Introduction
Seeds have quality properties that will determine their agronomic performance in the field. These properties include genetic and physical purity as well as absence of disease. Another factor that plays an important role in seed performance in the field is the physiological quality which consists in germination, vigor, desiccation tolerance, and longevity (Popinigis, 1985; Bewley et al., 2013). Seeds with superior physiological quality will show advantages compared with those with low quality, ensuring a successful plant stand. Furthermore, seedlings with high vigor have a greater capacity to withstand stresses, for instance low temperatures and water deficit stress. Rapid and uniform emergence of seedlings competes more efficiently with invasive plants for available resources required for growth and development (e.g. water and light) (Marcos-Filho, 2005; Carvalho & Nakagawa, 2012).
Germination is the first step of a seedling being generated, starting with the absorption of water by the seed and concluding with the radicle emergence (Bewley, 1997). Through a straightforward and simple test, the potential of a seed to germinate can be evaluated thus assessing in part the quality of that given seed. In combination with germination, another important physiological quality component is seed vigor. Vigor can be characterized by a combination of properties that will determine the performance of a seed during germination and seedling emergence (ISTA, 2015). Unlike germination, vigor is a sum of different properties of a seed and its evaluation is not done by a single standard test. There are a number of qualitative and quantitative tests available to assess vigor, and all tests are used to detect differences between lots or treatments. Percentage of normal and abnormal seedlings, radicle length, and seedling dry weight are some of the tests that can be performed to assess vigor. Normal seedlings are more likely to develop into successful plants when compared with abnormal seedlings. Kamara et al. (2017) assessed vigor in peanut seeds by comparing the number of normal, abnormal, and non-germinated seeds in plants treated with calcium and phosphorus. The authors observed that increased calcium fertilization in the field during peak flowering resulted in greater seed vigor on the next generation. In addition, the application of phosphorus increased the protein content of the seeds, which also indirectly contributed to greater seed vigor. Seedling weight and radicle length can also provide comparative information on vigor. Seedlings with greater biomass and longer radicles have been found to be more vigorous than those with less biomass and shorter radicles. Hamed (2021) measured length of radicle and seedling dry weight of three cultivars of wheat (Triticum aestivum L.) stored under different conditions for six months. Seeds stored at five Celsius degrees and 25 C indicated higher values for radicle length when compared with seeds stored under a metal shelter or in a sludge shelter both without control of temperature. The author also reported that seeds stored at 25 C presented the highest seedling weight. Radicle length and seedling dry weight were used as indicators of seed vigor, being positively correlated with the percentage of field emergence.
More attention has been given to seed quality recently especially due to the increasing pressure from biotic and abiotic factors in crop production, consequently affecting seed quality (Lamichhane et al., 2018; Racette et al., 2019). Among biotic factors, the effects of virus infection on seed quality have been investigated (Li et al., 2011; Anderson et al., 2017; Bueso et al., 2017). Anderson et al. (2017) studied the effects of soybean vein necrosis virus on the quality of soybean (Glycine max (L.) Merr.) seed and reported that the virus affected the seed quality, reducing oil content and increasing protein content in seeds from infected plants. Bueso et al. (2017) investigated seed deterioration in Arabidopsis (Arabidopsis thaliana) as affected by four different virus infections. Seeds infected with cucumber mosaic virus (CMV) and alfalfa mosaic virus (AMV) resulted in an increased tolerance to deterioration, increasing seed longevity, whereas cauliflower mosaic virus (CaMV) infection generated seeds more susceptible to deterioration, decreasing seed longevity. Evaluating the effect of peanut stripe virus (PStV) on peanut (Arachis hypogaea L.) seed composition, Ross et al. (1989) observed that seeds derived from plants infected by the virus showed an increase in the content of glucose, fructose, and raffinose, modifying the quality of the roasted peanut flavor. Furthermore, seeds also showed a decrease in potassium and magnesium content, indicating an adverse effect on the nutritional value of peanuts.
One of the main disease-related problems in peanut production in the southeast U.S. is tomato spotted wilt disease (TSW) caused by Tomato Spotted Wilt Virus (TSWV; genus Tospovirus, family Bunyaviridae) (Culbreath et al., 1992; Srinivasan et al., 2017; Tillman et al., 2018). When initially identified in the early 1990s, severe epidemics occurred in peanut fields causing considerable yield losses. Since then, a successful TSW management system was developed, with yield and production increasing consistently over the last decade.
Peanut plants infected with TSW usually exhibit concentric rings in the leaflets, moderate and severe plant growth and yield reductions, dwarfing, and bud necrosis (Culbreath et al., 2013). Although it is known that this virus is not transmitted by seed (Pappu et al., 1999), seeds from infected plants occasionally indicate symptoms such as reddish discoloration on seed coat and shriveled and smaller seeds (Tillman et al., 2018). The presence of virus symptoms in the seeds is common; however, only a few studies about the effect of TSW on seed quality were reported. Ali and Rao (1982) reported that peanut seeds from TSWV infected peanut plants had a reduced percentage of oil content when compared with seeds from non-infected plants. Sanders et al. (1994) evaluated the quality of peanut seeds with presence of red seed coat as a symptom of TSW and observed that these seeds had similar market quality as those without symptoms.
Considering that TSWV has not been eradicated from peanut production and there is limited information on the impact of this disease on peanut seed quality, studies are necessary to assess whether the presence of TSW symptoms on the seed coat can decrease seed quality. Therefore, this study aimed to evaluate the effect of TSW symptoms on the seed coat on the physiological seed quality of five peanut genotypes.
Materials and Methods
Seed Material and Treatment
Five peanut genotypes were chosen: one pre-breeding line (LPM 21-A; Suassuna et al., 2015) and two breeding lines (2013-424 and 2015-453) related with the putative wild ancestors of peanut A. ipaënsis and A. duranensis (Bertioli et al., 2019), ‘Sun Oleic 97R’ (Gorbet & Knauft, 2000) used as a susceptible cultivar to TSW, and ‘TifNV-High O/L’ (Holbrook et al., 2017) used as a resistant cultivar to TSW. The seeds of all genotypes were obtained from the same seed lot described by Suassuna et al. (2021). The experimental area was planted on April 18, 2018, an early planting date selected according to the Risk Mitigation Index (Peanut Rx; Kemerait et al., 2020) to increase risk for TSW incidence. All genotypes were planted in the same field and treated equally. The field was dug and inverted on September 6, 2018 (141 days after planting) and harvested after 5 days with a small-plot peanut thresher. All samples were stored in shell for three months at room temperature (23 C ±3 C) in the Grain and Seed Processing facility of the USDA-ARS, in Tifton, Georgia. The seeds of all genotypes exhibited incidence of TSW symptoms; TSWV was detected by PCR in the seeds from the lot produced in the field and used for the seed tests (Suassuna et al., 2019). After shelling the pods, peanut seeds from each genotype were classified into two groups: asymptomatic and symptomatic according to the absence or presence of TSW symptoms on the seed coat, respectively. The seeds were stored for two additional months under the same conditions at the Crop Physiology laboratory of the University of Georgia, Tifton Campus, until analyses were performed. All field production practices and post-harvest seed handling and storage were identical for all genotypes to eliminate potential confounding factors.
Seed quality
Germination and vigor tests were performed in the two symptomology groups of each genotype to assess the seed quality. Dormancy was naturally released during the storage period.
Standard germination
Germination tests were performed on the two symptomology groups for each genotype using four replications with 25 seeds each. Using the method ‘between paper’ (BP), seeds were placed between two layers of a germination paper with adequate moisture, rolled, and placed in an upright position inside an incubator for 10 consecutive days in a constant temperature of 25 C (ISTA, 2017). After 10 days, the germination rolls were opened and the number of seeds that had radicle protrusion with at least two mm in length was quantified. The seeds that did not germinate were considered dead and discarded. To calculate the percentage of germination, the total number of germinated seeds was divided by the total number of seeds per replication.
Seedling growth
Percentage of normal and abnormal seedlings was evaluated as a seedling growth parameter. Seedling growth was assessed on the same seeds used for the standard germination test. As described above for ‘standard germination’, seeds were incubated for 10 days. After the incubation period, seedling growth was measured immediately following the germination assessment. Seedlings with cotyledons remained attached as well as hypocotyl without lesions or malformation and presence of primary (not shortened or split) and secondary roots were considered as normal seedlings. The percentages of normal and abnormal seedlings were calculated based on the number of normal and abnormal seedlings divided by the total number of seeds per replication.
Vigor
To evaluate vigor, two different tests were performed: radicle length and total seedling weight. Using the seedlings classified as 'normal' in the seedling growth measurement, a vigor test was performed measuring the radicle length of each individual normal seedling and calculating the mean seedling radicle length for each replication. Furthermore, seedling dry weight was determined by placing all seedlings (normal + abnormal) of each replication in paper bags and dried in an air oven at 60 C for 24 hours. Thereafter, seedlings were weighed and results were used as a measure of vigor between symptomology groups.
Statistical Analysis
The experiment was treated as a completely randomized design with four replications. Symptomology (TSW symptom on the seed coat) and genotype were considered as fixed effects. The data were subjected to analysis of variance (ANOVA). When significant differences were observed, Fisher’s Protected LSD post hoc test at 5% probability was used to separate the means. All statistical analyses were done using JMP software v.14.1.0 (SAS Institute Inc., Cary, NC) and graphed using SigmaPlot v.14 (Systat Software, San Jose, CA).
Results and Discussion
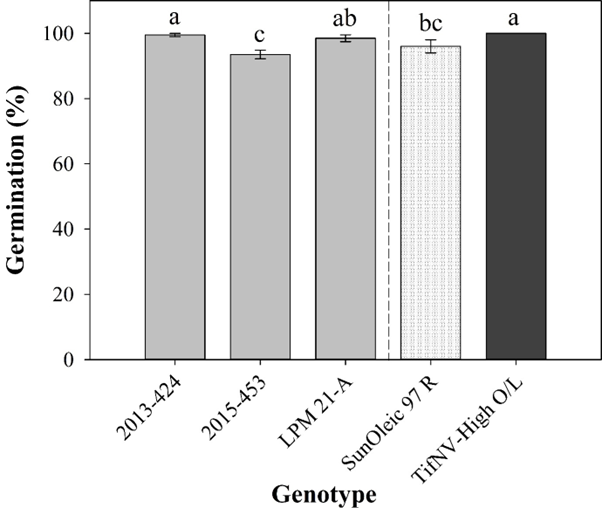
Seed germination was not affected by the interaction between TSW symptom and genotype or the presence of symptoms on the seed coat separately. However, germination was significantly different among the five genotypes (Fig. 1). The cultivar ‘TifNV-High O/L’ and the breeding line 2013-424 had the highest germination (100 and 99.5%, respectively), whereas 2015-453 had the lowest germination (Fig. 1). Among the breeding lines, 2013-424 and LPM 21-A indicated greater germination performance than 2015-453. In addition, ‘SunOleic 97 R’, a susceptible cultivar to TSW, had germination four percent lower than ‘TifNV-High O/L’, a resistant cultivar to TSW. Similar results were found by Groves et al. (2016), in which the presence of soybean vein necrosis virus symptoms on the seed coat did not affect germination of soybean.
Seed germination percentage among five peanut genotypes. Dashed line represents a visual separation between the cultivars (‘SunOleic 97R’, as susceptible to spotted wilt, and ‘TifNV-High O/L’, as resistant to spotted wilt) and breeding lines (2013-424, 2015-453, and LPM 21-A). All values are means ± standard errors. Bars followed by different letters are significantly different according to Fisher’s Protected LSD test at P ≤ 0.05.
Although differences were observed among the genotypes, overall, the germination percentage of all genotypes can be considered optimal, ranging from 93 to 100 %. The minimum germination percentage required to meet Georgia Crop Improvement Association’s seed certification standards for peanut is 75% but varies from state to state (Georgia Crop Improvement Association, 2016). Seeds with germination of 85 % or more have great likelihood of successful emergence and performance in the field. For germination between 75 to 85% the seeding rate is recommended to be increased to reach the optimal final plant stand. Due to the reduced quality, planting seeds with germination below 75 % increases risk of poor stand establishment, leading to further production problems such as TSWV incidence and weed competition (UGA Extension, 2020).
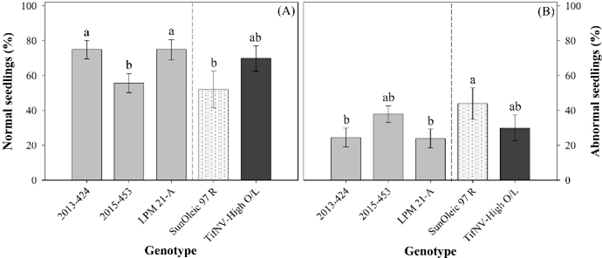
Spotted wilt symptoms on the seed coat or the interaction between TSW symptom and genotype did not affect the percentage of normal or abnormal seedlings. The percentage of normal seedlings was 61 and 70 for symptomatic and asymptomatic seeds, respectively, whereas for abnormal seedlings, the percentage was 36 for symptomatic and 28 for asymptomatic seeds. Similar results were found in a study assessing the effect of Cauliflower mosaic virus on physiological quality of canola (Brassica napus) seeds (Morais Junior, 2017). The author reported similar percentage of normal seedlings between seeds from infected and non-infected plants, with the virus not affecting seed vigor. Although the percentage of normal seedlings was not directly affected by TSW, when data for both symptoms were pooled, the genotypes indicated innate differences in the development of normal seedlings (Fig. 2A). Despite the high germination across all five genotypes (> 90%; Fig. 1), the breeding line 2013-424 and the pre-breeding line LPM 21-A showed the greatest percentage of normal seedlings (75% on average). The breeding line 2015-453 had 56% normal seedlings, which was similar to that for the susceptible cultivar to TSW ‘SunOleic 97R’ (52% normal seedlings) (Fig. 2A). The percentage of abnormal seedlings, as opposed to normal seedlings, was greatest for ‘SunOleic 97R’ (44%) and lowest for 2013-424 (25%) and LPM 21-A (24%) (Fig. 2B). This variation in seedling growth among genotypes are likely due to innate differences in the physiological seed quality. During seed formation, each genotype will develop the seed with specific genetic information expressed by the maternal plant. This transfer of genetic information has a direct impact on seed and seedling characteristics, such as seed size and seedling development, which will determine the seed quality of each genotype (Finch-Savage & Bassel, 2016; Singh et al., 2017).
(A) Percentage of normal seedlings and (B) abnormal seedling among the five genotypes. Dashed line represents a visual separation between the cultivars (‘SunOleic 97R’, as susceptible to spotted wilt, and ‘TifNV-High O/L’, as resistant to spotted wilt) and breeding lines (2013-424, 2015-453, and LPM 21-A). All values are means ± standard errors. Bars followed by different letters are significantly different according to Fisher’s Protected LSD test at P ≤ 0.05.
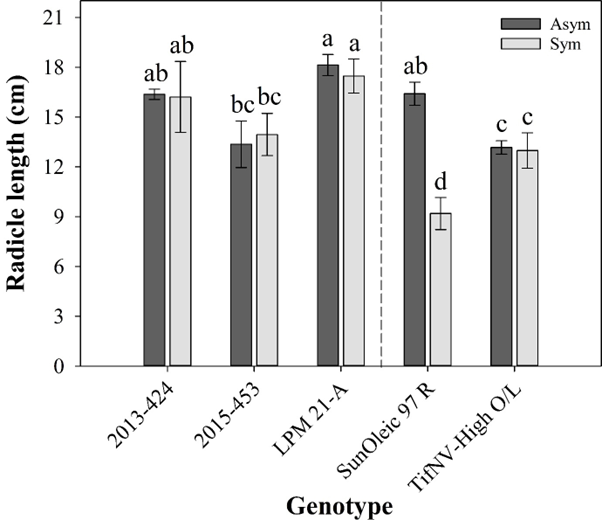
Significant differences in radicle length were observed for the interaction between genotype and TSW symptom on the seed coat (Fig. 3). Within asymptomatic seeds (Fig. 3, dark grey bars), LPM 21-A had the longest radicle, with an average of 18.1 cm, whereas the shortest radicle was observed for the cultivar ‘TifNV-High O/L’, with a measure of 13.2 cm. For symptomatic seeds (Fig. 3, light grey bars), LPM 21-A also showed longer radicle (average of 17.5 cm) than 2015-453, ‘TifNV-High O/L’ and ‘SunOleic 97R’, with average of 14, 13, and 9.2 cm, respectively.
Radicle length (cm) of seedlings from five peanut genotypes as affected by spotted wilt symptoms on the seed coat. Dark grey bars and light grey bars represent spotted wilt asymptomatic and symptomatic groups of seeds, respectively, for each genotype. Dashed line represents a visual separation between the cultivars (‘SunOleic 97R’, as susceptible to spotted wilt, and ‘TifNV-High O/L’, as resistant to spotted wilt) and breeding lines (2013-424, 2015-453, and LPM 21-A). All values are means ± standard errors. Bars followed by different letters are significantly different according to Fisher’s Protected LSD test at P ≤ 0.05.
All vigor measurements followed a similar trend and did not differ significantly between symptoms, except for ‘SunOleic 97R’ that was the only genotype showing significant differences in radicle length between asymptomatic and symptomatic seeds for TSW. Seeds without symptoms had the longest radicle, with an average of 16.4 cm, and symptomatic seeds had the shortest radicle (9.2 cm on average). Biotic stresses, such as TSWV infection, can result in impaired vegetative growth and reduced photosynthesis, affecting seed filling by reducing translocation of storage reserve to seeds (Culbreath et al., 2013; Gull et al., 2019). The stress results in multiple metabolic changes that can alter seed composition, affecting the seed physiological quality (Gooding et al., 2003). Although ‘SunOleic 97R’ is a susceptible cultivar to TSW, the physiological process behind the reduced radicle length in seedlings from TSW symptomatic seeds of this cultivar cannot be attributed solely to greater TSW incidence in the plants under field conditions, and requires further research. There were no differences in seed size due to TSW symptomology for ‘SunOleic 97 R’ or the other genotypes evaluated by Suassuna et al. (2021). Historically, seeds with TSW symptoms on the coat have not been discarded in the seed production system. As a consequence, the risk for low seedling emergence and poor stand establishment increases, thus favoring field TSW epidemics.
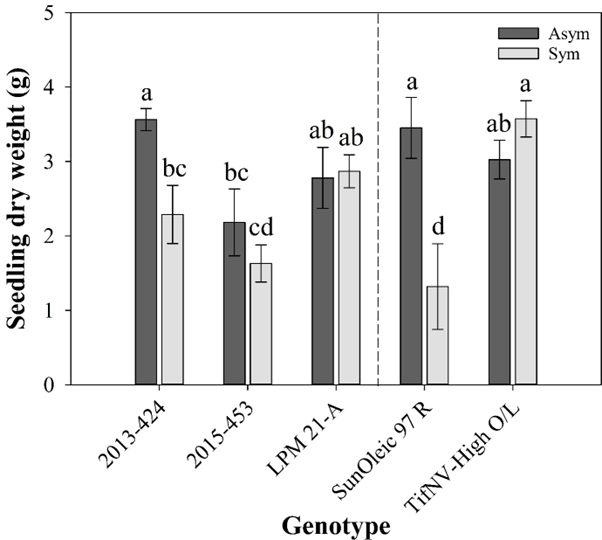
Similar to results observed for the radicle length, there was a significant interaction between TSW symptom and genotype for seedling dry weight (Fig. 4). The genotypes 2013-424 and ‘SunOleic 97R’ presented the highest seedling dry weight within asymptomatic seeds, with an average of approximately 3.5 g (Fig. 4, dark grey bars). ‘TifNV-High O/L’ was the genotype with the highest dry weight (3.6 g) within the symptomatic seed group, whereas ‘SunOleic 97R’ had the lowest dry weight, with an average of 1.3 g.
Seedling dry weight (g) from five peanut genotypes as affected by spotted wilt symptoms on the seed coat. Dark grey bars and light grey bars represent spotted wilt asymptomatic and symptomatic groups of seeds respectively for each genotype. Dashed line represents a visual separation between the cultivars (‘SunOleic 97R’, as susceptible to spotted wilt, and ‘TifNV-High O/L’, as resistant to spotted wilt) and breeding lines (2013-424, 2015-453, and LPM 21-A). All values are means ± standard errors. Bars followed by different letters are significantly different according to Fisher’s Protected LSD test at P ≤ 0.05.
The genotypes ‘SunOleic 97R’ and the 2013-424 presented a significant difference in dry weight between the two TSW symptomology groups (Fig. 4). The lowest seedling dry weight was observed on symptomatic seeds of ‘SunOleic 97R’, with a reduction of almost 60 % in dry weight compared to asymptomatic seeds from the same genotype. Similar results were observed for 2013-424, which exhibited a lower seedling dry weight on symptomatic seeds, with an average of 2.3 g, compared with asymptomatic seeds, with an average of 3.6 g.
Overall, all genotypes showed high seed quality, even when TSW symptoms were present in the seeds. The presence of the TSW symptoms did not affect germination and normal seedling development. The breeding line 2013-424 was the genotype with the highest seed quality and performance among the genotypes, responding similarly to ‘TifNV-High O/L’, used in this experiment as the TSW resistant cultivar. Although symptomatic seeds of 2013-424 had low seedling dry weight, the percentage of normal seedling was the highest, along with LPM 21-A and the cultivar resistant to TSW ‘TifNV-High O/L’. Alternatively, despite the high germination percentage of ‘SunOleic 97R’, this cultivar showed the lowest values for the vigor measurements, with the lowest seed quality and performance among all genotypes. The low seed quality of ‘SunOleic 97R’ is not likely related to the presence of symptoms on the seed coat alone. It could also be associated with the innate susceptibility of this cultivar to TSW. This results in a negative effect during seed formation, reducing the seed physiological quality. Measurements of vigor are better interpreted together. Examination of a given vigor measurement alone can lead to misinterpretation. For instance, lower seedling dry weight does not necessarily indicate lower seed vigor. A normal seedling can weigh less yet have longer radicle compared to an abnormal seedling with a higher dry weight and shorter radicle. In general, the differences in radicle length and seedling dry weight were mostly associated with factors influencing the acquisition of seed physiological quality during seed formation, with little impact of TSW symptoms on the seed coat. For this study, the measurements of normal seedling development, radicle length, and seedling dry weight were considered suitable parameters of seedling growth and vigor to compare TSW symptomology on the seed coat.
Summary and Conclusions
The presence of TSW symptoms on the peanut seed coat did not impact germination, one of the main components of the seed physiological quality. All genotypes had germination above 93% without significant effect of TSW symptoms. Vigor was mostly affected by the differences among the genotypes rather than the TSW symptoms found on the seed coat. High TSWV pressure during seed formation was found to be a factor that influenced final physiological quality of seeds from some peanut genotypes, as can be seen by the low vigor in symptomatic seeds of ‘SunOleic 97R’ . Overall, the presence of TSW symptoms on the seed coat did not affect the seed germination and seedling growth and had little effect on seed vigor, depending on the genotype and its susceptibility to TSW. Peanut seeds from genotypes with improved resistance to TSW had greater vigor than those from susceptible genotypes. This research provides inceptive knowledge on the impact of TSW infection on peanut seed quality. However, further research is required to investigate the effective contribution of innate differences in TSW susceptibility of genotypes and infection during seed formation on physiological quality of peanut seeds.
Acknowledgements
This research was funded by EMBRAPA, grants number 13.17.00.102.00.00 and 20.18.01.021.00.00.
Literature Cited
Ali M. I. M., & Rao R. D. P. ( 1982). Effect of tomato spotted wilt virus on the oil content of groundnut seeds. Madras Agric. J. 69(4): 269-270.
Anderson N. R., Irizarry M. D., Bloomingdale C. A., Smith D. L., Bradley C. A., Delaney D. P., Kleczewski N. M., Sikora E. J., Mueller D. S., & Wise K. A. ( 2017). Effect of soybean vein necrosis on yield and seed quality of soybean. Can. J. Plant Pathol. 39(3): 334-341.AOSA (2018). Seedling evaluation. In Rules for Testing Seeds ( vol. 4 pp. 57-60). Washington, D.C. : Association of Official Seed Analysts.
Bertioli D. J., Jenkins J., Clevenger J., Dudchenko O., Gao D., Seijo G., Leal-Bertioli S.C., Ren L., Farmer A.D., Pandey M.K. and Samoluk S.S. ( 2019). The genome sequencing of the segmental allopolyploid Arachis hypogaea. Nature Genetics, 51(5):877-887. doi: [: 10.1038/s41588-019-0405-z].
Bewley J. D. ( 1997). Seed germination and dormancy. The Plant Cell 9(7):1055-1066.
Bewley J. D., Bradford K. J., Hilhorst H. W. M., & Nonogaki H. ( 2013). Seeds – Physiology of Development, Germination and DormancySpringer.
Bueso E., Serrano R., Pallás V., & Sánchez-Navarro J. A. ( 2017). Seed tolerance to deterioration in arabidopsis is affected by virus infection. Plant Physiol. Biochem. 116: 1-8.
Carvalho N. M., & Nakagawa J. ( 2012). Seed Vigor. In Carvalho N. M. & Nakagawa J. (Eds), Seeds: science, technology, and production (pp. 224-242). FUNEP.
Culbreath A. K., Todd J. W., & Demski J. W. ( 1992). Productivity of Florunner peanut infected with tomato spotted wilt virus. Peanut Science 19(1): 11-14.
Culbreath A. K., Tubbs R. S., Tillman B. L., Beasley Jr. J. P., Branch W. D., Holbrook C. C., Smith A. R., Smith N. B. ( 2013). Effects of seeding rate and cultivar on tomato spotted wilt of peanut. Crop Protection 53, 118-124.
Finch-Savage W. E., & Bassel G. W. ( 2016). Seed vigour and crop establishment: extending performance beyond adaptation. J. Exp. Bot. 67(3): 567-591.
Georgia Crop Improvement Association. ( 2016). Peanut seed certification standards. http://www.georgiacrop.com/fullpanel/uploads/files/peanut-standards-rev2016.pdf.
Gooding M. J., Ellist R. H., Shewry P. R., & Schofield J. D. ( 2003). Effects of restricted water availability and increased temperature on the grain filling, drying and quality of winter wheat. J. Cereal Sci. 37: 295-309.
Gorbet D. W., & Knauft D. A. ( 2000). Registration of ‘SunOleic 97-R’ peanut. Crop Science 40: 1190.
Groves C., German T., Dasgupta R., Mueller D. S., & Smith D. L. ( 2016). Seed Transmission of Soybean vein necrosis virus: The First Tospovirus Implicated in Seed Transmission. PLoS ONE 11(1): e0147342.
Gull A., Lone A. A., & Wani N. U. I. ( 2019). Biotic and abiotic stresses in plants. In de Oliveira A. B (Ed.), Plants, Abiotic and Biotic Stress in Plants. IntechOpen. https://www.intechopen.com/chapters/66714.
Hamed M. A. ( 2021). Impact of Storage Conditions on Seed Vigor and Viability of Bread Wheat (Triticum aestivum) seeds. In IOP Conference Series: Earth and Environmental Science, vol. 761, n. 1, p. 012069. IOP Publishing.
Holbrook C. C., Ozias-Akins P., Chu Y., Culbreath A.K., Kvien C.K., & Brenneman T.B. ( 2017). Registration of ‘TifNV-High O/L’ peanut. Journal of Plant Registrations 11: 228-230.
ISTA (2015). Seed vigor testing. In International Rule for Seeds Testing (pp. 1-20). Brassersdorf: International Seed Testing Association.
ISTA (2017). The germination tests. In International Rule for Seeds Testing (pp. 1-62). Brassersdorf: International Seed Testing Association.
Kamara E. G., Olympio N. S., Asibuo J. Y., Kabbia M. K., Yila K. M., & Conteh A. ( 2017). Effect of calcium and phosphorus fertilizer on seed yield and nutritional quality of groundnut (Arachis hypogaea L.). Intl J. Agric. For. 6: 129-133.
Kemerait R., Culbreath A., Prostko E., Brenneman T., Tubbs S., Srinivasan R., Abney M., Monfort S., and Rabinowitz A.. 2020. Peanut Rx: Minimizing diseases of peanut in the southeastern United States. 2020 Peanut Update 36–55.
Lamichhane J. R., Debaeke P., Steinberg C., You M. P., Barbetti M. J., & Aubertot J. ( 2018). Abiotic and biotic factors affecting crop seed germination and seedling emergence: a conceptual framework. Plant Soil 432: 1-28.
Li L., Wang X. F., & Zhou G. H. ( 2011). Effect of Seed Quality on the Proportion of Seed Transmission for Sugarcane Mosaic Virus in Maize. Cereal Res. Commun. 39:257-266.
Marcos-Filho J. ( 2005). Seed physiology of cultivated plants. FEALQ.
Morais Junior I. J. ( 2017). Assessment of physiological quality of canola seeds produced from plants infected by Cauliflower mosaic virus (CaMV) [Undergraduate thesis, Federal University of Uberlandia]. Federal University of Uberlandia Repository. https://repositorio.ufu.br/handle/123456789/18992.
Pappu S. S., Pappu H. R., Culbreath A. K., & Todd J. W. ( 1999). Localization of Tomato spotted wilt virus (Genus Tospovirus, Family Bunyaviridae) in peanut pods. Peanut Science 26(2): 98-100.
Popinigis F. ( 1985). Seed Physiology. Agiplan.
Racette K., Rowland D., Tillman B., Erickson J., Munoz P., & Vermerris W. ( 2019). Transgenerational stress memory in seed and seedling vigor of peanut (Arachis hypogaea L.) varies by genotype. Environ. Exp. Bot 162: 541-549.
Ross L. F., Lynch R. E., Conkerton E. J., Demski J. W., Daigle D. J., & McCombs C. ( 1989). The effect of peanut stripe virus infection on peanut composition. Peanut Science 16(1): 43-45.
Sanders T. H., Schubert A. M., & Bett K. L. ( 1994). Quality characteristics of red testa peanuts associated with tomato spotted wilt virus infected plants. Peanut Science 21(2): 81-83.
Singh J., Michelangeli J. A. C., Gezan S. A., Lee H., & Vallejos C. E. ( 2017). Maternal Effects on Seed and Seedling Phenotypes in Reciprocal F1 Hybrids of the Common Bean (Phaseolus vulgaris L.). Front. Plant Sci. 8(42). doi: 10.3389/fpls.2017.00042.
Srinivasan R., Abney M. R., Culbreath A. K., Kemerait R. C., Tubbs R. S., Monfort W., & Pappu H. ( 2017). Three decades of managing Tomato spotted wilt virus in peanut in southeastern United States. Virus Research, 241. http://dx.doi.org/10.1016/j.virusres.2017.05.016.
Suassuna T.M.F., Suassuna N.D., Holbrook C.C., Culbreath A.K., Bag S., Deraniyagala A.S. ( 2019a, 9-11 July) Relationship among field and post-harvest evaluations of spotted wilt in Arachis germplasm [Poster presentation]. 2019 APRES Annual Meeting, Auburn, Alabama.
Suassuna T.M.F., Suassuna N.D., Moretzsohn M.C., Leal-Bertioli S.C.M., Bertioli D.J. & Medeiros E.P. ( 2015). Yield, market quality, and leaf spots partial resistance of interspecific peanut progenies. Crop Breeding and Applied Biotechnology 15: 1175-180.
Suassuna T.M.F., Grey T.L., Luo X., Culbreath A.K. & Pilon C. ( 2021) Physiological Quality of Peanut Seed Affected by Tomato Spotted Wilt Disease. Agronomy Journal, accepted.
Tillman B. L., & McKinney J. L. ( 2018). Relationships among symptoms of spotted wilt disease of peanut and their potential impact on crop productivity and resistance breeding. Plant Breeding 137(5): 757-762.
UGA (University of Georgia) Extension. ( 2020). “Potential Seed Quality issues in 2020 – What can I do to reduce the risk of having stand issues?”. University of Georgia Cooperative Extension. https://site.extension.uga.edu/benhillcoag/2020/03/potential-seed-quality-issues-in-peanut-what-you-can-do-to-reduce-the-risk
Notes
- First, and second authors: Graduate Student, Assistant Professor, Crop & Soil Sciences Dept., University of Georgia, Tifton, GA 31794; Third author: Researcher, Peanut Breeding Program, Embrapa Algodão, Campina Grande, PB, Brazil. [^] Corresponding author’s E-mail: leticiadeaguilamoreno@uga.edu