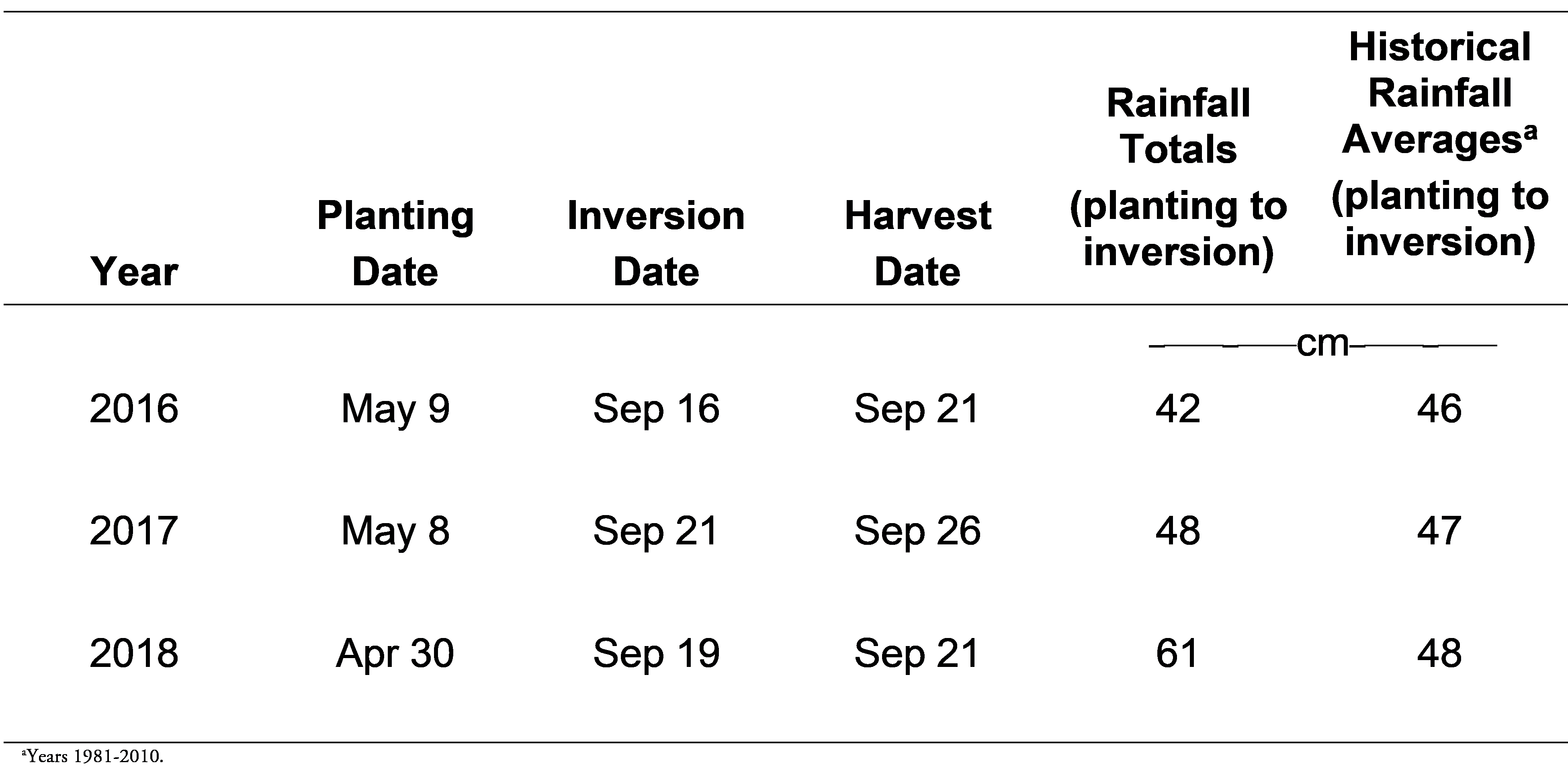INTRODUCTION
Watermelon (Citrullus lanatus Thunb.) is an important high-value crop in Georgia. In 2021, Georgia was the 3rd largest watermelon producing state, accounting for 20% of the total U.S. watermelon production, with a farm gate value of $103,742,000 (USDA 2022). Weed control is critical for producing a high yielding, quality, and profitable watermelon crop. Weeds can be a serious problem in watermelon because of initial slow plant growth, low plant populations, and limited ability for cultivation once plants are established (Elmstrom and Locascio 1974; Larson et al. 2017). Crop rotation, tillage, and a sound herbicide program are all critical components for long-term weed control success in watermelon (Culpepper and Smith 2020). An herbicide with long residual activity to control emerging weeds for the entire watermelon growing season is necessary (Elmstrom 1973). Several University of Georgia recommended watermelon weed control programs include terbacil for control of broadleaf weeds and it is currently being utilized on 70-75% of watermelon hectares (A.S. Culpepper, pers. commun. 2021; Culpepper and Smith 2020). Terbacil is registered for use in alfalfa (Medicago sativa L.), watermelon, caneberries (Rubus fruticosus L.), mint (Mentha species), peach [Prunus persica (L.) Batsch], and several other specialty crops and is sold under the trade name of Sinbar® 80WGD (Anonymous 2021).
Terbacil is a photosystem II inhibiting herbicide that is a member of the substituted uracil family (Shaner 2014). Terbacil is absorbed by roots and transported to its site of action in the mesophyll chloroplasts via the xylem, however it can also penetrate foliar tissue to reach the site of action (Barrentine and Warren 1970; Ashton and Monaco 1991). Its soil residual activity provides control of germinating weeds and germinated weed seedlings (Hu et al. 2016).
Soil organic matter and/or clay content can greatly influence the persistence of terbacil in the soil (Jensen and Kimball 1982; Raham 1977). The average field half-life for terbacil is 120 d (Shaner 2014). However, a half-life of up to six months was reported in a Butlertown silt loam (Gardiner et al. 1969). Another study reported that five to seven months was needed for the amount of soil surface terbacil to be reduced by 50% (Marriage et al. 1977).
Terbacil is primarily degraded by soil microbes (Gardiner et al. 1969; Wolf and Martin 1974). The driving variables for the microbial degradation of herbicides are soil moisture, soil temperature, soil aeration, soil pH, and the nutrient status of the soil (Anderson 1984).
Georgia peanut (Arachis hypogaea L.) growers commonly use crop rotation as a method to help successfully manage weeds, insects, and disease pressure (Higgs et al. 1990; Vencill et al. 2012). Rotating crops can be a difficult task with the variability in crop tolerances to the numerous herbicides that could potentially be used in agronomic and vegetable crop rotations. Currently, the terbacil herbicide label restricts the planting of peanut, along with all other row crops, for two years following the last application of terbacil (Anonymous 2021). The ability to plant peanut 12 months or earlier after a terbacil application would greatly increase the options that a Georgia watermelon grower has for rotational crops. Research was conducted to determine peanut response to terbacil with the ultimate goal of reducing the current two-year crop rotational restriction.
Materials and Methods
Small-plot replicated field trials were conducted at the Ponder Research Farm in Ty Ty, Georgia in 2016, 2017, and 2018 (31.5076540N, -83.6583950W) to determine the effects of direct terbacil applications to peanut. The soil type was a Fuquay sand with 96% sand, 0% silt, 4% clay, 0.57% organic matter, and a pH of 6.6. Conventional tillage practices were used and peanut (cv. Georgia-06G) (Branch 2007) peanut was planted using a vacuum planter calibrated to deliver 18 peanut seed/m at a depth of 5 cm. (Monosem Precision Planters, 1001 Blake St., Edwardsville, KS). Peanuts were planted in twin rows (90 cm X 22 cm spacing) with a plot size of 7.6 m X 0.9 m.
Treatments were arranged in a randomized complete block design and replicated four times. Terbacil was applied preemergence immediately after peanut planting at the following rates: 28, 56, 112, and 224 g ai/ha. Immediately following application, the plot area received 1.25 cm of overhead irrigation for soil incorporation. Terbacil use rates in watermelon range from 112 to 224 g ai/ha, with a maximum use rate per year of 224 g ai/ha (Anonymous 2021). Herbicide treatments were applied using a CO2 – pressurized backpack sprayer calibrated to deliver 140 L/ha at 4.8 km/hr. Plots were maintained weed-free throughout the season using a combination of herbicides (pendimethalin, diclosulam, flumioxazin, imazapic, and 2,4-DB) and hand-weeding. Supplemental over-head irrigation was applied as needed to maintain optimum peanut yields.
Data collected included peanut density (stand) at approximately 30 d after planting (DAP), visual estimates of crop injury, and peanut yield. Peanut density was obtained by counting the number of emerged plants/1 row m. Visual estimates of crop injury were obtained 14, 28, 50, and 100 DAP using a subjective scale of 0 to 100 percent (0=no injury; 100=plant death). Peanut yield data was obtained using commercial harvesting equipment. Yields were adjusted to 10% moisture. A complete summary of planting/harvesting dates and rainfall totals can be found in Table 1.
Data were subjected to ANOVA using PROC GLIMMIX in SAS, version 9.4 (SAS Institute, Cary, NC). Peanut injury, density, and yield were set as the response variables with year and replication within year included in the model as random factors. All data were combined over years. Including year as a random effect allows for estimations of the consistency of the treatment effects across study years, making the conclusion applicable to other years (Moore and Dixon 2014). All P-values for tests of differences between least-squares means were compared and separated using the Tukey-Kramer method (P<0.10).
Results and Discussion
Visual injury Visual injury ratings presented in Table 2 were collected 14, 28, 50, and 100 DAP. With the exception of the 56 g ai/ha rate at 28 DAP, the only rates of terbacil that caused significant visual peanut injury were the 112 g ai/ha and 224 g ai/ha rates. Injury at these rates ranged between 12% and 81%. The 224 g ai/ha rate caused more peanut injury than the 112 g ai/ha rate. Typical terbacil peanut injury symptoms include stunting, veinal chlorosis, and eventual necrosis of the leaf (Figure 1). Visual injury at these rates was anticipated based on previous soybean research (Rahman 1977).
Peanut density and yield Peanut density was not reduced by any rate of terbacil (Table 3). Peanut yields were significantly reduced by terbacil at 112 g ai/ha and 224 g ai/ha. Yield losses at these rates were 28% and 70% respectively. In a related study, soybean yield was reduced 81% from applications of 500 g ai/ha of terbacil on a sandy loam soil (Rahman et al. 1976). In our studies, peanut yield losses from terbacil were only observed from the typical range of watermelon field use rates. It is important to note that rainfall totals for these studies were above average in 2 of 3 years and that supplemental irrigation was applied as needed to maintain optimum peanut yields.
Summary and Conclusions
All rates of terbacil caused visual peanut injury. However, only the 2 highest rates (112 and 224 g ai/ha) resulted in significant peanut yield losses. Assuming normal temperatures, rainfall patterns, and supplemental irrigation programs, these data suggest that peanut could be planted following terbacil after approximately two field half-lives, or ~240 d have occurred in coarse textured soils. In Georgia, watermelon is planted earlier in the spring (March to Mid-April) than peanut (Late-April to Early-June). This would allow for the planting of peanut the following growing season ~365 d after an application of terbacil. If labeled rotation restrictions for terbacil were reduced, watermelon growers in Georgia could diversify their crop rotation options to include peanut. Since the persistence of terbacil is greatly influenced by organic matter, soil type, and microbial degradation, it is possible then that terbacil could persist longer than reported herein in areas with finer soils, higher organic matter contents, cooler temperatures, and less rainfall/supplemental irrigation.
Acknowledgements
This research could not have been conducted without the technical support of Charlie Hilton, Tim Richards, and Dewayne Dales.
Literature Cited
Anderson J.P.E. 1984. Herbicide degradation in soil: Influence of soil microbes. Soil Biol. Biochem. Vol. 16. 5:483-489.
Anonymous. 2021. Sinbar® herbicide product label. Tessenderlou Kerley Inc., Phoenix, AZ. at: https://s3-us-west-1.amazonaws.com/agrian-cg-fs1-production/pdfs/Sinbar_WDG_Label1o.pdf. Accessed: November 1, 2021.
Ashton F.M. and Monaco T.J.. 1991Weed Sci., Principles and Practices, 3rd ed., Wiley, New York.
Barrentine J.L. and Warren G.F.. 1970. Isoparaffinic oil as a carrier for chlorpropham and terbacil. Weed Sci. 18:365–372.
Branch W.D. 2007. Registration of ‘Georgia-06G’ peanut. J. Plant Registrations 1:120.
Culpepper A.S. and Smith J.C.. 2020. University of Georgia Weed Control Programs for Watermelon 2020. University of Georgia Ext. Pub. at: http://www.gaweed.com/HomepageFiles/2020watermelon.pdf. Accessed: November 1, 2021.
Elmstrom G.W. 1973. Evaluation of herbicides for watermelon. Proc. So. Weed Sci. Soc. 26:270-275.
Elmstrom G.W. and Locascio S.J.. 1974. Evaluation of herbicides for watermelon in Florida. Florida Agric. Exp. Sta. Journal Series. No. 5653, Pps. 179-184.
Gardiner J.A., Rhodes R.C., Adams J.B., Soboczenski E.J.. 1969. Synthesis and studies with 2-C-14 labeled bromacil and terbacil. Journal of Agric. and Food Chem. 17:980-986.
Higgs R.L., Peterson A.E., and Paulson W.H.. 1990. Crop rotation: Sustainable and profitable. J. Soil Water Conserv. 45:68–70.
Hu C., Mohsen M.M., and Doohan D.. 2016. Tolerance of strawberry cultivars to terbacil. International Journal of Fruit Sci. 17.1:20-28.
Jensen K.I. and Kimball E.R.. 1982. The comparative behavior of simazine and terbacil in soils. Weed Res. 22:7-12.
Larson B.C., Mossler M.A., and Nesheim O.N.. 2017. Florida Crop/Pest Management Profile: Watermelon. Circ. 1236. Gainesville, FL University of Florida. https://edis.ifas.ufl.edu/pdf/PI/PI03100.pdf. Accessed: November 1, 2021.
Marriage P.B., Khan S.U., and Saidak W.J.. 1977. Resistance and movement of terbacil in peach orchard soil after repeated annual applications. Weed Res. 17: 219–255.
Moore K.J. and Dixon P.M.. 2014. Analysis of combined experiments revisited. Agronomy J. 107:763-771.
Rahman A. 1977. Persistence of terbacil and trifluralin under different soil and climatic conditions. Weed Res. 17:145-52.
Rahman A., Burney B., Whitham J.M. and Manson B.E.. 1976. A comparison of the activity of herbicides in peat and mineral soils. New Zealand Journal of Exp. Agr. 4.1:79-84.
Shaner D.L. 2014Herbicide Handbook. 10th Edition, Weed Science Society of America, Champaign, IL. Pp. 513.
U.S. Department of Agriculture, National Agricultural Statistics Service. 2022. Vegetables 2021 Summary. at: https://downloads.usda.library.cornell.edu/usdaesmis/files/02870v86p/zs25zc490/9593vz15q/vegean22.pdf. Accessed: March 2, 2022.
Vencill, W.K., R.L. Nichols, T.M. Webster, J.K. Soteres, C. Mallory-Smith, N.R. Burgos, W.G. Johnson, and M.R. McClelland. 2012. Herbicide resistance: toward an understanding of resistance development and the impact of herbicide-resistant crops. Weed Sci.: Special Issue Vol. 60.1:2-30.
Wolf, D.C. and J.P. Martin. 1974. Microbial degradation of 2-carbon-14 bromacil and terbacil. Soil Sci. Soc. Am. Proc. 38:921-925.
Notes
Former Graduate Research Assistant, Graduate Research Assistant, and Extension Weed Specialist/Professor, Dept. of Crop Soil Sciences, The University of Georgia, Tifton, GA 31793.
Corresponding author’s Email: eprostko@uga.edu