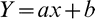Introduction
Historically, annual grasses have been considered to be among the most troublesome weeds of peanut in the U. S. (Hauser et al. 1973). Annual grasses reduce peanut yield primarily through interference and excessive harvest losses. Fall panicum [Panicum dichotomiflorum (L.) Michx.] was found to be highly competitive with peanut, reducing yield by 25% with a density of one weed per 4.9 m (York and Coble 1977). They also reported that peanut was a poor competitor with fall panicum and particularly vulnerable to interference from early season through pod fill. Chamblee et al. (1982) investigated the interference of broadleaf signalgrass [Brachiaria platyphylla (Griseb.) Nash] in North Carolina peanut and found that a density of 1.6 plants/m reduced peanut yield 28%. Their observations indicated that broadleaf signalgrass would over-top peanut canopy 8 wk after planting, eventually producing nearly twice the biomass as peanut by the end of the growing season. McCarty (1983) found that goosegrass [Eleusine indica (L.) Gaertn.] at a density of 3.2 plants/m reduced peanut yield by 20%.
Texas panicum is a troublesome weed, primarily on coarse-textured soils of the southeastern and southwestern U. S. Schroeder et al. (1990) found that Texas panicum will grow and produce seed under a wide range of soil moisture conditions, including drought. It has the ability to tolerate drought and thrive in coarse-textured soils, partially explaining the extreme competitiveness of the weed. Patterson (1990) found that maximum Texas panicum growth occurred with an average daily temperature of 28.7 C and it was hypothesized that greater weed growth would have occurred at higher temperatures.
Selection pressure from herbicide use patterns likely contributed to the development of Texas panicum as a troublesome weed of peanut in the southeastern U. S. Prior to widespread use of herbicides in peanut production, large crabgrass [Digitaria sanguinalis (L.) Scop.] and Florida pusley (Richardia scabra L.) were the most common weeds of peanut production. This phenomenon was reflected in early weed management studies conducted in the 1960's that reported large crabgrass and Florida pusley were the most common weeds present in those trials, but not Texas panicum (Hauser and Parham 1964, 1969; Pieczarka 1962). Afterwards, effective control of these shallow-germinating weeds allowed for establishment of deep-germinating Texas panicum that escaped control from preemergence herbicides (Chandler and Santelmann 1969, Santelmann 1974). Weed surveys conducted in the early-1970's indicated that large crabgrass continued to be among the most common weeds of peanut, but was no longer considered to be troublesome (Hauser et al. 1973). The same survey also reported that Texas panicum developed into one of the most common and troublesome weeds of peanut in the southeastern U. S. By 2005, surveys indicated that Texas panicum continued to be among the most common and troublesome weeds of peanut in the southeastern U. S. (Webster 2005), despite effective controls.
Texas panicum control is costly, often requiring dinitroaniline herbicides for residual control, followed by a postemergence graminicide to control escapes (Brecke and Currey 1980; Grichar 1991; Grichar and Boswell 1986; Johnson et al. 2002; Prostko et al. 2001). Ethalfluralin, pendimethalin, and trifluralin are applied preplant incorporated to control Texas panicum. Escapes are controlled with a postemergence application of either clethodim or sethoxydim. Peanut growers usually apply postemergence graminicides from mid- to late-season at which time the Texas panicum plants are very large. Control of robust Texas panicum plants with postemergence graminicides is inconsistent, even at elevated rates. Assuming acceptable efficacy using late-season graminicide applications, peanut yields will still be reduced compared to early-season control efforts (Grichar and Boswell 1986). This is likely due to unrecoverable yield losses from Texas panicum interference prior to control with a postemergence graminicide, although this has not been quantified.
Profits from peanut production are marginal due to commodity values lower than those in the 1990's and escalating costs of production. Weed management systems have become scrutinized as part of an overall effort to streamline production costs. Since Texas panicum management systems can involve two separate herbicide applications, there is interest in determining treatment thresholds to eliminate unnecessary costs. Trials were initiated in 2001 to determine the critical density of Texas panicum escaping earlier control efforts to justify treatment with a postemergence graminicide, the critical period that escaped Texas panicum be controlled without significant yield reduction, and to quantify harvest losses from Texas panicum interference.
Materials and Methods
Irrigated field trials were conducted from 2001 to 2003 at the Ponder Farm, a unit of the Coastal Plain Experiment Station, near Tifton, GA. Soil was a Tifton loamy sand (thermic, Plinthic Kandiudults) having 84% sand, 10% silt, and 6% clay, with 0.3% organic matter. This site was typical of the peanut-producing region of the southeastern U. S.
The experimental sites chosen had heavy natural populations of Texas panicum. The experimental sites were deep turned (23 cm deep) with a moldboard plow 1 wk before planting and tilled 7.6 cm deep with a power-tiller to condition and shape seedbeds. The peanut cultivar ‘C99R’ was planted in early May of each year. Small seeded dicot weeds were controlled with a preemergence application of alachlor (1.1 kg ai/ha). Alachlor does not control Texas panicum at this rate (personal observation). Bentazon (1.1 kg ai/ha) plus 2,4-DB (0.28 kg ai/ha) was applied for maintenance weed control and supplemented with hand-weeding as needed. Excluding weed management, peanut were managed according recommendations from the Georgia Cooperative Extension Service (Beasley et al. 1997).
Texas Panicum Density
The experimental design was a randomized complete block, with four replications. Plots were four rows wide (1.8 m) and 10 m in length. Rows were spaced 91 cm apart. Texas panicum were established in the middle two rows of the plot from natural infestations to the following densities: 0, 1, 2, 4, 8, 16, and 32 plants per plot (total of 20 m row) and allowed to interfere with peanut the entire season. The outside rows in each plot were maintained as weed-free borders. Texas panicum plants were chosen from those emerged two weeks after peanut emergence, with the density equally split between the two center rows in the plot and the weeds as evenly distributed as possible. The Texas panicum plants chosen were confined to those present in a 30 cm band centered over the drill. Any other Texas panicum plants emerging the remainder of the season were removed by hand-weeding.
Duration of Texas Panicum Interference
These trials were conducted using the same general protocol as described for the density studies. Texas panicum were established two weeks after peanut emergence from natural populations and maintained at a density of 8 plants/20 m. Treatments were the amount of time that Texas panicum was allowed to interfere with peanut at the established density until removal. The duration of Texas panicum interference was 2, 4, 6, 8, 10, 12, 14, 16, 18, and 20 weeks after peanut emergence, along with a season-long weed-free control. At the conclusion of the designated period of Texas panicum interference, weeds were removed by an application of clethodim (0.21 kg ai/ha) plus a crop oil concentrate adjuvant. This technique was used to remove emerged Texas panicum since clethodim is commonly used to control Texas panicum escapes, generally non-phytotoxic to peanut, and less disruptive to peanut growth than hand-removal of large Texas panicum.
Peanut yields in both trials were measured by digging, inverting, air-curing, and combining peanut using commercial two-row equipment, modified for small plot production. Yield samples were mechanically cleaned to remove foreign material (soil, mineral concretions, and roots). Final yield is reported as cleaned farmer stock peanut. A 500 g sub-sample was used to measure peanut grade, expressed as total sound mature kernels (TSMK). In the density trials, plots were re-established immediately after combining to measure harvest losses. In the center of each plot, 1 m2 of soil was sieved through hardware cloth to collect peanut pods lost during harvest due to Texas panicum interference. Peanut harvest losses were expressed in kg/ha.
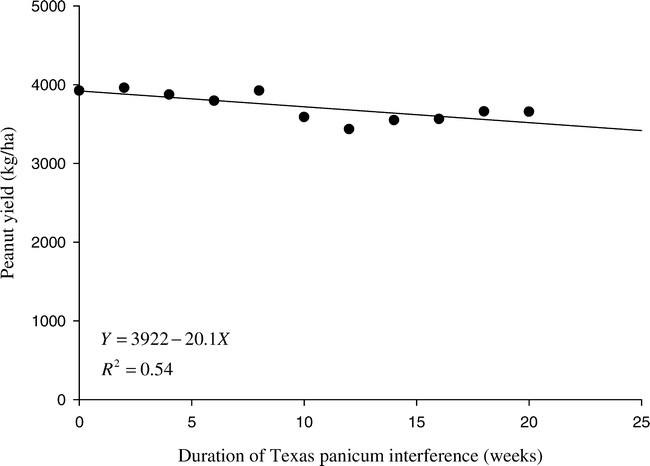
These data were regressed to determine the effect of Texas panicum density and duration of interference on the parameters measured. The regression analysis was based on the principles outlined by Draper and Smith (1981) using:
Where Y = parameters being measured, a = intercept, b = slope, and x = Texas panicum density or duration (weeks) of Texas panicum interference.
Results and Discussion
Statistical analysis showed nonsignificant treatment by year interactions for all parameters (data not shown). Therefore, all data are combined across years.
Texas Panicum Density
Yield data combined across 3 years indicated a linear response of peanut yield to Texas panicum density (Figure 1). Each Texas panicum plant per 20 m row reduced peanut yield by 25 kg/ha. Based on 91 cm row spacing and the 3 year average peanut yields in the weed free plots (3950 kg/ha), Texas panicum at 40 plants/20 m (2.2 plants/m2) was predicted to reduce peanut yield by 25%.
To compare the interference of various weeds with peanut requires conversion of reported weed densities to common units which are based on experimental technique, peanut row spacing, and extrapolation from regression curves. Data interpolation indicated that 1.1 goosegrass plants/m2 (McCarty 1983), 0.9 broadleaf signalgrass plants/m2 (Chamblee et al.1982), and 2.0 fall panicum plants/m2 (York and Coble 1977) reduced peanut yields by 25%. Our data shows that Texas panicum at 2.2 plants/m2 reduces peanut yield by 25%. It is prudent to use caution in comparing these critical densities among species since the earlier studies were conducted using what are now obsolete cultivars across an array of growing conditions, cultural practices, and irrigation regimes. However, these values are grossly similar among the annual grasses. In contrast, previous studies indicated that 0.3 common cocklebur (Xanthium strumarium L.) plants/m2 (Royal et al. 1997), 1.0 wild poinsettia (Euphorbia heterophylla L.) plants/m2 (Bridges et al. 1992), 2.3 bristly starbur (Acanthospermum hispidum DC) plants/m2 (Walker et al. 1989), 6.2 Florida beggarweed [Desmodium tortuosum (Sw.) DC] plants/m2 (Hauser et al. 1982), 7.2 sicklepod [Senna obtusifolia (L.) Irwin and Barneby] plants/m2 (Hauser et al. 1982), and 68.0 yellow nutsedge (Cyperus esculentus L.) plants/m2 (Johnson and Mullinix 2003) reduced peanut yield by 25%. Texas panicum and other annual grasses are among the most competitive weeds of peanut production.
Peanut grade (TSMK percentage) was not affected by Texas panicum density (data not shown), despite interference from varying densities of Texas panicum which reduced yield. Similarly, McCarty (1983) reported that goosegrass interference did not affect peanut grade.
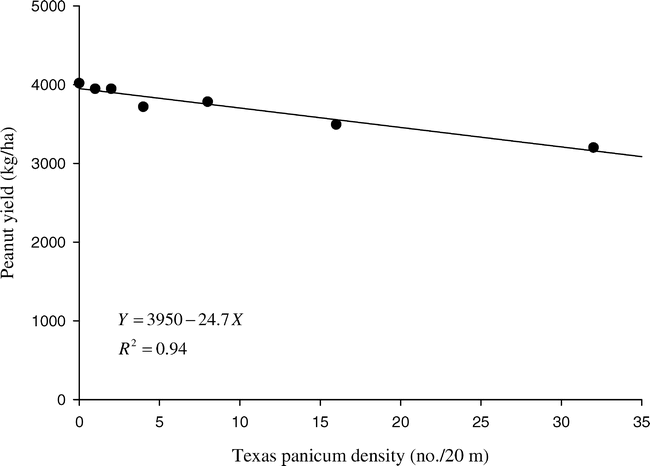
Texas panicum interference directly affects peanut harvest losses, with pods entangled in fibrous Texas panicum roots being torn from the plant during digging. As Texas panicum densities increased, peanut harvest losses increased at a linear rate (Figure 2). Even in the weed-free control, peanut harvest losses were 623 kg/ha. However, peanut harvest losses in plots with Texas panicum at 32 plants/20 m were 19% greater than the weed-free control. When considered with total yield reduction, a Texas panicum density of 40 plants/20 m is predicted to reduce peanut yields by 25% (Figure 1), resulting in peanut harvest losses of 836 kg/ha (Figure 2). Assuming peanut value of $0.47/kg, the value of these harvest losses is estimated to be $393/ha.
Texas Panicum Duration of Infestation
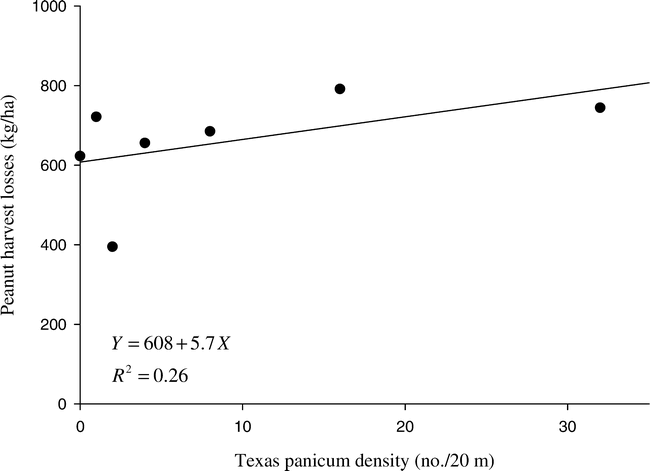
At the weed density evaluated in our trials (8 plants/20 m), each week of Texas panicum interference reduced peanut yield by approximately 20 kg/ha (Figure 3). Chamblee et al. (1982) reported that broadleaf signalgrass interference from a natural infestation of 1050 plants/10 m (2100 plants/20 m) for at least 4 to 6 wk after planting resulted in unrecoverable peanut yield reduction. York and Coble (1977) reported similar results for fall panicum interference at 243 plants/m2 (4423 plants/20 m). Our studies indicate that Texas panicum at 8 plants/20 m must be controlled no later than 9 to 10 wk after peanut emergence to prevent significant (P≤0.05) yield loss.
The previous research trials, as well as ours, had objectives and designs relevant to the era when they were conducted. The differences between our results with the duration of Texas panicum interference and previous studies on fall panicum (York and Coble 1977) and broadleaf signalgrass (Chamblee et al. 1982) interference are likely due to large differences in baseline weed densities and methods of weed removal. Our trials were designed to determine the critical density of Texas panicum escaping earlier control efforts for later control with a postemergence graminicide and the densities in our trials reflect that underlying premise. Control of annual grasses with a postemergence graminicide was not an option for peanut growers when the previous trials were conducted in the mid-1970's and early-1980's. We used applications of clethodim to remove Texas panicum, while the previous studies used handweeding to remove the fall panicum and broadleaf signalgrass at the desired intervals. Peanut is very tolerant of clethodim. However, hand removal of dense infestations of large grasses is very disruptive to the subterranean fruiting of peanut.
These results indicate that Texas panicum can be very competitive with peanut, which affects treatment decisions. Clethodim is commonly used to control Texas panicum escapes. Assuming an application of clethodim costs $51/ha (cost of herbicide, adjuvant, and application), peanut value of $0.47/kg, and average peanut yields in the weed free controls in this trial (3950 kg/ha), the minimum density of Texas panicum to justify control is 4.4 plants/20 m row. Using the results from the duration of interference trials, the critical period to control Texas panicum without non-recoverable yield reduction is no later than 9 to 10 weeks after emergence. Failure to control Texas panicum escapes in a timely manner will result in yield reduction and excessive harvest losses. The tendency of peanut growers to control Texas panicum escapes late in the season is not a sound practice due to inconsistent efficacy and potential for non-recoverable yield reduction.
Acknowledgements
We acknowledge the technical contributions of Andy M. Hornbuckle and Vann M. Jones in these trials.
Literature Cited
Beasley J. , Bader M. , Baldwin J. , Harris G. , Padgett B. , Brown S. L. , and MacDonald G. Peanut Production Field Guide. Ga. Coop. Ext. Ser. Bull 1997 1146 .
Brecke B. J. and Currey W. L. Weed control in peanuts with ethalfluralin. Peanut Sci 1980 7 : 124 – 127 .
Bridges D. C. , Brecke B. J. , and Barbour J. C. Wild poinsettia (Euphorbia heterophylla) interference with peanut (Arachis hypogaea). Weed Sci 1992 40 : 37 – 42 .
Chamblee R. W. , Thompson L. , and Coble H. D. Interference of broadleaf signalgrass (Brachiaria platyphylla) in peanuts (Arachis hypogaea). Weed Sci 1982 30 : 45 – 49 .
Chandler J. M. and Santelmann P. W. Growth characteristics and herbicide susceptibility of Texas panicum. Weed Sci 1969 17 : 91 – 93 .
Draper N. R. and Smith H. Applied Regression Analysis. 2nd ed 1981 New York John Wiley and Sons .
Grichar W. J. Control of Texas panicum (Panicum texanum) and southern crabgrass (Digitaria ciliaris) in peanuts (Arachis hypogaea) with postemergence herbicides. Peanut Sci 1991 18 : 6 – 9 .
Grichar W. J. and Boswell T. E. Postemergence grass control in peanut (Arachis hypogaea). Weed Sci 1986 34 : 587 – 590 .
Hauser E. W. , Buchanan G. A. , Nichols R. L. , and Patterson R. M. Effects of Florida beggarweed (Desmodium tortuosum) and sicklepod (Cassia obtusifolia) on peanut (Arachis hypogaea). Weed Sci 1982 30 : 602 – 604 .
Hauser E. W. and Parham S. A. Effects of annual weeds and cultivation on the yield of peanuts. Weed Res 1969 9 : 192 – 197 .
Hauser E. W. and Parham S. A. Herbicide mixtures for weed control in peanuts (Arachis hypogaea), 1961 – 1963. Weed Res 1964 4 : 338 – 350 .
Hauser E. W. , Santelmann P. W. , Buchanan G. A. , and Rud O. E. Controlling weeds in peanuts. Peanuts - Culture and Uses 1973 Stillwater, OK Amer. Peanut Res. Educ. Assoc., Inc 327 – 360 In .
Johnson W. C. and Mullinix B. G. Yellow nutsedge (Cyperus esculentus) interference in peanut (Arachis hypogaea). Peanut Sci 2003 30 : 14 – 18 .
Johnson W. C. , Prostko E. P. , and Mullinix B. G. Texas panicum (Panicum texanum) control in strip-tillage peanut (Arachis hypogaea) production. Peanut Sci 2002 29 : 141 – 145 .
McCarty M. T. Economic Thresholds of Annual Grasses in Agronomic Crops 1983 Raleigh, NC North Carolina State University 58 pp PhD dissertation.
Patterson D. T. Effects of day and night temperature on vegetative growth of Texas panicum (Panicum texanum). Weed Sci 1990 38 : 365 – 373 .
Pieczarka S. J. , Wright W. L. , and Alder E. F. Trifluralin as a soil-incorporated pre-emergence herbicide for agronomic crops. Proc. So. Weed Conf 1962 15 : 92 – 96 .
Prostko E. P. , Johnson W. C. , and Mullinix B. G. Annual grass control with preplant incorporated and preemergence applications of ethalfluralin and pendimethalin in peanut (Arachis hypogaea). Weed Technol 2001 15 : 36 – 41 .
Royal S. S. , Brecke B. J. , and Colvin D. L. Common cocklebur (Xanthium strumarium) interference with peanut (Arachis hypogaea). Weed Sci 1997 45 : 38 – 43 .
Santelmann P. W. Today's weed - Texas panicum. Weeds Today 1974 pp 21 Winter, 1974.
Schroeder J. , Dowler C. C. , and Stansell J. R. Texas panicum (Panicum texanum) growth as affected by irrigation management and planting date. Weed Sci 1990 38 : 374 – 378 .
Walker R. H. , Wells L. W. , and McGuire J. A. Bristly starbur (Acanthospermum hispidum) interference in peanuts (Arachis hypogaea). Weed Sci 1989 37 : 196 – 200 .
Webster T. M. Weed survey - southern states. Proc. So. Weed Sci. Soc 2005 58 : 291 – 306 .
York A. C. and Coble H. D. Fall panicum interference in peanuts. Weed Sci 1977 25 : 43 – 47 .
Notes
- 1Res. Agron., USDA-ARS and Agric. Res. Statistician; Coastal Plain Exp. Sta., Tifton, GA 31793-0748. [^] Corresponding author's Email: cjohnson@tifton.usda.gov.