INTRODUCTION
In Ghana, there has been an increase in the development and release of peanut cultivars in recent years. Ghana’s seed sector is characterized by two dominant seed systems from which peanut farmers obtain seeds for production: the formal seed system and the informal/farmers’ seed system. The formal seed system involves the production and purchase of commercial certified seed. Major activities under this system include development of crop varieties, approval and registration of varieties, production of breeder and foundation seed, production of commercial seeds, as well as quality control and certification of seeds (Etwire et al., 2013; Tripp and Mensah-Bonsu, 2013). Crops Research Institute (CRI) and Savanna Agricultural Research Institute (SARI) are responsible for developing improved varieties of peanut, while the National Variety Release and Registration Committee (NVRRC) releases the varieties. Typically, the Ministry of Food and Agriculture through the extension department are formally responsible for dissemination of the released improved varieties (Asiedu-Darko, 2014). For improved peanut cultivars, it is important to maintain genetic purity of cultivars so that yield and quality attributes are predictable throughout the peanut value chain. However, it is estimated that less than 5% of peanut cultivated in Ghana are certified seed sourced from the formal seed system (Puozaa et al., 2021) while the vast majority of seed is supplied by the informal seed system (e. g., farmer saved seeds, exchanges with other farmers and the grain markets).
Genetic quality entails the underlying genetic potential of the cultivar and determines yield potential (Biemond, 2013). A high level of genetic purity in crop cultivars or hybrids is required to ensure that the advances in productivity and quality imparted by breeders are delivered to the farmer and ultimately to the consumer (Smith and Register, 2008). Knowing the overall quality status of a seed helps the farmer with important crop input decisions during production. Farmers expect high-quality, genetically pure seeds. To ensure this, seed testing is required so that farmers receive the quality of seed they desire. Seed testing ensures that seeds meet minimum quality standards and minimizes the risk of crop failure and mitigate the negative effects of using seed contaminated with weed seeds, pathogens and inert matter (FAO, 2018). However, in Ghana, testing the genetic purity of seeds by farmers before planting is seldom implemented and thus genetic purity of peanut cultivars is unknown once received by farmers and incorporated in the informal seed system. According to Ibrahim and Florkowski (2015), most farmers involved in on-field trials with improved varieties share the seeds with other farmers even before the variety is released, which leads to the contamination of the seed prior to release. For the farmers who are able to purchase seeds of the improved peanut varieties from the formal seed system, it often takes about 10 years to replace initial seeds (Anonymous, 2016). Additionally, through seed diffusion between farmers and cultural practices including selection and seed storage conditions, diversity among seeds produced by individual plants within a cultivar can shift (Thomas et al., 2012). Ibrahim and Florkowski (2015) reported that both peanut farmers and traders in northern part of Ghana are not able to distinguish among varieties and often give descriptive names to varieties other than the original, official name. For instance, ‘Simbaligu’, which literally means ‘small kernels’ is the name given to Shitaochi because of its small kernel size.
Goals of cultivar testing includes identifying the cultivar, discriminating between cultivars, and documenting genetic purity of the cultivar (Powell, 2009). Depending on the objective, approaches to cultivar testing can include grow-out tests, biochemical methods, and DNA-based methods. Historically, genetic purity testing was achieved through plant grow-outs in the field that relies on the visual identification of plant morphological characteristics (Ballester and de Vincente, 1998; Della Vecchia et al., 1998). However, this method is time consuming, labor intensive, and space demanding (Liwang et al., 2004). Recently, genetic identity and purity testing have shifted to DNA-based molecular marker methods (Lai et al., 2015).
Molecular markers, which are based on variation in DNA sequence, provide an unbiased and objective way of identifying cultivars (Bora et al., 2016). While numerous molecular markers are available for variety identification and genetic purity studies, simple sequence repeats (SSR) have been widely employed. This is largely based on characterization by co-dominance, multiallelic variation, relative abundance, reproducibility, and good genome coverage (Lu et al., 2018; Stachel et al., 2000).
Simple sequence repeats are short tandem repeat motifs that may vary in the number of repeats at a given locus (Tautz, 1989). The use of SSR as markers is based on the differences in repetition of usually two or three nucleotide bases (Ghosh et al., 2014). SSR markers have been used to differentiate accessions of the peanut species (Kottapalli et al., 2007), genetic diversity in peanut (Oteng-Frimpong et al., 2015; Patel et al., 2015; Tang et al., 2007), QTL analysis (Khedikar et al., 2010; Zhao et al., 2016), and construction of genetic linkage maps (Li et al., 2019; Qin et al., 2012). Also, SSR markers have been successfully used to assess the genetic purity of farmer-saved seeds in soybean [Glycine max (L.) Merr.] (Meesang et al., 2001), commercial hybrids of soybean (Li et al., 2019; Zhang et al., 2014), corn (Zea mays L.) (Chaudhary et al., 2018; Daniel et al., 2012; Elçi and Hançer, 2015), and rice (Oryza sativa L.) (Kumar et al., 2012; Yashitola et al., 2002; Ye-Yun et al., 2005).
The cultivar Shitoachi was released in Ghana more than 40 years ago (NVRRC, 2019) and is currently estimated to be grown on about 80 to 90% of land area devoted to peanut cultivation in Ghana (Owusu-Akyaw et al., 2019). More recently, the cultivar Yenyawoso was released in Ghana in 2012 and is estimated to planted on a limited amount of ha in the country (Owusu-Akyaw et al., 2019). Using these two cultivars as references, the objective of this research was to estimate genetic purity of farmer-saved seeds using SSR markers for the cultivar Yenyawoso in a relatively short period of time after release. Should contamination of seed be observed for this cultivar, the cultivar Shitoachi would be the likely source of adulteration in the informal seed system in Ghana.
MATERIALS AND METHOD
Seeds of two commercially available peanut cultivars in Ghana were collected from farmers and research institutes to determine genetic similarity using SSR. Seeds from 28 farmers were collected with 20 being the cultivar Shitaochi and eight being Yenyawoso. One seed sample of Shitaochi and two samples of Yenyawoso were collected from the Council of Scientific and Industrial Research’s Savanna Agricultural Research Institute (CSIR-SARI) and Crops Research Institute (CSIR-CRI) as standards against the farmer-saved samples.
For each accession, fifteen seeds were randomly selected and sown in plastic containers in the greenhouse. DNA was extracted from three recently expanded leaflets from 10 individual plants for each sample. Total genomic DNA was isolated following the Cetyl-trimethyl ammonium bromide (CTAB) procedure (Cuc et al., 2008).
Quality and quantity of the extracted DNA were assessed using a UV-Vis Spectrophotometer Nanodrop 2000 (Thermo Scientific) and diluted to final concentration of 100 ng/µl. DNA from 10 plants representing one sample were bulked prior to Polymerase Chain Reaction (PCR) analysis.
A total of twenty-five SSR primer pairs were randomly selected from the existing literature on the basis of their polymorphic nature. Following screening for their ability to discriminate between Shitaochi and Yenyawoso cultivars, four polymorphic primers were selected for genetic analysis of the 28 farmer-saved seed samples (Table 1).
Polymerase Chain Reaction (PCR) amplification was performed using 20 µl of PCR mixture solution containing 10 µl of 1X PCR master mix (SBS), 1 µl of each primer (20 µM) (forward and reverse), and 100 ng of genomic DNA templates. The PCR program was set at initial denaturation temperature of 94 ℃ for 5 minutes, followed by denaturation at temperature of 95 ℃ for 1 minute, 51-60 ℃ for annealing for 45 seconds, based on the primers, 72 ℃ for 45 seconds for extension, all run for 35 cycles and final extension cycle at 72 ℃ for 8 minutes, and then an indefinite hold at 4 ℃.
Amplified products from 5 µl of the PCR reaction were separated by electrophoresis in a 6 % polyacrylamide gel together with a Quick-Load Purple 100 bp DNA ladder (New England Biolabs), for 75 minutes at 80 V and 300 amh in 1x TBE buffer. Gels were visualized using an Alpha Imager TM 2200 (AlphaInnotech Inc., San Leandro, CA) gel documentation system.
Gel images were scored for the presence (1) and absence (0) of SSR bands manually. The band scores were used to generate a similarity data matrix using Simple Matching (SM) coefficient. A dendrogram was constructed from the similarity coefficient values following the unweighted pair-group method with arithmetic average (UPGMA) technique in NTSYS-pc version 2.20v (Rohlf, 2009). The Sequential Agglomerative Hierarchical and Nested (SAHN) method was adopted for clustering.
RESULTS AND DISCUSSION
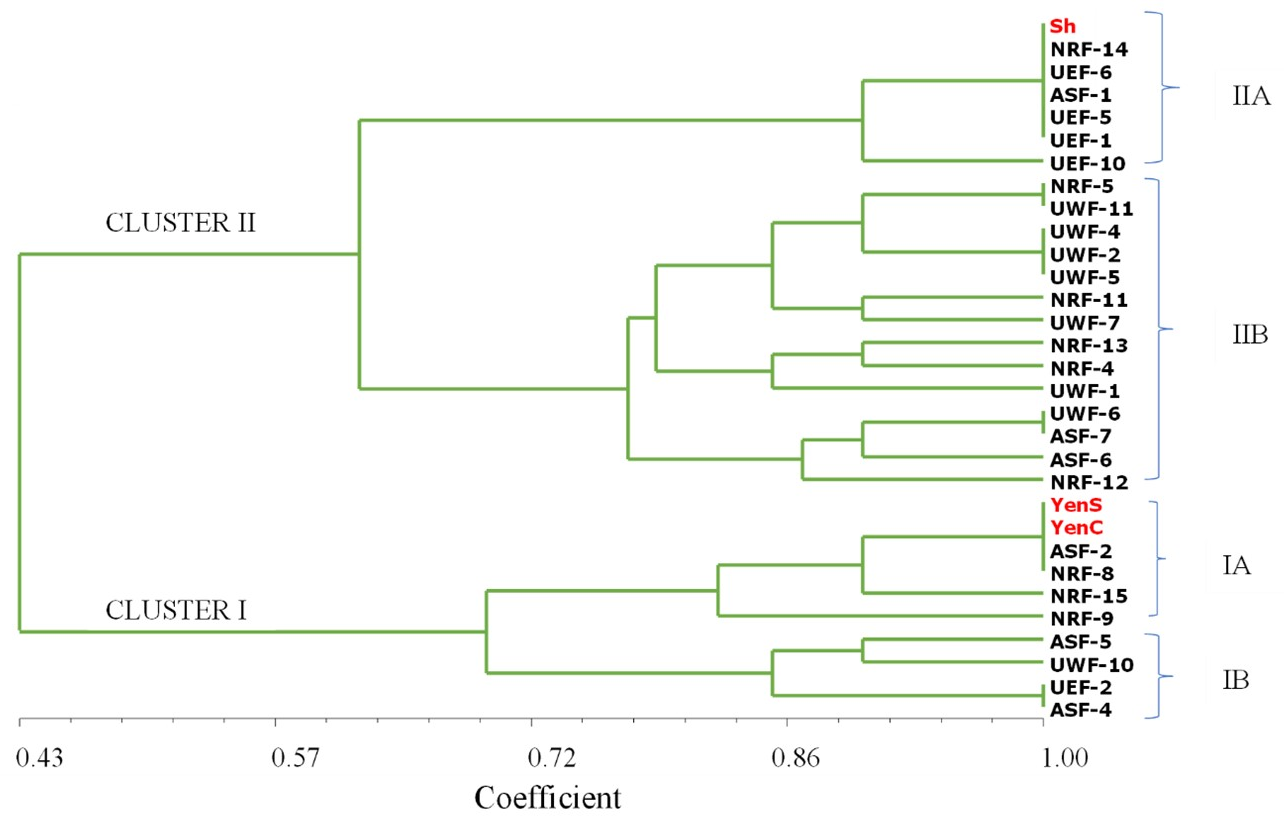
The UPGMA dendrogram based on simple matching coefficients of binary scores of polymorphic bands obtained from the 21 seed lots of Shitaochi and 10 seed lots of Yenyawoso is provided in Figure 1. Genetic similarity values ranged from 0.43 to 1.0. Overall, cluster analysis put the seed lots into one group at 43% similarity but subsequently separated them into two clusters at 0.6 coefficient of similarity. Two Yenyawoso seed lots from CSIR-SARI and CSIR-CRI (e. g., Yen-S and Yen-C, respectively) were grouped in Cluster I along with eight farmer-saved Yenyawoso seed lots (ASF-2, NRF-8, NRF-15, NRF-9, ASF-5, ASF-4 and UWF-10) and one farmer-saved Shitaochi seed lot (UEF-2). Cluster II similarly contained the Shitaochi seed lot obtained from CSIR-SARI (Sh), nineteen farmer-saved Shitaochi seed lots (NRF-14, UEF-6, ASF-1, UEF-5, UEF-1, UEF-10, NRF-5, UWF-11, UWF-4, UWF-2, UWF-5, NRF-11, UWF-7, NRF-13, NRF-4, UWF-6, ASF-7, ASF-6 and NRF-12), and one farmer-saved Yenyawoso seed lot (UWF-1).
Figure 1. Dendrogram showing genetic relationship among 31 peanut seed lots generated using UPGMA based on Simple Matching (SM) coefficient. Reference samples: Sh-‘Chitaochi’, YenS-YenyawosoSARI, YenC-YenyawosoCRI; Farmer samples: UWF-Upper West Farmer, UEF- Upper East Farmer, NRF-Northern Region Farmer, ASF-Ashanti Region Farmer.
In the screening of twenty-five SSR markers for polymorphic markers, only four (16%) discriminated between Shitaochi and Yenyawoso. These include Seq17F6, Seq5D5, PM50, and Seq2B10. This suggests there is a low level of polymorphism between these two peanut cultivars. The results, however, corroborate the findings of Gaikpa et al. (2015) who reported a low level of polymorphism between Shitaochi and Yenyawoso using the SSR primer Seq17F6.
Farmers select varieties on the basis of their agricultural characteristics such as resistance to biotic and abiotic stresses, or their productivity and the recognized value of their products (Powell, 2009). As such, it is pertinent that farmers obtain seeds of good genetic quality pertaining to their chosen variety. Out of the twenty seed lots reported by the farmers to be the Shitaochi peanut cultivar in this study, only five were completely genetically similar (NRF-14, UEF-6, ASF-1, UEF-5 and UEF-1) to the reference Shitaochi seed in cluster I. With regards to Yenyawoso farmer-saved seeds, only two samples were 100% genetically similar (ASF-2 and NRF-8) to the reference Yenyawoso seed samples. This is indicative that these seed samples are very similar to the original breeder seeds and thus have not changed in the hands of the farmers during seed production. This also means that the owners of these seed samples have accurately identified the cultivar. The other farmer-saved seed samples that were grouped in the two clusters at a coefficient of similarity less than 1.0 suggests that they are different from the seeds from SARI and CRI, and that their genetic purity has been lost in the informal seed chain. This perhaps is due to inadvertent errors in the identification of cultivars in the informal seed exchange system among farmers. Additionally, different selection methods used by farmers during seed saving processes could be responsible for the deterioration in genetic purity of some of the farmer-saved seeds. Samples UEF-2 and UWF-1 were identified by the farmers in this study to be Shitaochi and Yenyawoso, respectively. However, UEF-2 was grouped in the same cluster as the Yenyawoso reference samples while UWF-1 was grouped in the same cluster as the Shitaochi reference sample. This could be a result of misidentification of the samples by the farmers. Sometimes, farmers name cultivars apart the original name designated by the breeder on different basis: specific characteristics of the cultivar or the person/organization who introduced the cultivar to the community
In conclusion, these results indicate that Yenyawoso and Shitaochi cultivars in the hands of farmers are different genetically when compared to those from the research institutes. Results from this research have limitations due to several factors. First, specific practices for each farmer relative to how seeds were handled previously was not determined. The cause of impurity of seed noted through SSR markers may have been elucidated more clearly with greater information from farmers on their seed sourcing. Secondly, determining if phenotypic differences were present for plants grown from seed used for SSR markers would have been informative and is a common practice when determining genetic differences among cultivars. Additionally, future research should be conducted on the genetic purity of cultivars by employing a greater number of markers due to the low level of polymorphism detected by primer pairs used in this research. The low level of polymorphism was not unexpected based on the origin of Arachis hypogaea. (Seijo et al., 2007). There is also a need to screen more peanut cultivars that have been in cultivation for many years. None-the-less, results from this research provide information on genetic purity of cultivars in the informal seed system in Ghana.
Acknowledgements
This research was supported by the United States Agency for International Development, as part of the Feed the Future initiative, under the CGIAR Fund, award number BFS-G-11-00002, the predecessor fund the Food Security and Crisis Mitigation II grant, award number EEM-G-00-04-00013, and the Office of Agriculture, Research and Policy, Bureau of Food Security, U.S. Agency for International Development, under the terms of Award No. AID-ECG-A-00-07-0001 to The University of Georgia as management entity for U.S. Feed the Future Innovation Lab for Peanut. The opinions expressed herein are those of the authors and do not necessarily reflect the views of the U.S. Agency for International Development.
Literature Cited
Anonymous. 2016. Ghana early generation seed study. AGRA-SSTP. Available at: https://www.agrilinks.org/sites/default/files/resource/files/ghana_early_generation_seed_report.pdf.
Asiedu-Darko E. 2014. Farmers’ perception on agricultural technologies a case of some improved crop varieties in Ghana. Agriculture, Forestry and Fisheries. 3(1):13-16.
Ballester J. and de Vicente M. C.. 1998. Determination of F1 hybrid seed purity in pepper using PCR-based markers. Euphytica. 103(2):223-226.
Biemond P. C. 2013. Seed quality in informal seed systems. PhD thesis, Wageningen University, Wageningen, Netherlands. 120 pages.
Bora A., Choudhury P. R., Pande V., and Mandal A. B.. 2016. Assessment of genetic purity in rice (Oryza sativa L.) hybrids using microsatellite markers. 3 Biotech. 6(1):50.
Chaudhary S., Kharb P., Yashveer S., Prajapati D. R., and Kamboj M. C.. 2018. Utilization of SSR markers for studying genetic diversity of popular maize and their hybrids. Bull. Env. Pharmacol. Life Science. 7:36-42.
Cuc L. M., Mace E. S., Crouch J. H., Quang V. D., Long T. D., and Varshney R. K.. 2008. Isolation and characterization of novel microsatellite markers and their application for diversity assessment in cultivated groundnut (Arachis hypogaea). BMC Plant Biology. 8(1):1-11.
Daniel I. O., Adetumbi J. A., Oyelakin O. O., Olakojo S. A., Ajala M. O., and Onagbesan S. O.. 2012. Application of SSR markers for genetic purity analysis of parental inbred lines and some commercial hybrid maize (Zea mays L.). Journal of Experimental Agriculture International. 2(4):597-606.
Vecchia Della, P. T., C. A. R. Da Silva, and P. Terenciano-Sobrinho. 1998. Use of molecular marker techniques in seed testing by Brazilian seed companies. Scientia Agricola. 55:79-82.
El çi, E., and T. Hançer. 2015. Genetic analysis of maize (Zea mays L.) hybrids using microsatellite markers. Journal of Agricultural Sciences. 21:192-198.
Etwire P. M., Atokple I. D., Buah S. S., Abdulai A. L., Karikari A. S., and Asungre P.. 2013. Analysis of the seed system in Ghana. International Journal of Advance Agricultural Research. 1(1):7-13.
FAO [Food and Agriculture Organization]. 2018. Farmer seed systems and sustaining peace. Rome. 52 pages.
Ferguson M. E., Burow M. D., Schulze S. R., Bramel P. J., Paterson A. H., Kresovich S., and Mitchell S.. 2004. Microsatellite identification and characterization in peanut (Arachis hypogaea L.). Theoretical and Applied Genetics. 108:1064-1070.
Gaikpa D. S., Akromah R., Asibuo J. Y., and Nyadanu D.. 2015. Studies on molecular variation in commercially cultivated groundnuts (Arachis hypogaea L.) using SSR markers. The International Journal of Science and Technoledge. 3(2):80.
Ghosh J., Ghosh P. D., and Choudhury P. R.. 2014. An assessment of genetic relatedness between soybean [Glycine max (L.) Merr.] cultivars using SSR markers. American Journal of Plant Sciences. 5(20):3089.
Guo Y., Khanal S., Tang S., Bowers J. E., Heesacker A. F., Khalilian N., Nagy E. D., Zhang D., Taylor C. A., Stalker H. T., and Ozias-Akins P.. 2012. Comparative mapping in intraspecific populations uncovers a high degree of macrosynteny between A-and B-genome diploid species of peanut. BMC Genomics. 13(1):1-12.
He G., Meng R., Newman M., Gao G., Pittman R.N., and Prakash C.. 2003. Microsatellites as DNA markers in cultivated peanut (Arachis hypogaea L.). BMC Plant Biology. 3(1):1-6.
Ibrahim M. and Florkowski W.. 2015. Analysis of farmers’ willingness to adopt improved peanut varieties in northern Ghana with the use of baseline survey data. AgEcon Search: Research in Agricultural & Applied Economics. 10.22004/ag.econ.197049.
Khedikar Y. P., Gowda M.V.C., Sarvamangala C., Patgar K.V., Upadhyaya H.D., and Varshney R.K.. 2010. A QTL study on late leaf spot and rust revealed one major QTL for molecular breeding for rust resistance in groundnut (Arachis hypogaea L.) Theoretical and Applied Genetics. 121(5):971-984.
Kottapalli K. R., Burow M. D., Burow G., Burke J., and Puppala N.. 2007. Molecular characterization of the US groundnut mini core collection using microsatellite markers. Crop Science. 47(4):1718-1727.
Kumar M. C., Vishwanath K., Shivakumar N., Rajendra Prasad S., Radha B. N., and Ramegowda B. N.. 2012. Utilization of SSR markers for seed purity testing in popular rice hybrids (Oryza sativa L.). Annals of Plant Science. 1:1-5.
Lai K., Lorenc M. T., and Edwards D.. 2015. Molecular marker databases. Pages 49-62 In J. Batley, ed. Plant Genotyping: Methods and Protocols. Springer New York: New York, NY, USA.
Li F., Liu X., Wu S., Luo Q., and Yu B.. 2019. Hybrid identification for Glycine max and Glycine soja with SSR markers and analysis of salt tolerance. PeerJ. 7. e6483.
Li L., Yang X., Cui S., Mu G., Hou M., He M., and Liu L.. 2019. Construction of high density genetic map and mapping quantitative trait loci for growth habit related traits of groundnut (Arachis hypogaea L.). Frontiers in Plant Science. 10:745.
Liwang L., Xilin H., Yiqin G., Yunming Z., Kairong W., and Junfei Z.. 2004. Application of molecular marker in variety identification and purity testing in vegetable crops. Molecular Plant Breeding. 2(4):563-568.
Lu X., Adedze Y. M. N., Chofong G. N., Gandeka M., Deng Z., Teng L., and Li W.. 2018. Identification of high-efficiency SSR markers for assessing watermelon genetic purity. Journal of Genetics. 97(5):1295-1306.
Meesang N., Ranamukhaarachchi S. L., Petersen M. J., and Andersen S. B.. 2001. Soybean cultivar identification and genetic purity analysis using microsattelite DNA markers. Seed Science and Technology. 29(3):637-646.
Moretzsohn M. C., Hopkins M. S., Mitchell S. E., Kresovich S., Valls J. F. M., and Ferreira M. E.. 2004. Genetic diversity of peanut (Arachis hypogaea L.) and its wild relatives based on the analysis of hypervariable regions of the genome. BMC Plant Biology. 4:11.
NVRRC [National Variety Release and Registration Committee] 2019. Catalogue of crop varieties released and registered in Ghana. Available at: https://nastag.org/docx/resources/2019%20NATIONAL%20CROP%20VARIETY%20CATALOGUE.pdf.
Oteng-Frimpong R., Sriswathi M., Ntare B. R., and Dakor F. D.. 2015. Assessing the genetic diversity of 48 groundnut (Arachis hypogaea L.) genotypes in the Guinea savanna agro-ecology of Ghana, using microsatellite-based markers. African Journal of Biotechnology. 14(32):2485-2485.
Owusu–Akyaw M., Mochiah M. B., Asibuo J. Y., Osei K., Ibrahim A., Bolfrey Arku G., Lamptey J. N. L. Danyi A. A., Oppong A., Addo J. K., Boateng M. K., Adu-Dapaah H. K., Addy S., Amoah S., Osei-Yeboah S., Abudulai M., Denwar N., Naab J., Mahama G., Akroma R., Brandenburg R. L., Bailey J. E., Jordan D. L., Williams T. H., Hoisington D., and Rhoads J.. 2019. Evaluation and release of two peanut cultivars: a case study of partnerships in Ghana. Peanut Science. 46(1):37-41.
Patel S. V., Mandavia M. K., and Golakiya B. A.. 2015. Assessment of genetic diversity among groundnut (Arachis hypogaea L.) genotypes using molecular markers. Indian Journal of Agricultural Biochemistry. 28(1):70-76.
Puozaa D. K., Jinbaani A. N., Adogoba D. S., Busagri D., Rasheed M. A., Issah A. R., and Oteng-Frimpong R.. 2021. Enhancing access to quality seed of improved groundnut varieties through multi-stakeholder platforms in Northern Ghana. Pages 65-69 In Enhancing Smallholder Farmers' Access to Seed of Improved Legume Varieties Through Multi-stakeholder Platforms. Springer, Singapore.
Powell A. A. 2009. What is seed quality and how to measure it. Paper presented at the PROC. 2nd World Seed Conf. Responding to the challenges of a changing world: The role of new plant varieties and high-quality seed in agriculture, FAO Headquarters, Rome, Italy.
Qin H., Feng S., Chen C., Guo Y., Knapp S., Culbreath A., He G., Wang M. L., Zhang X., Holbrook C. C., and Ozias-Akins P.. 2012. An integrated genetic linkage map of cultivated groundnut (Arachis hypogaea L.) constructed from two RIL populations. Theoretical and Applied Genetics. 124(4):653-664.
Seijo G., Lavia G.J., Fernandez A., Krapovickas A., Ducasse D.A., Bertioli D.J., and Moscone E.A. . 2007. Genetic relationship between the cultivated peanut (A. hypogaea, Leguminosae) and its close relatives revealed by GISH. American Journal of Botany. 94 ():1963-1971.
Smith J., and Register J.. 1998. Genetic purity and testing technologies for seed quality: A company perspective. Seed Science Research. 8(2):285-294.
Stachel M., Lelley T., Grausgruber H., and Vollmann J.. 2000. Application of microsatellites in wheat (Triticum aestivum L.) for studying genetic differentiation caused by selection for adaptation and use. Theoretical and Applied Genetics. 100(2):242-248.
Tang R., Gao G., He L., Han Z., Shan S., Zhong R., Zhou C., Jiang J., Li Y., and Zhuang W.. 2007. Genetic diversity in cultivated groundnut based on SSR markers. Journal of Genetics and Genomics. 34(5):449-459.
Tautz D. 1989. Hypervariability of simple sequences as a general source for polymorphic DNA markers. Nucleic Acids Research. 17(16):6463-6471.
Thomas M., Demeulenaere E., Dawson J. C., Khan A. R., Galic N., Jouanne S.‐Pin, C. Remoué, C. Bonneuil, and I. Goldringer. 2012. On‐farm dynamic management of genetic diversity: the impact of seed diffusions and seed saving practices on a population‐variety of bread wheat. Evolutionary Applications. 5(8):779-795.
Tripp R., and Mensah-Bonsu A.. 2013. Ghana's commercial seed sector: New incentives or continued complacency? GSSP working papers 32, International Food Policy Research Institute (IFPRI). 22 pages. Available at: https://www.ifpri.org/publication/ghanas-commercial-seed-sector-new-incentives-or-continued-complacency.
Wang C. T., Yang X. D., Chen D. X., Yu S. L., Liu G. Z., Tang Y. Y., and Xu J. Z.. 2007. Isolation of simple sequence repeats from groundnut. Electronic Journal of Biotechnology. 10(3):473-480.
Yashitola J., Thirumurugan T., Sundaram R. M., Naseerullah M. K., Ramesha M. S., Sarma N. P., and Sonti R. V.. 2002. Assessment of purity of rice hybrids using microsatellite and STS markers. Crop Science. 42(4):1369-1373.
Ye-Yun X., Zhan Z., Yi-Ping X., and Long-Ping Y.. 2005. Identification and purity test of super hybrid rice with SSR molecular markers. Rice Science. 12(1):7-12.
Zhang C. B., Peng B., Zhang W. L., Wang S. M., Sun H., Dong Y. S., and Zhao L. M.. 2014. Application of SSR markers for purity testing of commercial hybrid soybean (Glycine max L.). Journal of Agricultural Science and Technology. 6(6):1389-1396.
Zhao Y., Zhang C., Chen H., Yuan M., Nipper R., Prakash C. S., and He G.. 2016. QTL mapping for bacterial wilt resistance in groundnut (Arachis hypogaea L.). Molecular Breeding. 36(2):13.
Notes
- Author Affiliations
- Department of Horticulture, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana; [^]
- Department of Crop and Soil Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana; [^]
- Department of Agricultural Economics, Agribusiness and Extension, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana; [^]
- Department of Biochemistry and Biotechnology, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana; [^]
- Council for Scientific and Industrial Research, Crops Research Institute, Kumasi, Ghana; [^]
- Feed the Future Innovation Lab for Peanut, University of Georgia, Athens, GA 30602; [^]
- Department of Crop and Soil Sciences, North Carolina State University, Raleigh, NC 27695. [^] Corresponding author’s e-mail: david_jordan@ncsu.edu