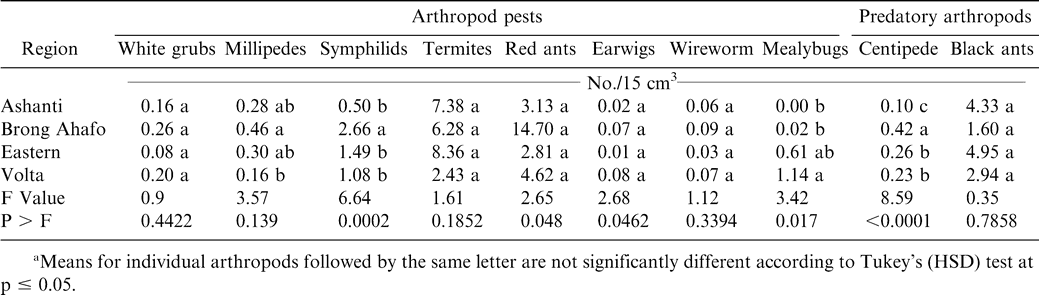
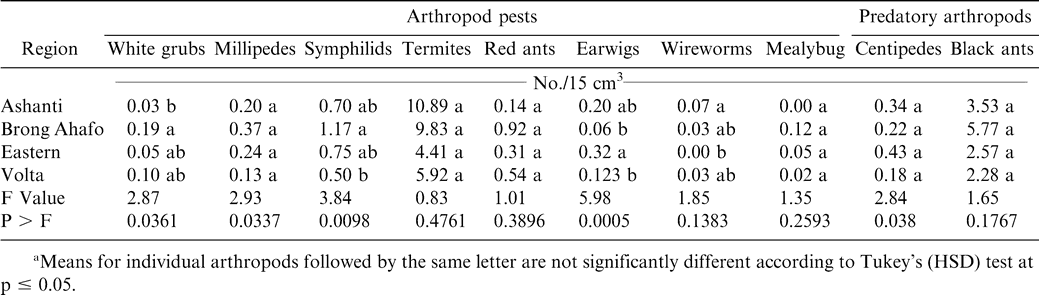
Soil arthropod pests observed in all the regions during both years included white grubs, millipedes, symphilids, termites, red ants, earwigs, wireworms, and mealybugs while predators included black ants and centipedes (Tables 2 and 3). Most of these damage-causing organisms have been previously reported by other researchers in peanut fields (Bowen and Mack, 1993; Feakin, 1973; Johnson et al., 1981; Lynch and Douce, 1992; Raheja, 1975; Sohati and Sithanatham, 1990; Tsigbey et al., 2003; Wightman et al., 1990; Wightman and Ranga Rao, 1993; Wightman and Wightman, 1994; Umeh et al., 2001). Termites were observed in all the regions during 1999 and 2001 (Tables 3 and 4). Although red ants were not present in 2001, they were present during 1999, and higher populations were noted in particular in the Brong Ahafo region (Tables 3 and 4). Symphilids were present during both years but at lower populations than termites or red ants in 1999 or termites in 2001 (Tables 2 and 3). Populations of centipedes were low compared to those of black ants. Low numbers of some of the arthropods may be attributable to the high populations of the predatory black ants (Table 3 and 4), which may suppress the populations of other arthropods. Sekamatte et al. (2000) and Erbaugh et al. (2003) also indicated that an increase in ant population and activity, through baiting, significantly reduced termite damage in corn (Zea mays L.).
The percentage of unfilled pods during 1999 and 2001 ranged from 41 to 62% (Tables 4 and 5). Percentages of insect-damaged pods and kernels were relatively low across all regions during both years. Damage may have resulted from low calcium in soil solution and the inconsistent maturity of the crop. Calcium is critical for adequate pod development and optimum yield (Barber, 1984; Smal et al., 1989; Summer et al., 1988). Nevertheless, the feeding holes of these arthropods could constitute routes of infection by aflatoxin-producing fungi, contaminating and reducing quality of kernels (Bowen and Mack, 1993; McDonald and Harkness, 1963).
Efficacy of Carbofuran and Chlorpyrifos Against Soil Arthropods
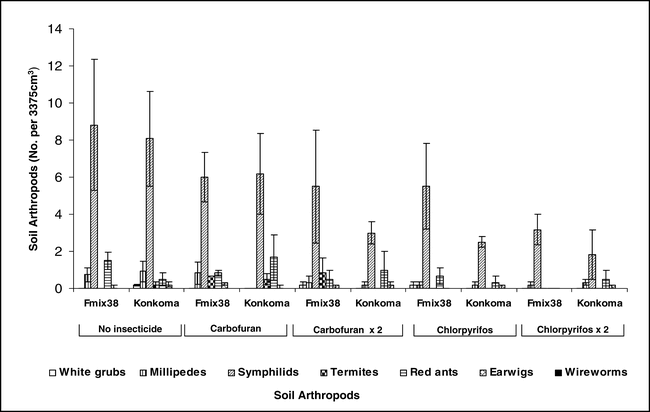
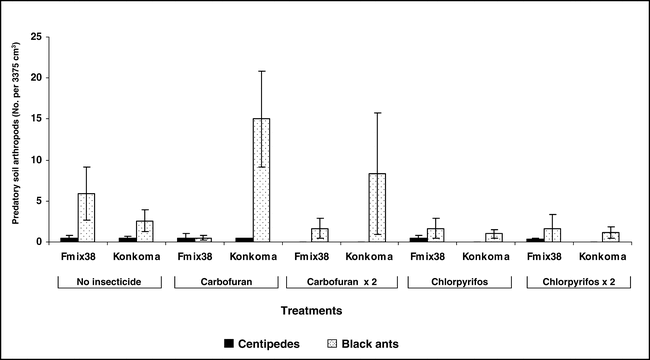
Populations of white grubs, millipedes, termites, red ants, earwigs, and wireworms were relatively low and few differences were noted when comparing among insecticide treatments during 2003 at Kwadaso (Figure 1). High populations of symphilids were noted, and chlorpyrifos usually demonstrated a trend to reduce populations of this soil arthropod. Carbofuran was generally less effective than chlorpyrifos and while trends were consistent, significant differences among treatments were not common. There was no advantage to applying chlorpyrifos both at planting and 60 days after planting compared with a single application at planting (Figure 1). During 2004 at Ejura, no differences in populations of white grubs, millipedes, symphilids, termites, earwigs, and wireworms were noted (data not presented). Although relatively high populations of termites were noted at this location, no treatment differences were observed when comparing non-treated peanut with either insecticide treatment (data not presented).
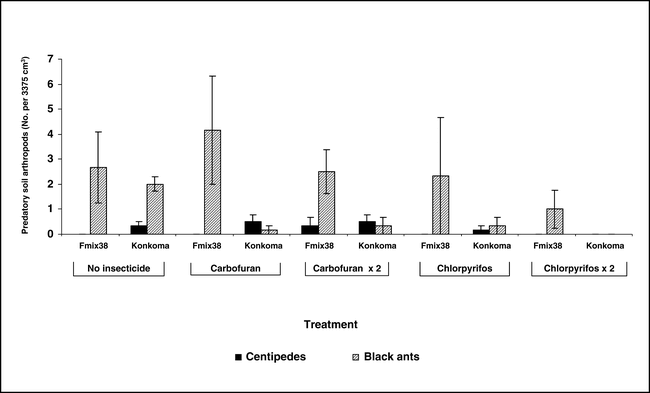
Populations of centipedes, a predatory arthropod, were low regardless of whether or not insecticide was applied at Kwadaso (Figure 2) and Ejura (data not presented). In contrast, high populations of black ants were noted for some but not all treatments. Surprisingly, the highest population was noted when carbofuran was applied at planting to the cultivar Konkoma. Black ant population was consistently low when chlorpyrifos was applied at planting only or when applied at planting and repeated 60 days after planting. Although considerable variation was noted for black ant populations at Ejura during 2004, fewer black ants were associated with the cultivar Konkoma (Figure 3).
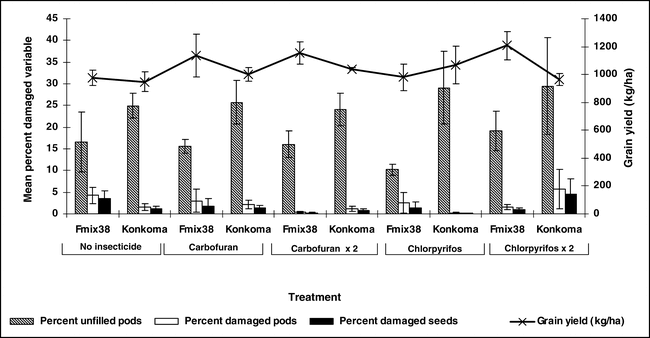
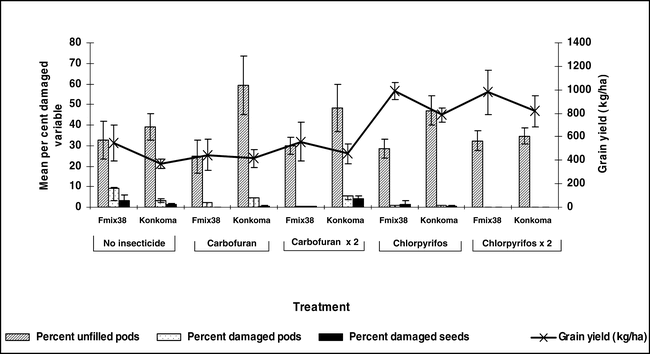
The percentage of unfilled pods ranged from 7 to 25% at Kwadaso during 2003 (Figure 4). Generally, a higher percentage of unfilled pods was noted for the cultivar Konkoma compared with FDRS-20 × F-MIX-38. However, no differences in the percentage of unfilled pods were noted when comparing insecticide treatments within a cultivar. Damage to pods and kernels from arthropods was less than 5% and no differences among insecticide treatments and cultivars were noted (Figure 4). A similar trend was noted at Ejura during 2004 where considerable variation in the percentage of unfilled pods was noted and few differences in percentages of damaged pods and kernels were noted (Figure 5). Limited pod fill is often associated with low calcium levels in soil (Barber, 1984, Summer et al., 1988 and Smal et al., 1989) and this was the case at both locations (S. Osei-Yeboah et al., unpublished data). These data also indicate that FDRS-20 × F-MIX-38 may be better suited for the region, at least with respect to pod fill. Additionally research comparing cultivar response to calcium is needed to determine the cause of differences noted in these experiments. Peanut was harvested at optimum maturity for each cultivar.
Pod yield was not affected by cultivar or insecticide treatment at Kwadaso during 2003 (Figure 4). Applying chlorpyrifos either at planting only or at planting followed by application 60 days after planting produced a trend for higher yield compared with non-treated peanut and peanut treated with carbofuran at Ejura in 2004. Yield of FDRS-20 × F-MIX-38 with one or two applications of chlorpyrifos was higher yield than the non-treated control or carbofuran treated FDRS-20 × F-MIX-38. When Konkoma was treated with chlorpyrifos in a similar fashion, yield was higher than yield of Konkoma treated with carbofuran or when Konkoma was not treated with insecticide. The yield increase associated with application of chlopyrifos did not appear to be associated with insect populations and subsequent damage to pods and kernels. However, chlorpyrifos has been shown to reduce incidence of southern stem rot (caused by Sclerotium rolfsii Sacc.) (Melouk et al., 1995).
Collectively, results from this survey indicate that a wide range of soil arthropods are present in fields associated with peanut production in southern Ghana. Despite low arthropod pest damage to pods, yield increased when chlorpyrifos was applied in one (Ejura) of the two locations but not when carbofuran was applied. This may be due to non-insecticidal effects including suppression of disease. While soil arthropods did not appear to play a significant role in yield reductions in the two locations evaluated in this study, the wide array of species and consistent occurrence would indicate that under certain environmental conditions, one or more species might reach significant pest status. This would be particularly true under hot and dry conditions for termites. Additional research is needed to compare peanut response to insecticides and document the role of these products in suppressing pests other than arthropods. Defining relationships among pest management practices and other agronomic inputs related to cultivar is needed to develop comprehensive production systems for peanut production in southern Ghana.
Acknowledgements
J. Ohene-Mensah, Adama Amadu, Augustine Agyekum, Michael Sarfo, and E. Asiedu provided technical assistance. This study was supported by funds administered through USAID Peanut CRSP (LAG –G-00-96-90013-00).
Literature Cited
Barber S. A. 1984 Soil nutrient bio-availability: A mechanistic approach John Wiley and Sons New York, NY .
Bowen K. L. and Mack T. P. 1993 Relationship of damage from the lesser cornstalk borer to Aspergillus flavus contamination peanuts. J. Entomol. Sci 28 : 1 .
Erbaugh J. M. , Kyamanywa S. , and Bigirwa G. 2003 Overview of the African site in Uganda, Eight Annual Report.
Feakin S. D. 1973 Pest control in peanuts. 3rd Edition PANS Manual No. 2 London, UK Center for Overseas Pest Research, London 197 .
Johnson R. A. and Gumel M. H. 1981 Termite damage and crop loss studies in Nigeria- the incidence of termite–scarified peanut pods and resulting kernel contamination in field and market samples. Tropical Pest Management 27 / 3 : 343 – 350 .
Johnson R. A. , Lamb R. W. , and Wood T. G. 1981 Termite damage and crop loss studies in Nigeria- a survey of damage to peanuts. Tropical Pest Management 27 / 3 : 325 – 342 .
Lynch R. E. and Douce G. K. 1992 Implementation of integrated pest management in peanut; current status and future direction. 267 – 278 In Nigam S. N. ed Peanut – a global perspective: proceedings of an international workshop, 22–29 Nov. 1991, ICRISAT Center, India. (Nigam, S. N. ed).
Lynch R. E. , Wilson D. M. , Ouedraogo A. P. , and Some S. A. 1989 Interrelationship between soil insect damage to peanut pods and aflatoxin in kernels. Proc. Am. Peanut Res. Educ. Soc 21 : 60 .
Martin P. M. D. and Gilman G. A. 1976 A consideration of the mycotoxin hypothesis with special reference to the mycoflora of maize, sorghum, wheat and peanuts Report Tropical Products Institute G105.
McDonald D. 1970 Fungal infection of peanut fruit before harvest. Transactions British Mycological Society 54 / 3 : 453 – 460 .
McDonald D. and Harkness C. 1963 Growth of Aspergllus flavus and production of aflatoxin in peanuts. Part 11. Tropical Science 5 / 3 : 143 – 154 .
Melouk H. A. and Blackman P. A. 1995 Management of soilborne pathogens. 75 – 82 In Melouk H. A. and Shokes F. M. eds. Peanut Health Management Am. Phytopath. Soc St. Paul, MN .
Perry D. A. 1967 Premature death of peanut plants in northern Nigeria. Experimental Agriculture 3 : 211 – 214 .
Porter D. M. , Wright F. S. , and Steele J. L. 1972 Relationship of shell damage to colonization of peanut seed by Aspergilus flavus. J. Amer. Peanut Res. Educ. Assoc 4 : 207 .
Raheja A. K. 1975 Millepedes on peanuts Arachis hypogaea L. Nigerian J. Plant Prot 1 : 91 – 92 .
Sandhu R. S. , Syamasonta B. , and Simvula E. 1986 Recent Developments in Peanut Improvement in Zambia. 45 – 50 In Second Regional Peanut Workshop for Southern Africa, Harare, Zimbabwe, International Crops Research Institute for the semi-Arid Tropics India .
SAS Institute 1999–2000 User's Guide: Statistics SAS Institute Cary, N. C .
Sekamatte B. , Latizo M. , Kyamanywa S. , and Wilson H. 2000 The use of protein and sugar-based baits to enhance predatory ant activity and reduce termite damage in maize in Uganda. Seventh Annual Report of the Integrated Pest Management Collaborative Research Support Program (IPM CRSP).
Smal H. , Kvien C. S. , Summer M. E. , and Csinos A. S. 1989 Solution calcium concentration and application date effects on pod calcium uptake and distribution in Florunner and Tifton-8. Peanut Journal of Plant Nutrition 12 : 37 – 52 .
Sohati P. H. and Sithanantham S. 1990 Damage and yield loss caused by insect pests on peanut in Zambia. 113 – 116 In Patacheru A. P. ed. Proc. of the fourth regional peanut workshop for southern Africa. March 19–23, 1990 Arusha, Tanzania ICRISAT: International Crops Research for the Semi-Arid Tropics .
Sokal R. R. and Rohlf F. G. 1981 The principles and practices of statistics in biological research. In “Biometry,” Second Edition 721 – 730 Freeman Company New York .
Summer M. E. , Kvien C. S. , Smal H. , and Csinos A. S. 1988 On the calcium nutrition of peanut (Arachis hypogea L.). 1. Operational model. J. of Fertilizer Issues 5 : 97 – 102 .
Tsigbey F. K. , Brandenburg R. L. , and Clottey V. A. 2003 Peanut Production Methods in Northern Ghana and Some Disease Perspectives. World Geography of the Peanut Knowledge Base website. http://lanra.anthro.uga.edu/peanut/knowledgebase/request.pdf. 9 pages.
Umeh V. C. , Waliyar F. , Traore S. , Chaibou I. M. , Omar B. , and Detognou J. 2001 Farmers' opinions and influence of Cultural practices on soil pest damage to peanut in West Africa. Insect Science and its Application 21 / 3 : 257 – 265 .
Wightman J. A. , Dick K. M. , Ranga Rao G. V. , Shanower T. G. , and Gold C. G. 1990 Pests of peanut in Semi-Arid Tropics. 242 – 320 In Singh S. R. ed. Insect pests of food legumes John Wiley Sons Ltd .
Wightman J. A. and Ranga Rao G. V. 1993 A peanut insect identification handbook for India. 38 – 51 In Patasheru A. P. ed Information Bulletin No. 39: 38–51 International Crops Research Institute for Semi-Arid Tropics India .
Wightman J. A. and Wightman A. S. 1994 An insect, agronomic and sociological survey of peanut fields in southern Africa. Agric., Ecosystems and Environ 51 : 311 – 331 .
Wilson C. 1945 A. Diplodia associated with concealed damage in peanuts. Phytopath 35 : 480 .
Notes
- 1CSIR-Crops Research Institute, P. O. Box 3785, Kumasi, Ghana. [^]
- 2Kwame Nkrumah University of Science and Technology, Kumasi, Ghana. [^]
- 3Department of Entomology, Box 7613, North Carolina State University, Raleigh, NC 27695-7613. [^]
- 4Department of Crop Science, Box 7620, North Carolina State University, Raleigh, NC 27695-7620. [^] *Corresponding author - Rick Brandenburg.