Introduction
Peanut production in Georgia is among the state’s top agriculture industries and comprises around 50% of the total acreage and production of all peanuts grown in the United States. In 2020, 323,887 ha of peanuts were harvested in Georgia contributing approximately 656,000,000 US dollars to the state’s agriculture economy (USDA-NASS, 2020). To protect crop yields and profits throughout the season, peanut growers rely heavily on pesticides for pest management every year. Among different pesticide applications in peanut, herbicides and fungicides are the most frequently used (applied to 94% and 88%, respectively of the planted acres), according to an agricultural chemical use survey conducted by USDA-NASS in 2018 in six states (including Georgia) that accounted for 93% of the 0.57 million hectares planted to peanuts in the United States (USDA-NASS, 2018). Considering the importance and volume of pesticide applications made each year in peanut production, maximizing the effectiveness of pesticide application remains a top priority for growers. Consequently, more research efforts have been focused on understanding the performance of spray equipment under different application conditions as well as evaluating technologies that can provide improved coverage and efficacy for effective pest management in peanuts.
Spray technology has evolved considerably over the last decade with a rising trend towards increased application widths and faster moving equipment to cover more acres per tank load (Etheridge et al., 1999). However, pesticide applications at faster speeds and improper selection of spray parameters have also raised concerns regarding off-target movement of pesticides to neighboring vegetation, water bodies and wildlife (de Snoo and de Wit, 1998; Freemark and Boutin, 1995; Rodrigues et al., 2020). Increased occurrence of off-target movement of chemicals during pesticide applications has resulted in more emphasis on controlling variables that can help minimize such issues. Selection of spray parameters (application speed, nozzle type, spray pressure and boom height) and environmental conditions (wind speed and direction, temperature, humidity) at time of application can influence spray performance and off-target movement (Creech et al., 2015). While environmental conditions such as wind speed and direction cannot be controlled by the operator, managing controllable factors such as nozzle type, pressure, speed, and boom height are important considerations in controlling off-target movement during pesticide applications.
When considering ways to control off-target movement, nozzle type is an important tool as it influences droplet size and spray distribution (Rogers and Maki, 1986; Taylor et al., 2004). Selection of correct nozzles is also considered important to attain desired spray coverage and product efficacy during pesticide applications. Generally, finer droplets are needed for increased coverage and efficacy (Etheridge et al., 2001), however they are also more susceptible to increased drift potential. Thus, a common practice to reduce off-target movement of spray particles is to increase droplet size by reducing pressure or by selecting a nozzle with larger orifice size (Creech et al., 2015; Hartley and Graham-Bryce, 1980). Though this conventional practice helps minimize off-target movement, it also influences application rate if not properly accounted for with additional changes to the application speed (Knoche, 1994) and potentially pesticide efficacy (in case of contact herbicides) as larger droplets provide less coverage (Spillman, 1984). To address this issue, nozzle manufacturers have designed new types of nozzles to achieve coarser spray droplets to decrease off-target movement while maintaining adequate droplet distribution at the same pressure for optimal pesticide efficacy (Piggot and Matthews, 1999; Ramsdale and Messersmith, 2001). Several researchers reported that air-induction nozzles exhibited improved drift control and similar/increased efficacy (coarser droplets) when compared to conventional flat fan nozzles (medium droplets) at a given pressure (Etheridge, 1999; Lund, 2000; Wolf, 2005; Sikkema et al., 2008).
Recently, increased occurrence of herbicide resistant weeds due to selection pressure has led agriculture companies to develop auxin-resistant corn, cotton and soybean (USDA-APHIS, 2015). Thus, auxin herbicides are extensively used in current production systems for weed control. However, other broadleaf plant species (not resistant to auxin herbicides) are highly sensitive and can exhibit significant damage at very low drift rates (Egan et al., 2014). Therefore, the current herbicide labels (for auxin tolerant crops) contain specific application requirements including considerations for wind speed, time of application, buffer zones, application speed, boom height, and nozzle type to mitigate any potential off-target movement of spray particles. Labels for auxin herbicides registered for use in tolerant crops require that the growers must use approved air-induction nozzles (labeled for use with auxin herbicides, hereby “AI nozzles”) within the recommended pressure range to produce the larger droplets (coarser) needed to minimize potential off-target movement during application. In the southeastern United States, the adoption of auxin-resistant technologies has been steadily increasing as more growers are planting auxin-resistant cultivars so they can utilize dicamba or 2,4-D as primary herbicides in their weed management programs. For examples, Georgia cotton producers planted auxin-tolerant varieties on 98% (96%-dicamba + 2%-2,4-D) of the total acres planted in cotton in 2020 (USDA-AMS, 2020).
In Georgia, peanut is an important rotational crop with cotton due to its many benefits such as its ability to provide nitrogen to the soil, reduced disease and insect pressure, and easier to manage weed pressure (Elkan, 1995; Vencill et al., 2012). Following label requirements for auxin herbicides in cotton also means that growers could utilize the recommended AI nozzles on peanuts as routinely changing nozzles between crops as well as between different pesticide applications is very uncommon for growers. Most pesticides used in peanut require medium spray droplets for optimum coverage; medium droplets are more prone to off-target movement than coarser droplets achieved with AI nozzles. Since larger droplets can decrease spray coverage (Spillman, 1984), pesticide applications in peanut with AI nozzles could mean compromised coverage resulting in reduced pest control, which can eventually lead to yield loss. Thus, peanut growers are questioning whether AI nozzles (labeled for use with auxin herbicides) are appropriate to apply all pesticides (herbicides, insecticides and fungicides) for effective pest control as compared to the conventional flat fan nozzles (“non-AI nozzles”).
Numerous studies have investigated the effect of nozzle type on droplet size, coverage, control and efficacy of pesticide applications in different crops (Sikkema et al., 2008; Meyer et al., 2016; Rodrigues et al. 2020). However, limited research exists on evaluating the effect of nozzle type on pest control and efficacy in peanut (Berger et al. 2014; Carter et al. 2017). Berger et al. (2014) reported no differences in herbicide efficacy between the non-AI and AI nozzles despite reduced coverage for the AI nozzle. Carter et al. (2017) compared three different nozzle types [AIXR11002 (coarse droplets), TTI11002 (ultra coarse droplets), and DG11002 (medium droplets)] for weed control in peanut herbicide systems. The authors reported reduced grass control (5-6%) with the TTI nozzle but no differences in Palmer amaranth control and yield were observed between the nozzle types. It should be noted that most studies on evaluating nozzle type are conducted utilizing small-plot research techniques with herbicide applications performed using a CO2-powered backpack sprayer. Consequently, results attained in these studies need to be verified in large-scale field trials using commercial application equipment typically used by growers. Additionally, most commercial applications are performed at speeds >16 km hr−1 which is quite difficult to simulate when implementing small-plot research trials. Moreover, the effect of nozzle type on pest management in peanut has not been fully investigated across all pesticide applications (herbicides, fungicides and insecticides). Therefore, the objective of this study was to evaluate and compare the performance of AI (very coarse to ultra coarse droplets) and non-AI nozzles (medium to coarse droplets) on pest control and efficacy in peanut through large-scale on-farm trials using commercial boom sprayers.
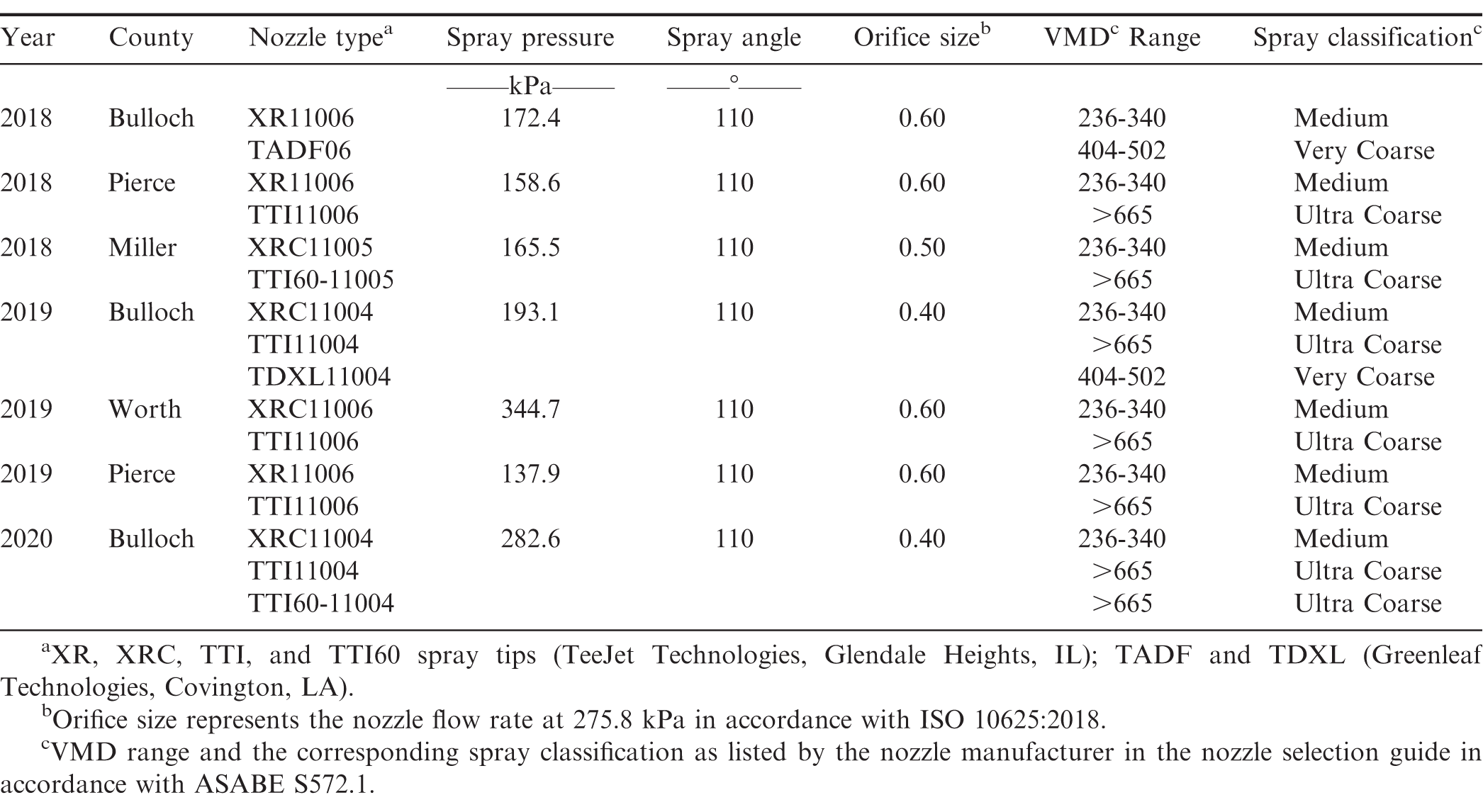
Materials and Methods
Seven field experiments were conducted in commercial peanut fields using commercial sprayers from 2018 to 2020 at four locations in Georgia. To compare the performance of non-AI (medium to coarse droplets) to AI nozzles (very coarse to ultra-coarse droplets), treatments for each experiment consisted of two to three different nozzle types with at least one nozzle representing a conventional non-AI nozzle [XR and XRC (TeeJet Technologies, Springfield, IL)], and one or two other nozzles representing an AI nozzle [TADF and TDXL (Greenleaf Technologies, Covington, LA 70433); TTI and TTI60 (TeeJet Technologies, Springfield, IL, 62701)]. Table 1 provides detailed information (by study year and location) on nozzle types, operating spray pressure, spray angle, nozzle orifice size, and spray classification for the nozzles used during each on-farm test. All nozzles tested in these studies had a 110° spray angle while the nozzle orifice sizes varied from 0.40 to 0.60 depending on the target application rate and spray pressure required to obtain that rate. Orifice size is related to the nozzle capacity (flow rate in gallons per minute) at 275.8 kPa as nozzle flow rate is a function of orifice size and pressure (ISO 10625, 2018). Spray classification in Table 1 represents the expected droplet size (VMD range) and spray quality listed by the nozzle manufacturer in the nozzle selection guide in accordance with the droplet size classification standard ASABE S572.1 (ASABE, 2018). VMD represents the median droplet size, in microns (μm), at which half of the spray volume is composed of spray droplets larger than the VMD and the other half of the spray volume with spray droplets smaller than the VMD. As listed in Table 1, the expected spray quality, based on the orifice size and operating pressure, from the non-AI nozzles (XR and XRC) is medium droplets whereas it is very coarse to ultra-coarse droplets for the AI nozzles (TTI, TTI60, TADF and TDXL) at the selected nozzle size and pressure.
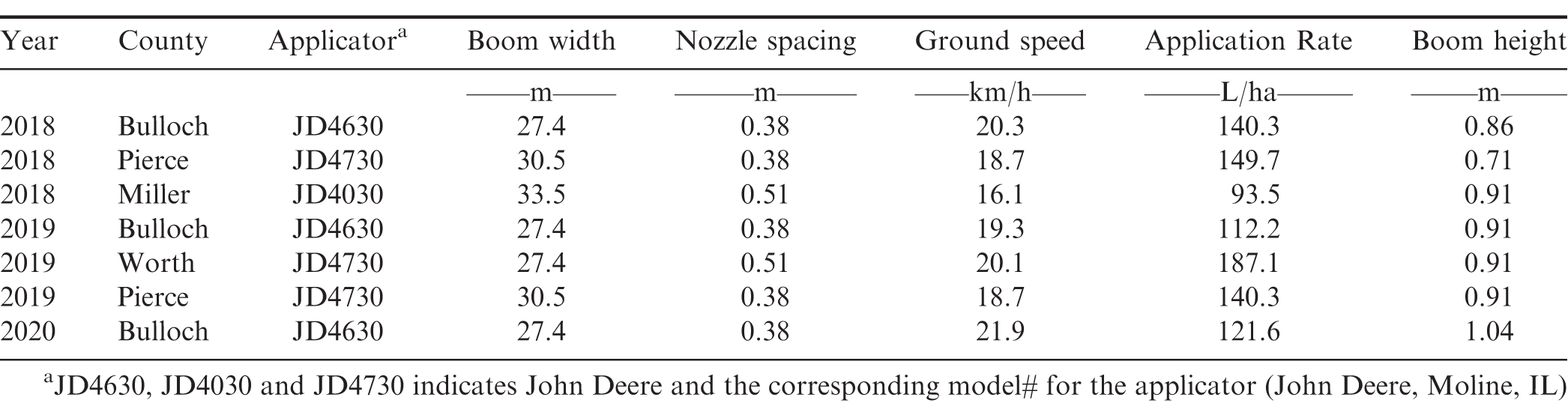
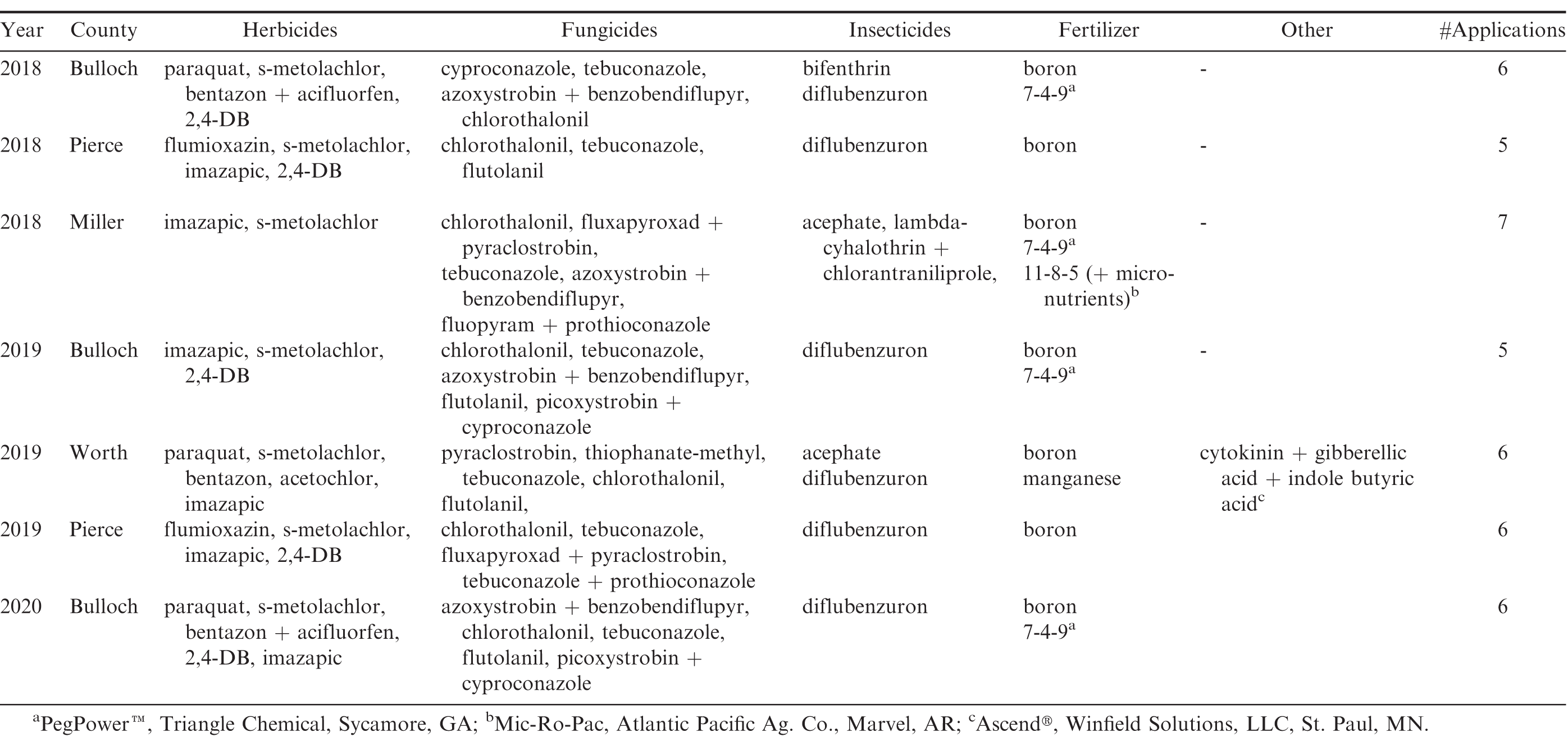
For each test, treatments - consisting of different nozzle types - were implemented in long replicated strips where each sprayer pass represented a nozzle treatment. Consequently, each sprayer pass was treated as an individual plot where plot width was equivalent to the boom width (27.4 – 33.5 m) and plot length equal to the length of the peanut field (304.8 – 607.4 m). Each nozzle treatment was replicated three to four times depending on available field space and all replications were randomized in the field. All application parameters except nozzle type remained constant between the treatments for each test and were selected by growers based on their preferred spray application practices. Table 2 presents information on applicator, boom width, nozzle spacing, ground speed, application rate, and boom height utilized during the on-farm nozzle tests conducted at each location. All agri-chemical applications for the entire production year were made by the grower and were applied according to the grower’s typical production practices (Table 3).
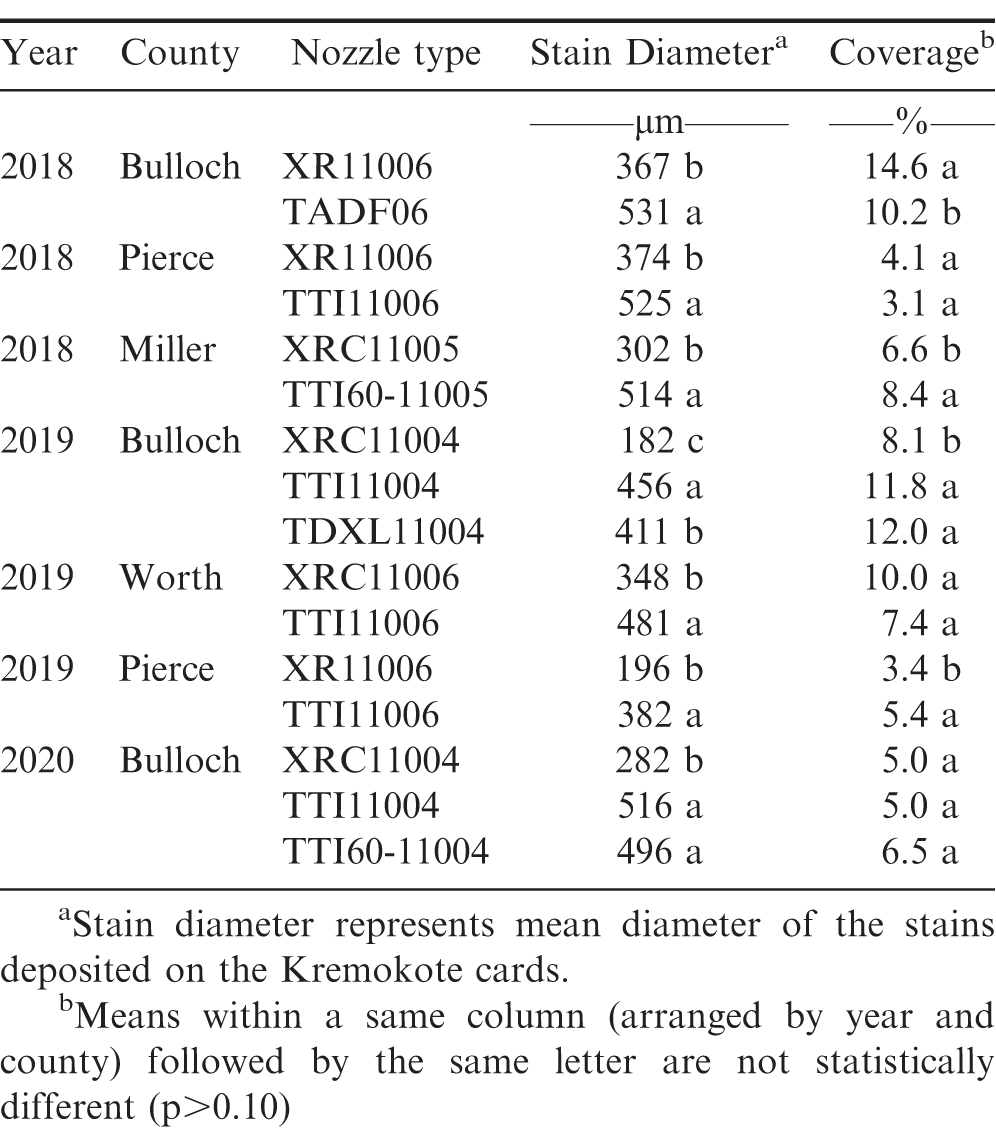
Prior to peanut planting and pesticide applications, spray deposition data (stain diameter and coverage) was collected using Kromekote cards that measured 5.1 by 7.6 cm. Kromekote cards are a specialty photo paper that stains when a spray solution containing dye is deposited on it (Johnstone 1960), and have been commonly used for spray deposition analysis by researchers. Kromekote cards were placed on the ground in a 3.1 × 3.1 m grid pattern in the field (5 to 9 cards along the spray width) and sprayed with water mixed with a dye (Vision Pink™, Garrco Product Inc., Converse, IN) using the selected nozzle types (Table 2) and same spray settings (Table 3) as utilized by the growers during actual pesticide applications. Spray quality data can vary between different pesticide solutions due to difference in viscosity and surface tension among the solutions. Although it is feasible to collect spray deposition data with actual pesticides (instead of water) using water-sensitive paper, considering the number of different agri-chemicals used and applications made throughout the season in each field, it was not feasible as well as beyond the scope of this study to collect spray quality data for all pesticides used in this study. For all tests, spray deposition data were collected three times (3 replications) for each nozzle providing 15 to 27 Kromecote cards for each treatment. After being sprayed with water and dye, the cards were allowed to dry and then carefully collected and stored in standard paper envelopes. A software program DropletScan™ (WRK of Oklahoma, Stillwater, OK) coupled with a high-resolution flatbed scanner CanoScan 5600F (Canon USA, Melville, NY) was used to assess stain size diameter and coverage on the Kromekote cards. The software was set to scan 75% of the card area and the resolution of the scanner was set to 30 μm per pixel.
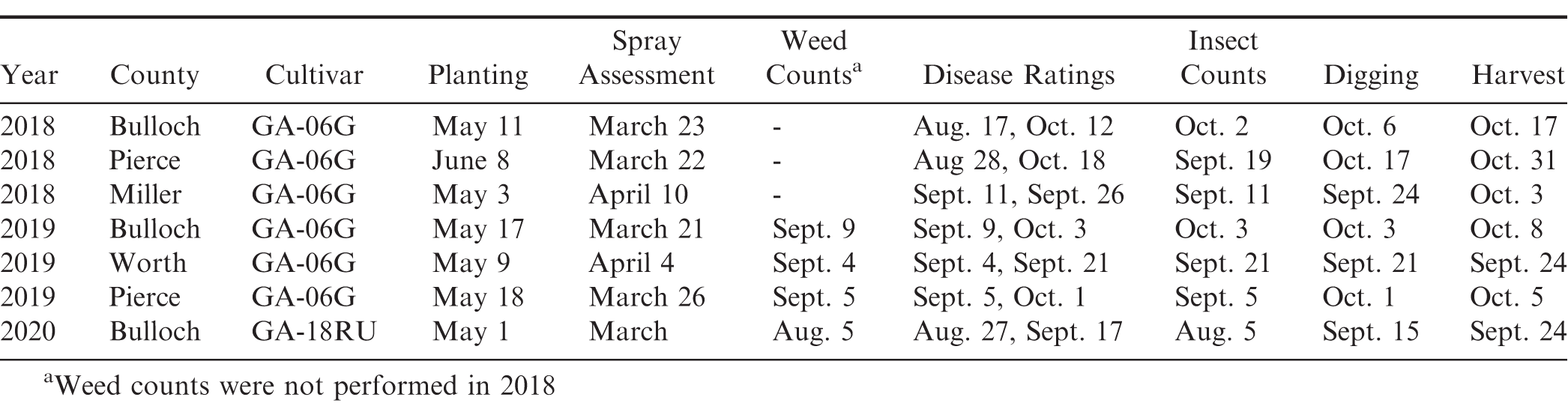
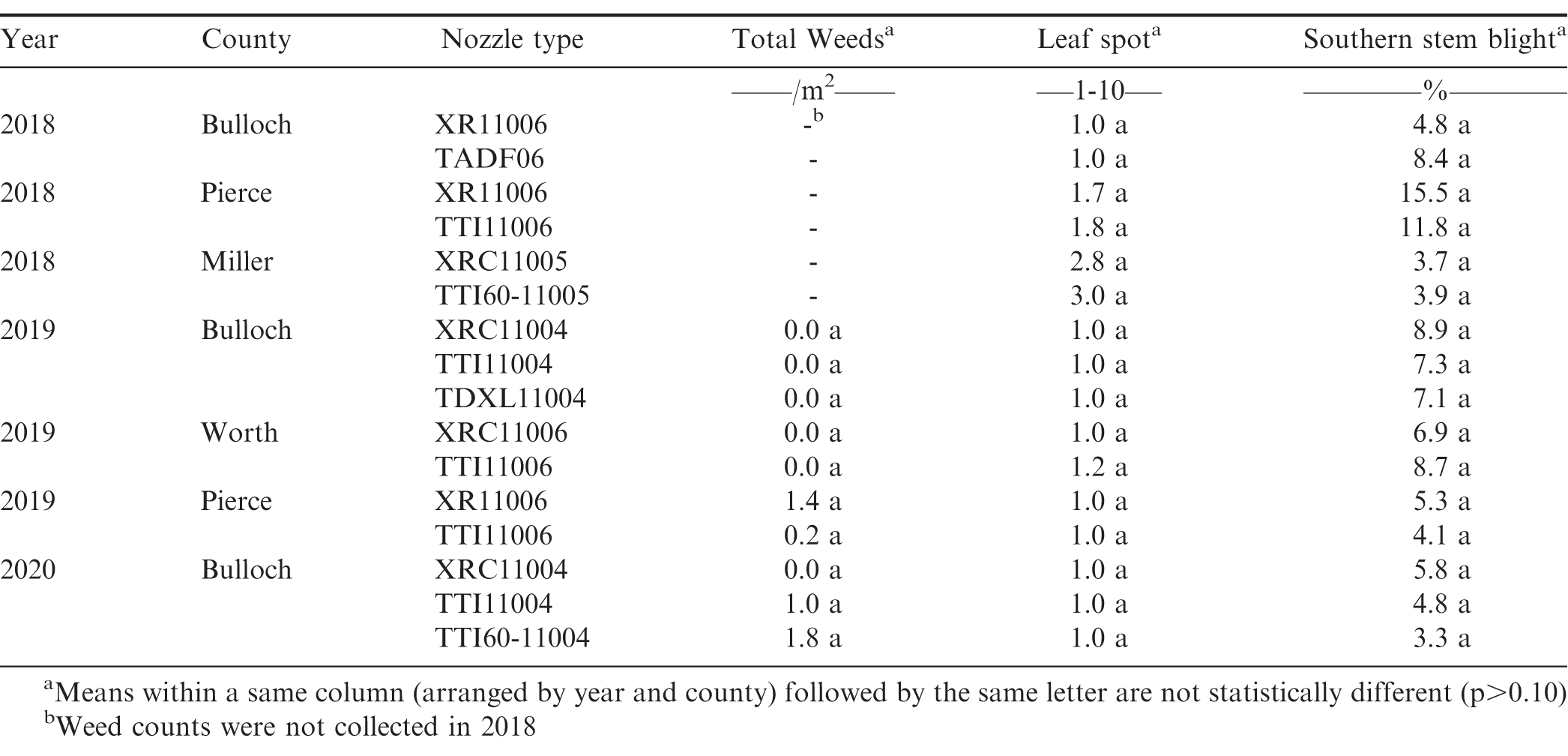
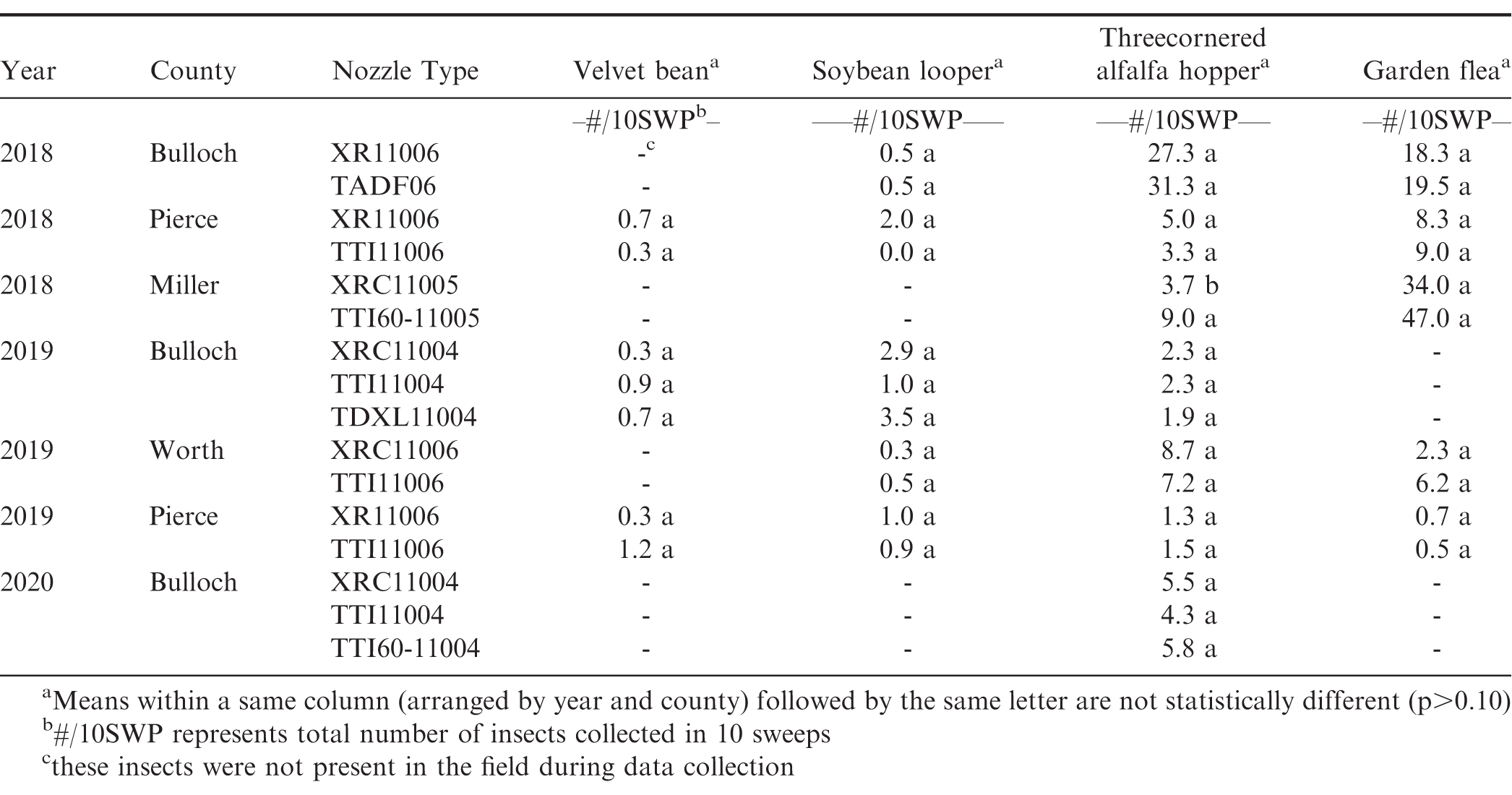
Weed density data were obtained by counting the number of weeds per m2 after all herbicides were applied. Insect data were obtained by counting the number of insects in 10-15 sweeps per plot (38 cm diameter sweep net) after all insecticides were applied. Insects commonly observed in the peanut fields during these studies were velvetbean caterpillar [Anticarsia gemmatalis (Hübner)], soybean looper [Chrysodeixis includens (Walker)], threecornered alfalfa hopper [Spissistilus festinus (Say)], and garden fleahopper [Halticus bractatus (Say)]. After all fungicide applications were made, early/late leaf spot [Cercospora arachidicola (Hori) U. Braun and Cercosporidium personatum (Berk. & Curt.)] severity was visually estimated using the Florida 1-10 leaf spot severity scale where 1 = no disease, 0% defoliation, and 10 = 100% defoliation, plants dead, killed by leaf spot (Chiteka et al., 1988). Southern stem blight or white mold [Sclerotium rolfsii (Sacc.)] severity was determined by counting the number of disease loci per 0.31 m (per foot) of row length and converting to the percent of row infected. Peanut yield data were collected by harvesting the center six rows (5.5 m, 6 ft) of each test strip using commercial harvesting equipment and a wagon equipped with scales that facilitates the weighing of large peanut plots (Figure 1). The harvest plot length varied between 304.8 and 607.5 m (1000 and 1993 ft) based on the field length. Peanut yields were adjusted to 10% moisture. Table 4 provides a complete timeline including peanut planting, spray assessment, weed and insect counts, disease ratings, and peanut digging and harvest dates for all on-farm nozzle tests conducted in Georgia from 2018 to 2020.
Data for each test location and year were analyzed separately due to differences in grower production practices and field conditions between the locations and study years. For each study, data were subjected to analysis of variance (ANOVA) using the PROC GLM procedure in SAS 9.4 (SAS Institute, Cary, NC 27511) where nozzle type was considered as an explanatory variable, and droplet size, coverage, weed, disease, insect data, and peanut yield were treated as response variables. Least square means for the response variables were compared using MEANS LSD option within the PROC GLM procedure using an alpha value of 0.10 (p≤0.10).
Results and Discussion
Spray Assessment
Table 5 presents the spray assessment data (stain diameter and coverage) for the different nozzle types evaluated during each on-farm test. From stain diameter, it can be noticed that the size of the spray droplets produced by the AI nozzles (TADF, TTI, TDXL and TTI60) were significantly larger than the spray droplets produced by the non-AI nozzles (XR and XRC) at the same orifice size and spray pressure. These results were expected as AI nozzles tend to produce larger droplets than non-AI nozzles at same nozzle size and pressure, and thus the reason they are recommended for spraying auxin herbicides to reduce off-target movement of spray particles. Creech et al. (2015) and Rodrigues et al. (2020) also reported similar findings where the AI nozzles (TTI and AIXR) produced larger droplets than the non-AI nozzles (XR).
Spray coverage results were variable among the nozzle tests. In three out of seven tests (Pierce – 2018, Worth – 2019, and Bulloch – 2020), spray coverage was not significantly different between the AI and non-AI nozzles, despite differences in droplet size between these nozzles. In general, smaller droplets (medium - coarse) are expected to provide better coverage than larger droplets (very coarse - ultra coarse) due to the presence of more fines; however, this was only noticed in one on-farm nozzle test (Bulloch - 2018) where the XR nozzle provided better coverage than the TADF nozzle. In three other tests (Miller – 2018, Bulloch – 2019, and Pierce - 2019), the spray coverage obtained from the AI nozzles (TTI60, TTI and TDXL, and TTI, respectively) was greater than the non-AI nozzles (XRC, XRC, and XR, respectively) used in these tests. Other studies have also reported similar inconsistent coverage results between these nozzle types, where in some cases the XR nozzles exhibited greater coverage than the TTI nozzles (Ferguson et al., 2020), and in other cases the coverage produced by the TTI nozzles was similar to the XR nozzles (Legleiter and Johnson, 2016).
Pest Control
Nozzle type had no effect on weed, disease, and insect control in any of the on-farm tests conducted in this study (Table 6 and Table 7). The only exception to this was the observed significant difference in threecornered alfalfa hopper counts between the XRC and TTI60 nozzle in 2018 for the nozzle test conducted in Miller county. This specific finding was unexpected and cannot be explained by the parameters measured in this trial. Spray coverage results for this field also showed greater coverage for the TTI60 nozzle than the XRC nozzle which suggests that the TTI60 nozzle would have provided better control due to increased coverage if nozzle type had any influence on insect control. Typically, smaller droplets produced by non-AI nozzles (XR and XRC) should provide better efficacy for non-systemic pesticides due to increased coverage (Ennis and Williamson, 1963, McKinlay et al., 1972); however, previous research on effect of nozzle type on pest control (specifically weed control) has reported varying results on correlation between droplet size and the pesticide efficacy (Shaw et al., 2000, Sikkema et al., 2008, Berger et al., 2014). These contrasting results could be attributed to interactions among pesticide efficacy and other factors such as weed species and density, tillage, and soil type (Franca et al., 2020).
One possible reason for the results observed in our study could be that the weed, disease and insect pressure in most of the fields during all three years was low to normal, which could have influenced the effect of nozzle type on pest control. A high or severe pest pressure could result in a different outcome on the influence of nozzle type than that observed in this study. Therefore, future research on nozzle evaluation could be expanded to include fields with a high weed, disease or insect pressure, and to collect additional nozzle performance data to measure spray coverage and penetration in the crop canopy during actual pesticide applications.
Peanut Yield
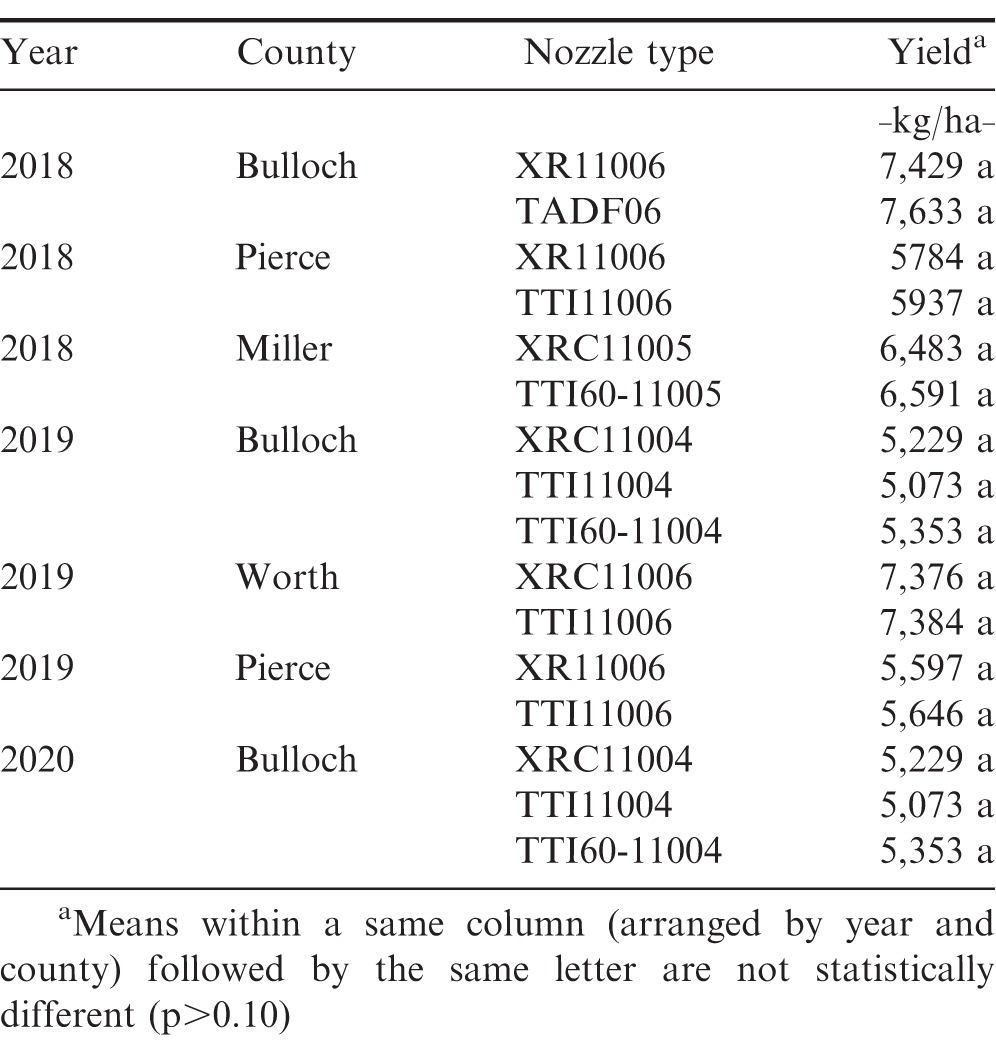
Nozzle type had no influence on peanut yield in any of the on-farm tests (Table 8). The yield results were somewhat expected as no differences in pest management (weeds, disease, and insect) were observed between the AI and non-AI nozzles in all on-farm trials. Carter et al. (2017) reported similar findings on the influence of nozzle type on peanut yield where no differences in peanut yields were observed between the nozzles that produced medium to coarse droplets (DG11002 and AIXR11002), and ultra-course spray droplets (TTI11002) at the selected pressure of 262 kPa. Though limited research exists on the influence of nozzle type on peanut yield, research studies on nozzle evaluation in other row-crops (corn, cotton, and soybeans) have reported varying results as crop yield is also influenced by pest species, density, application timing, and duration of interference (Undersander et al., 1985; Ferrell and Vencill, 2003; Sikkema et al. 2008; Everman et al., 2008).
Conclusions
The results of this research demonstrate that despite differences in droplet stain diameter (and coverage in some cases) between the non-AI (XR and XRC) and AI nozzles (TTI, TTI60, TADF and TDXL) tested in these on-farm studies, no significant differences in pest (weed, insect and disease) control and peanut yield were observed among the nozzle types. These results suggest that peanut growers who utilize AI nozzles (labelled for use with auxin herbicides) for pest control in cotton can utilize the same nozzles for pesticide applications in peanut without any concern about reduced pest control and/or yield. It should be noted that the spray performance results reported here were obtained in fields with low to average pest pressure during all study years. Presence of a high or above average pest pressure in a field could lead to different results with the same nozzles at the same or different application rates and/or spray pressures. It should also be noted that the spray deposition data in these trials were collected prior to any pesticide applications on a bare-ground surface using water mixed with dye, thus spray deposition results for the same nozzles could vary if collected using actual pesticide solution and/or in the presence of crop canopy. Future nozzle evaluation studies in peanut should investigate the influence of droplet size in combination with different spray volumes and pressures on spray coverage and penetration into crop canopy to better understand spray distribution and canopy penetration at different droplet sizes during pesticide applications.
Acknowledgements
The authors would like to greatly acknowledge the grower-cooperators who contributed to this project including the following: Greg Sikes – Bulloch County; Jim Waters – Pierce County; Kim Henley – Miller County; Steve and Gene Patterson – Worth County
Literature Cited
ASABE. 2018. ASABE S572.1 - Droplet Size Classification. Am. Soc. Agric. & Bio. Eng., St. Joseph, MI. 4 pp.
Berger S.T., Dobrow, M.H. Ferrell J.A. and Webster. T.M. 2014. Influence of carrier volume and nozzle selection on Palmer amaranth control. Peanut Sci. 41: 120– 123.
Carter O.W., Prostko E.P. and Davis. J.W 2017. The influence of nozzle type on peanut weed control programs. Peanut Sci. 44: 93– 99.
Chiteka Z. A., Gorbet, D.W. Shokes, F.M. Kucharek T.A. and Knauft. D.A. 1988. Components of resistance to late leafspot in peanut. I. Levels and variability-implications for selection. Peanut Sci. 15: 25– 30.
Creech C.F., Henry, R.S. Fritz B.K. and Kruger. G.R. 2015. Influence of herbicide active ingredient, nozzle type, orifice size, spray pressure, and carrier volume rate on spray droplet size characteristics. Weed Technol. 29: 298– 310.
de Snoo G.R. and de Wit. P.J. 1998. Buffer Zones for Reducing Pesticide Drift to Dicthes and Risks to Aquatic Organisms. Ecotoxicol. Environ. Saf. 41: 112– 118.
Egan J.F., Barlow K.M. and Mortensen. D.A. 2014. A meta-analysis on the effects of 2, 4-D and dicamba drift on soybean and cotton. Weed Sci. 62: 193– 206.
Elkan G.H. 1995. Biological nitrogen fixation in peanuts. 286-297 in Pattee H.E. and Stalker. H.T. (eds). Advances in Peanuts Science. American Peanut Research and Education Soc.
Ennis W.B.and Williamson. R.E. 1963. Influence of droplet size on effectiveness of low-volume herbicidal sprays. Weeds. 1: 67– 72.
Etheridge R.E., Hart, W.E. Hayes R.M. and Mueller. T.C. 2001. Effect of venturi-type nozzles and application volume on postemergence herbicide efficacy. Weed Technol. 15: 75– 80.
Etheridge R. E., Womac A.R. and Mueller. T.C. 1999. Characterization of the spray droplet spectra and patterns of four venturi-type drift reduction nozzles. Weed Technol. 13: 765– 770.
Everman W.J., Burke, I.C. Clewis, S.B. Thomas W.E. and Wilcut. J.W. 2008. Critical period of grass vs. broadleaf weed interference in peanut. Weed Technol. 22: 68– 73.
Ferguson J.C., Hewitt, A.J. O’Donnell C.C. and Kruger. G.R. 2020. Comparison of water-sensitive paper, Kromekote and Mylar collectors for droplet deposition with a visible fluorescent dye solution. J. Plant Prot. Res. 60 (1): 98– 105.
Ferrel J.A. and Vencill. W.K. 2003. Impact of adjuvants and nozzle types on cotton injury from flumioxazin applied post-directed. J. of Cotton Sci. 7: 242– 247.
Franca L.X., Dodds, D.M. Butts, T.R. Kruger, G.R. Reynolds, D.B. Mills, J.A. Bond, J.A. Catchot A.L. and Peterson. D.G. 2020. Evaluation of optimal droplet size for control of Palmer amaranth (Amaranthus palmeri) with acifluorfen. Weed Technol. 34: 511– 519.
Freemark K. and Boutin. C. 1995. Impacts of agricultural herbicide use on terrestrial wildlife in temperate landscapes: A review with special reference to North America. Agric. Ecosyst. Environ. 52: 67– 91.
Hartley G. S. and Graham-Bryce. I.J. 1980. Physical Principles of Pesticide Behaviour, Academic Press, Cambridge, MA.
ISO. 2018. ISO: 10625 - Equipment for Crop Protection. Sprayer Nozzles, Colour Coding for Identification. International Standard Organization, 12 pp.
Johnstone D.R. 1960. Assessment Techniques 2. Photographic Paper. CPRU Porton Report No. 177. Mimeographed, 13.
Knoche M. 1994. Effect of droplet size and carrier volume on performance of foliage-applied herbicides. Crop Prot. 13: 163– 178.
Legleiter T.R. and Johnson. W.G. 2016. Herbicide coverage in narrow row soybean as influenced by spray nozzle design and carrier volume. Crop Prot. 83: 1– 8.
Lund I. 2000. Nozzles for drift reduction. Aspects Appl. Biol. 57: 97– 102.
McKinlay K.S., Brandt, S.A. Morse P. and Ashford. R. 1972. Droplet size and phytotoxicity of herbicides. Weed Sci. 20: 450– 452
Meyer C.J., Norsworthy, J.K. Kruger G.R. and Barber. T.L. 2016. Effect of nozzle selection and spray tank volume on droplet size and efficacy of Engenia tank-mix combinations. Weed Technol. 30: 377– 390.
Piggott S. and Matthews G.A. 1999. Air induction nozzles: a solution to spray drift? Int. Pest Control. 41: 24– 28.
Ramsdale B. K. and Messersmith C. G., . 2001. Drift-Reducing Nozzle Effects on Herbicide Performance. Weed Technol. 15 (3): 453– 460
Rodrigues A.O., Campos, L. G. Creech, C. F. Fritz, B. K. Antuniassi U. R. and Kruger. G. R. 2020. Influence of Nozzle Type, Speed, and Pressure on Droplet Size and Weed Control from Glyphosate, Dicamba, and Glyphosate Plus Dicamba. In Pesticide Formulation and Delivery Systems: 38th Volume, Innovative Application, Formulation, and Adjuvant Technologies, ed. Fritz B. and Butts T. (West Conshohocken, PA: ASTM International, 2018), 61-75.
Rogers R.B. and Maki. R. 1986. The Effect of Drop Size on Spray Deposit Efficiency. St. Joseph, MO: American Society of Agricultural Engineering Paper No.86–1508.
Shaw D.R., Morris, W.H. Webster E.P. and Smith. D.B. 2000. Effects of spray volume and droplet size on herbicide deposition and common cocklebur (Xanthium strumarium) control. Weed Technol. 14: 321– 326.
Sikkema P.H., Brown, L. Shropshire, C. Spieser H. and Soltani. N. 2008. Flat fan and air induction nozzles affect soybean herbicide efficacy. Weed Biol. Manag. 8: 31– 38.
Spillman J.J. 1984. Spray impaction, retention and adhesion: an introduction to basic characteristics. Pest Manag. Sci. 15: 97– 106
Taylor W.A., Womac, A.R. Miller P.C.H. and Taylor. B.P. 2004. An attempt to relate droplet size to drift risk. Pages 210– 223 in Proceedings of the International Conference on Pesticide Application for Drift Management. Pullman, WA: Washington State University
[USDA-AMS] United States Department of Agriculture-Agricultural Marketing Service-Cotton and Tobacco Program. 2020. Cotton varieties planted-2020 crop. mp_cn833. https://www.ams.usda.gov/mnreports/cnavar.pdf
[USDA-APHIS] United States Department of Agriculture–Animal and Plant Health Inspection Service. 2015. Monsanto Co.; Determination of Nonregulated Status of Herbicide Resistant Soybean and Cotton. https://www.federalregister.gov/articles/2015/01/20/2015-00723/monsanto-co-determination-ofnonregulated-status-of-herbicide-resistant-soybean-and-cotton.
[USDA-NASS] U.S. Department of Agriculture-National Agricultural Statistics Survey. 2020. State Agriculture Review–Georgia 2020. https://www.nass.usda.gov/Quick_Stats/Ag_Overview/stateOverview.php?state=GEORGIA
[USDA-NASS] U.S. Department of Agriculture – National Agriculture Statistics Survey. 2018. 2018 Agricultural Chemical Use Survey – Peanuts. https://www.nass.usda.gov/Surveys/Guide_to_NASS_Surveys/Chemical_Use/2018_Peanuts_Soybeans_Corn/ChemUseHighlights_Peanuts_2018.pdf
Undersander D.J., Marek T.H. and Clark. R.N. 1985. Effect of nozzle type on runoff and yield of corn and sorghum under center pivot sprinkler systems. Irrigation Sci.. 6 (2): 107– 116.
Vencill W.K., Nichols, R.L. Webster, T.M. Soteres, J.K. MallorySmith, C. Burgos, N.R. Johnson W.G. and McClelland. M.R. 2012. Herbicide resistance: toward an understanding of resistance development and the impact of herbicide-resistant crops. Weed Sci. 60: 2– 30.
Wolf T. M., Liu, S.H. Caldwell B.C. and Hsiao. A.I. 1997. Calibration of greenhouse spray chambers –The importance of dynamic nozzle patternation. Weed Technol. 11 (3): 428– 435.
Notes
- First and second authors: Assistant Professor, and Professor, Dept. of Crop & Soil Sciences, University of Georgia, Tifton, GA 31793 [^]
- Third author: Professor, Dept. of Plant Pathology, University of Georgia, Tifton, GA 31793 [^]
- Fourth and fifth authors: Associate Professor and Professor, Dept. of Entomology, University of Georgia, Tifton, GA 31793 [^]
- Sixth author: Miller County Extension Agent, University of Georgia Cooperative Extension, Colquitt, GA 39837 [^]
- Seventh author: Worth County Extension Agent, University of Georgia Cooperative Extension, Sylvester, GA 31791 [^]
- Eighth author: Pierce County Extension Agent, University of Georgia Cooperative Extension, Blackshear, GA 31516 [^]
- Ninth author: Bulloch County Extension Agent, University of Georgia Cooperative Extension, Statesboro, GA 30458 [^] *Corresponding author Email: svirk@uga.edu
Author Affiliations