Introduction
Georgia is the top peanut (Arachis hypogaea L.) producing state in the U.S. accounting for roughly half of all peanuts produced each year (Archer, 2016). Within the state, approximately 40 to 60% peanut hectarage is irrigated in any given growing season depending upon increase or decrease in overall peanut supply and demand factors.
Each year, insect and disease problems can also affect overall peanut production. However, current insecticides and fungicides are very effective but can be costly. Unfortunately since the mid-1980s, tomato spotted wilt (TSW) disease caused by Tomato spotted wilt virus (TSWV) has persisted in the southeastern U.S. growing region (Branch and Culbreath, 2015). Cultivar resistance has been the primary control strategy for TSWV and is used in combination with other management practices like planting dates (Brown et al., 2005).
General or horizontal field resistance to TSWV should not be confused with specific or vertical resistance (Fehr, 1987; Vanderplank, 1984). Immunity to TSWV has not lasted very long in other crops, since it is normally controlled by single gene, which puts pressure on the virus to mutate (Moury et al., 1997; Roggero et al., 2002). In contrast, moderate to high general resistance is usually controlled by multiple genes, each acting in an additive manner which allows for both the virus and host plants to co-exist and reduces viral survival pressure. General field resistance to TSWV in peanut has been shown to be stable and long-lasting for >20 years (Branch and Culbreath, 2018). However under certain conditions, even the most resistant available cultivars may require an integrated management program to prevent yield loss due to TSWV.
The objective of this study was to assess the relative TSW incidence and general field performance among several commercially available peanut cultivars. Different testing environments were used to determine the effect of varying production practices on TSW incidence and field performance of current peanut cultivars over years and locations in Georgia.
MATERIALS AND METHODS
During three years (2017-19), 30 total field trials were conducted to determine TSW incidence and general field performance of 19 peanut cultivars. Four different production practices were compared across three Georgia locations (Tifton, Plains, and Midville).
Two-pairs of four field production practice trials were planted early in April each year at Tifton and Plains, GA. At each location, one field trial involved maximum-input production practices of recommended pesticides for weed, disease, and insect control with irrigation. The other early-planted field trial did not receive any foliar fungicides or insecticides and no irrigation.
Another set of paired production practice field trials had an optimum-planting date in May each year as recommended for better TSW control; whereas, April-plantings usually have higher TSW incidence. These statewide Official Variety Trials (OVT) utilized maximum-input production practices of pesticides both with and without irrigation at three Georgia locations (Tifton, Plains, and Midville).
A randomized complete block field design with six replications was used for each location. These four varying production practice field trials were conducted on a Tifton loamy sand soil type (fine-loamy, siliceous, thermic Plinthic Kandiudult) at Tifton, GA; a Greenville sandy clay loam (fine, kaolinitic, thermic Rhodic Kandiudult) at Plains, GA; and a Dothan sandy loam (fine-loamy, kaolinitic, thermic Plinthic Kandiudult) at Midville. All plots in each test consisted of two-rows, 6.1 m long x 1.8 m wide.
TSW disease occurrence included classical chlorotic ring spot symptoms on leaves of both stunted as well as normal size plants. TSW incidence (0-100%) was first assessed at approximately midseason (70 days after planting), when TSW is typically the only disease occurring during that part of the growing season. Additionally, percentages of total disease incidence were scored prior to digging, which included primarily TSW but also soilborne diseases. A disease “hit” equaled one or more diseased plants in a 30.5-cm section of row.
Each entry was individually dug near optimum maturity based upon the hull-scrape method from adjacent border plots (Williams and Drexler, 1981). After harvest, peanut pods from each plot were dried with forced warm-air to approximately 6% moisture. Pod samples were then cleaned before weighing for yield determinations. Grade samples were presized and shelled on federal state inspection service equipment accordingly for runner and virginia-type peanuts, respectively. Gross dollar values, were calculated from yield and grade based upon peanut loan schedules for each year.
Data from these tests were combined and statistically analyzed by analysis of variance (ANOVA) using general linear model. Waller-Duncan’s T-test (k-ratio = 100) were used for mean separation involving three or more means, and T-tests were used for only two mean comparisons in SAS (SAS Institute, Inc., Cary, NC).
RESULTS AND DISCUSSION
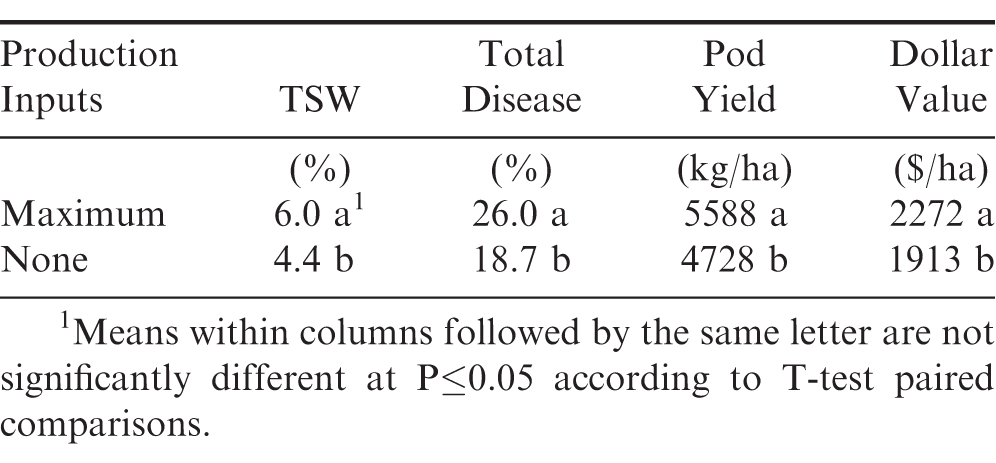
Field performance trials were conducted for three-years (2017-19) at Tifton and Plains, GA to compare two different early-planted tests involving maximum-input production practices with irrigation versus no-fungicides, no-insecticides, and without irrigation (Table 1). The maximum-input production practices with irrigation resulted in significantly (P≤0.05) higher percentage of TSW and total disease incidence, but as expected had the higher pod yields and dollar values. The highest TSW and total disease percentages in the maximum-input production practices with irrigation confirms an earlier report (Branch et al., 2003), which included different runner and virginia-type cultivars compared to this current study.
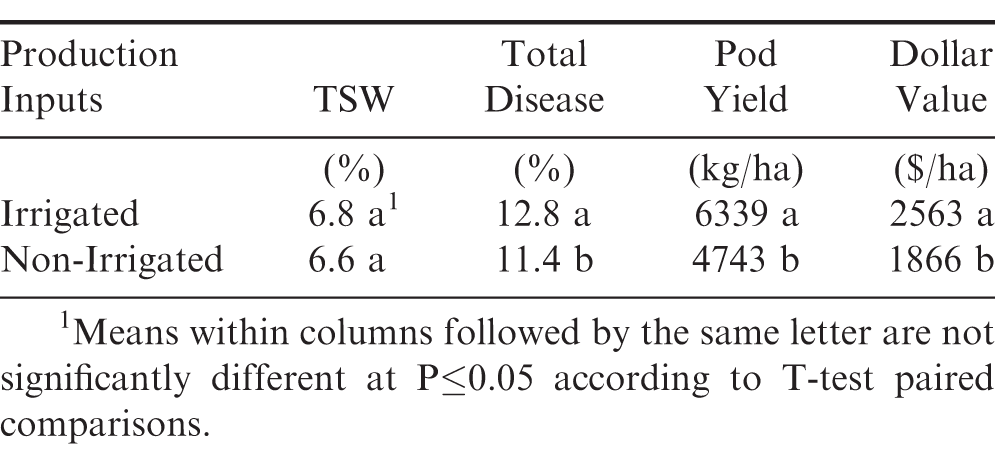
During 2017-19, field performance trials were likewise conducted at Tifton, Plains, and Midville locations to compare two optimum-planted tests for maximum-input production practices with and without irrigation (Table 2). The mid-season TSW incidence showed no significant differences between irrigated and non-irrigated trials. However, the end-of-season total disease percentages, predominantly consisting of TSW, did show significantly higher percentage in the irrigated tests compared to the non-irrigated tests involving both maximum-input production practices. Similar to the early-planted tests, the average pod yields and dollar values were significantly higher in the irrigated tests compared to the non-irrigated tests. These results agree with a previous report involving the Georgia peanut OVT irrigated test having a significantly higher pod yields and dollar values compared to the non-irrigated test at these same locations (Isleib et al., 2014).
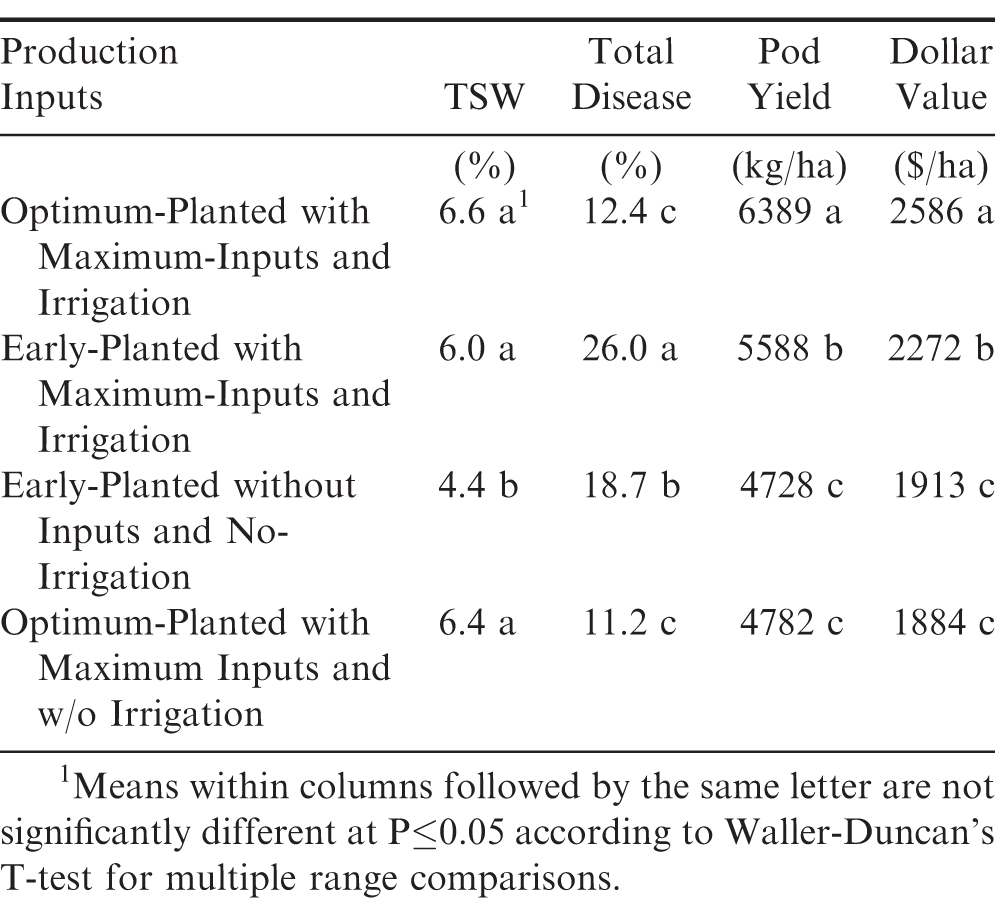
In the overall four tests comparison between each of the two early-planted tests and each of the two optimum-planted tests, disease results showed significantly lower TSW incidence in the early-planted tests without fungicide, insecticide, or irrigation compared to the other three test conditions (Table 3). As recommended for planting dates by the Georgia Cooperative Extension Service, average field performance for pod yields and dollar values were significantly highest in the optimum-planted tests with maximum-input production practices including irrigation in comparison to the other three testing environments. These findings show that the highest average field performance for pod yield and dollar value also had among the lowest total disease incidence and among the highest TSW incidence which does not agree with an earlier report (Brown et al., 2005). Additional reports have also noted earlier April planting dates had more TSW compared to optimum-planting dates in May (Culbreath et al., 2010; Nuti et al., 2014; Tillman et al., 2007). It should be pointed out that in this study, the TSW percentages at mid-season are relatively low at <10%. In addition, another probable explanation is that the current peanut cultivars have much higher TSWV general field resistance than past cultivars.
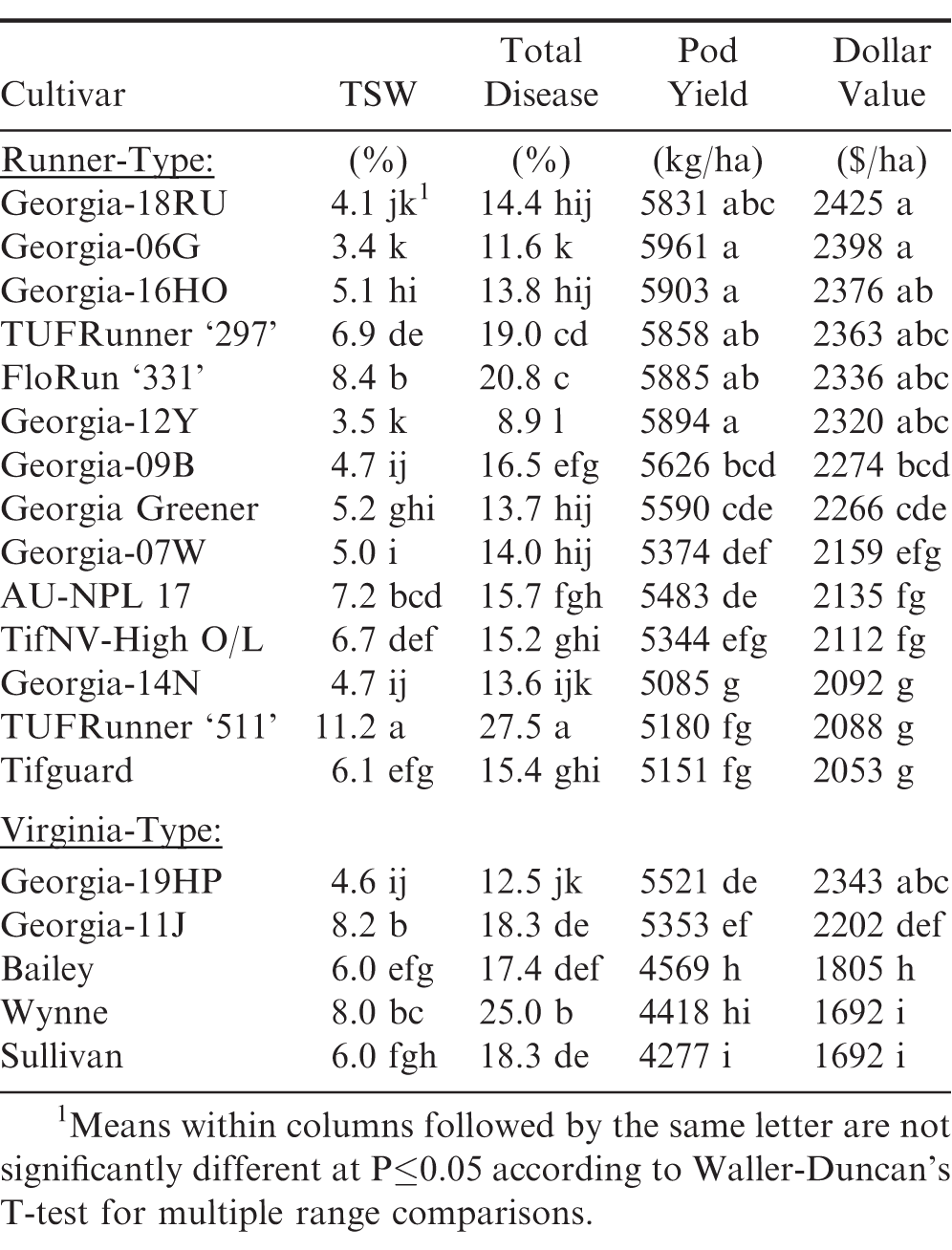
Table 4 shows the three-year relative TSW incidence and field performance of 14 current runner and five virginia market type peanut cultivars averaged over these 30 tests. Significant differences were found among these 19 peanut cultivars for TSW and total disease incidence as well as pod yields and dollar values. ‘Georgia-06G’ (Branch, 2007), ‘Georgia-12Y’ (Branch, 2013), and ‘Georgia-18RU’ (Branch, 2019) had the lowest TSW incidence at midseason compared to other runner cultivars; whereas, ‘Georgia-19HP’ (Branch and Brenneman, 2020) had the lowest TSW and total disease incidence among the virginia-type cultivars. Among the runner-type cultivars, Georgia-12Y had the lowest total disease incidence which included predominantly TSW but also any soilborne diseases (i.e. white mold or stem rot) observed at the time of rating. Since Georgia-12Y has good resistance to Sclerotium rolfsi, Sacc., it is not surprising to see these results at the end of the growing season.
The highest pod yield was found with Georgia-06G, ‘Georgia-16HO’ (Branch, 2017), and Georgia-12Y, but these three cultivars were not significantly different in yield from Georgia-18RU, FloRun ‘331’ (Tillman, 2021), and TUFRunner ‘297’ (Tillman, 2018). Likewise, Georgia-18RU and Georgia-06G had the highest dollar values, but these two runner-type peanut cultivars were not significantly different from Georgia-16HO, TUFRunner ‘297’, FloRun ‘331’, and Georgia-12Y. The new virginia-type peanut cultivar Georgia-19HP, which also had the highest pod yield and dollar value among the virginia-types, was not significantly different from ‘Georgia-11J’ (Branch, 2012) in pod yield.
It is interesting to note that among these six runner cultivars and two virginia cultivars with the highest field performance for pod yield and dollar values, the Georgia cultivars had the lowest TSW and total disease incidence. The highest disease percentages were found with the more susceptible TUFRunner ‘511’; cultivar (Tillman and Gorbet, 2017) which had 11.2% TSW and 27.5% total disease incidence at the end of the growing season, respectively.
TSW persists as an important disease in southeastern U.S. peanut production areas, however, general TSWV field resistance among several of these cultivars appears to be significantly better than many of the more susceptible cultivars. The TSWV resistance of Georgia-06G has previously been shown to be very stable across many years (Branch and Culbreath, 2018), and now shows stable TSWV resistance across different input production practices. Because TSW incidence can be somewhat sporadic in any given test, location, and year, evaluations should include pooled data across as many different environments as possible for an accurate comparison of relative general TSWV resistance in peanut.
Literature Cited
Archer, P. 2016. Overview of the peanut industry supply chain. Chap. 9 pp 253-266. In: Stalker H. T. and Wilson R. F. (ed.) Peanuts Genetics, Processing, and Utilization. AOCS Monograph Series on Oilseeds, Acad. Press., and Amer. Peanut Res. Educ. Soc., Univ. of Georgia, NESPAL Bldg., Tifton, GA 31793.
Branch, W. D. 2007. Registration of ‘Georgia-06G’ peanut. J. Plant Reg. 1: 120.
Branch, W. D. 2012. Registration of ‘Georgia-11J’ peanut. J. Plant Reg. 6: 281– 283.
Branch, W. D. 2013. Registration of ‘Georgia-12Y’ peanut. J. Plant Reg. 7: 151– 153.
Branch, W. D. 2017. Registration of ‘Georgia-16HO’ peanut. J. Plant Reg. 11: 231– 234.
Branch, W. D. 2019. Registration of ‘Georgia-18RU’ peanut. J. Plant Reg. 13: 326– 329.
Branch, W. D. and Brenneman. T. B. 2020. Registration of ‘Georgia-19HP’ peanut. J. Plant Reg. 14: 306– 310.
Branch, W. D., Brenneman T. B. and Culbreath. A. K. 2003. Tomato spotted wilt virus resistance among high and normal O/L ratio peanut cultivars with and without irrigation. Crop Protection 22: 141– 145.
Branch, W. D. and Culbreath. A. K. 2015. Stability of TSWV general field resistance in the ‘Georgia Green’ peanut cultivar. Plant Health Progress 16: 95– 99.
Branch, W. D. and Culbreath. A. K. 2018. Transgressive segregation and long-term consistency for high TSWV field resistance in the ‘Georgia-06G’ peanut cultivar. Plant Health Progress 19: 201– 206.
Brown, S. L., Culbreath, A. K. Todd, J. W. Gorbet, D. W. Baldwin, J. A. and Beasley. J. P. 2005. Development of a method of risk assessment to facilitate intergrated management of spotted wilt of peanut. Plant Dis. 89: 348– 356.
Culbreath, A. K., Tillman, B. L. Tubbs, R. S. Beasley, J. P. Kemerait, R. C.and Brenneman. T. B. 2010. Interactive effects of planting date and cultivar on tomato spotted wilt of peanut. Plant Disease 97: 898– 904.
Fehr, W. R. 1987. Breeding for pest resistance. Pages 304-314 in: Principles of Cultivar Development, Vol. 1 Macmillan Publ. Co., New York.
Isleib, T. G., Day, J. L. Coy, A. E. Beasley, J. P.and Branch. W. D. 2014. Genotype-by-irrigation interaction in the Georgia peanut official variety test. Peanut Sci. 41: 1– 7.
Moury, B., Palloix, A., Selassie, K.G., and Marchoux, G. 1997. Hypersensitive resistance to tomato spotted wilt virus in three Capsicum chinense accessions is controlled by a single gene and is overcome by virulent strains. Euphytica 94: 45– 52.
Nuti, R. C., Chen, C. Y. Dang, P. M. and Harvey. J. E. 2014. Peanut cultivar response to tomato spotted wilt over five planting dates. Peanut Sci. 41: 32– 41.
Roggero, P., Masenga, V., and Tavella, L. 2002. Field isolates of tomato spotted wilt virus overcoming resistance in pepper and their spread to other hosts in Italy. Plant Dis. 86: 950– 954.
Tillman, B. L. 2018. Registration of TUFRunner ‘297’ peanut. J. Plant Reg. 12: 31– 35.
Tillman, B. L. 2021. Registration of FloRun ‘331’ peanut. J. Plant Reg. 15: (in press).
Tillman, B. L. and Gorbet. D. W. 2017. Registration of TUFRunner ‘511’ peanut. J. Plant Reg. 11: 235– 239.
Tillman, B. L, Gorbet, D. W. and Anderson. P. C. 2007. Influence of planting date on yield and spotted wilt of runner market type peanut. Peanut Sci. 34: 79– 84.
Vanderplank, J. E. 1984. Horizontal and vertical resistance. Pages 57-81 in: Disease Resistance in Plants, 2nd Ed. Academic Press, New York, NY.
Williams, E. J. and Drexler. J. S. 1981. A non-destructive method for determining peanut pod maturity. Peanut Sci. 8: 134– 141.
Notes
- Professor, Dept. of Crop & Soil Sci., University of Georgia, Tifton, GA 31793 [^]
- Assistant Research Scientist, Dept of Crop and Soil Sci., University of Georgia, Tifton, GA 31793 [^]
- Public Service Assistant, Dept. of Crop & Soil Sci., University of Georgia, Griffin, GA 30223 [^]
- Professor, Dept. of Plant Pathology., University of Georgia, Tifton, GA 31793 [^] *Corresponding author Email: wdbranch@uga.edu
Author Affiliations