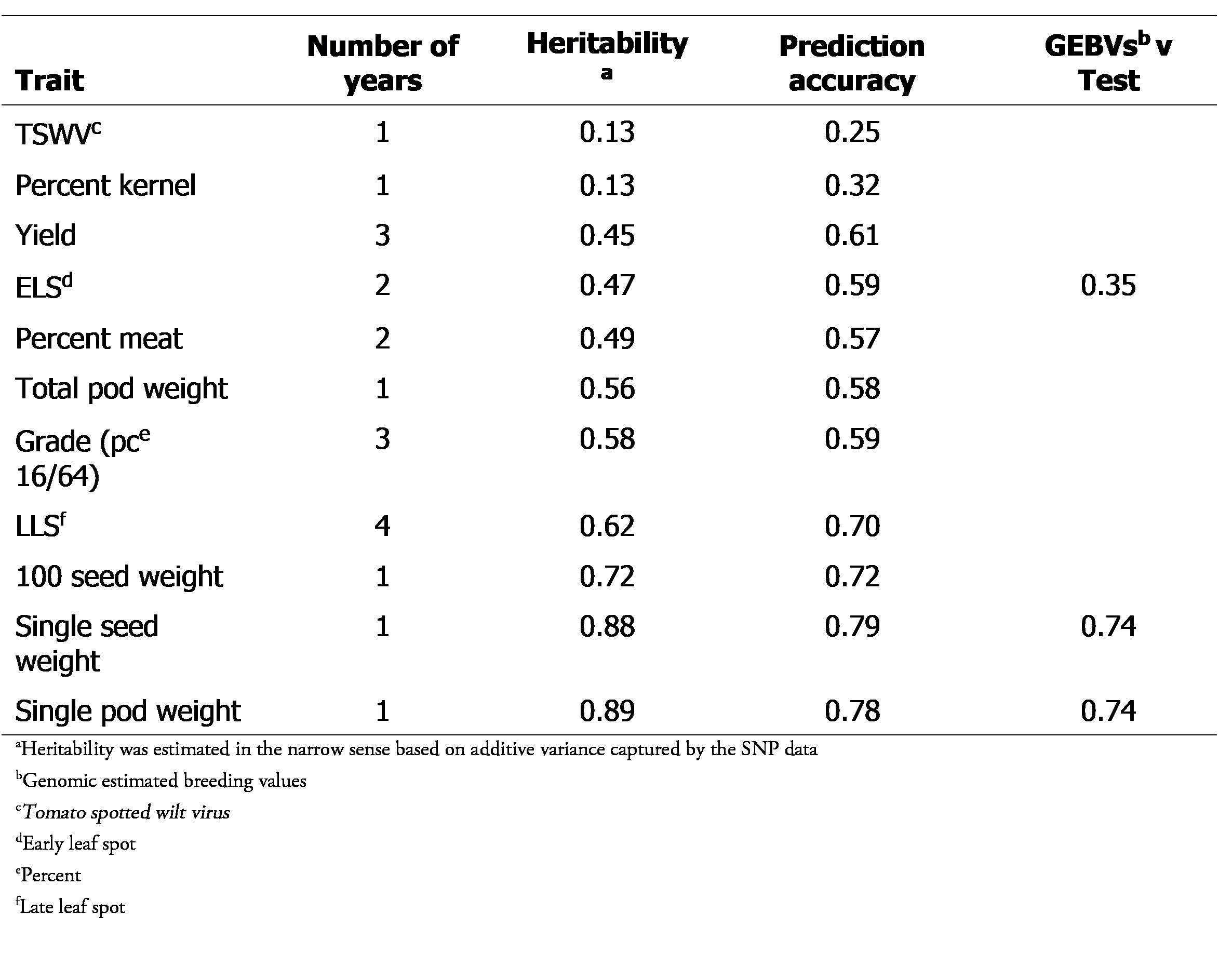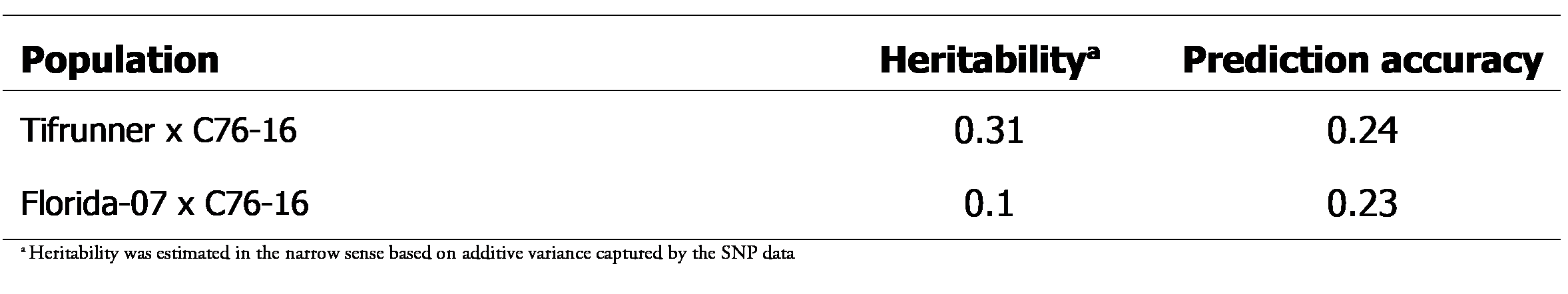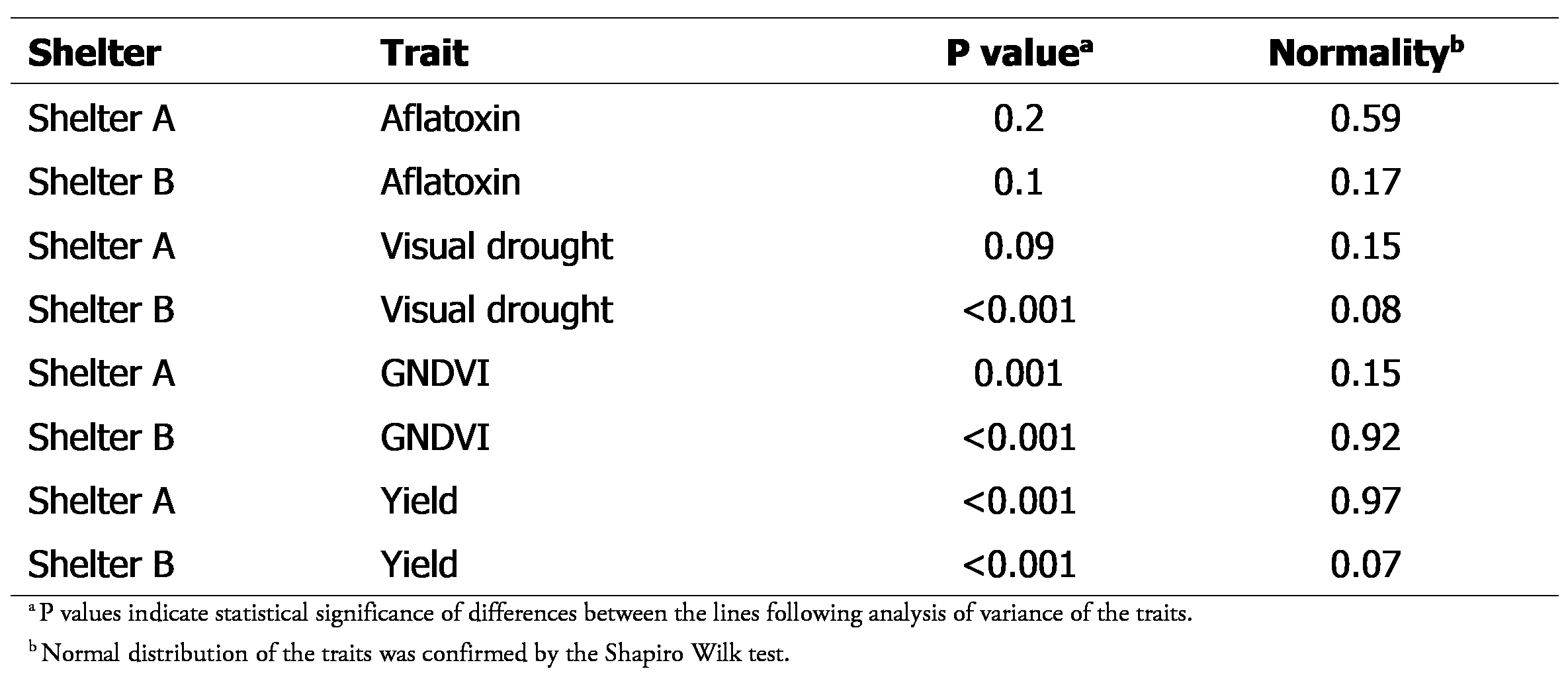Introduction
Peanut is an important oil seed legume that is widely grown in all tropical and sub-tropical regions in the world (Bertioli et al., 2011). An important constraint to its production is contamination by aflatoxins, which are secondary metabolites mainly produced by Aspergillus flavus and Aspergillus parasiticus (Khlangwiset et al., 2011). The fungi occur ubiquitously in the soil and can colonize the seeds of a diverse number of crops such as peanut, maize, soybean, sorghum, cotton seed, cassava, chilies and tree nuts. At optimum conditions, typically temperatures between 24 and 35°C and humidity above 7%, the fungi can grow and produce aflatoxins on many commodities (Williams et al., 2004; Klich, 2007). A. flavus produces the aflatoxins B1 and B2 while A. parasiticus can also produce aflatoxins G1 and G2. Exposure to small amounts of these aflatoxins results in development of chronic aflatoxicosis while acute aflatoxicosis is the consequence of exposure to high concentrations of the toxins (Bhatnagar-Mathur et al., 2015). Both levels of exposure have serious consequences to health, since aflatoxin is the most potent naturally occurring carcinogen. It has been linked to development of liver cancer, suppression of the immune system, and nutritional interference, which in turn leads to severe weight loss and stunting especially in children (Williams et al., 2004). It is a serious problem especially in developing countries where high levels of exposure begin in utero and is associated with severe developmental challenges as well as growth impairment (Gong et al., 2002; Khlangwiset et al., 2011).
In many developing countries, there is high contamination of staple crops such as maize and peanut due to absence of regulations to control exposure (Wild and Gong, 2010). Combined with the challenges of frequent food shortages, consumption of highly aflatoxin-contaminated food is common (Guo et al., 2009). This was tragically illustrated in the 2004 aflatoxicosis outbreak in rural Kenya that resulted from consumption of maize with up to 1000 ppb of aflatoxin, leading to 125 deaths (Lewis et al., 2005). In developed countries, the law regulates the maximum aflatoxin contamination allowed. For instance in the US, only 0.5 ppb, 20 ppb, and up to 300 ppb respectively, is permissible in milk, food and feed products, respectively, while in the EU, limits as low as 4 ppb of total aflatoxins are allowed (Guo et al. 2009). In peanut, higher contamination rates result in destruction of nuts leading to losses that have been estimated at around $20 million annually in the southeastern US (Lamb and Sternitzke, 2001).
The health and economic consequences of aflatoxin contamination makes its prevention a key concern for the peanut industry (Holbrook et al., 2009). While it is possible to mitigate aflatoxin contamination by optimizing post-harvest cultural practices such as curing, drying and storage, a more desirable approach is the identification of genetic-based resistance to pre-harvest aflatoxin contamination (PAC). Ideally, resistant genotypes would be those that either mitigate fungal colonization, inhibit the formation of aflatoxin or exhibit resistance to abiotic stresses such as drought (Liang et al., 2006;Guo et al., 2008). Particularly for peanut, development of cultivars with resistance to PAC is desirable (Anderson et al., 1995; Holbrook et al., 2009; Wang et al., 2010). The geocarpic nature of the peanut plant, that is the habit of flowering above ground and production of fruit below the soil surface (Barker, 2005), makes the fruit particularly vulnerable to PAC.
The correlation of drought stress with development of PAC has been well established (Pettit et al., 1971; Hill et al., 1983; Wilson and Stansell, 1983; Holbrook et al., 2000a). The combination of water stress and exposure of developing peanut pods to high soil temperature, triggers the contamination of intact undamaged pods with aflatoxin prior to harvest. This is because A. flavus and A. parasiticus optimally grow at temperatures of 25 to 42°C, being able to thrive in soils with moisture potential as low as -35 MPa. Under these conditions, the fungi become extremely competitive and may become the dominant fungal species in the soil. Elevation of soil temperature occurs as solar radiation reaches the soil surface due to receding of the peanut canopy during severe drought (Blankenship et al., 1984; Cole et al., 1985; Payne, 1998). It is thus not surprising that in peanut, aflatoxin contamination becomes prevalent during prolonged exposure to heat and drought stress, with drought susceptible genotypes manifesting higher susceptibility to PAC (Holbrook et al., 2000a; Girdthai et al., 2010). As a result of the significance of aflatoxin, screening and breeding of cultivars with resistance especially to PAC is a key objective in peanut breeding (Holbrook et al., 2009; Wang et al., 2010).
Several studies have attempted to address the challenge of aflatoxin contamination, with availability of resistant genotypes being reported (Mixon and Rogers, 1973; Mehan et al., 1981; Mixon, 1986). These resistant genotypes were identified through in vitro seed colonization by A. flavus (IVSCAF) lab assays. However, when tested in field studies under drought conditions, these genotypes have not demonstrated clear genetic resistance to aflatoxin contamination (Blankenship et al., 1985; Anderson et al., 1995). Nonetheless, consistent with the association between drought stress and PAC, other studies have demonstrated that more drought tolerant genotypes accumulate less aflatoxin than drought susceptible genotypes. This was noted for example by Holbrook et al. (2000a) in a study that also noted positive correlations between PAC and leaf temperature measurements as well as visual stress ratings. Due to their cost effectiveness and ease of measurement, the latter two were considered to be potential proxies for selection of PAC resistance.
Due to the importance of the high oleic trait in peanut, studies have been done to determine whether differential accumulation of aflatoxin occurs with high oleic compared to high linoleic peanut. High oleic lines were shown to have high post-harvest growth of Aspergillus and increased aflatoxin contamination (Xue et al., 2003). However, a previous field-based study of high oleic peanut subjected to drought stress and aflatoxin inoculation did not show any differences in PAC as a result of fatty acid differences (Holbrook et al., 2000b). In a quest to find out if genetic resistance to other fungi that infect peanut can confer protection against Aspergillus, Holbrook et al. (1997) tested genotypes that are resistant to late leaf spot and white mold. Unfortunately, they did not observe reduced PAC in any of the genotypes tested, thus invalidating resistance to other fungi as an indirect selection method for PAC.
Recent studies of PAC in peanut seem to suggest the key to genetic resistance is not so much the prevention of fungal invasion but the diminution of aflatoxin production. For instance, using automated rainout shelters to induce aflatoxin contamination and RNA sequencing of susceptible genotypes, Clevenger et al. (2016) demonstrated that the permissive state for PAC occurs as a result of alteration of abscisic acid biosynthesis and signaling by the ABR1 gene. On the other hand, Korani et al. (2017) observed that under both field pre-harvest inoculation of A. flavus under drought stress and post-harvest inoculation, the drought tolerant genotype ICG 1471, was colonized with the fungus comparably with the susceptible genotypes, however, it accumulated less aflatoxin than the susceptible genotypes suggesting an underlying genetic mechanism that restricted aflatoxin accumulation.
In this study, we used a genomic selection (GS) strategy to screen two populations subjected to terminal drought stress for aflatoxin resistance. This took advantage of the availability of high density SNP chips for peanut (Clevenger et al., 2017; Clevenger, et al., 2018) and phenotype data from previous years that were used to derive prediction models for breeding values used for selection. The use of GS was first reported in cattle (Meuwissen et al., 2001), and has subsequently been used in rice (Spindel et al., 2015), wheat (Poland et al., 2012; Rutkoski et al., 2015; Battenfield et al., 2016), maize (Bernardo and Yu, 2007; Albrecht et al., 2011; Technow et al., 2013) and soybean (Zhang et al., 2016) breeding. The potential utility of GS in studying PAC resistance lies in the fact that it does not capitalize on detection of few large effect QTLs. Rather, it uses low cost genome spanning markers to detect even small effect variations that may not be captured by other QTL detection strategies (Heffner et al., 2009; Jannink et al., 2010). For studying PAC in peanut, this is useful, given the dearth of genetic variation for the trait (Anderson et al., 1995; Nigam et al., 2009). In addition to direct measurements of aflatoxin contamination, visual as well as high-throughput ratings were taken as easier to evaluate correlates for aflatoxin contamination.
Materials and methods
Genomic selection
We applied GS to three peanut recombinant inbred line (RIL) populations. The first population, Florida 07 X GP-NC WS 16, had 383 RILs; the second population, Tifrunner X C76-16, had 152 RILs; and the third population, Florida-07 X C76-16, had 242 RILs.
Florida-07 X GP-NC WS 16 population
All of the Florida-07 X GP-NC WS 16 population had previously been genotyped with the Affymetrix v1 SNP array (Clevenger et al., 2017; Pandey et al., 2017) resulting in 999 SNP markers while phenotypic data on 11 traits had been collected for half of the population (192 lines) over a period of 1 to 4 years. These traits included late leaf spot resistance (LLS; 4 years), grade (3 years), yield (3 years), early leaf spot resistance (ELS; 2 years), percent meat (2 years), Tomato spotted wilt virus resistance (TSWV; 1 year), total and single pod weights (1 year), single and 100 seed weights (1 year) and percent kernel (1 year). The phenotype data were subjected to mixed model analysis using the R package lme4 (Bates et al., 2015) as implemented in lmerTest (Kuznetsova et al., 2017) and the resultant coefficients were used for GS model training and testing.
Tifrunner X C76-16 and Florida-07 X C76-16 populations
Phenotype data for aflatoxin collected over three and two years for Tifrunner X C76-16 and Florida-07 X C76-16 respectively, were available. These were the results of assays done using the AflaTest immune-affinity columns (Vicam, Milford, MA). Aflatoxin data were log + 1 transformed to normalize their distribution. Following linear regression, the resultant coefficients were used for subsequent GS. All 394 lines were genotyped using the Affymetrix v1 and v2 SNP chips (Clevenger et al., 2017, 2018). The SNPs were filtered to retain polymorphic markers with minor allele frequency (MAF) > 0.05. For both chips, the SNP clustering was manually inspected to ensure expected clustering profiles. SNPs with call rate below threshold were eliminated for chip 2 only since the polymorphic marker density was higher for the v2 chip. SNPs from both chips were combined and pruned using SNPRelate (Zheng et al., 2012) at a threshold of 0.2 to eliminate SNPs in linkage disequilibrium.
For all populations, GS models were developed and heritability estimates derived using subsets of the populations that were both genotyped and phenotyped (training population) using rrBLUP (Endelman, 2011). To test the models, 10-fold cross validation was performed. In this case, the phenotype data for a random tenth part of the training population (TP) was masked and the rest of the TP was used to calculate the genomic estimated breeding values (GEBVs), which were then correlated to the actual phenotype values. This was performed 1000 times and the mean of the correlations was calculated, resulting in the prediction accuracy. The models were used to estimate aflatoxin GEBVs for the genotyped portion of the population without phenotype data (test population). The populations were combined, ranked on GEBVs and the top and bottom performers of each trait were selected based on seed availability for a rainout shelter study.
Population phenotyping
Phenotypic evaluation was performed in two rainout shelters that constituted two drought stress environments. Each rainout shelter had 100 1.5m long microplots and were located at Gibbs farm in the University of Georgia, Tifton Campus (31°26'5"N; 83°35'20"W). The soil type was Tift loamy sand (fine-loamy, kaolinitic, thermic Plinthic Kandiudults). Seeds were sown in the microplots in a randomized complete block design (RCBD), with 10 replications and 5 cm spacing between the seeds. The plants were managed following conventional agronomic practices for peanut including irrigation and fungicide applications. The tractor-mobile shelters were pulled over the plots 40 days prior to harvest to keep out the rain and impose drought and heat stress.
Fungal inoculation
Fungal inoculum was prepared by inoculating heat-sterilized cracked corn with a seven-day old A. flavus and A. parasiticus spore suspension containing 1 × 106 conidia/ml of water. The inoculum was incubated at 25 °C for 3 d then stored at 4 °C until used for field inoculation (Will et al., 1994). Inoculation was accomplished by sprinkling 28g of corn infested with A. flavus and 28g of corn infested with A. parasiticus on the plant foliage and on the soil surface approximately 60 days after planting (Holbrook et al., 1997).
Rating for drought stress
Two weeks prior to harvest, manual drought stress ratings were done. A scale of 1 to 5 as described by Luis et al., (2016) was used to rate as follows: 1) healthy plants with no drought-related symptoms, 2) upper branches bend downwards, 3) whole plant bends downwards and leaves start to dry and turn brown, 4) upper canopy dries up with leaves becoming brittle and thin and 5) plants are severely wilted and/or dead. A day before harvest, aerial high-throughput ratings of the plots were done using a multispectral camera (Parrot Sequoia, MicaSense, Seattle, WA) mounted on a 3DR Solo quadcopter (3D Robotics, Berkeley, CA).
Aflatoxin assay
After harvest, the shelled peanuts from each plot were ground, mixed and 100 g collected for aflatoxin content assay using the Vicam fluorometry method. The ground samples were mixed with 10 g NaCl and 200 ml of methanol/water (80:20 v/v), homogenized using a blender and filtered through a filter paper. Five ml of the filtrate was diluted with 20 ml HPLC water and filtered again. Ten ml of the filtrate was purified using AflaTest immunoaffinity columns containing aflatoxin-specific (B1, B2, G1 and G2) monoclonal antibodies and washed with 10 ml HPLC water. The column was washed with 10 ml HPLC water and aflatoxin was eluted using HPLC grade methanol. Aflatoxin was measured fluorometrically.
Post-harvest analysis
Aerial images were stitched using the photogrammetry software Pix4D (Pix4D S.A., Prilly, Switzerland) and resulted in whole field orthomosaics for each band: green, red, red-edge, and near-infrared. From these, the Green Normalized Difference Vegetation Index (GNDVI) was derived using ArcGIS (ESRI, 2011). Quantitative scores for the index were obtained by manually drawing boundaries to delineate each plot in the fields with appropriate buffering to ensure no overlap between plots. Pixels outside the plot boundaries were eliminated. Within the plots, thresholds of pixels representing soil were manually determined using the identity function and eliminated. Canopy pixels were averaged to determine the score for each line.
The index, manually collected drought scores, aflatoxin content and plot yield data were analyzed using Spatial Analysis of field Trials with Splines (SpATS) models (Rodríguez-Álvarez et al., 2018) as implemented in the R package statgenSTA (van Rossum, 2020). All analyses were done in R (R Core Team, 2013).
Results and discussion
Pre-harvest aflatoxin contamination is a serious constraint in peanut production as it renders the commodity unacceptable for trade and unfit for consumption as food or feed (Waliyar et al., 2016). While management practices are integral to amelioration of this problem, identification of genetic sources of resistance is desirable (Holbrook et al., 2009). This study attempted to take advantage of the numerous genomic resources now available for peanut to apply the novel method of GS in selecting for PAC tolerance. Genomic selection has been implemented with varying levels of success in studying traits with various levels of complexity in several species. For instance, prediction accuracies ranging from 0.16 to 0.34 for dry weight and 0.52 to 0.56 for days to heading have been reported in perennial ryegrass (Faville et al., 2016). In wheat, prediction accuracies of 0.35 for Fusarium head blight resistance, 0.62 for flour yield (Hoffstetter et al., 2016) and 0.28 to 0.45 for grain yield (Poland et al., 2012) have been reported. In maize, prediction accuracies of 0.28 to 0.49 for grain yield and 0.35 to 0.6 for plant height have been reported (Zhang et al., 2015), while in sugarcane, values of 0.22 to 0.45 for commercially extractable sucrose have been reported (Deomano et al., 2020). In peanut, cross validation analysis with multiple models have yielded prediction accuracies of 0.4 to 0.6 for pods per plant and shelling percentage, and above 0.6 for high heritability traits such as days to 50% flowering, days to maturity, 100 seed weight, oleic acid, rust resistance and late leaf spot (Pandey et al., 2020). Similarly, cross validation analysis of sting nematode resistance in peanut yielded prediction accuracies of 0.18 to 0.53 (Ravelombola et al., 2022).
In this study we began by implementing cross validation analysis on the Florida-07 X GP-NC WS 16 population. This population was comprised of 383 RILs that were developed to introduce TSWV as well as early and late leaf spot resistance to a cultivated background (Holbrook et al., 2013). Prediction accuracies ranged from 0.25 for TSWV to 0.79 for single seed weight (Table 1).
The tendency of traits with high heritability yielding higher correlation values was observed, which is in line with trends observed in other studies (Lin et al., 2014; Pandey et al., 2020). This was corroborated by available test data for ELS, single seed weight and single pod weight. For these traits, the GEBVs were correlated with the test data (Table 1). ELS which had moderate heritability had the lowest correlation between GEBVs and test data. Single seed and pod weights had high heritability. Correspondingly, their prediction accuracies were high and the correlations between GEBVs and test data were also high. The trend of prediction accuracy increasing with higher heritability indicated that GS was effective for selection of traits with a clear genetic basis. However, such traits are also easily amenable to conventional marker-assisted selection precluding the necessity of using GS, which would be more suitable for more complex, hard to phenotype traits such as aflatoxin.
The Tifrunner X C76-16 and Florida-07 X C76-16 populations were used to evaluate the possibility of using GS in breeding for the more challenging trait of PAC resistance. These populations have elite runner backgrounds (Tifrunner for Tifrunner X C76-16 and Florida-07 for Florida-07 X C76-16) with the un-adapted C76-16 used as a source of variability for drought tolerance (Holbrook et al., 2013).
Genotyping of the populations resulted in a total of 2204 and 1781 polymorphic SNPs for Tifrunner X C76-16 and Florida-07 X C76-16 populations, respectively (Table 2). The SNPs were evenly distributed in the chromosomes. For Tifrunner X C76-16, the percentage of polymorphic SNPs in each chromosome relative to total polymorphic SNPs ranged from 3.1% (A10) to 8.5% (B9) while for Florida-07 X C76-16 the range was 1.9% (A8) to 8.4% (B9) (Table 2). The genotype data were used for cross validation analysis. The prediction accuracy for PAC was 0.24 and 0.23 for Tifrunner X C76-16 and Florida-07 X C76-16, respectively. These low values corresponded to the low heritability scores of 0.31 and 0.1 for the two populations respectively (Table 3). Despite the low scores, the GEBVs were used to rank the populations, with the intention of testing the best and worst performers. Unfortunately, there was limited seed availability which impeded selection solely on the basis of the computed GEBVs. Twenty lines were selected for planting with a majority (15) being derived from the Tifrunner X C76-16 population. There were sufficient quantities of seed for 13 of the lines to be planted in two rainout shelters while the rest were planted in only one shelter (Table 4).
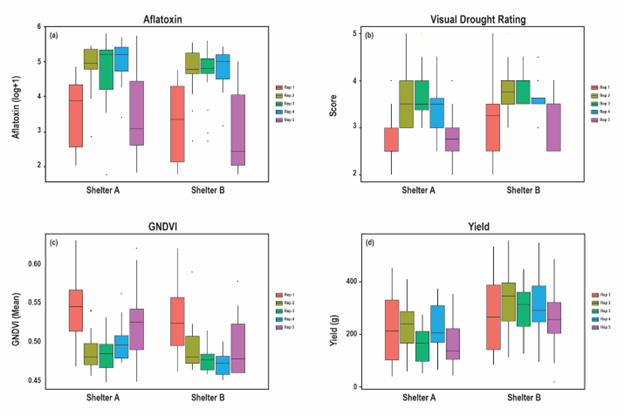
Preliminary exploration of data collected at the end of the season showed that all data were normally distributed. However, there were no significant differences between the lines for aflatoxin in both shelters and for visual drought ratings in shelter A (Table 5). In addition, spatial trends were observed, with most traits having higher or lower scores in plots along the edges of the shelters (Figure 1). This necessitated the use of models that accounted for spatial trends in the data for downstream analysis.
Figure 1. Box plots of end season aflatoxin (a), visual drought ratings (b), GNDVI (c) and yield (d). In each shelter, the left most and right most plots represent rep 1 and rep 5 respectively, which were situated on the outer edges of the shelters. Clear spatial trends in trait scores were apparent particularly for lines sown on the edges.
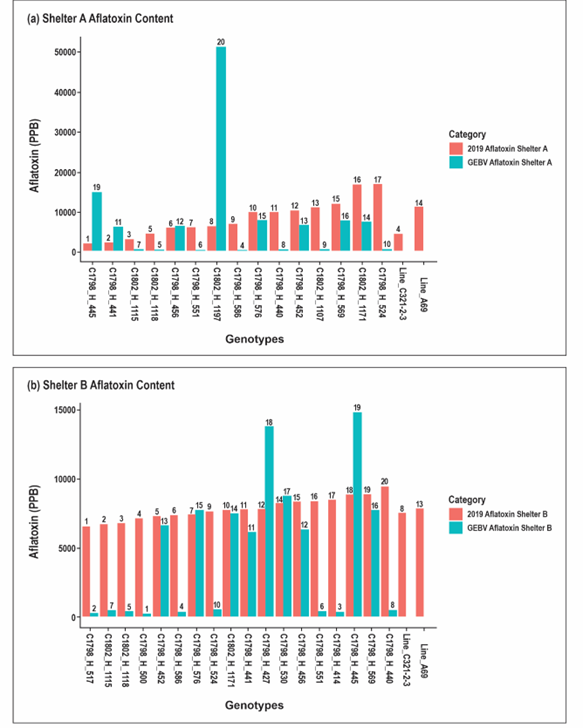
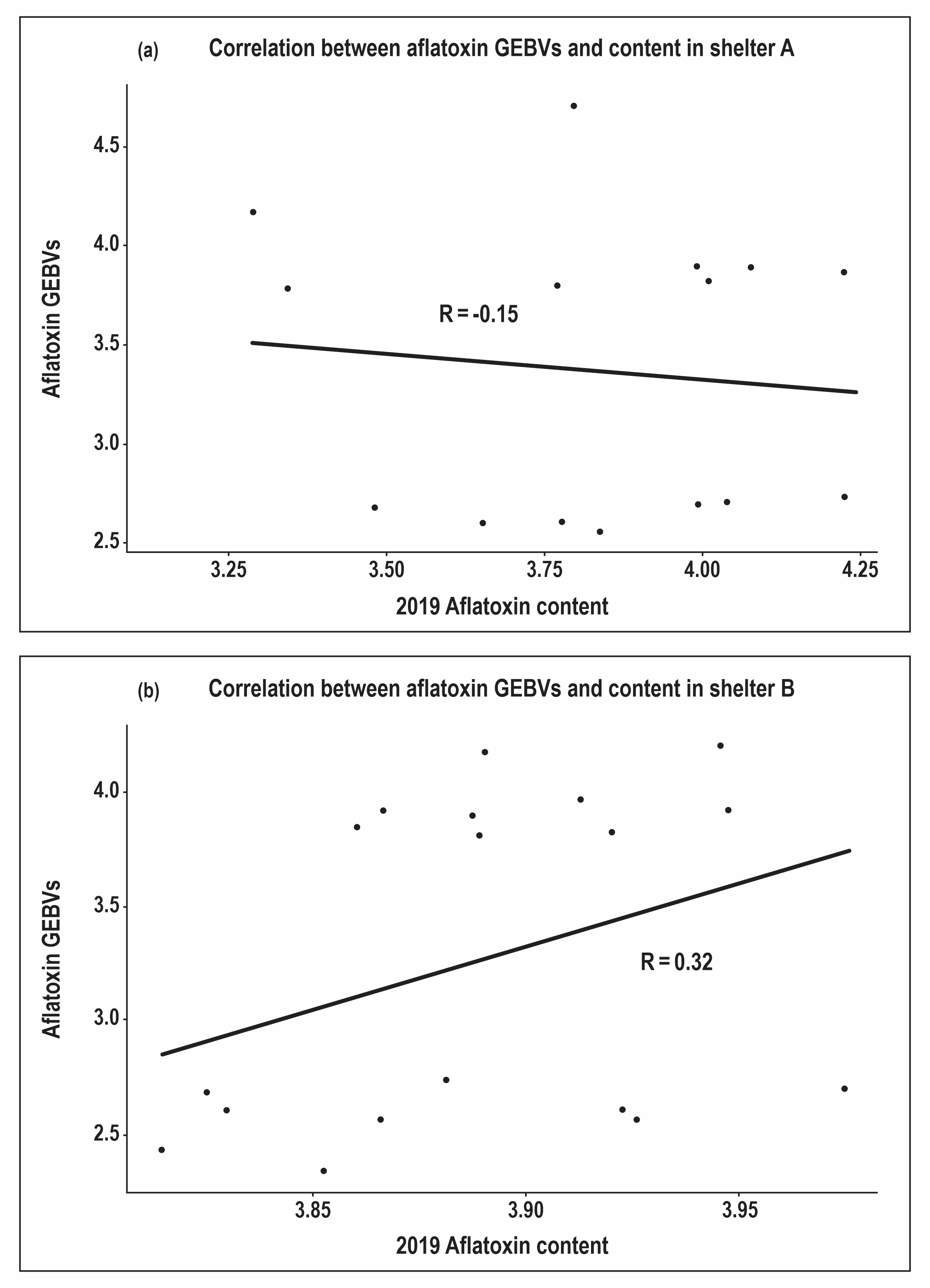
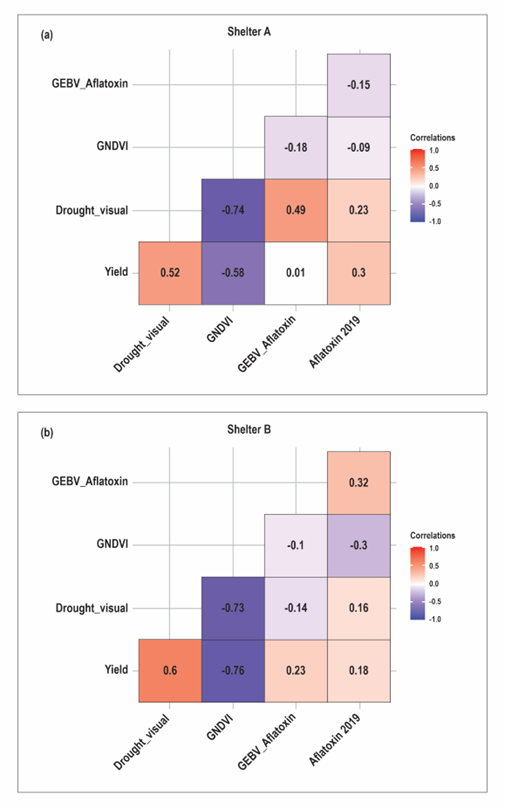
Rankings of aflatoxin content were inconsistent between the shelters. Similarly, the performance of the lines differed from the GEBVs, which were the basis of selection (Figure 2). In both shelters, differences were apparent both in rank and magnitude between the aflatoxin GEBVs and the assayed aflatoxin content values. These differences were more pronounced in shelter A. Correlation analysis showed low positive correlation (0.32) between the GEBVs and shelter B aflatoxin content. However, this was not the case for shelter A where the correlation was hardly discernible (-0.15) (Figure 3). These results serve to underscore the challenge of studying PAC which is extremely variable even with setups designed to induce heat and drought stress over extended periods as is necessary for consistent aflatoxin contamination (Holbrook et al., 1994, 2009).
Figure 2. Shelter rankings for GEBVs and end season aflatoxin content in shelter A (a) and shelter B (b). Numbers above the bars indicate the rank assigned to each line by the GEBVs (blue bars) and the actual 2019 aflatoxin content (pink bars). Lines prefixed C1798 constitute the Tifrunner X C76-16 population while those prefixed C1802 constitute the Florida-07 X C76-16 population. The last two pink bars with no corresponding GEBV data are the check varieties.
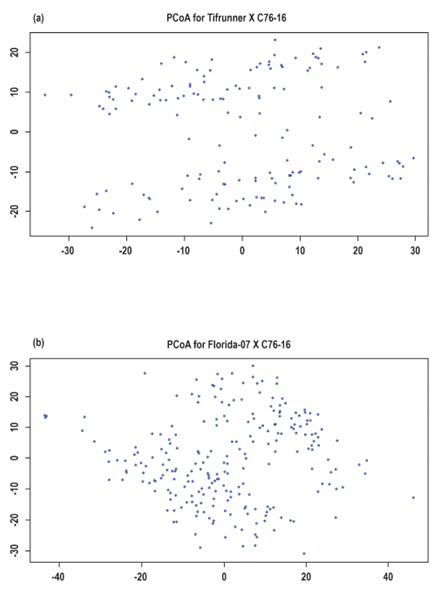
Ideally, a difficult to phenotype trait such as PAC resistance would be preferable for implementing GS, since it uses whole genome spanning markers developed at low cost to detect small effect genetic variations that influence the trait (Heffner et al., 2009; Jannink et al., 2010). Despite the fair distribution of SNPs along the chromosomes of the two populations (Table 2), the parents of these populations have been shown to have generally low polymorphism (Chu et al., 2018). In addition, the process of advancing the population during RIL development may have favored selection of plants with minimal linkage drag from the common parent (C76-16), thus imposing population structure. Visual inspection of principal coordinate analysis plots of the SNP data revealed the likely presence of population structure in both populations (Figure 4). However, factoring this in the computation of GEBVs did not improve the analysis. Moreover, selection was done under irrigated conditions with no aflatoxin pressure. It is not known if genetic components from C76-16 with good aflatoxin or drought resistance made it to the RILs. These factors likely compromised the genetic architecture of PAC resistance, which of itself is a low heritability trait. Hence, in this study, GS does not confer clear advantages over conventional selection for PAC resistance.
In other studies, low visual stress ratings and leaf temperature - which are less variable and cheaper to measure - have been associated with reduced PAC in peanuts (Holbrook et al., 2000a; Girdthai et al., 2010). In this study, it was not possible to make such an association since the correlations between visual drought ratings and PAC were low (0.23 and 0.16 for shelter A and B, respectively). However, strong inverse correlations were observed between GNDVI and visual drought ratings (~ -0.7 in both shelters). In shelter B, GNDVI had comparable correlation with aflatoxin content as the GEBVs (Figure 5). Even though GNDVI is a high throughput phenotyping method that can substitute manual, subjective and labor-intensive drought rating methods, its predictive ability for aflatoxin resistance cannot be ascertained since the results did not replicate between the shelters. It is worth noting that GNDVI was the trait with the strongest correlation with yield in both shelters. Given that both traits are statistically significant, it can be deduced that GNDVI can be a good predictor of yield under drought conditions.
Summary and conclusion
To the best of our knowledge, this is the first report of deployment of GS to tackle the challenge of breeding for PAC resistance in peanut. We also report the first use of GEBVs for selection in peanut. The use of genome spanning SNP markers ideally facilitates the detection of genetic variations that individually have a small effect on phenotype and are difficult to capture by other methods. Consequently, the method was demonstrated to be effective for traits with high heritability. Despite its potential usefulness for PAC resistance studies, the low heritability of the trait in the two populations studied resulted in weak prediction accuracies, suggesting that the challenge of genomic aided breeding for PAC resistance still remains. In addition, despite using rainout shelters to impose terminal drought stress, extreme variability of the trait was still observed. Lack of clear association of both visual drought ratings and GNDVI with PAC hindered the use of these strategies as better correlates for the trait.
Acknowledgements
The authors would like to thank Jason Golden, Shannon Atkinson, Betty Tyler and Kathy Marchant for technical assistance. The project was funded by the Georgia Peanut Commission. The authors declare no competing interests
Literature Cited
Albrecht T., Wimmer V., Auinger H-J., Erbe M., Knaak C., Ouznova M., Simianer H. and Schon C-C.. 2011. Genome-based prediction of testcross values in maize. Theoretical and applied genetics, 123(2): 339–50.
Anderson W.F., Holbrook C.C., Wilson D.M., and Matheron M.E.. 1995. Evaluation of preharvest aflatoxin contamination in several potentially resistant peanut genotypes. Peanut Science, 22(1): 29–32.
Barker N.P. 2005. A review and survey of basicarpy, geocarpy, and amphicarpy in the African and Madagascan flora. Annals of the Missouri Botanical Garden, 92(4): 445–462.
Bates D., Mächler M., Bolker B., and Walker S.. 2015. Fitting linear mixed-effects models using lme4. Journal of Statistical Software, 67(1): 1–48.
Battenfield S.D., Guzmán C., Gaynor R.C., Singh R.P., Peña R.J., Dreisigacker S., Fritz A.K , and Poland J.A.. 2016. Genomic selection for processing and end-use quality traits in the CIMMYT spring bread wheat breeding program. The Plant Genome, 9(2): 0.
Bernardo R., and Yu J.. 2007. Prospects for genomewide selection for quantitative traits in maize. Crop Science, 47(3): 1082.
Bertioli D.J., Seijo G., Freitas F.O., Valls J.F.M. and Leal-Bertioli S.C.M.. 2011. An overview of peanut and its wild relatives. Plant Genetic Resources: Characterisation and Utilisation, 9(1): 134–149.
Bhatnagar-Mathur P., Sunkara S., Bhatnagar-Panwar M., Waliyar F., and Sharma K.K.. 2015. Biotechnological advances for combating Aspergillus flavus and aflatoxin contamination in crops. Plant Science, 234: 119–132.
Blankenship P.D., Cole R.J., and Sanders T.H.. 1985. Comparative susceptibility of four experimental peanut lines and the cultivar Florunner to preharvest aflatoxin contamination. Peanut Science, 12(2): 70–72.
Blankenship P.D., Cole R.J., Sanders T.H., and Hill R.A.. 1984. Effect of geocarposphere temperature on pre-harvest colonization of drought-stressed peanuts by Aspergillus flavus and subsequent aflatoxin contamination. Mycopathologia, 85(1–2): 69–74.
Chu Y., Holbrook C.C., Isleib T.G., Burow M. and Culbreath A.K.. 2018. Phenotyping and genotyping parents of sixteen recombinant inbred peanut populations. Peanut Science, 45(1): 1–11.
Clevenger J., Chu Y., Chavarro C., Agarwal G. and Bertioli D.J.. 2017. Genome-wide SNP genotyping resolves signatures of selection and tetrasomic recombination in peanut. Molecular Plant, 10(2): 309–322.
Clevenger J.P., Korani W., Ozias-Akins P., and Jackson S.. 2018. Haplotype-based genotyping in polyploids. Frontiers in Plant Science, 9: 564.
Clevenger J., Marasigan K., Liakos V., Sobolev V. and Vellidis G.. 2016. RNA Sequencing of contaminated seeds reveals the state of the seed permissive for pre-harvest aflatoxin contamination and points to a potential susceptibility factor. Toxins, 8(11): 317.
Cole R.J., Sanders T.H., Hill R.A., and Blankenship P.D.. 1985. Mean geocarposphere temperatures that induce preharvest aflatoxin contamination of peanuts under drought stress. Mycopathologia, 91(1): 41–46.
Deomano E., Jackson P., Wei X., Aitken K. and Kota R.. 2020. Genomic prediction of sugar content and cane yield in sugar cane clones in different stages of selection in a breeding program, with and without pedigree information. Molecular Breeding, 40(4): 38.
Endelman J.B. 2011. Ridge Regression and other kernels for genomic selection with R package rrBLUP. The Plant Genome Journal, 4(3): 250.
ESRI. 2011. ArcGIS Desktop.
Faville M.J., Ganesh S., Moraga R., Easton H.S. and Jahufer M.Z.Z.. 2016. Development of genomic selection for perennial ryegrass. In: Roldán-Ruiz I., Baert J., Reheul D. (eds) Breeding in a World of Scarcity. Springer, Cham. p. 139–143.
Girdthai T., Jogloy S., Vorasoot N., Akkasaeng C. and Wongkaew S.. 2010. Associations between physiological traits for drought tolerance and aflatoxin contamination in peanut genotypes under terminal drought. Plant Breeding, 129(6): 693–699.
Gong Y.Y., Cardwell K., Hounsa A., Egal S. and Turner P.C.. 2002. Dietary aflatoxin exposure and impaired growth in young children from Benin and Togo: cross sectional study. BMJ, 325(7354): 20.
Guo B., Chen Z.Y., Lee R.D., and Scully B.T.. 2008. Drought stress and preharvest aflatoxin contamination in agricultural commodity: Genetics, genomics and proteomics. Journal of Integrative Plant Biology, 50(10): 1281–1291.
Guo B., Yu J., Holbrook C.C., Cleveland T.E. and Nierman W.C.. 2009. Strategies in prevention of preharvest aflatoxin contamination in peanuts: aflatoxin biosynthesis, genetics and genomics. Peanut Science, 36(1): 11–20.
Heffner E.L., Sorrells M.E., and Jannink J-L.. 2009. Genomic selection for crop improvement. Crop Science, 49(1): 1.
Hill R.A., Blankenship P.D., Cole R.J., and Sanders T.H.. 1983. Effects of soil moisture and temperature on preharvest invasion of peanuts by the Aspergillus flavus group and subsequent aflatoxin development. Applied and environmental microbiology, 45(2): 628–33.
Hoffstetter A., Cabrera A., Huang M., and Sneller C.. 2016. Optimizing training population data and validation of genomic selection for economic traits in soft winter wheat. G 3: Genes, Genomes, Genetics, 6(9): 2919–2928.
Holbrook C.C., Guo B.Z., Wilson D.M., and Timper P.. 2009. The U.S. Breeding program to develop peanut with drought tolerance and reduced aflatoxin contamination. Peanut Science, 36(1): 50–53.
Holbrook C.C., Isleib T.G., Ozias-Akins P., Chu Y. and Knapp S.J.. 2013. Development and phenotyping of recombinant inbred line (RIL) populations for peanut (Arachis hypogaea). Peanut Science, 40: 89–94.
Holbrook C.C., Kvien C.K., Rucker K.S., Wilson D.M. and Hook J.E.. 2000a. Preharvest aflatoxin contamination in drought tolerant and drought intolerant peanut genotypes. Peanut Science, 27(2): 45–48.
Holbrook C.C., Matheron M.E., Wilson D.M., Anderson W.F. and Will M.E.. 1994. Development of a large-scale field system for screening peanut for resistance to preharvest aflatoxin contamination. Peanut Science, 21(1): 20–22.
Holbrook C.C., Wilson D.M., Matheron M.E., and Anderson W.F.. 1997. Aspergillus colonization and aflatoxin contamination in peanut genotypes with resistance to other fungal pathogens. Plant Disease, 81(12): 1429–1431.
Holbrook C.C., Wilson D.M., Matheron M.E., Hunter J.E. and Knauft D.A.. 2000b. Aspergillus colonization and aflatoxin contamination in peanut genotypes with reduced linoleic acid composition. Plant Disease, 84(2): 148–150.
Jannink J-L., Lorenz A.J., and Iwata H.. 2010. Genomic selection in plant breeding: from theory to practice. Briefings in functional genomics, 9(2): 166–77.
Khlangwiset P., Shephard G.S., and Wu F.. 2011. Aflatoxins and growth impairment: A review. Critical Reviews in Toxicology, 41(9): 740–755.
Klich M.A. 2007. Aspergillus flavus: The major producer of aflatoxin. Molecular Plant Pathology, 8(6): 713–722.
Korani W.A., Chu Y., Holbrook C., Clevenger J., and Ozias-Akins P.. 2017. Genotypic regulation of aflatoxin accumulation but not Aspergillus fungal growth upon post-harvest infection of peanut (Arachis hypogaea L.) seeds. Toxins, 9(7): 218.
Kuznetsova A., Brockhoff P.B., and Christensen R.H.B.. 2017. lmerTest package: tests in linear mixed effects models. Journal of Statistical Software, 82(13): 1–26.
Lamb M.C., and Sternitzke D.A.. 2001. Cost of aflatoxin to the farmer, buying point, and sheller segments of the southeast United States peanut industry. Peanut Science, 28(2): 59–63.
Lewis L., Onsongo M., Njapau H., and Schurz-Rogers H.. 2005. Aflatoxin contamination of commercial maize products during an outbreak of acute aflatoxicosis in eastern and central Kenya. Environmental Health Perspectives, 113(12): 1763-1767.
Liang X.A., Luo M., and Guo B.Z.. 2006. Resistance mechanisms to Aspergillus flavus infection and aflatoxin contaminatin in peanut (Arachis hypogaea). Plant Pathology Journal, 5: 115-124.
Lin Z., Hayes B.J., and Daetwyler H.D.. 2014. Genomic selection in crops, trees and forages: a review. Crop Pasture Science. 65(11): 1177–1191.
Luis J.M., Ozias-Akins P., Holbrook C.C., Kemerait, Jr. R.C., Snider J.L., and Liakos V.. 2016. Phenotyping peanut genotypes for drought tolerance. Peanut Science, 43: 36-48.
Mehan V., McDonald D., Nigam S., and Lalitha B.. 1981. Groundnut cultivars with seed resistant to invasion by Aspergillus flavus. Oleagineux, 36(10): 501–507.
Meuwissen T.H., Hayes B.J., and Goddard M.E.. 2001. Prediction of total genetic value using genome-wide dense marker maps. Genetics, 157(4): 1819–29.
Mixon A.C. 1986. Reducing Aspergillus species infection of peanut seed using resistant genotypes. Journal of Environment Quality, 15(2): 101.
Mixon A.C., and Rogers K.M.. 1973. Peanut accessions resistant to seed infection by Aspergillus flavus. Agronomy Journal, 65(4): 560.
Nigam S.N., Waliyar F., Aruna R., Reddy S. V. and Kumar P.L.. 2009. Breeding peanut for resistance to aflatoxin contamination at ICRISAT. Peanut Science, 36(1): 42–49.
Pandey M.K., Agarwal G., Kale S.M., Clevenger J. and Nayak S.N.. 2017. Development and evaluation of a high density genotyping “Axiom-Arachis” array with 58 K SNPs for accelerating genetics and breeding in groundnut. Scientific Reports, 7: 1–10.
Pandey M.K., Chaudhari S., Jarquin D., Janila P., Crossa J., Patil S.C., Sundravadana S., Khare D., Bhat R.S., Radhakrishnan T., Hickey J.M., Varshney R.K.. 2020. Genome-based trait prediction in multi-environment breeding trials in groundnut. Theoretical and Applied Genetics, 133:3101.
Payne G.A. 1998. Process of contamination by aflatoxin-producing fungi and their impact on crops. In: Sinha K.K. and Bhatnagar D. (eds), Mycotoxins in agriculture and food safety. Marcel Dekker, New York. p. 279–306.
Pettit R.E., Taber R.A., Schroeder H.W., and Harrison A.L.. 1971. Influence of fungicides and irrigation practice on aflatoxin in peantus before digging. Applied Microbiology, 22(4): 629–634.
Poland J., Endelman J., Dawson J., Rutkoski J. and Wu S.. 2012. Genomic selection in wheat breeding using genotyping-by-sequencing. The Plant Genome Journal, 5(3): 103.
Core Team R. 2013. R: A language and environment for statistical computing. R Foundation for statistical computing, Vienna, Austria, https://www.R-project.org/.
Ravelombola W., Cason J., Tallury S., Manley A., Pham H.. 2022. Genome-wide association study and genomic selection for sting nematode resistance in peanut using the USDA public data. https://doi.org/10.1080/15427528.2022.2087127.
Rodríguez-Álvarez M.X., Boer M.P. , van Eeuwijk F.A. , andEilers P.H.C. . 2018. Correcting for spatial heterogeneity in plant breeding experiments with P-splines. Spatial Statistics, 23: 52–71.
Rutkoski J., Singh R.P., Huerta-Espino J., Bhavani S. and Poland J.. 2015. Genetic gain from phenotypic and genomic selection for quantitative resistance to stem rust of wheat. The Plant Genome, 8(2): 1-10.
Spindel J., Begum H., Akdemir D., Virk P. and Collard B.. 2015. Genomic selection and association mapping in rice (Oryza sativa): effect of trait genetic architecture, training population composition, marker number and statistical model on accuracy of rice genomic selection in elite, tropical rice breeding lines. PLoS Genetics, 11(2).
Technow F., Bürger A., and Melchinger A.E.. 2013. Genomic prediction of northern corn leaf blight resistance in maize with combined or separated training sets for heterotic groups. G 3: Genes, Genomes, Genetics, 3(2): 197–203.
van Rossum B. J. 2020. statgenSTA: single trial analysis (STA) of field trials. R package version 1.0.4. https://CRAN.R-project.org/package=statgenSTA.
Waliyar F., Kumar K.V.K., Diallo M., Traore A. and Mangala U.N.. 2016. Resistance to pre-harvest aflatoxin contamination in ICRISAT’s groundnut mini core collection. European Journal of Plant Pathology, 145(4): 901–913.
Wang T., Zhang E., Chen X., Li L., and Liang X.. 2010. Identification of seed proteins associated with resistance to pre-harvested aflatoxin contamination in peanut (Arachis hypogaea L). BMC Plant Biology, 10:267.
Wild C.P., and Gong Y.Y.. 2010. Mycotoxins and human disease: a largely ignored global health issue. Carcinogenesis, 31(1): 71–82.
Will M.E., Holbrook C.C., and Wilson D.M.. 1994. Evaluation of field inoculation techniques for screening peanut genotypes for reaction to preharvest A. flavus group infection and aflatoxin contamination. Peanut Science, 21(2): 122–125.
Williams J.H., Phillips T.D., Jolly P.E., Stiles J.K. and Jolly C.M.. 2004. Human aflatoxicosis in developing countries : a review of toxicology , exposure , potential health consequences , and interventions. American Journal of Clinical Nutrition, 80(5): 1106–1122.
Wilson D.M., and Stansell J.R.. 1983. Effect of irrigation regimes on aflatoxin contamination of peanut pods. Peanut Science, 10(2): 54–56.
Xue H.Q., Isleib T.G., Payne G.A., Wilson R.F. and Novitzky W.P.. 2003. Comparison of aflatoxin production in normal and high-oleic backcross-derived peanut lines. Plant Disease, 87(11): 1360–1365.
Zhang X., Pérez-Rodríguez P., Semagn K., Beyene Y. and Babu R.. 2015. Genomic prediction in biparental tropical maize populations in water-stressed and well-watered environments using low-density and GBS SNPs. Heredity, 114(3): 291–299.
Zhang J., Song Q., Cregan P.B., and Jiang G.-L.. 2016. Genome-wide association study, genomic prediction and marker-assisted selection for seed weight in soybean (Glycine max). Theoretical and Applied Genetics, 129(1): 117–130.
Zheng X., Levine D., Shen J., Gogarten S.M. and Laurie C.. 2012. A high-performance computing toolset for relatedness and principal component analysis of SNP data. Bioinformatics, 28(24): 3326–3328.
Notes
- First, fifth and eighth authors: Former Graduate Student, Assistant Professor and Professor, Institute of Plant Breeding Genetics and Genomics, University of Georgia, Tifton, GA 31793; Second author: Associate Professor, Institute of Plant Breeding Genetics and Genomics, University of Georgia, Athens, GA 30602; Third author: Supervisory Research Geneticist, United States Department of Agriculture - Agricultural Research Service, Tifton GA, 31793; Fourth author: Professor Emeritus, Crop and Soil Sciences Department, North Carolina State University, Raleigh NC, 27695; Sixth and seventh authors: Assistant and Associate Professors, Crop and Soil Sciences Department, University of Georgia, Tifton, GA 31793. [^] Corresponding author’s E-mail: pozias@uga.edu