Introduction
A mycotoxin is any secondary fungal metabolite that is toxic to humans or animals. Aflatoxin is a mycotoxin produced by the fungus Aspergillus flavus and the closely related Aspergillus parasiticus and is arguably the most problematic mycotoxin known given its impact on global health and the world economy (Coulibaly, et al., 2008). The fungi producing aflatoxin are ubiquitous in much of the tropical and subtropical latitudes, and under certain conditions, can readily colonize several global staples including corn, tree nuts and peanuts, among others. This colonization can lead to aflatoxin contamination in these important food and feed sources. Aflatoxin is highly toxic to humans and many animals, and as such, most governments have adopted strict regulatory limits for foods/feeds at risk for this toxin (JECFA, 2017).
In the United States, the USDA adopted the first regulatory limits for peanuts (Arachis hypogaea) entering edible markets in the mid 1960’s, not long after aflatoxin was discovered, and these limits and the sampling plans to deliver these measurements have continued to the present time and evolved to ever more aggressive limits. Since its discovery 60 plus years ago, the US peanut industry has made extensive investments across the supply chain to minimize, or preferably eliminate, aflatoxin from the crop. There are numerous points from seed to finished product where if conditions are not carefully controlled, aflatoxin can contaminate peanuts.
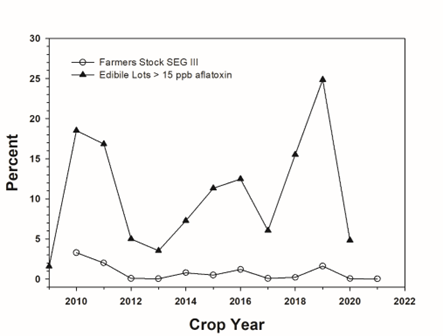
Even if all best practices are followed, there are still situations where pre-harvest aflatoxin levels (developed in the field) can be extremely high. Yearly rejection rates for shelled kernels in the United States exceeding 15 ppb total aflatoxin, which is the USDA allowable limit for edible, shelled peanuts, over the past 10 plus crop years is provided in Figure 1 (Georgia Federal-State Inspection Service 2020). Large fluctuations in rejection rates are observed and elevated aflatoxin contamination correlates with weather conditions, especially elevated temperatures during production and late season drought stress. Such stress predisposes the weakened plants for aflatoxin contamination and while adequate irrigation is a critically important mitigation strategy for reducing aflatoxin, such irrigation is often not available. During the 2019 growing season, the southeastern US peanut crop was produced under unusually high temperatures and late season drought stress, resulting in approximately 30% of the shelled runner peanut lots in the SE being rejected due to excessive aflatoxin levels (USDA, 2022). As discussed in more detail later, such losses are devasting economically for all segments of the industry and include rejected product, lost markets, increased processing costs due to additional processing to remove aflatoxin from contaminated streams, and elevated testing regimes just to name a few. Furthermore, anecdotal observations from many in the industry seem to indicate that the frequency of “bad aflatoxin years” is increasing.
Given the impact of aflatoxin on the US peanut crop, the American Peanut Council’s Aflatoxin Task Force was organized at the Winter Conference in Dec 2020 with the specific purposes of (1) evaluating the state of the art in production, transportation, handling, storage, and processing, (2) identifying the gaps in knowledge, and (3) prioritizing the importance of the knowledge gaps in relation to risk of aflatoxin contamination. As a means toward meeting these objectives, the Aflatoxin Task Force organized a Peanut Quality Symposium held in Tifton, Georgia on November 29-30, 2021. This symposium was open to all interested stakeholders.
Peanut Quality Symposium
The symposium was organized with a general session of speakers to provide context on the status of the negative impacts of aflatoxin across the entire US peanut industry. Speakers in the general session included Mr. Karl Zimmer, Chairman of the Aflatoxin Task Force, Dr. Marshall Lamb, Research Leader at the USDA, ARS National Peanut Research Laboratory, Dr. David Hoisington, Director of the Feed the Future Innovation Lab for Peanut at the University of Georgia, and Julie Adams, Vice President, Global Technical & Regulatory Affairs, Almond Board of California. The general session was followed by moderated breakout sessions. The breakout sessions were in the general areas of 1) Production and Pre-Harvest, 2) Post-Harvest Handling and Storage, 3) Shelling, Manufacturing, and Processing, and 4) Regulatory and Health and Nutrition
Symposium participants were divided into four groups approximately equal in size and sent to one of the four breakout sessions. After about an hour in the session, the groups rotated to a new breakout session. The rotation continued until all participants rotated through all four breakout topic areas. Notes were recorded by session moderators and a session recorder for each group in each topic session. There were two moderators for each of the breakout sessions who were considered knowledgeable in the field represented by the breakout session topic. Breakout session moderators then presented the consensus of the discussions in a wrap-up session. Following the summary presentations, summary points from each breakout topic were placed on posters around the auditorium. Each participant voted for five (5) summary points they considered the most important or the place where the largest knowledge gap in controlling aflatoxin contamination and its effects existed. The following is a summary of the general session speakers’ comments and the participants’ votes regarding knowledge gaps and research priorities.
General Session
Impact of Aflatoxin Contamination
Mr. Karl Zimmer and Dr. Marshall Lamb focused on the economic costs of aflatoxin across the US peanut value chain. Analysis of the 1993-1996 peanut crops in the southeastern US demonstrated that aflatoxin cost the US peanut industry an average of $25.53 per metric ton (MT) or $69.34/ha (Lamb and Sternitzke, 2001). A follow up survey for the 2016-2019 US peanut crops found that aflatoxin contamination cost the US industry an average of $32.95/MT or $134.96/ha. In 2017, a year with overall low aflatoxin contamination frequency, the industry lost approximately $18.45/MT and in 2019, which was the worst aflatoxin year in recent US history, the industry lost $52.28/MT (Lamb, et al., 2021).
The cost of aflatoxin is carried by all segments of the peanut industry, including the growers, shellers and manufacturers. Arguably, the largest portion of the aflatoxin cost is carried by the shelling segment of the peanut industry given the disconnect between measuring aflatoxin risk at harvest versus measuring the aflatoxin risk after shelling. According to USDA regulations (USDA 2017), farmers’ stock peanuts are graded into three segregation classifications: Segregation 1 (SEG I) are farmers’ stock peanuts with not more than 3.49 percent damaged kernels, no more than 1.00 percent concealed damage caused by rancidity, mold, or decay and are free from visible A. flavus. Segregation 2 (SEG II) peanuts are farmers’ stock peanuts with more than 3.49 percent damaged kernels, or more than 1.00 percent concealed damage caused by rancidity, mold, or decay and are free from visible A. flavus. Segregation 3 (SEG III) peanuts are farmers’ stock peanuts with visible A. flavus. The lowest quality and highest aflatoxin risk is thought to be in SEG III peanuts, followed by SEG II peanuts. SEG I peanuts are thought to have the highest quality and lowest aflatoxin risk. However, years of industry experience have demonstrated the presence of visible A. flavus or A. parasiticus does not always guarantee the presence of the toxin, which is not visible, and quite commonly the toxin is present (as detected chemically) but the mold is not visible.
Furthermore, under current marketing regulations, when a load receives a SEG III designation, the grower has the option to have the load cleaned and regraded. Public records do not indicate what portion of SEG I peanuts initially graded as SEG III, were cleaned and subsequently regraded as SEG I. The current system of determining the value of peanuts provides an incentive to clean and regrade peanuts determined to be SEG III in hope of regrading as SEG I. SEG III peanuts are automatically valued at 35% of the average loan value (USDA, 2019).
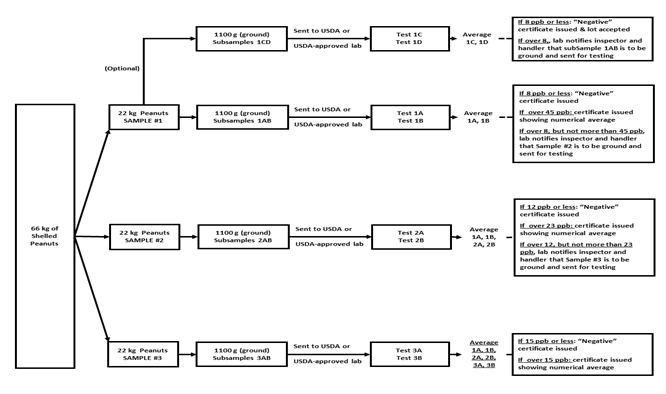
After storage, farmers’ stock peanuts are cleaned, shelled, and separated into 20-MT lots of shelled peanuts according to size and market type. Each lot of shelled peanuts is sampled by capturing three 22-kg samples during the filling process using a USDA-approved automated sampler or using a manual vacuum probe after filling. Each 20-MT lot and the associated samples are positive lot identified. A USDA or USDA-approved laboratory receives sample #1, grinds the sample, collects an 1100-g sample of the ground material, then divides it into an A and B subsample. Both subsamples are chemically tested using approved testing methods. The lot is “negative” for aflatoxin if the average aflatoxin content of the A and B subsamples is 8 ppb or less. If the average is over 45 ppb, then a report is issued showing the numerical average. If the average is over 8 but not more than 45 ppb, then Sample 2 is ground and analysed. The results of the 1A, 1B, 2A, and 2B analyses are averaged and compared to thresholds. If the average is greater than 12 but not more than 23 ppb, then Sample 3 is ground and analysed. The results of all six tests (1A, 1B, 2A, 2B, 3A, 3B) are averaged. The lot is “negative” if the average is less than or equal to 15 ppb. If the average is more than 15 ppb, then a report showing the numerical average is issued (Figure 2).
Comparison of shelled lots not meeting minimum edible USDA specifications for aflatoxin after chemical testing versus percentage of farmers’ stock lots grading as SEG III (Georgia Federal-State Inspection Service 2020) are provided in Figure 1 where it’s observed there is little to no correlation between the two measurements. This could be from multiple sources of variation including but not limited to discrepancies in testing methodology (visible mold versus chemical test), sampling plans (amount of sample that is taken, prepared, and measured) or natural sampling variation.
While aflatoxin contamination is worse in some years than others, it is not a single year issue. Figure 1 shows the percent of shelled edible peanut lots in each crop year from 2009 – 2020 that exceeded the maximum threshold of 15 ppb aflatoxin (USDA, 2022). For instance, more than 15% of the finished peanut lots from the 2010, 2011, 2018, and 2019 crops failed to meet aflatoxin specifications. Peanuts from the 2013, 2017, and 2020 crops failed to meet aflatoxin specifications 6% of the time or less. Figure 1 also shows the percent of farmers’ stock tons that had a final designation of SEG III (Georgia Federal-State Inspection Service, 2020). A SEG I designation indicates that A. flavus or A. parasiticus was not seen in the official grade sample. A SEG III designation indicates that at least one kernel with either A. flavus or A. parasiticus was seen. However, under current marketing regulations, when a load receives a SEG III designation, the grower has the option to have the load cleaned and regraded. Note that all segregation designations are the result of visual inspection and not any sort of chemical analysis. Therefore, a SEG I designation does not guarantee an absence of aflatoxin. For example, only 1.6% of the 2019 crop had a final designation of SEG III, yet the more than 25% of the finished lots exceeded 15 ppb. Public records do not indicate what portion of SEG I peanuts initially graded as SEG III, were cleaned, and subsequently regraded as SEG I. The current system of determining the value of peanuts provides an incentive to clean and regrade peanuts determined to be SEG III in hope of regrading as SEG I. SEG III peanuts are automatically valued at 35% of the average loan value (USDA 2019).
Given the potential for aflatoxin risk at harvest to vary widely depending on local growing conditions and crop inputs, and the lack of reliability in SEG III status to identify true aflatoxin risk, most shellers chemically test the loose shelled kernels (LSK), other kernels (OK), and damaged kernels (DK) retained from the farmers stock grade samples of every load purchased at a buying point. These kernel categories are tested as they are established as the highest risk for aflatoxin in a given load of incoming farmers’ stock (Whitaker, et al., 1999) and these categories are present/isolated in graded farmers’ stock typically at a size (<100 gram total) that is conducive to various quick testing aflatoxin kits available. Using this information along with other aflatoxin risk identification tools (rainfall/drought monitoring, field irrigation history, etc), many shellers then segregate their incoming farmers’ stock in warehouses according to expected aflatoxin risk at shelling. This is done at a substantial cost to the shelling community (added transportation and warehousing), but it can result in much more efficient and predictable shelling performance if peanut quality is largely segregated according to aflatoxin risk. Many shellers commonly perform an unofficial re-grade and chemical aflatoxin test of each load after it is removed from farmers stock storage and before entering the shelling plant. This confidential data has sometimes shown significant increases in aflatoxin contamination during farmers stock storage. Numerous causes for such increases in post-harvest aflatoxin are possible, but generally indicate inadequate storage (leaking roof for example) or equipment failure (inadequate ventilation for example) allowing moisture content/water activity of the stored material to exceed thresholds supporting mold growth and possible toxin formation.
While the sheller segment carries much of the aflatoxin costs, all segments are impacted. Growers can have load value reduced to 35% of the loan value if the load is designated SEG III at intake (USDA, 2019). Furthermore, the manufacturing sector can realize an inability to acquire sufficient inventory to produce products, lost finished product that does not meet finished aflatoxin specifications (FDA regulations) and/or possible product recalls due to aflatoxin contamination. While not required by law to measure aflatoxin in every lot of finished peanut product, if finished product is checked for aflatoxin by state or national authorities and found to be over the legal FDA limit (20 ppb total aflatoxin), the company would then be responsible for a recall in that situation. As such, most manufacturers using raw, shelled peanuts commonly perform in-house testing and/or use a third-party laboratory to ensure finished product meets acceptable minimum standards, which could be government mandated or customer mandated.
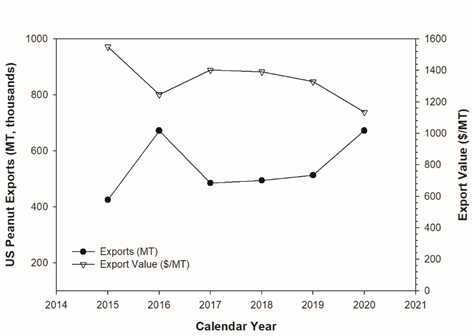
Zimmer discussed the overall negative impact of aflatoxin on the US peanut industry, specifically peanut exports. The per capita domestic peanut consumption is at an all-time high of approximately 3.6 kg (American Peanut Council, 2022); however, the U.S. peanut industry is heavily reliant on the export of U.S. peanuts. According to National Agricultural Statistics Service, the U.S. exports of peanuts between 2015 and 2020 averaged 543,434 MT and ranged from 424 to 672 thousand MT (American Peanut Council, 2022). The average value of the peanut exports was $1342/MT but has shown a general decline from a high of $1550/MT in 2015 to a $1134/MT in 2020 (Figure 3). While the decline in export value is not caused entirely by aflatoxin contamination, it is linked to aflatoxin contamination and the loss of high value markets with strict aflatoxin limits (the EU) to lower value markets with less stringent aflatoxin limits (peanuts to be crushed for oil in China).
The export market is highly competitive with other origins such as Argentina competing directly in the premium markets of the EU which require extremely low allowable aflatoxin limits, i.e. 2 ppb Aflatoxin B1 or 4 ppb total aflatoxin after testing an additional 20+ kg of peanuts above and beyond the USDA domestic program. Customers’ specifications, both foreign and domestic, are increasingly restrictive related to aflatoxin limits, with the USDA limits of 15 ppb total aflatoxin being well above most customers’ specifications. As such, the US peanut industry must continue to invest across the supply chain for solutions to minimize, or preferably eliminate aflatoxin to stay competitive with other world origins.
Dr. Hoisington discussed the impact of aflatoxin contamination on human health and nutrition, especially in the populations of developing nations. Peanuts are a global “brand” that is desired as a low-cost, plant-based protein source. In the developing economies, peanut is a high-value commodity that may be the basis for families’ economic survival. Aflatoxin is one of the most highly regulated mycotoxins. It is the only mycotoxin that has been classified as a Class I carcinogen (World Health Organization. International Agency for Research on Cancer, 1993). Prolonged low dose exposure can result in immunosuppression, nutritional dysfunction, and cancer while acute high dose exposure can result in liver damage and death (Marchese, et al., 2018). As a result, the peanut “brand” must be protected and through collaborative research address the health and nutrition concerns related to aflatoxin contamination. Continued research is necessary to provide data for U.S. trade negotiators and industry partners to advocate for science-based aflatoxin limits as opposed to arbitrarily low limits, that may not have a basis in achievable limits, detection limits, or sampling error (EFSA, 2018).
Perspective from the US Almond Industry
Finally, in the general session, Adams, Vice President for Global Technical & Regulatory Affairs, Almond Board of California, presented the perspective of the U.S. almond industry’s experience in dealing with aflatoxin. As background, California produces approximately 80% of the global supply of almonds. Europe (5%), Australia (6%), and the other producers (9%) make up the remaining 20% of the global almond supply (Almond Board of California 2022). Michailides, et al. (2007) collected soil samples from 28 Nonpareil almond orchards in northern, central, and southern California over a 5-yr period from 2007 to 2011 (excluding 2009) and examined the prevalence of A. flavus and A. parasiticus. During the study, both toxigenic and atoxigenic isolates were found demonstrating that soil can serve as a reservoir of Aspergillus. In addition, the study showed that the proportion of toxigenic isolates found varied across regions and years. They also demonstrated that the common pest in almonds and pistachios, navel orangeworm (NOW) (Amyelois transitella), that emerged from almond mummies may be contaminated with A. flavus. In one study, up to 8% of NOW adults removed from sticky traps from an almond orchard were infested with A. flavus/A. parasiticus. Michailides, et al. (2007) concluded that NOW may play an active role in vectoring and spreading aflatoxigenic fungi in almonds. This is similar to the soilborne insects such as lesser cornstalk borer (Elasmopalpus lignosellus (Zeller)) vectoring aflatoxigenic fungi in peanut (Lynch and Wilson, 1991). Whitaker, et al. (2010) demonstrated a correlation between aflatoxin and insect as well as other damage and confirmed the use of sorting techniques as an effective means for lowering aflatoxin levels in a lot of product. As a result of this and other NOW research conducted over many years, the Almond Board of California developed comprehensive guides for growers to minimize the risk of aflatoxin contamination which included minimizing NOW damage. Those practices include 1) winter orchard sanitation to remove mummy nuts – nuts that remain on the tree and become harborage for overwintering, 2) early harvest – harvesting as soon after maturity as possible, 3) in-season treatment for NOW, and 4) good practices for in-hull stockpile management.
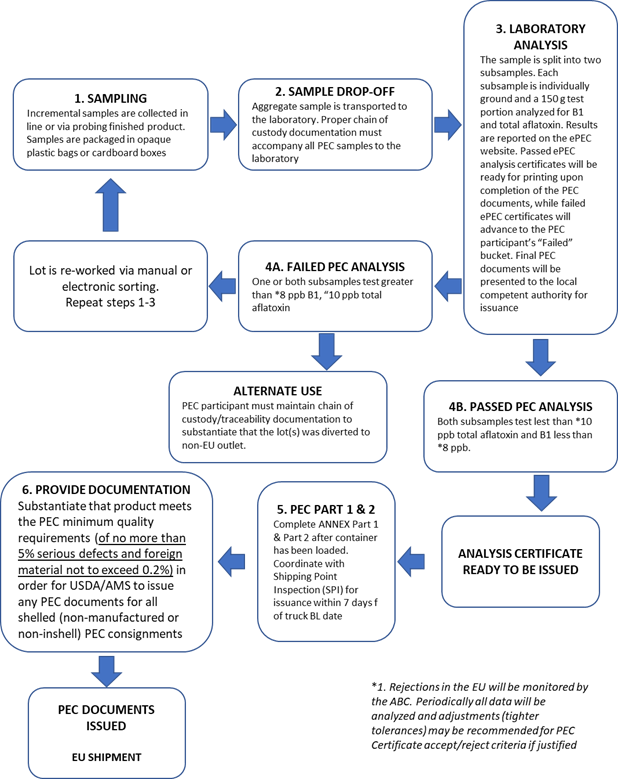
In 2007 to address the rejections in Europe, the Almond Board of California (ABC) worked with a consultant to develop and implement a voluntary pre-export chemical testing and uniform documentation program for aflatoxin based on Codex sampling plans. The aflatoxin criteria for the Pre-Export Checks (PEC) Program was revised based on the adopted Codex/EU limits for aflatoxin in ready-to-eat almonds. The stated purpose of the PEC program is “to provide an aflatoxin sampling plan for the analysis of ready to eat product with equivalency to that being used by the European Union (EU) for official testing of incoming consignments.” (Almond Board of California, 2020). Exporters volunteer to participate in the PEC Program and sign a memorandum of understanding stating that they will follow the internal controls, documentation, inspection, sampling and testing protocols outlined in the PEC Program Manual (Figure 4) The PEC Program Manual covers sample collection based on the lot size of almonds with lot identification for each consignment and associated aflatoxin sample. Samples are collected by personnel that are trained directly by the ABC or an ABC-certified trainer. Samples are delivered to a USDA-approved aflatoxin lab for testing; lab results are entered in the electronic PEC system (ePEC) administered by ABC. The PEC certificate which is generated for compliant lots aligns with EU import controls requirements and includes the analytical results. The analytical results must show that aflatoxin B1 is less than/equal to 8 ppb and total aflatoxin is less than/equal to 10 ppb. California shipping point inspection (SPI) personnel (by delegated authority from USDA) certify compliance with PEC aflatoxin limits and that the shipper has completed two other pieces of documentation. The PEC certificate must be signed by SPI no more than seven (7) days before the shipping date documented on the trucking bill of lading moving the container from the packer facility to the port. Another unique feature of the PEC program is that the Board monitors the aflatoxin levels in accepted/rejected consignments reported through the ePEC system along with EU rejection rates and may reduce the aflatoxin limits below the current 8 ppb B1 and 10 ppb total to minimize EU rejections.
Breakout Sessions
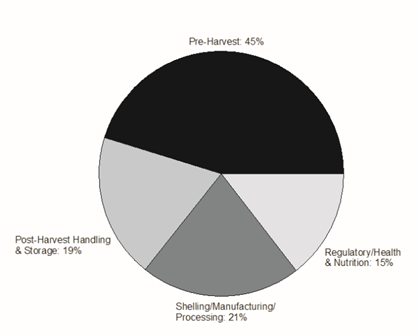
There were several areas of concern and knowledge gaps under each topic area and voting results for each are shown in Table 1. A total of 241 votes were cast in ranking the areas of concern. Overall, 45% of the votes were cast for Pre-Harvest, 19% for the Post-Harvest Handling and Storage area, 21% in the Shelling, Manufacturing and Processing, and 15% of the votes cast were in the Regulatory, Health, and Nutrition discussion area (Figure 5).
Pre-Harvest
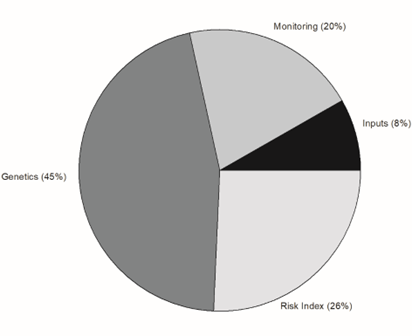
The Pre-Harvest category had four primary areas of concern, 1) Inputs, 2) Monitoring, 3) Genetics, and 4) Risk Index. Genetics received 50 votes with Developing/Improving Aflatoxin Risk Indices receiving 28 votes, Monitoring receiving 22 votes (Figure 6). The Inputs category received the least number of votes (9 votes).
The overall area of genetics received 6 votes in addition to the subtopics listed under genetics, Drought Tolerance, Seed Coat/Shell Characteristics, Nematode/Insect Tolerance, and Maturity. Genetic development of drought tolerance received 30 out of 52 votes in Genetics while seed coat/shell characteristics had 9 votes and transgenic technology received 4 votes.
The next largest area (28 votes) in the Pre-Harvest topic area was the development or improvement of a pre-harvest aflatoxin risk index for the purpose of applying or performing in-season mitigation practices to reduce aflatoxin contamination. Monitoring field conditions (what, when, and where) to manage aflatoxin risk garnered 14 out of the 22 overall votes in the Monitoring category.
Post-Harvest Handling and Storage
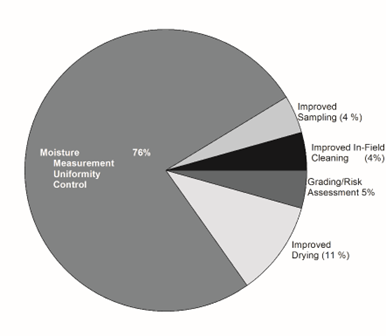
The Post-harvest Handling and Storage category received a total of 46 votes (Table 1) with the 74% (35) cast in the area moisture measurement, uniformity, and control. Five votes (11%) were cast for improved drying (Figure 7) which could be included in the broad category of moisture control and uniformity. Other areas discussed in the breakout sessions were improved sampling, in-field cleaning, and the general category of grading with each receiving 2 votes each (4.3%). In the discussions of grading in the breakout sessions it was noted that loads designated as SEG III loads upon initial farmers stock inspection and then subsequently cleaned, re-inspected, and re-classified as SEG I could pose a higher risk than those loads that initially graded as SEG I. The unanswered question was raised in at least one of the rotations, “What is the specific range of storage temperature and relative humidity coupled with peanut moisture content and/or water activity that causes mold growth and aflatoxin accumulation?”
Shelling, Manufacturing, and Processing
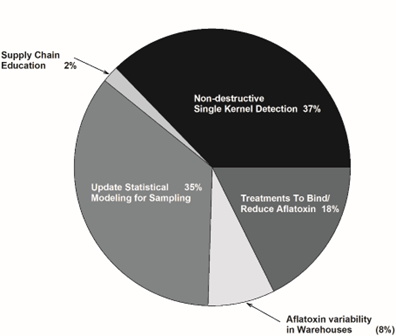
The relative voting for the Shelling/Manufacturing/Processing topic area is shown in Figure 8. There was some topics in Shelling, Manufacturing, and Processing area that overlapped with topics of discussion in the Post-Harvest Handling and Storage area. For instance, aflatoxin variability in farmers stock warehouses could have been moved into the Post-Harvest Handling and Storage topic area received four votes. The topics of statistical modelling to develop sampling plans (18 votes) and development/implementation of non-destructive, high-throughput single kernel aflatoxin detection (19 votes) had similar results. A distant second place in the polling was the uses of chemical treatments to reduce aflatoxin contamination in shelled product (9 votes).
Regulatory, Health, and Nutrition
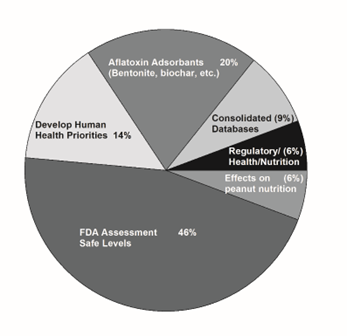
The item that drew the most interest (46%) was the need for FDA to conduct a risk assessment relative to human health and establish acceptable levels of aflatoxin. There was also interest (7 votes or 20%) in the effects aflatoxin on human health including cancer and the acute and chronic illnesses associated with ingestion of contaminated products. Adding inert ingredients such as bentonite clay and activated charcoal to adsorb and bind the aflatoxin received 18% of the votes category of Regulatory/Health and Nutrition (Figure 9). However, this could be combined with use of chemical treatments to reduce aflatoxin in shelled product for a total of 16 votes (Table 1).
Summary
The Peanut Quality Symposium was an industry effort to describe the challenge that aflatoxin contamination presents to the peanut industry and to identify gaps in technology and knowledge required to minimize aflatoxin’s impact on the industry. While attendees represented all segments of the peanut industry, the survey results presented here represent the consensus of only those attending the symposium. In summary, a consensus of those in attendance was that aflatoxin contamination in peanut is an ongoing and economic problem that must be addressed in a concerted ongoing effort by all the industry. It is in the best interest of growers, shellers, manufacturers, and consumers to minimize aflatoxin contamination at each step in the chain of custody. Focused research efforts were identified in production, harvesting, handling and processing. Genetics especially in drought tolerance, building on the previous genomics work that the industry has invested in should be a continued focus in the pre-harvest area. Research to develop and or improve risk indices that can pre-emptively allow growers to modify production and/or harvest practices is needed. In the area of post-harvest processing and storage, moisture management is crucial including systems to measure/monitor and provide uniformity. There is considerable interest in the development of and use of non-destructive single kernel detection of aflatoxin and should be pursued. Finally, in the regulatory and nutrition realm, standards for detection and specification should be science-based including obtaining and testing a sample that is fully representative of the peanut lot. While research in genetics and variety development was indicated as the best hope for managing aflatoxin contamination, this workshop identified several other areas where research is needed and improvements in the current systems can be made to mitigate aflatoxin contamination.
Literature Cited
Almond Board of California. 2022. Global Markets Overview. 07 Jun. https://www.almonds.com/almond-industry/regulatory-and-trade/global-markets-overview.
Almond Board of California. 2020. “Pre-Export Checks (PEC) Program Manual.” https://www.almonds.com/sites/default/files/2020-08/PEC%20Program%20Manual%20v.11.0.pdf.
American Peanut Council. 2022. The Peanut Industry. 06 Jun. https://www.peanutsusa.com/about-apc/the-peanut-industry.html.
Coulibaly, O, K, Bandyopadhyay, R Hell, S Hounkponou, and J F Leslie. 2008. “Economic impact of aflatoxin contamination in Sub-Saharan Africa.” In Mycotoxins: Detection Methods, Management, Public Health and Agricultural Trade, edited by J F Leslie, R Bandyopadhyay and A. Visconti, 67-76. CAB International.
EFSA. 2018. “Scientific statement on the effect on public health of a possible increase of the maximum level for ‘aflatoxin total’ from 4 to 10 µg/kg in peanuts and processed products thereof intended for direct human consumption or use as an ingredient in foodstuffs.” EFSA Journal (European Food Safety Authority) 16: 5175.
Georgia Federal-State Inspection Service. 2020. National Peanut Tonnage Report: Crop Year 2019. Georgia Federal-State Inspection Service. https://www.gafsis.com/tonnage/US/TN11/US2019Final.pdf.
International Agency for Research on Cancer 2012. Aflatoxins, IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 100F. Lyon, France: World Health Organization. https://publications.iarc.fr/123.
JECFA. 2017. Evaluation of certain contaminants in food. Eighty-third report of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). Food and Agriculture Organization of the United Nations, WHO Technical Report Series 1002. Geneva: World Health Organization.
Lamb M.C., and Sternitzke D.A.. 2001. “Cost of Aflatoxin to the Farmer, Buying Point, and Sheller Segments of the Southeast United States Peanut Industry.” Peanut Sci. 28 (2): 59-63. doi:10.3146/i0095-3679-28-2-4.
Lamb M.C., Sorensen R.B., and Butts C.L.. 2021. “Cost of Aflatoxin to the United States Industry.” Annual Meeting of the Am. Peanut Res. Educ. Soc. Online Conference: Am. Peanut Res. Educ. Soc. https://apresinc.com/wp-content/uploads/2021/06/LambM-Abstract-2021.pdf.
Lynch R E, and D M Wilson 1991. “Enhanced Infection of Peanut, Arachis hypogaea L., Seeds with Aspergillus flavus Group Fungi Due to External Scarification of Peanut Pods by the Lesser Cornstalk Borer, Elasmopalpus lignosellus (Zeller).” Peanut Sci 18 (2): 110-116. doi:https://doi.org/10.3146/i0095-3679-18-2-13.
Marchese, S, A Polo, A Ariano, S Velotto, S Costantini, and L Severino. 2018. “Aflatoxin B1 and M1: Biological Properties and Their Involvement in Cancer Development.” Toxins 10 (6): 214. doi:https://doi.org/10.3390/toxins10060214.
Michailides T J, Siegel J, Doster M A, Boekler L, Eveillard H, Charbaut T, Felts D, and Reyes H. 2007. “Aspergillus flavus/A. parasiticus Levels in Almond Orchards and on Navel Orangeworm (A Major Pest of Almonds and Pistachios).” Proceedings of the 2007 Annual Multicrop Aflatoxin/Fumonisin Elimination & Fungal Genomics Workshop. Atlanta, GA: USDA, ARS. 67. https://www.ars.usda.gov/ARSUserFiles/20300530/AEW/AEW2007.pdf.
USDA. 2017. Farmers Stock Peanuts: Inspection Instructions. Washington, DC: Specialty Crops Program, USDA, Agricultural Marketing Service, 90.
USDA. 2019. FSA Handbook: Peanut Buyers and Handlers Program Guidelines for 2019 and Subsequent Crop Years. Washington, DC: USDA, Farm Service Agency. https://www.fsa.usda.gov/Internet/FSA_File/1ppg-a1.pdf.
USDA. 2020. Milled Peanuts: Inspection Instructions. Government, Washington, DC: USDA, Agricultural Marketing Service, Fruit and Vegetable Division, p. 75.
USDA. 2022. Percent of Edibile Peanut Lots Exceeding 15 ppb Aflatoxin. Washington, DC: US Department of Agriculture, Agricultural Marketing Service, Special Crops Procurement, Market Development Division.
Whitaker T. B., Birmingham T., Adams J., Jacobs M., and Gray G.. 2010. “Correlation Between Aflatoxin Contamination and Various USDA Grade Categories of Shelled Almonds.” J. AOAC Int. 93 (3): 943-947.
Whitaker T.B., Hagler W.M., and Giesbrecht F.G.. 1999. “Performance of Sampling Plans to Determine Aflatoxin in Farmers' Stock Peanut Lots by Measuring Aflatoxin in High Risk Grade Components.” J. AOAC Int. 82 (2): 264-270.
World Health Organization. International Agency for Research on Cancer. 1993. Aflatoxins. Vol. 56, in IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, 245-395. Lyon, France: IARC.
Notes
- Research Agricultural Engineer (retired) and Research Leader, respectively, USDA ARS National Peanut Research Laboratory, Dawson, GA 39842 [^]
- President/CEO and Director of Sales and Industry Relations, Premium Peanut, Douglas, GA 31535 [^]
- Director, Feed the Future Innovation Lab for Peanut, University of Georgia, Athens, GA 30602 [^]
- Vice President for Global Technical & Regulatory Affairs, Almond Board of California, Modesto, CA 95354 [^]
- Director of Food Safety and Quality, Birdsong Peanuts, Blakely, GA 39823 [^]
- Professor, Agronomy, University of Florida, North Florida Research and Education Center, Marianna, FL 32446 [^]
- Professor, Plant Pathology, University of Georgia-Tifton Campus, Tifton, GA 31793 [^]
- Professor and consultant, Lubbock Christian University, Lubbock, TX 79407 [^]
- President and Director Technical Services, respectively, JLA International, Albany, GA 31721 [^]
- Research Director, The Peanut Institute, Albany, GA 31708 [^]
- Owner, JEE Food Consulting, LLC, Cincinnati, OH 45241 [^] Corresponding Author email: fullmoonengineer@att.net