Introduction
Weeds such as Palmer amaranth ( Amaranthus palmeri S. Wats), pitted morningglory ( Ipomoea lacunosa L.), purple nutsedge ( Cyperus rotundus L.), smellmelon ( Cucumis melo L.) Texas millet [ Urochloa texana (Buckl.)], and yellow nutsedge ( Cyperus esculentus L.) are a continuing problem in peanut ( Arachis hypogaea L.) growing areas of the southwestern US ( Grichar and Dotray, 2013; Grichar et al., 2019 ; Baughman et al., 2018 ; Dotray et al., 2018 ). These weeds can be found in all peanut growing areas of this region (Grichar et al., 1999).
Control of many broadleaf weeds and annual grasses can be obtained with a preplant application of a dinitroaniline herbicide such as ethalfluralin (Sonalan®), pendimethalin (Prowl® or Prowl H 20®), or trifluralin (Treflan®) ( Wilcut et al., 1995 ). However, weeds such as Palmer amaranth and Texas millet can escape control due to extremely high weed populations, improper soil incorporation, large seed size, and/or an inadequate herbicide rate ( Wilcut et al., 1995 ; Grichar and Colburn, 1996). Dinitroaniline herbicides alone do not adequately control any of these weeds for the entire growing season ( Wilcut et al., 1995 ; Grichar et al., 1999 ; Grichar and Dotray, 2013; Baughman et al., 2018 ; Dotray et al., 2018 ; Grichar et al., 2019 ).
Diclosulam was labeled for use in peanut in 2000 in all states but was restricted from use in New Mexico, Oklahoma, and Texas in 2001 ( Anonymous, 2017). This herbicide is a triazolopyrimidine sulfonanilide herbicide developed for use in soybean ( Glycine max L.) and peanut ( Gander et al., 1997 ; Sheppard et al., 1997 ). As a preplant incorporated (PPI) or preemergence (PRE) treatment, diclosulam controls many weeds found in soybean and peanut, including common cocklebur ( Xanthium strumarium L.), morningglory spp. (Ipomoea spp.), common ragweed ( Ambrosia artemisiifolia L.), pigweed spp. (Amaranthus spp.), common lambsquarters (Chenopodim album L.), prickly sida ( Sida spinosa L.), Florida beggarweed [Desmodium tortuosum (Sw) DC.], bristly starbur (Acanthospermum hispidum DC.), and yellow nutsedge ( Braxton et al., 1997 ; Richburg et al., 1997 ; Sheppard et al., 1997 ; Prostko et al., 1999 ; Wilcut et al., 1999 ; Bailey et al., 2002 ; Main et al., 2005 ; Grichar et al., 2008 ; Anonymous, 2017).
Yellow nutsedge control has been reported to vary with diclosulam rate and method of application ( Grichar et al., 1999 ; Bailey et al., 1999a; b). Diclosulam at increased rates provided 89% control of yellow nutsedge but was less consistent when applied postemergence (POST) ( Langston et al., 1997 ; Grichar et al., 1999 ). When diclosulam was added to metolachlor PRE, yellow nutsedge control was greater than 95% ( Scott et al., 2001 ). Grey et al. (2001) reported that increased rates of diclosulam applied PPI from 0.009 to 0.052 kg ai/ha resulted in increased yellow nutsedge control but additional POST herbicides were needed for acceptable control.
In earlier work in south Texas, no problems were reported with diclosulam ( Grichar et al., 1999 ). However, in west Texas, diclosulam caused peanut stunting and reduction in yield (Grichar et al., 2001 ; Karnei et al., 2001 , 2002; Murphree et al., 2003 ). Karnei et al. (2001, 2002) reported that under weed-free conditions, diclosulam at 0.062 kg ai/ha caused 8 to 10% late-season peanut injury while rates lower than 0.062 kg ai/ha resulted in less than 3% injury. They also reported that plots treated with diclosulam applied PPI at 0.062 kg ai/ha yielded 538 kg/ha less than diclosulam at 0.021 kg ai/ha. While the untreated check plot yielded greater than 3300 kg/ha, plots treated with diclosulam at 0.028 kg ai/ha applied PPI produced 2690 kg/ha, and plots treated with diclosulam at the same rate applied PRE yielded 2914 kg/ha. In growth chamber studies, Grichar et al. (2001) reported that diclosulam rate was a factor in reduced peanut germination in only one of three studies. In that study, germination decreased as diclosulam rate increased. They concluded that poor seed quality could reduce peanut seed germination. Murphree et al. (2003) reported diclosulam applied PRE at 0.031 kg ai/ha injured peanut 15 to 40% when rated 14 days after treatment (DAT) in 2001, but injury was less than 8% in 2002. When rated late-season, all injury decreased to less than 5% and peanut yields were not affected.
Research is limited since the early 2000’s in Texas and Oklahoma evaluating peanut variety response to diclosulam. Therefore, the objective of this research was to evaluate some of the newer released peanut varieties for tolerance to diclosulam in the Texas and Oklahoma peanut growing areas.
Materials and Methods
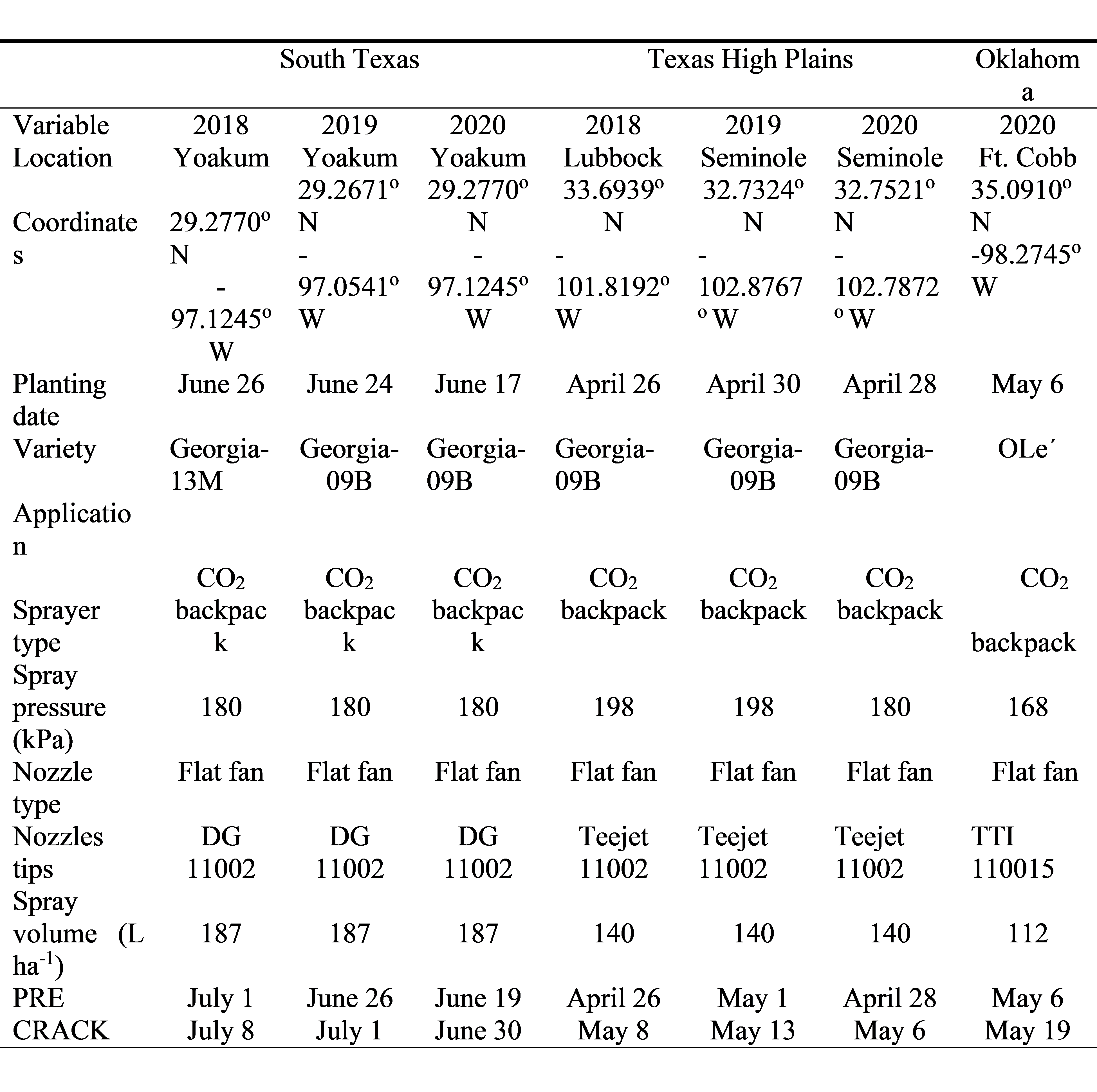
Peanut tolerance studies were conducted at the Texas A&M AgriLife Research site in south Texas near Yoakum (29.0369° N, 97.2616° W), in the Texas High Plains during the 2018 season near Lubbock at the Texas A&M AgriLife Research and Extension Center (33.6939° N, 101.8192° W) and in 2019 and 2020 in a producer’s field in Gaines County near Seminole (32.7429° N, 102.8253° W). The study in Oklahoma was conducted only in 2020 at the Oklahoma State University Caddo Research Station near Ft. Cobb (35.091° N, 98.275°W) in southwestern Oklahoma. Soils at Yoakum were a Denhawken sandy clay loam (fine, smectitic, hyperthermic, Vertic Haplustepts) with less than 1.0 % organic matter and pH 7.6 while near Lubbock soils were a Acuff loam (fine-loamy, mixed, superactive, thermic Aridic Paleustalls) with 1% organic matter and a pH 7.5 while soils at the Seminole location were a Patricia loamy fine sand (fine-loamy, mixed, superactive, thermic Aridic Paleustalfs) with 1.4% organic matter and a pH 7.9. Soils at Ft. Cobb were a Cobb fine sandy loam (fine-loamy, mixed, active, thermic Typic Haplustalfs) with less than 1% organic matter and a pH 7.3.
Treatments consisted of a factorial arrangement of two herbicide treatments (diclosulam at 0.026 or 0.052 kg ai/ha) and two application timings [PRE or at peanut cracking (CRACK)]. The CRACK application was applied 7 to 13 days after planting depending on location. An untreated check was included in each study and each treatment was replicated three to four times depending on location. Specifics of each study can be seen in Table 1.
Peanut cultivars evaluated were those commonly grown in each production area. In south Texas, Georgia-13M ( Branch, 2014) was evaluated in 2018 while Georgia-09B ( Branch, 2010) was evaluated in 2019 and 2020 while at the High Plains locations Georgia-09B was evaluated all 3 years. The Spanish cultivar OLe´ ( Anonymous, 2015) was evaluated at the Ft. Cobb location.
Each plot consisted of two rows spaced 97 cm apart and 7.6 m long at Yoakum. At the Texas High Plains locations plot size was 4 rows spaced 102 cm apart and 7.6 m long in 2018 and 9.1 m long in 2019 and 2020 but only the center two rows received the herbicide treatment. The Oklahoma location consisted of 2 rows spaced 91 cm apart and 7.6 m long. Traditional production practices were used to maximize peanut growth, development, and yield. Plots at the Texas locations received either ethalfluralin or pendimethalin applied PPI to control early season weeds. In 2018 at the High Plains location, lactofen plus S-metolachlor was applied 30 days after planting as a blanket treatment over the entire test area. In south Texas, clethodim and 2,4-DB were used POST to control any late season weed infestations. Hand-weeding was used exclusively in Oklahoma and only at the Texas locations if necessary. Insecticides were not needed at any location in any year. Herbicides for the small plots were applied using water as a carrier with a CO 2-pressurized backpack sprayer ( Table 1).
At Yoakum, lateral hand moved irrigation lines were used and irrigation was applied as needed throughout the growing season. At the Lubbock location in 2018, furrow irrigation was applied as needed while in 2019 and 2020 at the Seminole location and at the Ft. Cobb location, a center pivot irrigation system was used to apply water as needed.
Peanut stunting was based on visual subjective estimates using a scale of 0 (no peanut stunting) to 100 (peanut death). Peanut yield was determined by digging the pods based on maturity of non-treated control plots, air-drying in the field for 6 to 10 days, and harvesting with a small-plot thresher. Yield samples were cleaned and adjusted to 10% moisture. Pod, shell, and peanut kernal weight were determined from each sample. Grades [percent sound mature kernels (SMK) plus sound splits (SS)] were determined for a 200-g pod sample from each plot following procedures described by the Federal-State Inspection Service ( Anonymous, 2019). Grade data was collected at Yoakum and the High Plains locations.
Data for percentage of peanut stunting were transformed to the arcsine square root prior to analysis; however, nontransformed means are presented because arscine transformation did not affect interpretation of the data. Data were subjected to ANOVA and analyzed using the SAS PROC MIXED procedure 23 (SAS, 2019). Treatment means were separated using Fisher’s Protected LSD at P < 0.05. The untreated check was used for peanut yield and grade calculation comparison and as a visual comparison for stunting. However, the results from the untreated check plots for stunting were not included in that analysis but were included in peanut yield and grade analysis.
Results and Discussion
Peanut injury (plant stunting) was estimated visually throughout the growing season at all locations, however, only the 30 and 90 days after planting (DAP) evaluations are presented.
Stunting. Stunting was not observed in any year at the Yoakum location or in 2018 at the Lubbock location (data not shown). In 2018 at the Lubbock location, furrow irrigation was used to apply water as needed and this limited herbicide movement to the peanut plant while in 2019 and 2020 the plots were located in a field with an overhead center-pivot irrigation system which readily moved the herbicide down to the peanut root system. The south Texas results are similar to that seen in previous studies with little to no injury from diclosulam applications ( Grichar et al., 1999 ; 2008)
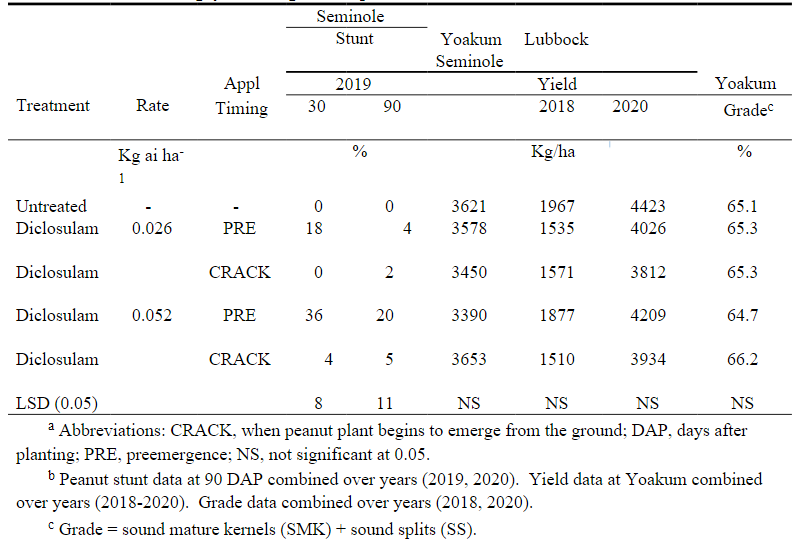
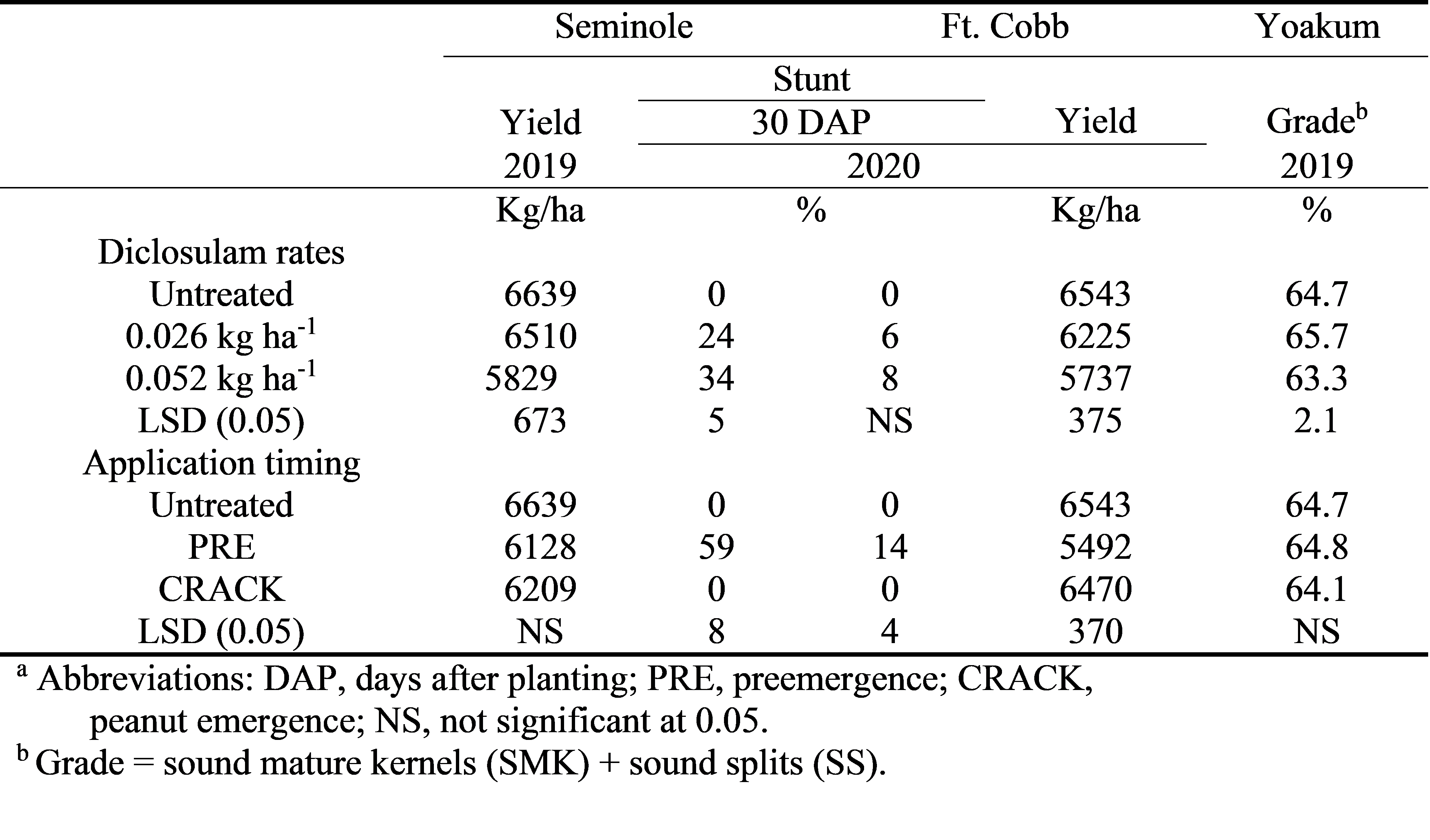
At Seminole in 2019 at the 30 DAP application, there was a diclosulam rate by application timing interaction ( Table 2). Diclosulam applied PRE at 0.026 or 0.052 kg/ha resulted in 18 and 36% stunting while diclosulam at 0.026 or 0.052 kg/ha applied at CRACK caused 0 and 4% stunt, respectively. In 2020 at the 30 DAP evaluation, there was not a diclosulam rate by application timing interaction although the diclosulam rate of 0.026 kg/ha resulted in 24% stunting while the diclosulam rate of 0.052 kg/ha resulted in 34% stunt ( Table 3). The PRE application caused 59% stunting compared with no stunting seen with the at CRACK diclosulam application. The 90 DAP evaluation from 2019 and 2020 was combined over years due to a lack of rate by timing by year interaction ( Table 2). Diclosulam at 0.052 kg/ha applied PRE resulted in 20% stunting while either diclosulam at 0.026 kg/ha applied PRE or at CRACK or diclosulam at 0.052 kg/ha applied at CRACK resulted in < 5% stunting.
In 2020 at Ft. Cobb, early-season stunting was only affected by application timing ( Table 3). Diclosulam applied PRE resulted in 14% stunting with no visible stunting noted with the at CRACK application. By the 90 DAP evaluation no peanut stunting was detected (data not shown).
In earlier work, peanut injury with diclosulam at many locations was minor and transient (Bailey et al., 2000 ; Grey et al., 2001 ; Lancaster et al., 2007 ; Price et al., 2002 ); however, in the High Plains of Texas, severe injury following diclosulam was reported (Karnei et al., 2001 ; 2002; Murphree et al., 2003). In later work, diclosulam at 0.018 and 0.027 kg/ha caused 7 to 33% stunting in west Texas but peanut injury was not observed in south and central Texas ( Grichar et al., 2008 ). In recent work, Meena et al. (2021) reported that diclosulam at 0.022 or 0.026 kg/ha did not cause any phytotoxic symptoms and diclosulam residues were below the detection level.
Yield . Since there was no diclosulam rate by application timing by year effect on peanut yield at Yoakum data were combined over years. No differences in yield between treatments were noted ( Table 2).
No diclosulam rate by application timing effects were noted at Lubbock in 2018 or Seminole in 2020; however, there was a year effect so years are presented separately ( Table 2). Yields in 2018 were extremely low due to furrow irrigation which resulted in uneven water distribution to the peanut plants (author’s personal observations). In 2019 diclosulam application timing did not have an effect on yield ( Table 3). However, peanut yield decreased as diclosulam rate increased.
At the Ft. Cobb location, a diclosulam rate and application timing effect were noted ( Table 3). As diclosulam rate increased peanut yield decreased and the PRE application greatly reduced yield when compared to the CRACK application.
In previous work in the High Plains region of Texas under yellow nutsedge pressure, diclosulam applied PRE at 0.018 kg/ha resulted in 11 to 21% injury and diclosulam at 0.027 kg/ha resulted in 18 to 33% injury but no yield differences were noted from the untreated check. In south Texas with diclosulam at either 0.009, 0.018, or 0.023 kg/ha applied PPI, PRE, or POST peanut yields increased over the untreated check with all diclosulam treatments except diclosulam applied PPI at 0.009 kg/ha ( Grichar et al., 2008 ). In recent work in India, Menna et al. (2021) reported no negative yield response when using diclosulam at 0.022 and 0.026 kg/ha. Earlier work in the Southeastern US has shown no negative yield response when using diclosulam (Bailey et al., 1999; Everman et al., 2006 ; Lancaster et al., 2007 ; Main et al., 2002 ). Bailey et al. (1999a) reported excellent tolerance to diclosulam at 0.017, 0.026, or 0.035 kg/ha while Lancaster et al. (2007) reported that imazapic treatments resulted in peanut yields greater than diclosulam treatments at 0.009 and 0.051 kg/ha but this difference in yield was more likely due to weed interference, nonvisible crop injury, or some other factor. Everman et al. (2006) reported, under weed-free conditions that diclosulam applied POST at 0.004 to 0.027 kg/ha did not adversely affect peanut yield.
Grade. At Yoakum, no diclosulam rate by application by year interaction were noted in 2018 or 2020; therefore, that data is combined over years with no differences noted ( Table 2). However in 2019, diclosulam rate did have an effect on grade with the grade decreasing as diclosulam rate increased. At the High Plains location, no interactions were noted; therefore, data were combined over years with no differences noted. No other studies could be found that reported on diclosulam effect on peanut grade.
Conclusions
The results of these studies indicate that diclosulam issues still exist in the Texas and Oklahoma peanut growing regions. Although no stunting or yield effects with diclosulam were noted in south Texas, stunting and yield reductions were noted in the Texas High Plains and Oklahoma. POST applications of diclosulam reduced the chance of injury or yield reductions. Therefore, more research is needed on the conditions that are responsible for peanut injury that can be seen in certain areas of the southwestern US peanut production area. Also, more research needs to be conducted with POST applications of diclosulam that may help reduce peanut injury or yield reductions.
Literature Cited
Anonymous. 2019. Farmer’s stock peanuts. Inspection Instructions. USDA. Online. Accessed at ams.usda.gov/sites/default/files/media/FarmersStockPeanutsInspectionInstructions.pdf. Accessed: June 21, 2021.
Anonymous. 2017. Strongarm product label. Indianapolis, IN. Dow AgroSciences, 5 p.
Anonymous. 2015. OLe´: A new Spanish peanut high in oleic acid. Agric. Res. 63 ( 2). USDA. Online. Accessed at agresearchmag.ars.usda.gov/2015/feb/peanut/. Accessed: June 22, 2021.
Bailey W. A., Wilcut J. W., Jordan D. L., Askew S. D., Hinton J. D., and Langston V. B. . 1999a. Weed management in peanut (Arachis hypogaea) with diclosulam preemergence. Weed Technol. 13: 450- 456.
Bailey W. A., Wilcut J. W., Jordan D. L., Swann C. W., and Langston V. B. . 1999b. Response of peanut (Arachis hypogaea) and selected weeds to diclosulam. Weed Technol. 13: 771- 776.
Bailey W. A., Wilcut J. W., Spears J. F., Isleib T. G., and Langston V. B. . 2000. Diclosulam does not influence yields in eight Virginia market-type peanut (Arachis hypogaea) cultivars. Weed Technol. 14: 402- 405.
Bailey W. A. and Wilcut J. W. . 2002. Diclosulam systems for weed management in peanut (Arachis hypogaea L.). Weed Technol. 16: 807- 814.
Baughman T. A., Grichar W. J., and Dotray P. A. . 2018. Weed control and peanut tolerance using pyroxasulfone in Oklahoma. J. Exp. Agric. Internatl. 21( 3): 1- 11. ArticleNo. [: JEAI. 39881].
Branch W. B. 2010. Registration of ‘Georgia-09B’ peanut. J. Plant Registration. 4( 3): 175.
Branch W. B. 2014. Registration of ‘Georgia-13M’ peanut. J. Plant Registration. 8( 3): 253.
Braxton L. B., Barrentine J. L., Geselius T. C., Grant D. L., Langston V. B., Nolting S. P., Redding K. D., Richburg, III J. S., and Sheppard B. R. . 1997. Efficacy and crop tolerance of diclosulam soil-applied in peanuts. Proc. South Weed Sci. Soc. 50: 162.
Dotray P. A., Baughman T. A., Grichar W. J., and Woodward J. E. . 2018. Performance of pyroxasulfone to control Amaranthus palmeri and Salsola kali in peanut . J. Exp. Agric. Internatl. 23( 2): 1- 10. Article No. [: JEAI. 41505].
Everman W. J., Clewis S. B., Taylor Z. G., and Wilcut J. W. . 2006. Influence of diclosulam postemergence application timing on weed control and peanut tolerance. Weed Technol. 20: 651- 657.
Gander J. R., Oliver L. R., and Wallace D. M. . 1997. Soybean weed control programs with diclosulam. Proc. South Weed Sci. Soc. 50: 22.
Grey T.L., Bridges D. C., and Eastin E. F. . 2001. Influence of application rate and timing of diclosulam on weed control in peanut (Arachis hypogaea L.). Peanut Sci. 28: 13- 19.
Grichar W. J. and Colburn A. E. . 1996. Flumioxazin for weed control in Texaspeanuts (Arachis hypogaea L.). Peanut Sci. 23: 30- 36.
Grichar W. J., Dotray P. A., and Sestak D. C. . 1999. Diclosulam for weed control inTexas peanut. Peanut Sci. 26: 23- 28.
Grichar W. J., Gerngross C. A., Lemon R. L., Senseman S. A., Besler B. A., and Langston V. B. . 2001. Interaction of peanut variety, diclosulam rate, and temperature on seed germination. Proc. South. Weed Sci. Soc. 54: 200.
Grichar W. J., Dotray P. A., and Prostko E. P. . 2008. Using diclosulam for yellow ( Cyperus esculentus) and purple ( Cyperus rotundus) nutsedge control in peanut . In Berklian Y. U. (ed) . Crop Rotation. Nova Science Publishers, Inc. p. 123- 140.
Grichar W. J. and Dotray P. A. . 2013. Smellmelon control and peanut response to flumioxazin and paraquat alone and in combination. Peanut Sci. 40: 135- 141.
Grichar W. J., Dotray P. A., and Baughman T. A. . 2019. Evaluation of weed control efficacy and peanut tolerance to pyroxasulfone herbicide in the south Texas peanut production area. J. Exp. Agric. Internatl. 29( 2): 1- 10. Article No. [: JEAI. 45347]. doi: [: 10.9734/JEAI/2019/45347].
Karnei J. R., Dotray P. A., Keeling J. W., and Baughman T. A. . 2001. Diclosulam performance in Texas High Plains peanut. Proc. South. Weed Sci. Soc. 54: 37.
Karnei J. R., Dotray P. A., Keeling J. W., and Baughman T. A. . 2002. Weed control and peanut response to diclosulam. Proc. South. Weed Sci. Soc. 55: 32.
Lancaster S. R., Beam J. B., Lanier J. E., Jordan D., and Johnson P. D. . 2007. Weed and peanut (Arachis hypogaea) response to diclosulam applied POST. Weed Technol. 21: 618- 622. DOI: [: 10.1614/WT-06-151.1]
Langston V. B., Braxton L. B., Barrentine J. L., Sheppard B. R., Nolting S. B., Richburg, III J. S., Grant D. L., Redding K. D., and Geselius T. C. . 1997. Efficacy and crop tolerance of diclosulam POST applied in peanuts. Proc. South. Weed Sci. Soc. 50: 162.
Main C. L., Ducar J. T., and MacDonald G. E. . 2002. Response of three runner market-type peanut cultivars to diclosulam. Weed Technol. 16: 593- 596.
Main C. L., Tredaway-Ducar J., Whitty E. B., MacDonald G. E. . 2005. Weed management in southeastern peanut with diclosulam and flumioxazin. Weed Technol. 19: 870- 874.
Menna H. N., Yadav R. S., Jain N. K., and Yadav M. . 2021. A novel pre-emergence herbicide (diclosulam) as an environmentally friendly weed management option in peanut and its phytotoxicity evaluation. Weed Biology and Manage. 21: 19- 27. https://doi.org/10.1111/wbm.12219.
Murphree T. A., Dotray P. A., Keeling J. W., Baughman T. A., and Grichar W. J. . 2003. Response of five peanut varieties to diclosulam and flumioxazin in Texas peanut. Proc. South. Weed Sci. Soc. 56: 34.
Price A. J., Wilcut J. W., and Swann C. W. . 2002. Weed management with diclosulam in peanut ( Arachis hypogaea) . Weed Technol. 16: 724- 730.
Prostko E. P., Grichar W. J., Baughman T. A., Brewer K. D., Besler B. A., and Lemon R. L. . 1999. Cadre and Strongarm comparisons for nutsedge ( Cyperus spp.) control in peanuts-1998 Proc . Amer. Peanut Educ. Res. Soc. 31: 27.
Richburg J.S., III , Barrentine J. L., Braxton L. B., Geselius T. C., Grant D. L., Langston V. B., Redding K. D., Sheppard B. R., and Nolting S. P. . 1997. Performance of diclosulam on key broadleaf weeds in southern soybeans. Proc. South. Weed Sci. Soc. 50: 161.
SAS Institute. 2019. SAS® Enterprise Guide 8.2 User’s Guide. Cary, NC.
Scott G. H., Askew S. D., and Wilcut J. W. . 2001. Economic evaluation of diclosulam and flumioxazin systems in peanut ( Arachis hypogaea) . Weed Technol. 15: 360- 364.
Sheppard B. R., Braxton L. B., Barrentine J. L., Geselius T. C., Grant D. L., Langston V. B., Redding K. D., Richburg, III J. S., and Roby D. B. . 1997. Diclosulam, a new herbicide for broadleaf weed control in soybeans and peanuts. Proc. South. Weed Sci. Soc. 50: 161.
Wilcut J. W., York A. C., Grichar W. J., and Wehtje G. R. . 1995. The biology andmanagement of weeds in peanut ( Arachis hypogaea) . In Pattee H. E. and Stalker H. T., eds. Advances in Peanut Science Amer. Peanut Res. Educ. Soc., Inc., Stillwater, OK. pp. 207- 244.
Wilcut J. W., Richburg, III J. S., and Braxton L. B. . 1999. Behavior of Strongarm in purple and yellow nutsedge. Proc. Amer. Peanut Educ. Soc. 31: 29.