INTRODUCTION
Effective weed control is considered critical to maximize the yield of peanut (Arachis hypogaea L.) (Leon et al., 2019). In Ghana, where herbicides are seldom used by smallholder farmers, yield losses due to weed interference are estimated to reach 50 to 80% (Dankyi, 2014; Dankyi et al., 2005; Dzomeku et al., 2009; Leon et al., 2019). Hand-weeding can be effective at controlling weeds; however, the time required to hand weed peanut in Ghana is a substantial component of the overall cost of peanut production (Dankyi, 2014; Dankyi et al., 2005). Adequate labor is often not available especially during the first 3-6 weeks of the season when it is most needed to prevent yield loss in peanut (El Naim et al., 2010; Everman et al., 2008). In countries where herbicides are available and affordable, farmers usually apply these products to the soil at planting and after weeds emerge later in the season to control weeds and protect yield (Leon et al., 2019). When comparing herbicide applications versus hand-weeding, studies in northern Ghana demonstrated that herbicides are effective in controlling weeds while minimizing the labor needed for hand weeding (Abudulai et al., 2017; Leon et al., 2019).
In addition to weeds, annual yield loss of peanut in Ghana from disease has been estimated to be as high as 50% (Nutsugah et al., 2007a 2007b). Early and late leaf spot, peanut rosette virus (Umbravirus) vectored by aphids (Aphis craccivura), and rust (Puccinia arachidis) are among the most important peanut diseases in Ghana and other countries in sub-Saharan Africa (Abudulai et al., 2007; Gaikpa et al., 2015; Nigam et al., 2018; Nutsugah et al., 2007a; Waliyar et al., 2000). Naab et al. (2005) demonstrated the value of incorporating fungicides into pest management strategies in peanut, but most smallholder farmers do not currently have access to fungicides or they do not have sufficient credit to purchase fungicides. Therefore, resistant cultivars are the most applicable strategy to minimize the negative impact of diseases on peanut (Gaikpa et al., 2015). However, high yielding and disease resistant cultivars are often not available on a wide scale in Ghana due to a lack of quality seed (Nigam et al., 2018; Owusu-Akyaw et al., 2019).
Recently, Abudulai et al. (2017) examined interactions of weed management practices that included herbicides and fungicides applied to protect peanut from leaf spot disease and improve production. In some instances, leaf spot disease was affected by weed management practice while disease management had no impact on weed management. For example, less canopy defoliation caused by leaf spot disease was observed in absence of fungicides when weeds were present compared with instances where weeds were controlled effectively by herbicides or hand-weeding. A limitation to these studies was the lack of comparison of the financial returns from the alternative treatments. Therefore, research was conducted to define interactions of weed management practices (e.g., combinations of herbicides and hand weeding) with fungicide treatments to control leaf spot in peanut. Possible interactions of pest management practices were also compared using measurements of weed and disease response, peanut pod yield, and estimated financial returns. The trials were conducted on-station and on-farm in the southern region of Ghana.
Materials and Methods
Field experiments were conducted during 2015 and 2016 near CSIR- Crops Research Institute Kwadaso Station (06°40'42.161"N 001°40'34.902"W), the Fumesua Station (06°43'24.588"N 001°31'58.164"W), and in a farmer’s field near Ejura (7°27'18.3"N 001°18'37.1"W). Soils were sandy loam alfisols at all locations. The improved leaf spot resistant cultivar Yenyawoso was used in one experiment in 2015 and two experiments in 2016, and the traditional leaf spot susceptible peanut cultivar Konkoma in two experiments in 2015 (Gaikpa et al., 2015; Owusu-Akyaw et al., 2019). The cultivars were in different experiments and were not compared directly. The cultivar Yenyawoso has expressed tolerance to both early leaf spot and late leaf spot (Gaikpa et al., 2015; Owusu-Akyaw et al., 2019). The number of days from planting to optimum kernel and pod maturity is approximately 90 days and is similar for both cultivars (Owusu-Akyaw et al., 2019). All trials were planted in early June in plots with 8 rows spaced 50 cm apart and 8 m in length. Seed was planted in conventionally-prepared flat seedbeds at an in-row density of 5 seeds per m of row.
Weed management consisted of: 1) no weed control; 2) hand-weeding at 3 and 6 weeks after planting (WAP); 3) a preemergence (PRE) application immediately after planting; 4) a postemergence (POST) application 4 WAP; 5) a PRE herbicide supplemented by hand-weeding 6 WAP; 6) a POST herbicide supplemented by hand-weeding at 6 WAP; 7) PRE and POST herbicides; and 8) PRE and POST herbicides supplemented by hand-weeding at 6 WAP (Table 1). The PRE herbicide metolachlor (Maestro 960 EC, Shandong Weifang Rainbow Chemical Co., LTD, Shandong, China) was applied at 1.15 kg ai/ha within 2 days after planting. The POST herbicide imazethapyr (Vezir 240 SL, ADAMA Makhteshim, Kiryat, Israel) was applied at 70 g ai/ha 4 WAP at the R1 or R2 growth stage (Boote, 1982). Nonionic surfactant at 0.25% (v/v) was included with imazethapyr.
Fungicide treatments consisted of: 1) no fungicide or 2) sequential fungicide applications at 4 and 6 WAP. In 2015, the fungicide program consisted of sequential applications of tebuconazole (Raintebzol 430SC, Shandong Weifang Rainbow Chemical Co., LTD, Shandong, China) at 100 g ai/ha. The fungicide program in 2016 consisted of sequential applications of azoxystrobin plus difenoconazole (Fivestar 325 SC, Shandong Weifang Rainbow Chemical Co., LTD, Shandong, China) applied at 0.09 plus 0.04 kg ai/ha, respectively, at the R2 growth stage (Boote, 1982). The formulated product in 2016 was used because it contained two fungicides and would serve as a more effective resistance management option (Shew, 2020). These fungicides are considered efficacious against the pathogens that cause leaf spot disease (Johnson et al., 2018; Shew, 2020). When combined with a fungicide treatment, the POST herbicide was applied 3 days prior to the fungicide. All herbicides and fungicides were applied at a carrier volume of 145 L/ha aqueous solution using a hand-held, backpack sprayer equipped with a single flat fan nozzle (Solo Backpack Sprayers, Newport News, VA).
The experimental design was a randomized complete block with 4 replications. Each combination of weed management practice and fungicide program was randomly assigned to plots in each replication. Visual estimates of leaf spot severity were determined from 10 randomly selected plants 12 WAP using an ordinal scale of 1 to 5 where 1 = plants with no visible lesions caused by leaf spot disease, 2 = 1 to 20% of peanut leaflets expressing lesions, 3 = 21 to 50% of leaflets with lesions, 4 = 51 to 70% of leaflets with lesions, and 5 = 71% to 100% of leaflets with lesions. The mid-point value for each category of the ordinal scale was used for statistical analysis (Chiang et al., 2014). Predominant weeds at Ejura, Fumesua, and Kwadodo are provided in Table 2. Dry weight of weed biomass and pod yield were determined from the 4 center rows (50-cm spacing) of each plot with a length of 4 m 10 WAP. Pod yield was adjusted to 8% moisture.
Financial return was calculated using a base cost excluding weed and disease control, of US $145/ha. Cost to dig pods and to remove pods from vines was $0.075/kg unshelled peanut for both operations based on information provided by local farmers and Ministry of Agriculture extension agents. Cost of shelling peanut was $0.075/kg shelled peanut based on discussions described previously. Cost of metolachlor and imazethapyr was $10/ha and $8/ha, respectively, based on costs quoted from local retailers in Ghana. Similarly, cost and application of tebuconazole and azoxystrobin plus difenoconazole was $60/ha and $40/ha, respectively. Hand-weeding labor cost was set at $1/hr with the amount of time required to remove weeds within each plot recorded. Cost of labor to apply pesticides was included in the cost for herbicides and fungicides. Cost of weed and disease control was determined and added to the base cost and costs associated with digging peanut, removing pods from plants, and shelling to establish total cost of production for each combination of weed and disease management practices. The price of peanut was set at $1.20/kg shelled peanut. Gross return was calculated as the product of grain yield assuming a shell out rate of 65% and price. Financial return was determined by subtracting total cost of production from gross return. Financial returns do not include cost of land or renting land. Household labor is also not included.
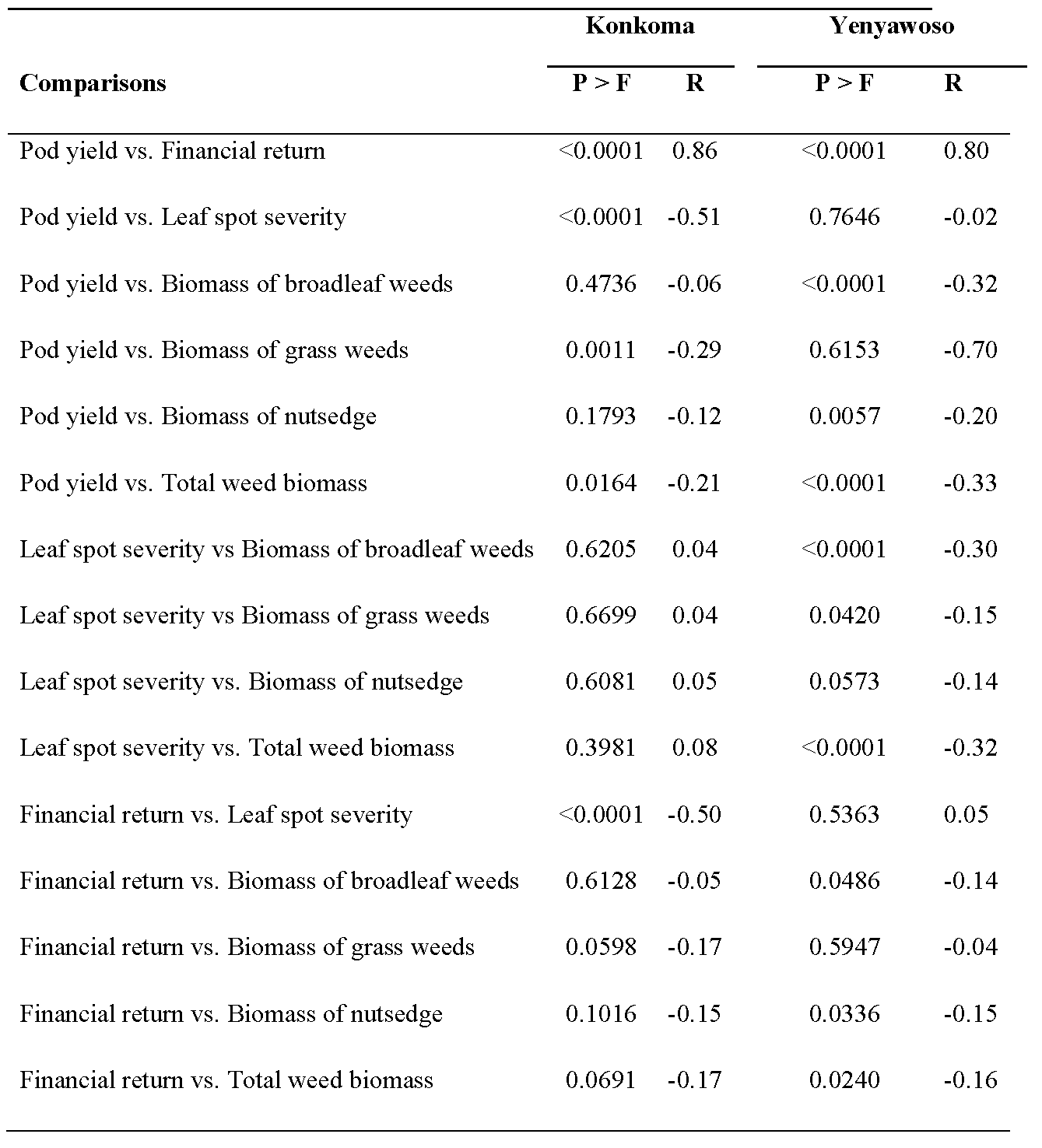
Data for weed biomass, leaf spot disease severity, pod yield, and financial return were subjected to ANOVA using the PROC GLIMMIX procedure in SAS (2002) considering the factorial treatment arrangement. Combinations of year and location (Yenyawoso cultivar during 2015 and 2016) or locations during 2015 with the cultivar Konkoma were defined as experiments and considered random effects along with replication within an experiment. The initial ANOVA indicated that the interaction of experiment × weed management practice × fungicide treatment was not significant for most measurements, and therefore data were pooled over experiments for each cultivar for AVOVA using the PROC GLIMMIX procedure in SAS. Means of significant main effects and interactions were separated using Fisher’s Protected LSD test at P ≤ 0. 05 when appropriate for data pooled over experiments for each cultivar. Pearson correlation coefficients (P < 0.05) were used to determine the relationships among leaf spot severity, weed biomass, pod yield, and financial returns pooled over experiments for each cultivar.
Results and Discussion
A significant and positive correlation of yield and financial return was observed for both cultivars (Table 3). This was expected as yield is a major contributor to financial return even though cost of pest management can affect financial return. Yield and financial return were negatively correlated with leaf spot severity for Konkoma but not for Yenyawoso. Leaf spot disease can reduce yield if effective disease management is not imposed when severity is high. Total weed biomass was negatively correlated with yield for both Konkoma and Yenyawoso. Financial return was negatively correlated with total weed biomass for Konkoma but not Yenyawoso. Decreasing weed interference with peanut often increases yield and financial return (Leon et al., 2019).
Results of ANOVA revealed an interaction of weed management practices × fungicide treatment was not significant in most instances for each cultivar (susceptible vs. resistant to leaf spot). However, the main effect of herbicide program was significant for biomass of broadleaf weeds, grass weeds, nutsedge, pod yield, and financial return regardless of cultivar. The interaction of weed management practices and fungicide program was significant for leaf spot severity for both cultivars. The main effect of fungicide program was significant for pod yield and financial return for the leaf spot susceptible cultivar Konkoma. However, when the leaf spot resistant cultivar Yenyawoso was grown, the main effect of fungicide was not significant for grain yield and financial returns. Differences among weed management practices for weed biomass, yield, and financial return were expected due to previous research with these approaches to weed management (Abudulai et al., 2017; Leon et al., 2007; Naab et al., 2005). Differences in leaf spot severity were somewhat surprising even though in most cases the differences were subtle. None-the-less, some practices may move soil on plants, and weeds may affect movement of spores onto the peanut canopy. It is also postulated that weeds could interfere with deposition of fungicide in the peanut canopy. Lack of response of Yenyawoso to fungicide was expected because of leaf spot tolerance reported in this cultivar (Owusu-Akyaw et al., 2019). The positive response of the cultivar Konkoma to fungicide was expected because this cultivar is susceptible to leaf spot disease (Owusu-Akyaw et al., 2019).
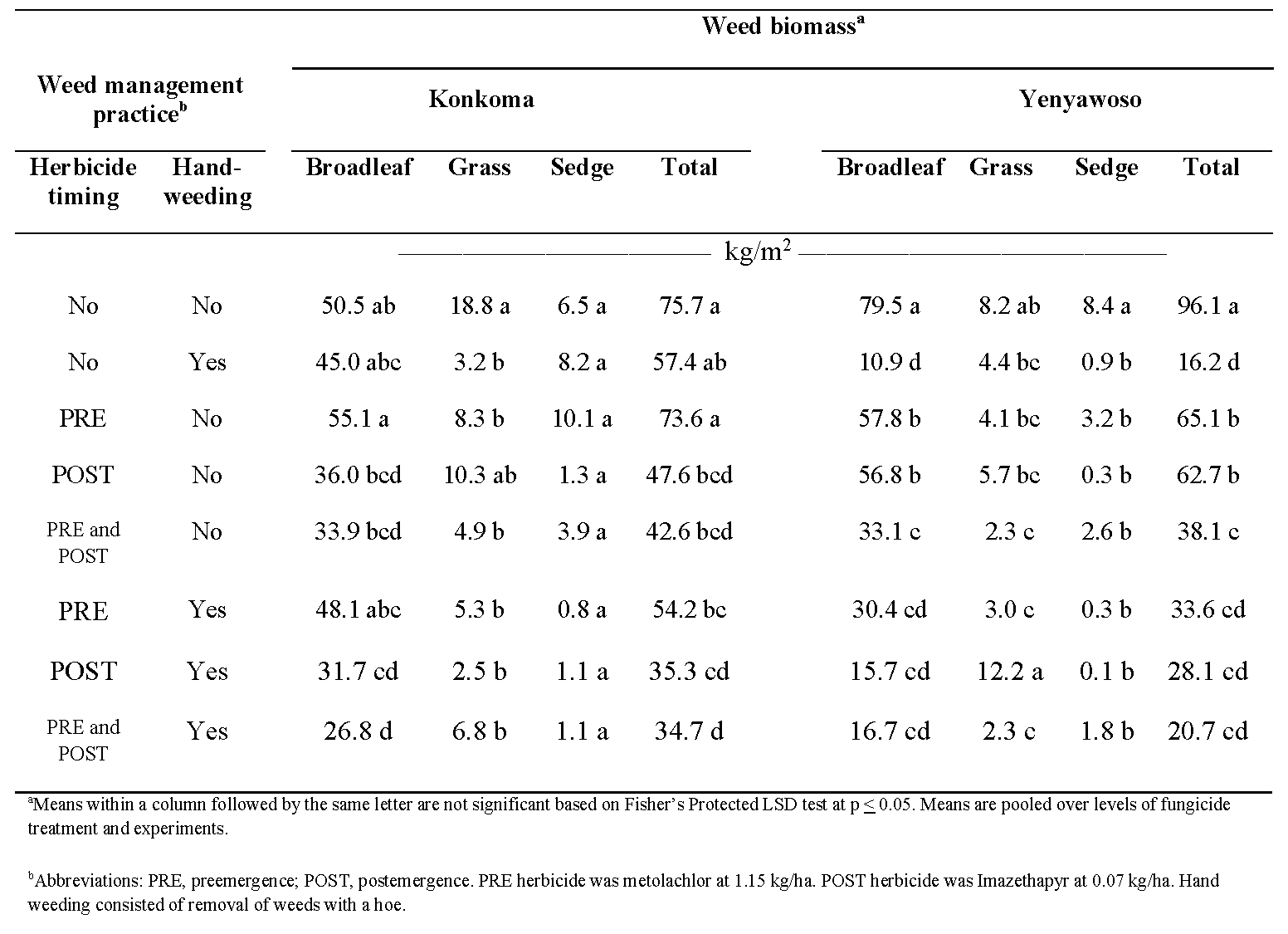
When comparing weed management practices, total weed biomass was similar for hand-weeding only (57.4 kg/m2), PRE herbicide only (73.6 kg/m2), and the non-treated control (75.7 kg/m2) for Konkoma (Table 4). In general, total weed biomass decreased as the number of weed management practices increased for this cultivar. For Yenyawoso, all weed management practices resulted in less total weed biomass than the non-treated control. The least effective weed management approach, with respect to total weed biomass, was noted when herbicides were applied PRE only (65.1 kg/m2) or POST only (62.7 kg/m2). A single weed management practice is often insufficient to control weeds completely (Hare et al., 2019). For example, employing two practices resulted in similar biomass reductions with values of 28.7 to 38.1 kg/m2. Hand-weeding only or in combination with PRE and/or POST herbicides resulted in weed biomass values of 16.2 to 33.6 kg/m2).
In absence of fungicides, differences in leaf spot severity ranged from 68 to 76% across weed management practices for Konkoma and 32 to 52% for Yenyawoso (Table 5). Although differences in weather conditions across the experiments were noted and the experimental design does not allow direct comparisons, the lower leaf spot incidence for Yenyawoso was expected because this cultivar expresses a greater degree of tolerance to leaf spot disease compared with the susceptible cultivar Konkoma (Owusu-Akyaw et al., 2019).
The interaction of weed management practice × fungicide treatment was significant for leaf spot severity for both Konkoma and Yenyawoso. Applying fungicide reduced leaf spot severity for the cultivar Konkoma except when weed management consisted of a PRE herbicide only or when no weed management (Table 5). Leaf spot severity ranged from 14% to 27% for the other weed management practices when fungicide was applied. The relatively poor weed control provided by PRE only weed management or no weed control may have resulted in limited deposition of fungicide into the peanut canopy compared with deposition when greater weed management was included. Greater deposition of fungicide would result in more effective pathogen control and less disease. Also, greater weed biomass may have limited air flow in the peanut canopy resulting in a more favorable environment for pathogens and subsequent disease progression. However, the mechanism of greater leaf spot severity for PRE only management and the treatment without weed management was not elucidated in this research. These results do suggest that additional research defining the mechanism of weed and disease management interactions would be informative. In contrast to results with Konkoma, no difference in leaf spot severity was observed for fungicide-treated and non-treated peanut for the PRE only treatment or when a weed control practice was not employed for Yenyawoso (Table 5). Applying fungicide resulted in lower severity of leaf spot when hand-weeding only was used or when two strategies were used for weed management. When PRE and POST herbicides were applied along with hand-weeding there was no difference in leaf spot severity when comparing fungicide treatments.
Pod yield and financial return were not affected by the interaction of weed management practice and fungicide program regardless of cultivar. However, both fungicide program and weed management practice affected these measurements. When data were pooled over weed management practices, grain yield and financial return were greater when fungicides were applied to Konkoma (Table 6). However, this was not the case for the cultivar Yenyawoso where yield and financial returns were similar for Yenyawoso regardless of fungicide treatment.
Pod yield for the non-treated control and use of a PRE herbicide without additional weed management for both cultivars was similar and less than 660 kg/ha (Table 7). For both cultivars, yield following hand-weeding only, PRE or POST herbicides followed by hand-weeding were similar. Including both PRE and POST herbicides plus hand-weeding resulted in similar yields compared with hand-weeding alone for Yenawoso.
With Konkoma, all treatments resulted in greater financial returns than the non-treated control (Table 7). Weed management including POST or PRE and POST, both without hand-weeding, resulted in lower financial returns that POST plus hand-weeding. With Yenyawoso, all treatments resulted in greater financial returns than the non-treated control. The PRE only treatment provided lower financial returns than all other treatments.
Two-hundred forty six and 308 hours were required to hand-weed peanut when herbicide was not included for the cultivars Konkoma and Yenyawoso, respectively (Table 8). When hand weeding was combined with a PRE herbicide only, a POST herbicide only, or the combination of a PRE and POST herbicide, the amount of labor required to apply herbicides and hand-weed was reduced 69 to 75% for Konkoma and 61 to 70% for Yenyawoso. These data demonstrate the value of using herbicides, especially in instances where labor to hand remove weeds may be limited.
Results from these experiments demonstrate the financial value of using herbicides in peanut production systems in Ghana. The greater financial returns noted when herbicides were used in combination with hand-weeding resulted in relatively high financial returns due to less labor requirements. However, availability of herbicides for smallholder farmers and credit to purchase herbicides continue to be a challenge in many areas of Ghana and most of Africa more generally. Results also demonstrate the financial value of fungicides for the traditional cultivar Konkoma that is susceptible to early and late leaf spot (Owusu-Akyaw et al., 2019). Differences in leaf spot severity were noted based on weed management practices for the leaf spot susceptible cultivar Konkoma. However, response to fungicide was similar across all weed control practices with respect to yield and financial return. Although differences in leaf spot severity were noted for the Yenyawoso, a cultivar that expresses some tolerance to leaf spot disease (Owusu-Akyaw et al., 2019), unlike Konkoma, these differences did not translate into differences in yield or financial return.
In addition to the lack of availability of herbicides and fungicides to smallholder farmers and limitations with respect to credit, overall yields in these experiments were considerably higher than those observed for the average smallholder farmer. Future research is needed with smallholder farmers to determine the value of using herbicides and fungicides in the broader context of peanut production in Ghana. None-the-less, incorporating herbicides and fungicides into production systems where appropriate could lead to greater yields and financial return and contribute to food security (Walker et al., 2014). In the meantime, the use of disease resistant, high yielding cultivars and timely hand-weeding is recommended to maximize yields and financial returns to smallholder farmers.
Acknowledgements
This publication was made possible in part through support provided by the Office of Agriculture, Research and Policy, Bureau of Food Security, U.S. Agency for International Development, under the terms of Award No. AID-ECG-A-00-07-0001 to The University of Georgia as management entity for U.S. Feed the Future Innovation Lab on Peanut Productivity and Mycotoxin Control (2012-2017). The opinions expressed herein are those of the authors and do not necessarily reflect the views of the U.S. Agency for International Development. Appreciation is expressed to technical staff and farmers for assistance with this research.
Literature Cited
Abudulai M., Dzomeku I. K., Salifu A. B., Nutsugah S. K., Brandenburg R. L., and Jordan D. L.. 2007. Influence of cultural practices on soil arthropods, leaf spot, pod damage, and yield of peanut in Northern Ghana. Peanut Sci. 34:72-78.
Abudulai M., Naab J., Seini S. S., Dzomeku I., Boote K., Brandenburg R., and Jordan D.. 2017. Peanut (Arachis hypogaea) response to weed and disease management in northern Ghana. International J. Pest Management. 63:1-6.
Boote K. J. 1982. Growth stages of peanut (Arachis hypogaea L.). Peanut Sci. 935-40.
Chiang K. S., Liu S. C., Bock C. H., and Gottwald T. R.. 2014. What interval characteristics make a good categorical disease assessment scale? Phytopathology. 104:575-585.
Dankyi A. A., Owusu-Akyaw M., Anchirinah V. M., Adu-Mensah J., Mochiah M. B., Moses E., Afun J. F. K., Bolfrey-Arku G., Osei K., Osei-Yeboah S., Adama I., Brandenburg R. L., Bailey J. E., and Jordan D. L.. 2005. Survey of production and pest management practices for peanut (Arachis hypogaea L.) in selected villages in Ghana, West Africa. Peanut Sci. 32:91-97.
Dankyi A. A. 2014. Economics of adopting IPM production practices. Pages 92-98 in M. Owusu-Akyaw, M.B. Mochiah, S. Gyasi-Boakye, and J.N. Asafu-Agyei, eds. Integrated Practices to Manage Diseases, Nematodes, Weeds and Arthropod Pests of Groundnut in Ghana. Council for Scientific and Industrial Research (CSIR) - Crops Research Institute (CRI), Kumasi, Ghana, West Africa and North Carolina State University (NCSU), USA. Available at: https://davidjordan.wordpress.ncsu.edu/files/2017/11/CRI-groundnut-manual-14_opti.pdf, 2014.
Dzomeku I. K., Abudulai M., Brandenburg R. L., and Jordan D. L.. 2009. Survey of weeds and management practices in peanut (Arachis hypogaea L.) in the savanna ecology of Ghana. Peanut Sci. 36:165-173.
Hare A.T., Jordan D.L., Leon R.G., Edmisten K.L., Post A.R., Mahoney D.J., and Washburn D.. 2019. Impact of weed management on peanut yield and weed populations the following year. Peanut Sci. 46:182-190.
Johnson III W. C., Culbreath A. K., and Luo X.. 2018. Interactive effects of cultivation, insect control, and fungal disease control in organic peanut production. Peanut Sci. 45:38-44.
El Naim A. M., Eldoma M. A., and Abdulla A. E.. 2010. Effect of weeding frequencies and plant density on the vegetative growth characteristic in groundnut (Arachis hypogaea L.) in North Kordofan of Sudan. International J. Applied Biology and Pharmaceutical Technology. 1:1118 -1193.
Everman W. J., Clewis S. B., Thomas W. E., Burke I. C., and Wilcut J. W.. 2008. Critical period of weed interference in peanut. Weed Technology. 22:63-67.
Gaikpa D.S., Akromah R., Asibuo J. Y., Appiah-Kubi Z., and Nyadanu D.. 2015. Evaluation of yield and yield components of groundnut genotypes under Cercospora leaf spots disease pressure. International J. Agronomy and Agricultural Research. 7:66-75.
Leon R. G., Jordan D. L., Bolfrey-Arku G., and Dzomeku I.. 2019. Sustainable weed management in peanut. Pages 345-366 In N.E. Korres, N.R. Burgos, and S.O. Duke SO, eds. Weed Control: Sustainability, Hazards and Risks in Cropping Systems Worldwide. CRC Press/Taylor and Francis Group.
Naab J. B., Tsigbey F. K., Prasad P. V. V., Boote K. J., Bailey J. E., and Brandenburg R. L.. 2005. Effect of sowing date and fungicide application on yield of early and late maturing peanut cultivars grown under rainfed conditions in Ghana. Crop Protection. 24:325-332.
Nigam S. N., Jordan D. L., and Janila P.. 2018. Improving cultivation of groundnuts”, Pages 155-180, In S. Sivasankar, D. Berguinson, P. Gaur, S. Kumar, S. Beebe, and M. Tamó, eds., Achieving Sustainable Cultivation of Grain Legumes; Improving Cultivation of Particular Grain Legumes. Volume 2. Burleigh Dodds Series in Agricultural Science, Burleigh Dodds Science Publishing, Cambridge, UK.
Nutsugah S. K., Abudulai M., Oti-Boateng C. , Brandenburg R. L., and Jordan D. L.. 2007a. Management of leaf spot diseases of peanut with fungicides and local detergents in Ghana. Plant Pathology J. 6:248-253.
Nutsugah S. K., Oti-Boateng C., Tsigbey F. K., and Brandenburg R.L.. 2007b. Assessment of yield losses due to early and late leaf spots of groundnut (Arachis hypogaea L.). Ghana J. Agricultural Science. 40:21-27.
Owusu–Akyaw M., Mochiah M. B., Asibuo J. Y., Osei K., Ibrahim A., Bolfrey Arku G., Lamptey J. N. L., Danyi A. A., Oppong A., Addo J. K., Boateng M. K., Adu-Dapaah H. K., Addy S., Amoah S., Osei-Yeboah S., Abudulai M., Denwar N., Naab J., Mahama G., Akroma R., Brandenburg R. L., Bailey J. E., Jordan D. L., Williams T. H., Hoisington D., and Rhoads J.. 2019. Evaluation and release of two peanut cultivars: a case study of partnerships in Ghana. Peanut Sci. 46:37-41.
SAS. 2002. Proc. GLIMMIX procedure, Version 9.4. SAS Institute, Cary, NC.
Shew B. 2020. Peanut disease management. Pages 101-140 In D. Jordan, Coordinating Author, 2020 Peanut Information, North Carolina Cooperative Extension Service AG-331. 186 pages.
Walker S. C., Culbreath A. K., Gianessi L., and Godfrey L. D.. 2014. The contributions of pesticides to pest management in meeting the global need for food production by 2050. Council for Agricultural Science and Technology. Issue Paper No. 55, 28 pages.
Waliyar F., Adomou M., and Traore A.. 2020. Rational use of fungicide applications to maximize peanut yield under foliar disease pressure in West Africa. Plant Disease. 84:1203-1211.
Notes
- Council for Scientific and Industrial Research-Crops Research Institute, Kumasi, Ghana; [^]
- Kwame Nkrumah University of Science and Technology, Kumasi, Ghana; [^]
- Department of Crop and Soil Sciences, North Carolina State University, Raleigh, NC; 4Department of Entomology and Plant Pathology, North Carolina State University, Raleigh, NC; 5Agricultural and Biological Engineering Department, University of Florida, Gainesville, FL; 6Department of Agricultural and Resource Economics, University of Connecticut, Storrs, CT; 7Feed the Future Innovation Lab for Peanut, University of Georgia, Athens, GA. [^] Corresponding author’s e-mail: david_jordan@ncsu.edu