Introduction
Cultivated peanut (Arachis hypogaea L.) is an annual legume grown in semi-arid tropic and sub-tropic regions in the world ( Naidu et al., 1999 ). The United States is the fifth largest peanut producer in the world (FAO, 2020) and the total annual peanut production in the world was more than 45.9 million tons in 2018. Peanut seeds have high oil content (45-52%) and are used for direct human consumption as well as for cooking oils. Peanut proteins are nutritious and are the principal plant-based protein source in some developing countries (Savage and Keenan, 1994). Peanut production is affected by many viral, fungal and bacterial diseases throughout the world ( Kokalis-Burelle et al., 1997 ). Spotted wilt disease, caused by Tomato spotted wilt virus, (TSWV, genus Tospovirus, family Bunyaviridae), is one of the most common diseases in the United States and can cause significant peanut yield loss. The total cost of losses from TSWV alone was estimated at $1.4 billion USD in the United States from 1996 to 2006 ( Riley et al., 2011 ).
Typical symptoms of spotted wilt on peanut are yellowing, stunting, concentric ringspots, chlorosis, and necrosis of leaflets ( Culbreath et al., 2003 ). TSWV has a very wide host range including both dicots and monocots in at least 92 families. More than 1000 plant species including many economically important field crops such as tobacco and peanut, and vegetables such as tomato, pepper, potato, and eggplant are hosts ( Jones and Baker, 1990; Peters, 1998). TWSV is transmitted only by thrips ( Frankliniella spcs.) of which two predominate as vectors: Tobacco thrips ( F. fusca (Hinds)) and Western flower thrips ( F. occidentalis (Pergande)) ( Todd et al., 1995 ; Kresta et al., 1995 ). Thrips acquire TSWV during the larval stage by feeding on infected host plants, but only transmit the virus during adult stages. TSWV particles are retained inside the vectors in a persistent manner which means that they can replicate their viral genomes inside the thrips ( German et al., 1992 ).
Many factors affect the severity of spotted wilt including peanut cultivar, planting date, plant population, row pattern, crop rotation and tillage. No single method can effectively control the impact or severity of spotted wilt. Methods involving these major factors have been combined into an integrated index tool to manage the risk of spotted wilt in peanut ( Brown et al., 1995 ; Culbreath et al., 2003 ). Among the factors in the spotted wilt index for peanut, cultivar or host resistance is the most important one for managing disease risk. Hence, development of spotted wilt resistance has become a major breeding objective in peanut breeding programs in the United States. Several cultivars have been released with moderate field resistance to spotted wilt ( Branch, 2002, 2007, 2010; Gorbet and Shokes, 2002; Gorbet, 2007; Gorbet and Tillman, 2008, 2009; Holbrook et al., 2008 ). However, no cultivar has been found to be immune to TSWV and all suffer significant yield losses when disease pressure is high ( Culbreath and Srinivasan, 2011).
Florida-EPTM ‘113’ is a runner-type cultivar released by the University of Florida Peanut Breeding Program that has superior spotted wilt resistance ( Tillman and Gorbet, 2012). It was derived from a cross between NC94022 and ANorden. NC94022 is a breeding line with excellent field resistance to spotted wilt. This resistance was theorized to have come from PI 576638, a varietal type of peanut known as hirsuta ( A. hypogaea subsp . hypogaea var. hirsuta) ( Barrientos-Priego et al., 2002 ). The hirsuta types might provide a special resource for spotted wilt resistance (Culbreath et al., 2005).
Florida-EPTM ‘113’ has been tested under favorable conditions for spotted wilt epidemics, i.e. with an earlier planting date (April) and reduced seed density (13.1 seed per meter). It showed excellent resistance to spotted wilt perhaps sufficient to obviate the high risk situations for most other cultivars even with an earlier planting date and lower seed density ( McKinney and Tillman, 2017). Immunostrip tests for TSWV and visual ratings also showed a significantly lower infection frequency (less than 10%) on both foliar symptomology and systematic infection. The other two cultivars tested, Florida-07 and Georgia Green, had 44% and 67% infection frequency, respectively. Other studies have reported several breeding lines that have lower TSWV infection, but the frequency was not as low as in Florida-EPTM ‘113’ ( Tillman and McKinney, 2018).
Much research has been focused on the virus itself, for example, the viral structure and genetics, but less research on the interaction between the virus and the host. Both the mechanisms and genetics related to spotted wilt resistance in peanut remain to be determined. The TSWV is mainly transmitted by adult viruliferous thrips, growing from larvae, which had fed on TSWV infected plants. After the infected thrips feed on the initial bud terminals (folded quadrifoliates), the presence of TSWV can be detected in the newly developed leaves. Subsequently, the virus moves down the plant and accumulates in the root crown. It is then transported back to young leaves leading to systemic spread ( Kresta et al., 1995 ; Rowland et al., 2005; Murakami et al., 2006 ). Initial spotted wilt symptoms have been observed as early as 30 days after planting (DAP) under high disease pressure and different cultivars have clearly distinct responses. Spotted wilt disease progress is slow at the beginning, but increases throughout the remainder of the growing season ( Culbreath et al., 1992 ).
Florida-EPTM ‘113’ displays a significantly lower spotted wilt incidence than other cultivars which are considered to be field resistant to TSWV. However, the mechanism of resistance is still unknown. The assessment of viral development pattern in Florida-EPTM ‘113’ and other existing cultivars with different levels of resistance is a prerequisite to understand the mechanism of resistance. In order to address this issue, an experiment was designed to evaluate the presence of TSWV in various plant tissues over the growing season. The objectives of this study were: 1) to evaluate TSWV presence in different cultivars throughout the growing season by immunoassays, 2) to evaluate TSWV presence in different plant tissues, and 3) to gain an understanding of the mechanism(s) controlling spotted wilt resistance in Florida-EPTM ‘113’.
Materials and Methods
Experimental Design
Field experiments were conducted at the North Florida Research and Education Center (NFREC) near Marianna, Florida (29°23‟ N, 82°12‟ W) in 2012 and 2014. Chipola loamy sands and Orangeburg loamy sands are the two major types of soils at the NFREC farm. Before the experiments were performed, maize ( Zea mays L.) and cotton ( Gossypium hirsutum L.) were planted for crop rotation. The fields were managed similarly to commercial peanut production and according to standard IFAS Extension recommendations; however, no in-furrow insecticide and early planting date were applied in order to maximize the occurrence of spotted wilt disease. The field plots were planted in mid-April, which is a window of high risk for spotted wilt as later planting dates tend to have lower disease pressure ( Culbreath et al., 2010 ). Overhead center pivots provided irrigation as needed.
The experimental design was a randomized complete block (RCBD) with four cultivars and three replications. Each plot was 1.8 m wide and 4.5 m long with two rows of the same cultivar spaced 0.9 m apart. The seed planting density was one seed per 0.3 m. The four cultivars (genotypes) evaluated were Florida-EPTM ‘113’ (Tillman and Gorbet, 2012), Florida-07 ( Gorbet and Tillman, 2009), Georgia Green ( Branch, 1996), and Georgia Valencia ( Branch, 2001).
Florida-07 is a high-oleic, runner-type cultivar with medium-late maturity and resistance to spotted wilt ( Gorbet and Tillman, 2009). Georgia Green is also a runner type cultivar ( Branch, 1996) and had moderate field resistance to spotted wilt, but it was more susceptible than Florida-07 ( Culbreath et al., 2008 ). According to the Peanut Rx spotted wilt risk index, Georgia Green had 30 points and Florida-07 scored 10 points ( Culbreath et al., 2010 ), where higher points translate to an increased risk of spotted wilt. Georgia Valencia is a large-podded Valencia market type cultivar used for boiling peanuts in fresh markets in the southeastern United States ( Branch, 2001) and appears to be spotted wilt susceptible ( Tseng et al., 2016 ).
Tissue Collection
Four types of tissues were collected: young leaf, old leaf, stem, and root crown. Young leaf was collected from the first unfolded leaf on the main stem. Old leaf was the last nodal position leaf, which was still attached to the main stem. Stem was the internode between the first node and the second node counted from the base. Root crown was collected under the soil surface after removing the lateral roots.
Tissue collection time points were 30, 60, 90, 120 days after planting (DAP), and prior to harvest. Four individual plants were randomly selected from each plot and entire plants were dug up. Young leaves, old leaves, stems, and root crowns were collected from each plant, separated and the same types of tissues from four different plants were pooled together for testing. The selected plants at each date were destructively sampled. There were 480 plants collected for testing (4 plants x 4 cultivars x 5 time points x 3 replicates x 2 years). All tissues were put into a freeze dryer to remove water and were stored under room temperature conditions with silica gel to control moisture.
Immunostrip Testing
A total of 480 samples (4 cultivars x 4 types of tissue x 5 time points x 3 replicates x 2 years) were tested for presence of TSWV using ImmunoStrip Kits (Agdia Inc., Elkhart, IN, United States). The kits contained TSWV specific monoclonal antibodies as the capture reagent and were used as an on-site tool to quickly identify virus in plants. The strip had two indication lines. The upper line was a control line and the lower line was the test line. If only the upper line (control line) displayed, no TSWV was detected; however, if two lines (control line and test line) were displayed, TSWV was detected in the sample. If neither line was displayed, the test was invalid. Score for the tissue was 1 if the virus was detected or 0 if no virus was detected.
Statistical Analysis
Data were analyzed by using the GLIMMIX procedure of SAS 9.4 (SAS version 9.4; SAS Institute, Cary, NC). Since the data were categorical with a binomial distribution, a generalized linear model was applied with logit function. The assessment date, tissue type, cultivar and their interaction were considered fixed effects, and all interactions with year and replication were considered random effects. Least squares means (LS-means) were computed to evaluate the treatment mean for time, tissue, and cultivar effect. If p-value was less than 0.05, treatment pairs were considered different from each other.
Results and Discussion
Cultivar and Temporal effects
The analysis of variance with p-values of each main factor and the interactions indicated that the main factors (date, tissue and cultivar) and interactions (date*tissue, date*cultivar, and tissue*cultivar) significantly ( p<0.05) affected the incidence of TSWV infection ( Table 1). The virus development was changing through different time points, by cultivar, by tissue type, and their various interactions. Varietal differences confirmed that host resistance is an important factor in disease development.
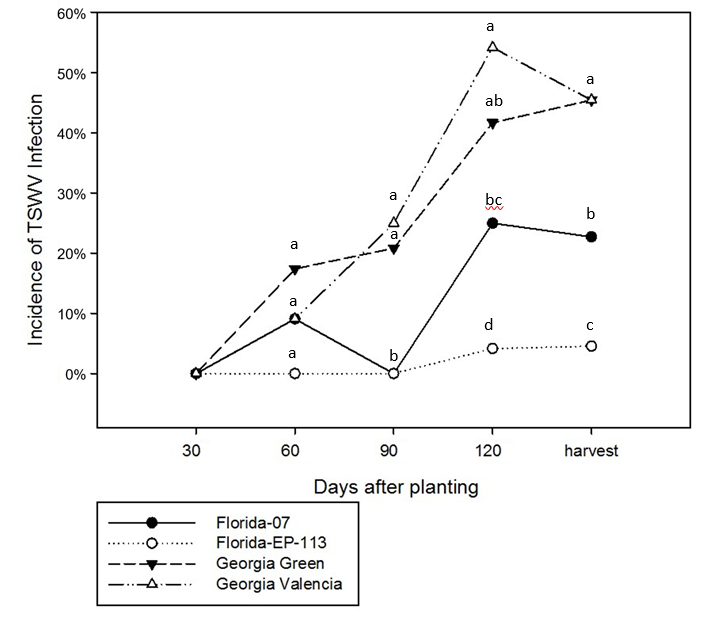
TSWV infection in Florida-EPTM ‘113’ was consistently low across different tissues and time points with an average of 1.72% infection frequency tested by immunostrips. Georgia Valencia had the highest infection frequency with an average of 28%, which is comparable to 25% in Georgia Green, and much higher than Florida-07 with 11% infection frequency. The average infection frequency differed ( p<0.05) among all the cultivars ( Figure 1), even though at certain time points some cultivars had similar infection frequency, especially Georgia Green and Georgia Valencia.
At 30 DAP, no TSWV was detected in any of the four cultivars. At 60 DAP, an average of 9% samples combined across tissue types were TSWV positive ( Figure 1). Disease incidence progressed with time and reached 12% at 90 DAP, 31% at 120 DAP, and remained near the same level (30%) at harvest (~140 DAP). TSWV incidence at 90, 120 DAP and harvest were different ( p<0.05).
Averaged over tissue types, Florida-EPTM ‘113’ showed a lower infection frequency as compared to the other three cultivars and it was the most consistent among different assessment dates with infection frequencies below 5% for all time points ( Figure 1). Georgia Green and Georgia Valencia had a higher frequency of infection. As the growing season progressed, the frequency of infection increased as well. There was no difference in infection frequency among cultivars at 30 DAP ( p>0.05) when no TSWV was detected. At 60 DAP, viruses were detected in Georgia Green, Georgia Valencia and Florida-07, but the differences were not significant ( p>0.05). At 90 DAP, there was no difference in infection frequency between Georgia Green and Georgia Valencia. Florida-07 and Florida-EPTM ‘113 had the lowest infection frequency. At both 120 DAP and harvest, there was no difference in infection frequency between Georgia Green and Georgia Valencia. Florida-07 had an intermediate level of infection. Florida-EPTM ‘113’ still had the lowest TSWV infection ( Figure 1). In general, the results from 120 DAP agreed with previously reported genotypic risk to spotted wilt disease.
Tomato spotted wilt virus infection detected by immunostrip at different assessment dates from four different peanut cultivars planted at North Florida Research and Education Center in 2012 and 2014. Analysis of variance showed that the interaction between cultivar and sampling date was significant ( p<0.05). Each data point is an average of 12 samples in each season. Different letters indicate different groups ( p<0.05).
Results of the present research resembled disease progress described by Culbreath et al. (1992) . Disease progress was linear over time but was lower in the more tolerant/resistant cultivar compared to the susceptible cultivar. In the current study, no TSWV was detected in any of four cultivars at 30 DAP with the virus being first detected at 60 DAP. The exact date of TSWV infection was not known and can only be estimated to be between 30 and 60 DAP.
A report from Murakami et al. (2006) showed similar results, i.e. they did not detect virus using ELISA at 30 DAP under low disease pressure in 1998 while they observed around 8% TSWV infection rate at 60 DAP. However, under high disease pressure in 1999, 10% of plants were detected with TSWV at 30 DAP ( Murakami et al., 2006 ). Rowland et al. (2005) also reported that there was almost no TSWV detected by ELISA at 25 DAP compared to that observed at 43 DAP. Even though the assessment dates were not exactly the same in different reports, there is consistency in that at approximately 25 DAP, the incidence of plants with TSWV starts to increase and continues to increase through the remainder of the season. In our study, between 60 and 120 DAP, the infection frequency of plants jumped from 10 to 30%. The infection frequencies were also much higher at 100 DAP ( Murakami et al., 2006 ) and at 70 DAP (Rowland et al., 2005) compared to the preceding assessment dates.
Tissue and Temporal effects
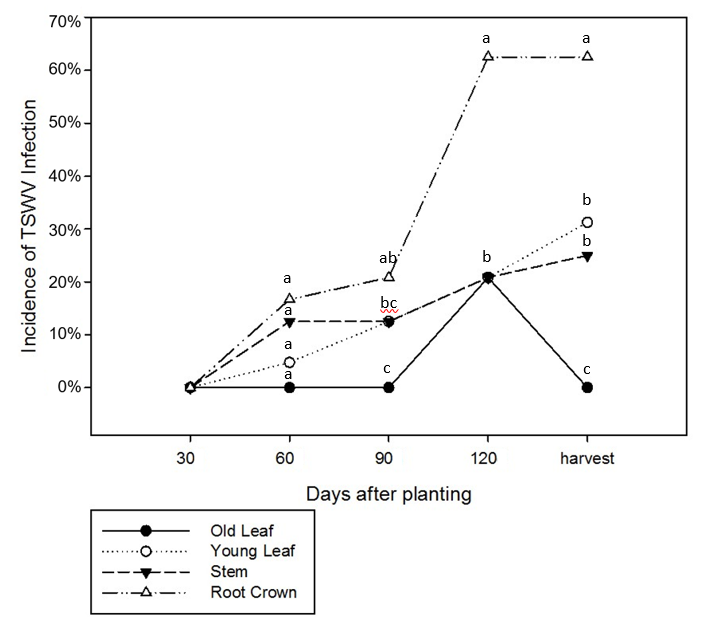
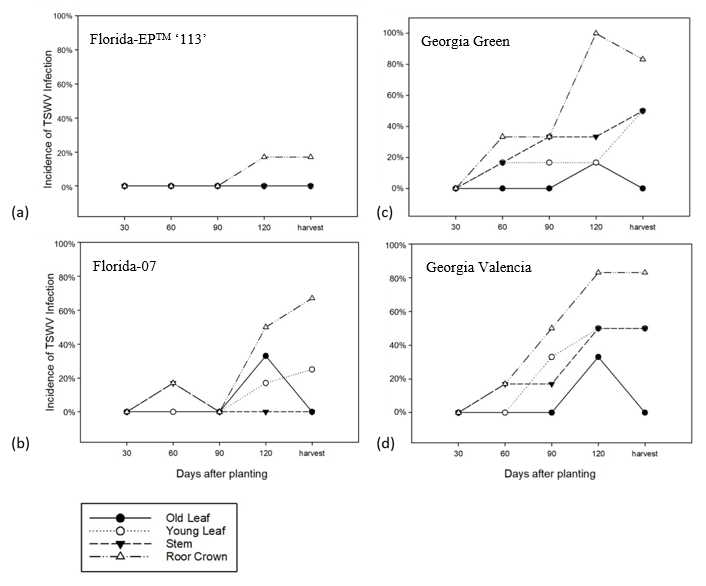
At 30 DAP, no TSWV was detected on any type of tissues. At 60 DAP, viruses were detected in young leaves, stems and root crowns, but the differences were not significant ( p>0.05). At 90 DAP, there was no difference among infection of roots, stems and young leaves ( p>0.05). Old leaves showed no TSWV infection. At 120 DAP, roots had the highest infection rate (63%, p<0.05) among all tissues with three times higher than at 60 DAP (17%) and 90 DAP (21%). At harvest, tissues had different levels of infection ( p<0.05). Roots had the highest infection frequency at 64% ( p<0.05) and old leaves had the lowest frequency (0%, p<0.05) ( Figure 2). Old leaves showed virus infections at 120 DAP only and no infection on other assessment dates. The lack of TSWV positive old leaves at harvest is likely due to sampling error because the frequency of TSWV positive old leaves was low and the sample size was limited.
Tomato spotted wilt virus infection detected by immunostrip at different assessment dates from four different peanut tissue types planted at North Florida Research and Education Center in 2012 and 2014. Analysis of variance showed that the interaction between tissue type and sampling date was significant ( p<0.05). Each data point is an average of 12 samples in each season. Different letters indicate different groups ( p<0.05).
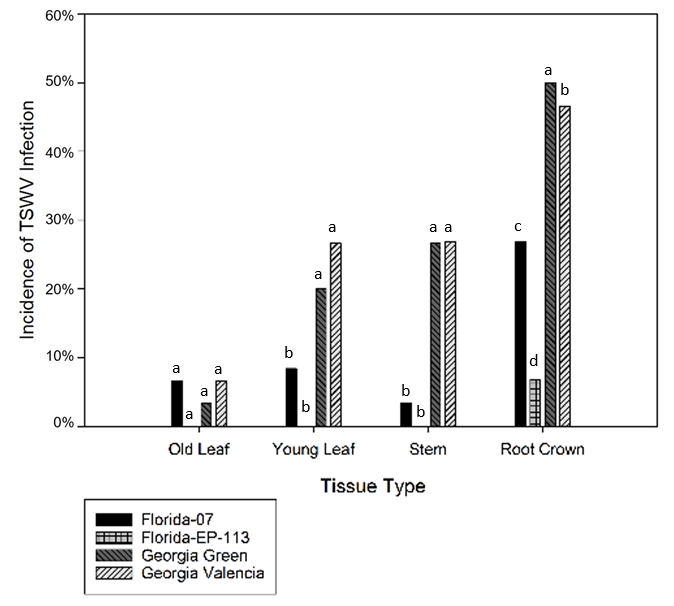
In each of the four cultivars, root crowns showed the highest infection frequency, 33% ( p<0.05) among all tissue types and old leaves had the lowest infection frequency, 4% ( p<0.05) ( Figure 3). The infection frequency of young leaves (13%) and stems (14%) were intermediate to the levels between root crowns and old leaves. There was no significant difference ( p>0.05) in incidence of TSWV infection between young leaves and stems.
In this study, the root crown was observed to have much higher TSWV frequency across all four cultivars as compared to leaves and stems. It has been reported that TSWV was easily detected in underground plant tissues (roots) ( Culbreath et al., 1991 ). If plants were infected early in the season, stunting was common and could result in cessation of plant growth. Research has shown that root systems were affected more by late season infection which resulted in yield loss ( Culbreath et al., 1992 ; Lyerly et al., 2002 ). However, in this research, TSWV was detected in roots at an early stage (60 DAP) in the more susceptible cultivars, but not in roots of Florida-EPTM ‘113’ until 120 DAP when the TSWV was detected on root crowns only and at a much lower frequency than all other cultivars ( Figure 3).
Cultivar effect on Spotted Wilt
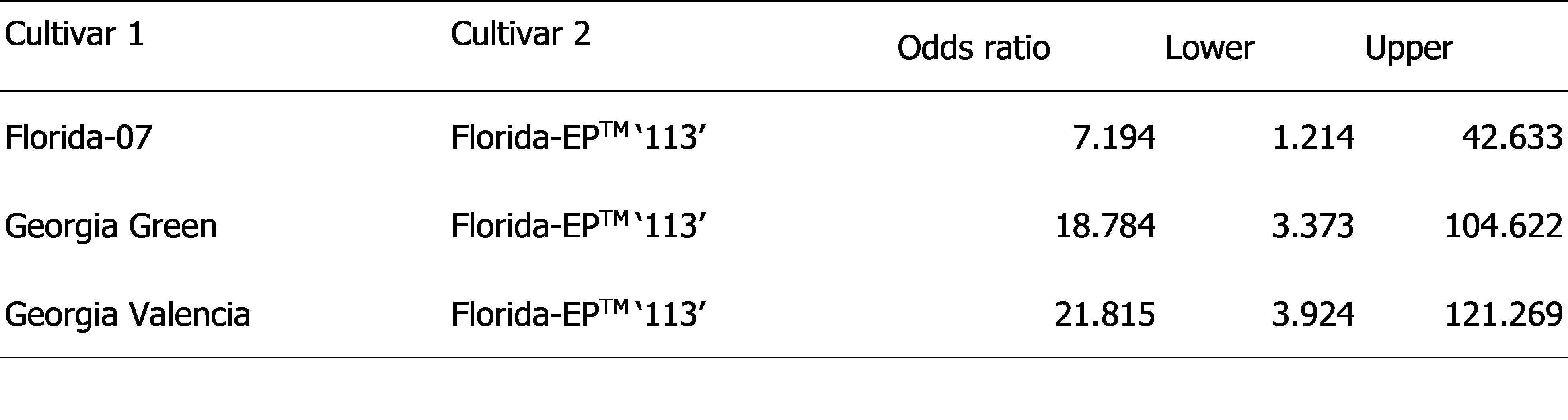
Florida-EPTM ‘113’ was used as a standard and the chance (odds) of TSWV infection incidence were compared to Florida-07, Georgia Green and Georgia Valencia ( Table 2). The data indicated that Florida-07 had seven times higher odds to be infected by TSWV than Florida-EPTM ‘113’. Georgia Green and Georgia Valencia possessed even greater ratios, 18 times and 21 times higher, respectively.
Florida-EPTM ‘113’ had the lowest TSWV infection frequency on all tissue types (old leaf, young leaf, stem, and root crown) ( Figure 3). TSWV infection in Florida-EPTM ‘113’ was observed only in roots, and not in leaves or stems. Georgia Green and Georgia Valencia displayed relatively higher infection frequency among all tissues. Florida-07 was at the intermediate level. On roots, all cultivars differed in infection frequency ( p<0.05) and roots showed highest infection frequency among all tissue types. In young leaves and stems, there was no difference between Florida-07 and Florida-EPTM ‘113’ ( p>0.05) and between Georgia Green and Georgia Valencia ( p>0.05). In old leaves, there was no significant difference in four cultivars ( p>0.05) ( Figure 3).
Tomato spotted wilt virus infection detected by immunostrip from different tissue types from four peanut cultivars planted at North Florida Research and Education Center in 2012 and 2014. Analysis of variance showed that the interaction between cultivar and tissue types was significant ( p <0.05). Each data point is an average of 15 samples in each season. Different letters indicate different groups ( p<0.05).
In peanut, TSWV is transmitted by thrips, and these vectors prefer to feed on younger and softer plant tissues which is why thrips are commonly found in the leaf terminals ( Smith Jr and Sams, 1977). Studies have shown that at early stages of plant development, viral distribution was not uniform throughout individual plants and was concentrated mainly in the leaf terminals ( Kresta et al., 1995 ; Hoffmann et al., 1998 ). After local infection from thrips feeding, the virus moves down to the roots. Then, TSWV is either accumulated in the root crowns or goes back to the growing points, causing systemic infection ( Kresta et al., 1995 ).
Cultivar and Tissue effects on Viral Detection
The incidence of TSWV infection rate on four cultivars are shown by the interaction between tissue types and time points ( Figure 4). In the present study, only Georgia Green showed TSWV infection in three tissues: roots, stems and young leaves at an early stage (60 DAP) compared to Georgia Valencia with TSWV present only in roots and stems, Florida-07 with TSWV present in roots only and TSWV undetected in Florida-EPTM ‘113’. The virus was most often detected in roots, which follows from the understanding that viruses move from cell-to-cell via plasmodesmata then into the phloem and down to the roots ( Harries and Ding, 2011).
Florida-07 is a spotted wilt tolerant/resistant cultivar; however, it still had high infection incidence, especially in the late season. At 30 and 90 DAP, no virus was detected, but roots and stems showed infection at 60 DAP. Since sampling was destructive, different samples were collected at each time point. At 60 DAP, the positive immunostrip result indicated at least one of the 12 samples contained TSWV, but none had TSWV at 90 DAP. This suggests that, at an early stage, Florida-07 can be infected with TSWV.
Short distance movement of viruses is slow, i.e, cell-to-cell movement, and the virus needs to spread through plasmodesmata ( Gunning and Overall, 1983). Comparatively, long distance movement is more rapid and most of time occurs through phloem after virus leaves the initial infection site ( Atabekov and Dorokhov, 1984). In tobacco ( Nicotiana tabacum), TSWV caused discrete individual spots by local infection, whereas virual particles were transported to the root system where they can trigger systemic infection. The symptoms of systemic infection are necrosis on leaves and apical buds. In this study, the intital thrips feeding time points were around 30 to 60 DAP and after that the virus presumably moved down to roots. This is apparently why the three more susceptible cultivars (Florida-07, Georgia Green, Georgia Valencia) had more TSWV infection in roots, but less in leaves.
It has been hypothesized that peanut cultivars which have greater TSWV field resistance restrict long-distance movement of the virus, thereby minimizing systemic infection ( Mandal et al., 2002 ). Culbreath et al. (1997) noted that peanut cultivar is an important factor affecting the progress of spotted wilt epidemics. Although Florida-EPTM ‘113’ has relatively low TSWV infection (5%) compared to other cultivars in this study, it is not immune to TSWV. The distinct differences between Florida-EPTM ‘113’ and other cultivars is the lack of TSWV detected in stems and leaves, the reduction in the frequency of root crown infection and the delayed onset of TSWV detection.
Resistance in Florida-EP TM ‘113’
Florida-EPTM ‘113’ displayed a very slow infection progress resulting in very low infection frequency compared to other cultivars. TSWV was detected in other cultivars at 60 DAP, but TSWV was not detected in Florida-EPTM ‘113’ until 120 DAP, a delay of at least 60 days. Also, the frequency of detection was much lower in Florida-EPTM ‘113’. Florida-EPTM ‘113’ appears to have a unique mechanism that delays short and/or long distance pathogen movement. There are two basic requirements to establish a systemic infection: (1) hosts have to support viral replication, and (2) the virus must move through plasmodesmata/vascular vessels to other cells/organs ( Cruz et al., 1998 ). A major QTL related to spotted wilt resistance has been reported on the A01 chromosome using Florida-EPTM ‘113’ as material (Tseng et al., 2016), however, the mechanism of resistance of Florida-EPTM ‘113’ and the gene(s) responsible have not been identified.
The resistance in Florida-EPTM ‘113’ possibly came from PI 576638. It is believed that lines derived from PI 576638 have better TSWV resistance than those derived from PI 203396 ( Culbreath et al., 2005 ). PI 203396 is a common source of spotted wilt resistance in runner type peanuts and many cultivars with moderate spotted wilt resistance have PI 203396 in their pedigree, including DP-1( Gorbet and Tillman, 2008), C-99R ( Gorbet and Shokes, 2002), Georgia-01R ( Branch, 2002), Florida-07 ( Gorbet and Tillman, 2009) and Tifguard ( Holbrook et al., 2008 ). These two PI accessions may contain different resistance genes and might have different resistance mechanisms ( Culbreath et al., 2005 ).
Summary and Conclusions
The immunostrip method can detect the presence of the virus in plants with visible foliar symptoms and in those that are asymptomatic. Compared to other cultivars, TSWV infection of Florida-EPTM ‘113’ was observed only later in the season and only in the root tissues. Systemic root infection was observed in less than 20% of tissue samples of Florida-EPTM ‘113’, similar to that reported by McKinney and Tillman (2018). However, TSWV was detected in over 60% of roots in the other three cultivars. Additionally, short or/and long-distance movement within the plant was apparently delayed and helped to minimize virus infection in root crowns and other tissues in Florida-EPTM ‘113’. Compared to other cultivars, virus detection was delayed up to at least 120 days in Florida-EPTM ‘113’. This characteristic is more similar to resistance as compared to the apparent tolerance to TSWV of the other cultivars and helps plants to maintain normal functions under disease pressure until harvest.
Acknowledgements
The research presented in this article was sponsored by Plant Molecular Breeding initiative (PMBI) at University of Florida and Southeastern Peanut Research Initiative (SPRI). The field technical support was provided by the peanut breeding group at Marianna, FL.
Literature Cited
Atabekov J.G., and Dorokhov Y.L. . 1984. Plant virus-specific transport function and resistance of plants to viruses. Adv Virus Res. 29: 313- 364.
Barrientos-Priego L., Isleib T.G., and Pattee H.E. . 2002. Variation in oil content among Mexican and Peruvian hirsuta peanut landraces and Virginia-type hypogaea lines 1 . Peanut Sci. 29: 72– 77.
Branch W.D. 1996. Registration of ’Georgia Green’ Peanut. Crop Sci 36: 806.
Branch W.D. 2001. Registration of ‘Georgia Valencia’ Peanut. Crop Sci. 41: 2002.
Branch W.D. 2002. Registration of ‘Georgia-01R’ Peanut. Crop Sci. 42: 1750.
Branch W.D. 2007. Registration of ‘Georgia-06G’ Peanut. J. Plant Regist. 1: 120.
Branch W.D. 2010. Registration of ‘Georgia-09B’ Peanut. J. Plant Regist. 4( 3): 175– 178.
Brown S.L., Todd J.W., and Culbreath A.K. . 1995. Effect of selected cultural practices on incidence of Tomato spotted wilt virus and populations of thrips vectors in peanuts . Acta Hortic. 431: 491– 498.
Cruz S., Roberts A., Prior D., Chapman S., and Oparka K. . 1998. Cell-to-cell and phloem-mediated transport of potato virus X. The role of virions. Plant Cell 10: 495– 510.
Culbreath A., Beasley J., Kemerait B., Prostko E., Brenneman T., Smith N., Tubbs S., Olatinwo R., Srinivasan R., and Tillman B. . 2010. Peanut Rx: minimizing diseases of peanut in the southeastern United States, pp. 81- 96. In Beasley J.P. (eds.) 2010 Peanut Update, University of Georgia Cooperative Extension Publication CSS-10-0125 , Athens, GA .
Culbreath A.K., Csinos A.S., Brenneman T.B., Demski J.W., and Todd J.W. . 1991. Association of Tomato spotted wilt virus with foliar chlorosis of peanut in Georgia . Plant Dis. 75: 863.
Culbreath A. K., Gorbet D.W., Martinez-Ochoa N., Holbrook C.C., Todd J.W., Isleib T.G., and Tillman B. . 2005. High levels of field resistance to Tomato spotted wilt virus in peanut breeding lines derived from hypogaea and hirsuta botanical varieties . Peanut Sci. 32: 20– 24.
Culbreath A. K., and Srinivasan R. . 2011. Epidemiology of spotted wilt disease of peanut caused by Tomato spotted wilt virus in the southeastern U.S . Virus Res. 159: 101– 109.
Culbreath A. K., Tillman B.L., Gorbet D.W., Holbrook C.C., and Nischwitz C. . 2008. Response of new field-resistant peanut cultivars to twin-row pattern or in-furrow applications of phorate for management of spotted wilt. Plant Dis. 92: 1307– 1312.
Culbreath A K., Todd J.W., and Brown S.L. . 2003. Epidemiology and management of tomato spotted wilt in peanut. Annu. Rev. Phytopathol. 41( 134): 53– 75.
Culbreath A.K., Todd J.W., Demski J.W., and Chamberlin J.R. . 1992. Disease progress of spotted wilt in peanut cultivars Florunner and Southern Runner. Phytopathology 82: 766– 771.
Culbreath A. K., Todd J.W., Gorbet D.W., Shokes F.M., and Pappu H.R. . 1997. Field response of new peanut cultivar UF 91108 to Tomato spotted wilt virus . Plant Dis. 81: 1410– 1415.
German T.L., Ullman D.E., and Moyer J.W. . 1992. Tospoviruses: diagnosis, molecular biology, phylogeny, and vector relationships. Annu. Rev. Phytopathol. 30: 315– 348.
Gorbet D.W. 2007. Registration of “ANorden”peanut. J. Plant Regist. 1: 123– 124.
Gorbet D.W., and Shokes F.M. . 2002. Peanut, Registration of “C-99R.” Crop Sci. 42: 343– 366.
Gorbet D.W., and Tillman B.L. . 2008. Registration of “DP-1”peanut. J. Plant Regist. 2: 200– 204.
Gorbet D.W., and Tillman B.L. . 2009. Registration of “Florida-07” Peanut. J. Plant Regist. 3: 14.
Gunning B.E.S., and Overall R.L. . 1983. Plasmodesmata and cell-to-cell transport in plants. Bioscience 33: 260– 265.
Food and Agriculture Organizaion of United Nations, 2020. FAOSTAT. Crop and liverstock products. http://www.fao.org/faostat/en/#data/QCL 2021 Dec. 9.
Harries P., and Ding B. . 2011. Cellular factors in plant virus movement: at the leading edge of macromolecular trafficking in plants. Virology 411: 237– 243.
Hoffmann K., Geske S.M., and Moyer J.W. . 1998. Pathogenesis of Tomato spotted wilt virus in peanut plants dually infected with Peanut mottle virus . Plant Dis. 82: 610– 614.
Holbrook C.C., Timper P., Culbreath A.K., and Kvien C.K. . 2008. Registration of “Tifguard” Peanut. J. Plant Regist. 2: 92.
Jones R.K., and Baker J.R.. 1990. TSMV: symptoms, host range and spread. USDA Workshop "Virus-Thrips-Plant Interactions of Tomato spotted wilt virus," April 18-19, 1990, Beltsville, Maryland.
Kokalis-Burelle N., Porter D.M., Rodriguez-Kabana R., Smith D.H., and Subrahmanyam P.. 1997. Compendium of Peanut Diseases (Vol. 2). American Phytopathological Society. APS Press, St. Paul, MN.
Kresta K.K., Mitchell F.L., and Smith Jr. J.W. 1995. Survey by ELISA of thrips (Thysanoptera: Thripidae) vectored Tomato spotted wilt virus distribution in foliage and flowers of field-infected peanut . Peanut Sci. 22: 141– 149.
Lyerly J.H., Stalker H.T., Moyer J.W., and Hoffman K. . 2002. Evaluation of Arachis species for resistance to Tomato spotted wilt virus . Peanut Sci. 29: 79– 84.
Mandal B., Pappu H.R., Culbreath A.K., Holbrook C.C., Gorbet D.W., and Todd J.W. . 2002. Differential response of selected peanut ( Arachis hypogaea) genotypes to mechanical inoculation by Tomato spotted wilt virus . Plant Dis. 86: 939– 944.
McKinney J. L., and Tillman B.L. . 2017. Spotted wilt in peanut as impacted by genotype resistance, planting date, and plant population. Crop Sci. 57: 130- 136.
Murakami M., Gallo-Meagher M., Gorbet D.W., and Meagher R.L. . 2006. Utilizing immunoassays to determine systemic Tomato spotted wilt virus infection for elucidating field resistance in peanut . Crop Prot. 25: 235– 243.
Naidu R.A., Kimmins F.M., Deom C.M., Subrahmanyam P., Chiyembekeza A.J., and Van der Merwe P.J.A. . 1999. Groundnut rossette: a virus disease affecting groundnut production in sub-saharan Africa. Plant Dis. 83: 700– 709.
Peters D. 1998. An updated list of plant species susceptible to tospoviruses. p. 107–110. 4th International Symposium on Tospoviruses and Thrips in Floral and Vegetable Crops.
Riley D. G., Joseph S. V., Srinivasan R., and Diffie S. . 2011. Thrips vectors of tospoviruses. Journal of Integrated Pest Management 2: I1- I10.
Rowland D., Dorner J., Sorensen R., Beasley Jr J. P., and Todd J. . 2005. Tomato spotted wilt virus in peanut tissue types and physiological effects related to disease incidence and severity . Plant Pathology 54: 431- 440.
Savage G. P., and Keenan J.I. . 1994. The composition and nutritive value of groundnut kernels. In: Smart J., Ed., The Groundnut Crop: Scientific Basis for Improvement, Chapman and Hall, London, 173- 213. http://dx.doi.org/10.1007/978-94-011-0733-4_6
Smith Jr J.W., and Sams R.L. . 1977. Economics of thrips control on peanuts in Texas. S.W. Entomol. 2: 149– 154.
Tillman B.L., and Gorbet D.W. . 2012. Peanut cultivar UFT113. U.S. Patent No. 8,178,752.
Tillman B. L., and McKinney J. L. . 2018. Relationships among symptoms of spotted wilt disease of peanut and their potential impact on crop productivity and resistance breeding. Plant Breeding 137: 757- 762.
Todd J. W., Culbreath A.K., Chamberlin J.R., Beshear R.J., and Mullinix B.G. . 1995. Colonization and population dynamics of thrips in peanuts in the southern United States, pp. 453– 460. In Parker B.L., Skinner M. and Lewis T. (eds.) Thrips biology and management . Springer, Boston, MA .
Tseng Y-C, Tillman B. L., Peng Z. and Wang J. . 2016. Identification of major QTLs underlying Tomato spotted wilt virus resistance in peanut cultivar Florida-EPTM ‘113’ . BMC Genet. 17: 128.
Notes
- Assistant Professor, Agronomy Dept., National Chiayi University, Chiayi City, Taiwan 60004 [^]
- Professor, Professor, and Associate Professor, Agronomy Dept., University of Florida, Gainesville, FL 32611 [^]
- Professor, College of Natural Sciences, Forestry, and Agriculture, University of Maine, Orono, ME 04469 [^] Corresponding author’s E-mail: yct@mail.ncyu.edu.tw