Introduction
Peanut (Arachis hypogaea L.) is an important leguminous crop in smallholder integrated crop-livestock production systems because of its nutritious grain, highly palatable fodder and positive contribution to soil fertility through natural nitrogen (N) fixation (Bell et al., 1994; Konlan et al., 2013). Manure obtained from animals fed peanut fodder is also comparable in quality to synthetic fertilizers and is very useful for improving soil fertility because of a high level of N excreted in the feces and urine (Ansa et al., 2019; Decandia et al., 2011). In west Africa, peanut is grown primarily for its grain as food and the fodder as a supplementary feed to natural pasture. Compared to other leguminous forages such as cowpea (Vigna unguiculata L.) and pigeon pea (Cajanus cajan L.), demand and market value for peanut fodder is higher because of its superior nutritional value and is often sold to supplement household income in the lean dry season (Konlan et al., 2018; Samireddypalle et al., 2017). Peanut fodder consists of leaves, stems, and roots, and is obtained by sun-curing the fresh forage on the farm field after harvesting and plucking off the pods. The dry matter (DM) yield of 4547 kg per ha and crude protein (CP) content of 19% DM for annual peanut fodder (Larbi et al., 1999) is superior to other tropical leguminous fodder such as cowpea (16% DM) and pigeon pea (13% DM) (Foster et al., 2011), and comparable to full-bloom alfalfa hay in chemical composition (Yang, 2005). Peanut fodder is therefore an important feedstuff in sub-Saharan Africa (Larbi et al., 1999), parts of the USA (Johnson, 1979; Foster et al., 2011) and semi-arid regions of Asia (Suttie, 2000).
Inter-row plant spacing affects grain and fodder yields of peanut (Giayetto et al., 1998; Jaaffar and Gardner, 1988; Onat et al., 2007). The recommended inter-row plant spacing for both erect and runner cultivars of peanut in Ghana ranges from 30 (Konlan et al., 2013) to 70 cm (Dapaah et al., 2014). Inter-row spacing of 46 cm has been reported to improve canopy closure, light interception, and DM yield than wider spacing of 91 cm (Jaaffar and Gardner, 1988). Closer plant spacing increases plant densities and thus have additional agronomic benefits of weed smothering. However, increased plant density increases intraspecific competition for resources and early canopy closure (Jaaffar and Gardner, 1988). Canopy closure causes shading which may reduce growth rate resulting in thinner stems and leaves thereby reducing biomass yield. Shading however reduces cellulose and lignin concentration, resulting in increased ruminal organic matter digestibility of forages whereas wider plant spacing increases radiation penetration into the crop thereby increasing the temperature of the crop and the concentration of indigestible cell wall fractions (Buxton and Fales, 1994). Plant spacing also affects the amount of N fixed in the vegetative part of the crop which affects the nutritional quality of the fodder. Konlan et al. (2013) noted that planting peanut at an inter-row spacing of 30 and 40 cm increased the amount of N fixed in the haulm than planting at 50 cm. In contrast, Giayetto et al. (1998) reported that an equal proportion of DM is partitioned to pods and vegetative parts irrespective of planting spacing.
Peanut fodder has been used as sole (Blümmel et al., 2005; Prasad et al., 2010;) or supplementary (Ayantunde et al., 2017; Etela and Dung, 2011; Khan et al., 2013; Manyuchi et al., 1997) diet to increase the productivity of sheep (Ovis aries L.) raised on natural pasture or low-quality forages. In Ghana, the productivity of sheep is usually low because of the poor quality of natural pasture in the rainy season and their destruction by wildfires in the dry season. The quality, especially CP content, of tropical natural pasture is therefore often inadequate to support the growth of sheep without supplementary forage legume high in CP (Ayantunde et al. 2007). Peanut planting systems that concomitantly increase fodder yield and quality without adversely affecting the nutritional and market value of the grains can help address the challenges of meeting the nutrient requirements of sheep especially in the dry season. Even though Larbi et al. (1999) has established a significant and positive relationship between grain and fodder yields in peanut, the effect of plant spacing on the nutritional quality of peanut fodder in terms of animal output such as daily weight gain, a function of the quantity (yield) and quality (CP, intake and digestibility of the fodder, and the quality of manure, have not been adequately studied in Ghana.
Three experiments were conducted to test the hypotheses that 1) compact inter-row plant spacing increases grain and forage yields (agronomic experiment) of peanut, 2) the digestibility (in situ experiment) and growth rate (growth performance experiment) of sheep fed peanut fodder obtained from compact inter-row plant spacing is greater than those planted at wider spacing.
Materials and Methods
Agroclimatic characteristics of the study area
The study was conducted on-farm in four peanut-growing communities (Tingoli, Cheyohi No. 2, Duko and Tibali) in the northern region of Ghana (Table 1) during the 2018 crop growing season. Tingoli and Cheyohi No. 2 communities are located in the Tolon District whereas Duko and Tibali are located in Savelugu District. The main source of livelihood of the people is smallholder crop-livestock farming. The northern region has a long and severe dry season (April−November) with a unimodal rainfall pattern which commences in late April and ends in October. The average daily temperature was approximately 28.1 °C, annual average rainfall is 1100 mm whereas relative humidity is 70 to 90% in the raining season and 20% during the dry season (Attandoh et al., 2013).
Agronomic Experiment
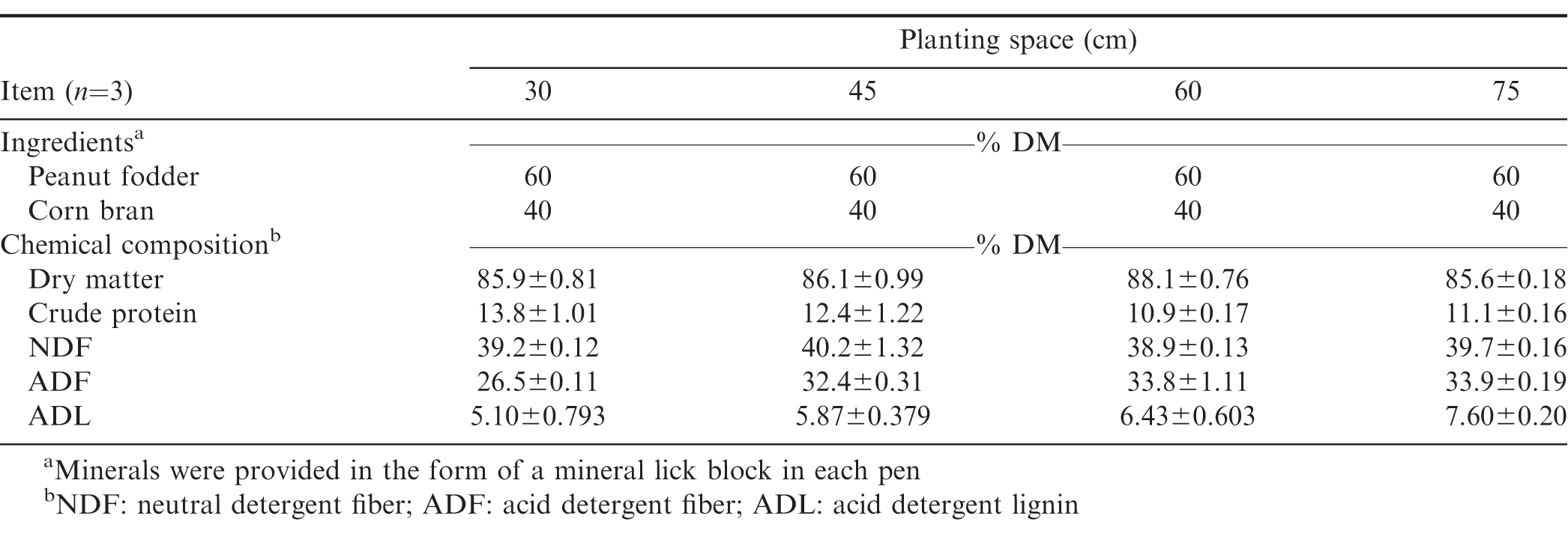
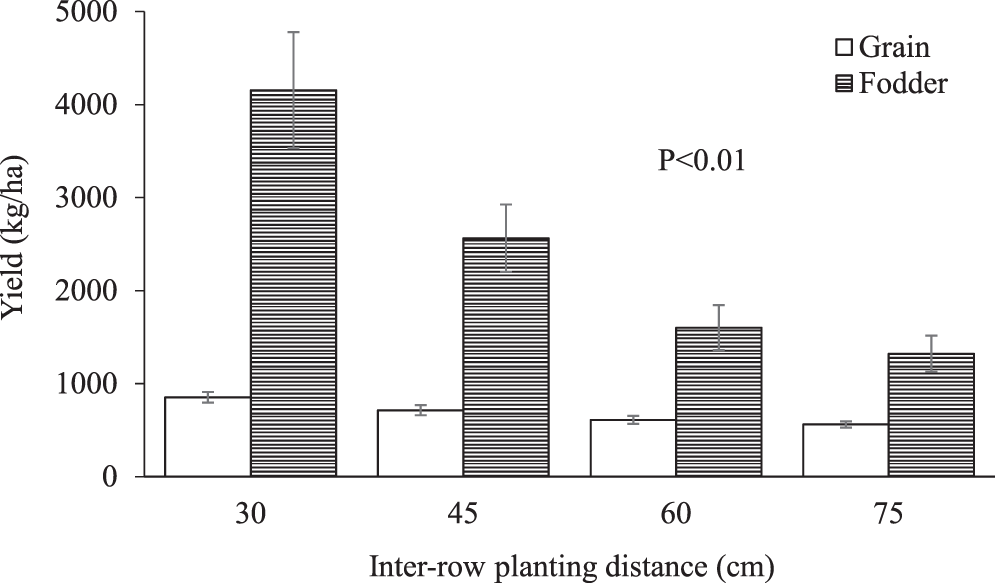
The experiment was arranged as a randomised complete block design with each treatment replicated twice in each community (Table 1). Peanut was planted on each farmer’s field at 30, 45, 60, and 75 cm apart between rows (inter-row) and 15 cm apart within rows (intra-rows), resulting in four experimental treatments per farmer in each community. The soils in the study areas had a pH of 5.6−6.3 with low organic carbon (5.5−9.7 g/kg), total nitrogen (0.5−0.9 g/kg), available phosphorus (9.5−16.6 mg/kg) and soil texture of loam to sandy loam (Tetteh et al., 2016). Data on grain and fodder yields were measured by harvesting peanut plants in the two middle rows of each treatment plot. After harvest, the peanut pods were removed manually, oven-dried (65°C) for 48 h to 87% DM and the pods manually cracked to measure grain yield. The remaining biomass obtained from the two middle rows of each treatment plot were similarly oven-dried (65°C) for 48 h to 87% DM for subsequent determination of fodder yield. The rest of the forage on each farmer’s plot was harvested and used in a growth performance experiment.
Growth Performance Experiment
The growth performance experiment was arranged as a randomised complete block design with four treatments (30, 45, 60, and 75 cm) and three replicate farmers. Each community received all four treatments. Peanut fodder was collected from each farmer’s field in the agronomic experiment (experiment I) where they were planted at 30, 45, 60 and 75 cm apart between rows (inter-rows) and 15 cm apart within rows (intra-rows).
Triplicate farmers were sampled from the agronomic experiment in each community (Duko, Tibali and Tingoli). Five Djallonké sheep of similar body weight (19.0±2.0 kg) were selected from each farmer’s flock for the growth performance experiment and were fed for 70 d. This resulted in a total of 12 farmers and 60 sheep (36 females and 24 males). Animals were weighed consecutively for 2 d at the beginning and end of the experiment. The averages of these measurements were used as the initial and final weights, respectively. Female sheep involved in the experiment were continually observed for signs of pregnancy or oestrus so that any of them found to have conceived prior to the experiment could be culled from the experiment.
The peanut fodder obtained from farmers’ fields was chopped to a theoretical length of 3-5 cm after manually pulling pods and sun-drying on large tarpaulin sheets spread on concrete floors. The fodder was turned-over to ensure uniform drying. Fodder was placed in sacks and stored. Corn (Zea mays L.) bran was purchased from local corn millers, dried and stored in sacks. The fodder and corn bran were mixed by weight into a ration consisting of 60% DM peanut fodder and 40% DM corn bran. Sheep were managed semi-intensively and kept in their pens (4 × 8 m) at about 6:00 and offered their rations until 14:00 the next day but allowed to graze natural pasture in the evening from 14:00 to 18:00. They were offered measured quantities of their diets (Table 2) daily between 6:30 and 9:30 each morning. Quantity of feed offered daily was adjusted based on individual daily intake of each sheep. Each pen was made of concrete floor with rice (Oryza sativa L.) straw beddings and was cleaned every morning. The predominant species in the natural pasture was northern gamba grass (Andropogon gayanus Kunth) and Stylosanthes sp. Prior to the commencement of the experiment, the sheep were managed according to the procedure outlined in the manual of sheep production in the humid tropics of Africa (Charry et al., 1992). They were adapted to their pens and feeding regimes for 10 d. Each pen was provided with a mineral lick block. Leftovers were weighed each morning prior to the supply of the feed in the morning. The ingredients (fodder and corn bran), feed offered and leftovers were sampled weekly for determination of dry matter. This data was used to calculate feed intake. A portion (∼500 g) of each of the fodder sampled weekly was combined into bi-weekly samples and stored for subsequent determination of DM degradability in an in situ digestibility experiment. The sheep were also weighed weekly for the estimation of weight gain and average daily weight gain (ADG) over a period of 70 days.
Manure samples (∼500 g) were collected from each pen every two ws, cleaned of bedding material and other debris and stored (-40°C). The samples collected were subsequently pooled into a composite sample per pen. The composited samples were stored in a freezer (-40°C) until analysed for determination of N, phosphorus, potassium, polyphenol and organic carbon concentration, and pH.
In Situ Digestibility Experiment
Fodder sampled (∼500 g) during the feeding trial in the growth performance experiment was used for the in situ digestibility experiment. The samples were pooled bi-weekly, frozen (−40°C) and subsequently dried and ground through a 2-mm screen. Ground samples of each spacing were mixed thoroughly and weighed (2 g per bag) into monofilament nylon bags (5 × 10 cm; 50 μm pore size; Ankom Technology Corp., Fairport, NY). Bags were tightly sealed by heat and placed in the rumens of two ruminally cannulated Nungua Black Head ewes (∼28 kg) housed in individual pens at the Livestock and Poultry Research Centre of the University of Ghana. Cannulated sheep were used to facilitate DM disappearance (DMD) of peanut fodder at 24 and 48 h in a Randomised Complete Block Design with each cannulated sheep as a block. Samples were placed in an oven at 80°C for 24 h and allowed to cool at room temperature in a desiccator. Triplicate nylon bags containing fodder samples for incubation at 24 and 48 h were placed into the rumens of the cannulated sheep by the reversed sequential order, according to incubation time, and were all removed at same time (Silva et al., 1997). Each nylon bag was tied to a 25-cm nylon string and anchored to the cannula cap before being placed deep into the rumen of the cannulated sheep. Triplicate bags per treatment for each incubation period of 0, 24 and 48 h (Silva et al., 1997) were inserted into the rumens of each sheep at 8:00 prior to feeding in the morning. On the first day of the 48-h incubation period, triplicate samples of each plant spacing designated for 48 h incubation period were marked and placed deep into the rumen of each sheep. On the next morning (day 2), samples designated for 24 h were similarly placed the rumen. All the bags (including the zero-hour samples) were retrieved after 48 h and immediately washed with cold water for about 30 minutes under running tap water while rubbing gently between the thumb and fingers until the water ran clear. The washed bags containing the residues after incubation were dried in the oven at 80°C for about 48 h and allowed to cool in a desiccator. The residues of replicate bags from the two cannulated sheep were then pooled according to spacing and incubation period and ground through a 1-mm screen. Ground samples of the pooled residues and unincubated haulms (0 h) were analyzed for DM concentration and used to estimate in situ DMD calculated as the difference in weight of the samples before and after incubation.
The experimental protocol for the in situ digestibility and growth performance experiments were approved by the Research Ethics Committee of the University for Development Studies and the sheep were cared for and managed according to the guidelines of the Research Ethics policy handbook of the University for Development Studies.
Laboratory Analyses
Feed samples collected weekly were pooled into replicate treatments samples for determination of DM and CP concentrations according to the official methods of analysis described by Association of Official Analytical Chemist (AOAC, 2005). All analyses were done in duplicates. For determination of DM, a sub-sample (20 g) of the dietary ingredients, manure or rations were determined in a forced air oven at 60°C for 48 h. Total N was determined by the Khejedal method and crude protein was estimated as total N × 6·25.
The rations were ground through a 1-mm screen for analysis of neutral detergent fiber (NDF) and acid detergent fiber (ADF) using an Ankom 200 system (Ankom Technology Corp., Fairport, NY). Neutral detergent fibre was analyzed with the addition of sodium sulphite and a-amylase, whereas ADF was analyzed without a-amylase. After determination of ADF, the residue was used for the determination of acid detergent lignin (ADL) by digesting the residue with 2% H2SO4 according to a procedure previously described by AOAC (1991).
Statistical analyses
Data from the three experiments were analysed by the MIXED procedure of SAS (version 9.4; Statistical Analysis System Inc., Cary, USA). Data on the growth performance experiment were analysed with each farmer’s pen as the experimental unit and each farming community (Tingoli, Cheyohi No. 2, Duko or Tibali) as a block or random variable in the model. The data on growth performance were adjusted for the effect of initial weight using initial weight as covariate in the model equation (Kaps and Lamberson, 2009) as yijk= μ + Di + β(χij) + Ck + ϵij with equation parameters as follows:
yijk is the dependent variable (weight gain, ADG) measured in the ith treatment (i.e. plant spacing; 30, 45, 60 and 75 cm) on jth sheep used in the growth performance experiment; μ is overall mean effect, β is the linear regression coefficient indicating the dependency of yijk on χij; Di is the effect of the ith treatment (ie planting space; 30, 45, 60, and 75 cm); χij is the independent continuous variable (initial weight) used as a covariate to control error and adjust the least square means of the dependent variable in the ith treatment; Ck is the random effect of each farming community (Tingoli, Cheyohi No. 2, Duko or Tibali); ϵij is the random error;
Data on the agronomic and DMD experiments were analysed as a Randomised Complete Block Design with each cannulated sheep as a block, and each replicate field and nylon bag as the experimental units, respectively in the model equation: yij=μ + Di + bj + ϵij with equation parameters as follows: yij is the dependent variable (manure quality, grain yield, DMD) measured in the ith treatment (ie planting space; 30, 45, 60, and 75 cm); Di is the effect of plant spacing in the ith treatment as in equation (1); bj is the random effect of each community in the agronomic experiment or cannulated sheep in the in situ experiment whereas ϵij is the random effect of error. Least square means with significant differences were separated at a probability level of 0.05.
Results and Discussion
Agronomic Experiment
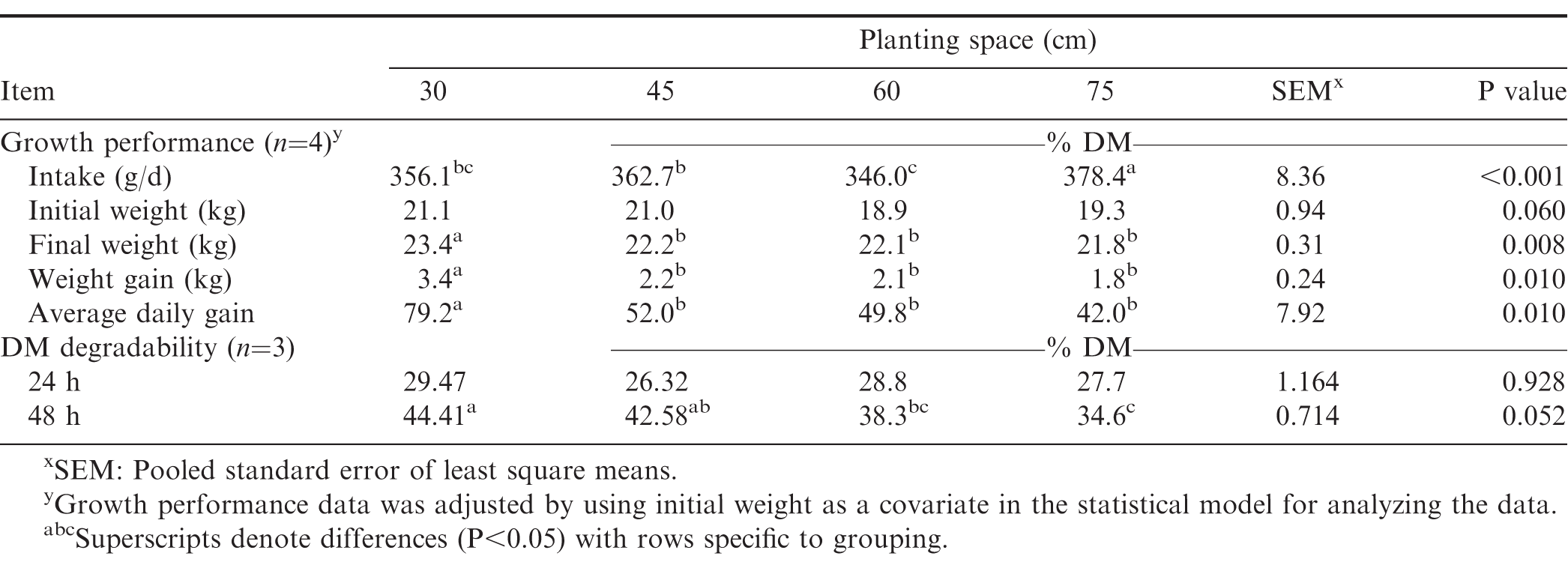
Planting peanut at closer inter-row spacing produced higher grain yield and more fodder than the other inter-row spacing as grain and fodder yields of peanut planted at 30 cm were higher (P=0.001) than those planted at 45, 60, and 75 cm (Fig. 1). These could be attributed to increased peanut plant density which resulted in more grains and fodder. This observation supports earlier reports that planting peanut at closer spacing increases pod and fodder yields (Giayetto et al., 1998; Tubbs et al., 2011), due to higher crop population. In similar studies, Jaaffar and Gardner (1988) found that planting peanut at closer spacing increased grain and DM yields without adverse effects on market quality of the grains. However, in other studies (Onat et al., 2017), decreasing plant spacing rather decreased grain and fodder yields because of reduced photosynthesis attributed to shading and competition for nutrients. This reported reduction in yield due to intraspecific competition for light and nutrients in compact plant spacing may however occur only at the plant level because at the population level, compact plant spacing produces more DM than wider plant spacing. This is because the reduction in yield due to intraspecific competition can be adequately compensated by the increased density of plants and greater interception of solar radiation in compact spacing, resulting in greater fodder and grain yields (Giayetto et al., 1998; Konlan et al., 2013).
Differences in grain and fodder yields between 60 and 75 were not significant (P=0.216). Fodder yield increased with decreasing plant spacing. A comprehensive elucidation of the dichotomy in these observations requires an understanding of the complex interaction between related factors such as light interception and radiation use efficiency, and how these affect fodder and grain yields of peanut (Kinirya et al., 2005). The results of this study suggest that farmers could dually increase grain yield and still benefit from increased fodder yield if peanut is planted at an inter-row spacing of 30 cm. This observation is consistent with findings by Larbi et al. (1999) which indicated a linear relationship between grain and fodder yields for different cultivars of peanut. In the present report, the fodder yield increased from 1,322, 1,602, and 2,563 kg DM/ha for inter-row spacings of 75, 60 and 45 cm to 4,154 kg DM/ha for an inter-row spacing of 30 cm. Larbi et al. (1999) assessed the fodder yield of 38 cultivars of peanut and found a maximum of forage yield of 6,075 kg DM/ha and a minimum of 2,106 kg DM/ha.
Growth performance experiment
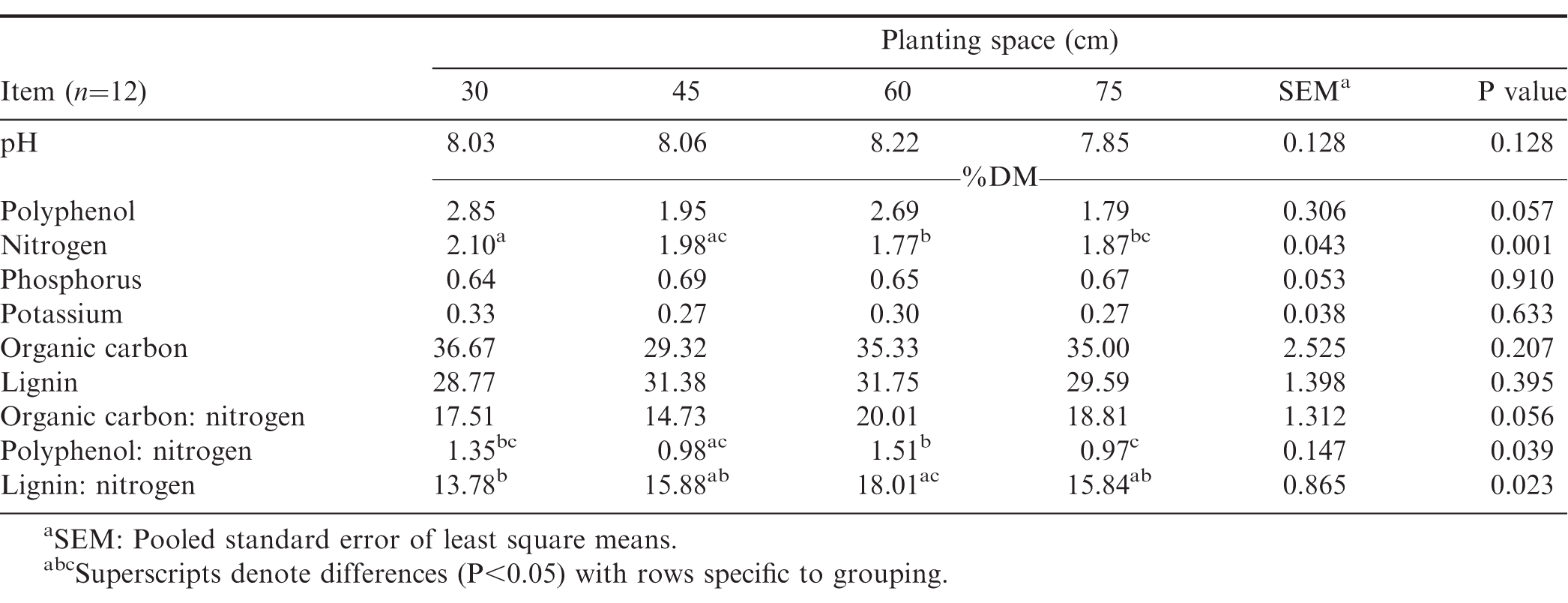
Dry matter intake, in situ digestibility and growth performance of sheep supplemented with the peanut fodder are shown in Table 3. Feed intake of sheep fed the 30 cm fodder was lower (P=0.001) than those fed 75 cm fodder but did not differ from those fed 45 (P=0.2614) and 60 cm (P=0.0917) fodder. The superior digestibility of the 30 cm fodder may account for the lower intake of the diet and the corresponding higher ADG of the sheep. In ruminants, intake of forage diets is generally limited by gut-fill due to bulkiness and other factors, however, when digestibility of the forage is higher, intake may be controlled less by forage factors and reticulo-rumen capacity but more by nutrient requirements (Horrocks and Vallentine, 1999). Crude protein content of fodder obtained from 30 cm plant spacing had comparatively higher CP and lower ADL than the other fodder (Table 2). The lower intake of the 30 cm diet, and its higher CP content and DMD, compared to the other fodder suggest that the sheep in that group were meeting their requirements for growth as shown by the greater ADG whereas the higher concentration of ADIN and ADL in the other fodder (45, 60, and 75 cm; Table 1) are suggestive of poor quality and the sheep had to consume more to meet their requirements. The data on DM intake and N content of the diets in this study are however inconsistent with those reported by Khan et al. (2013) who found DM intake to increase with increasing dietary N as wheat straw was replaced with peanut fodder. In contrast to feed intake, ADG of sheep fed the 30 cm fodder was higher (P=0.010) than those supplemented with the other types of peanut fodder. The ADG values observed in this study for the 30 and 45 cm spacing are higher than 1.4, 19.3, and 40.2 g/d reported by Ayantunde et al. (2007) when bush hay was supplemented with 150, 300 and 450 g/d of peanut hay, respectively, but are comparable to the ADG of sheep fed the 60, and 75 cm diets. When different varieties of peanut fodder was evaluated on growing Deccani sheep as a sole diet, the ADG ranged from 65 to 137 g/d (Prassad et al., 2010). Differences in the growth performance of sheep in the literature in response to supplementation of native pasture with peanut fodder should be interpreted cautiously because variations in the botanical and nutritional composition of native pasture could confound such comparisons because of the associative effects of the native pasture on ruminal digestibility of the supplementary fodder offered.
Manure quality
Chemical composition of manure obtained from sheep fed peanut fodder diets planted at different inter-row spacings is shown in Table 4. Nitrogen concentration of manure was greater (P=0.001) for sheep fed the 30 cm peanut fodder than for those fed the other fodder. Differences in the N content of the manure may be related to differences in the nutritional quality of the fodder obtained from the different plant spacing. Palm et al. (2001) classified the quality of manure based on the N content as either low (N < 2.5%) or medium (N > 2.5%). Although the range of N content of manure produced from the peanut fodder fed to the sheep in this study (Table 3) could be categorized as low quality (Palm et al., 2001), the N content of the manure produced from the 30 cm plant spacing was higher than those of the other plant spacing. The dietary CP concentration of the 30 cm diet was 17% greater than the CP content of the 45 cm diet and 27% higher than both the 60 cm and 75 cm diets (Table 1). The minimum dietary CP required for effective rumen function is estimated at 6−8% (Minson, 1990) whereas the National Research Council (NRC, 1985) recommends a CP of 14.5% for sheep fed for maximum growth. The CP of the 30 cm diet was 14% (Table 1), notwithstanding the additional CP intake from natural pasture. There is a general agreement that when the rate of ruminal dietary protein degradation exceeds the rate of carbohydrate fermentation, large amounts of N compounds are excreted via urine and faeces (Decandia et al., 2011; Woyengo et al., 2004). Though the lower DM intake and increased N in the manure may be irreconcilable, the results are consistent with some literature. A review of 154 mean values concerning diet composition, nutrient intake and digestibility, and faecal and urinary N excretion at different physiological stages in sheep, suggest that the faecal excretion of N is particularly high when the diet is high in soluble N (Decandia et al., 2011). The latter found that, in sheep, faecal N excretion increases exponentially as dietary CP is increased. Foster et al. (2009) evaluated four warm-season legumes (annual peanut, perennial peanut, cowpea, pigeon pea) as supplementary protein for sheep and found that N intake and faecal N were highest in annual peanut compared to the other legumes. These observations appear to explain the results of the present study because peanut fodder has a lower concentration of ruminally fermentable carbohydrate (Foster et al., 2011) but a higher concentration of soluble N (Khan et al., 2013; Larbi et al., 1999; Yang, 2005). This lack of adequate synchronization of energy and protein could account for the excretion higher amounts of N in the manure of sheep fed the 30 cm fodder.
Supplementation of maize stover plus cassava/yam peels with either cowpea or peanut fodder increased the manure N of Djallonké sheep by 67% and 63%, respectively, compared to the N content of the manure of the control group (Ansa et al., 2019). Supplementation of bush hay with increasing levels of peanut fodder also increased N intake and N excreted in the faces of sheep without adverse effects on ADG (Ayantunde et al., 2007). Though high N in the manure generally has negative effects on the environment, under small-holder crop-livestock integrated farming systems where inorganic fertilizers are economically inaccessible, manure with higher N content is very useful for improving soil fertility where soils have been exhaustively used and depleted of N.
Other chemical constituents and pH did not differ among planting spaces. Lignin: N of the 30 cm manure was lower (P=0.003) than manure from 60 cm but did not differ (P≥0.091) from the other types of manure. Polyphenol: N of 60 cm manure was also higher and differed from 45 cm (P=0.021) and 75 cm (P=0.017) manure but was similar (P=0.091) to 30 cm manure. Bioavailability of N to plants is controlled by many chemical indices such as lignin, polyphenols and N concentration. However, the mineralization and utilization of N by plants is greatly influenced by polyphenol:N ratio than by these other indices (Oglesby and Fownes, 1992). Livestock manure is particularly important in Africa because N input from livestock manure is by far the largest source of N input to African soils representing about 87% of total N input compared to synthetic fertilizers (FAO, 2018).
In situ digestibility experiment
The DMD obtained after 24 and 48 h of in situ incubation in this study are within ranges previously reported by Yang (2005) after 48 of incubation in situ (Table 3). In situ digestibility of peanut at 24 h was not affected (P=0.928) by plant spacing, however, planting peanut at 30 and 45 cm produced fodder that was more (P=0.052) digestible at 48 h than those obtained from peanut planted at 75 cm. The average DMD of thirty-eight cultivars of peanut after 48 h of incubation was 54.2% and 42.9% for leaves and stems, respectively (Larbi et al., 1999). Acid detergent lignin concentration was generally lower in the 30 cm diet but increased with increasing plant spacing (Table 1). Dietary ADL reduces fiber digestibility and therefore growth performance of animals. Previous studies have shown that ADL concentration of peanut fodder alone accounted for 58 and 72% of the variation in organic matter digestibility and ADG of sheep, respectively, with 80% of the variation in N retention being attributable to the combined effect of N and ADL concentration of the fodder (Prasad et al., 2010). Carvalho et al. (2017) reported that the poorer digestibility of tropical forages in sheep may be related to their higher concentration of ADIN and lignin. The manner in which ADL limits ruminal digestion has long been known in literature; ADL provides limited sites for rumen microbial colonization (Akin, 1989; Van Soest, 1982). In legumes, shading of lower parts of a plant by upper parts of the same plant or neighbouring plants in compact planting arrangements reduces the interception of solar radiation, and the biosynthesis and accumulation of lignin (Wei-gua et al., 2014). This alters the nutritional composition, morphological development, biomass yield and digestibility of forages. Lower shading in sparsely planted forages is however expected to increase the concentration of indigestible cell wall fraction such as lignin, resulting in poorer DMD as observed in 45 and 65 cm fodders. Stem elongation in legumes increases with increasing plant density as a result of shading and elongated stems have lower concentration of recalcitrant cell walls (Buxton and Fales, 1994). Differences within compact (30 and 45 cm) and wider (60 and 7 cm) spacing categories were not however significant as the differences between 45 and 60 cm, and between 60, and 75 cm was not significant (P≥0.06; Table 3).
Summary and Conclusions
Planting peanut at an inter-row spacing of 30 cm resulted in improved grain and fodder yields. The nutritional quality of fodder obtained from 30 cm inter-row plant spacing was also superior, resulting in increased in situ DMD and growth performance of sheep. This study recommends an inter-row plant spacing of 30 cm for the dual benefits of increasing grain and fodder yields as well as improving the nutritional quality of fodder as supplementary fed to poor natural pasture. The high concentration of N in the manure of sheep fed peanut fodder obtained from 30 cm inter-row plant spacing may have additional benefits of improving soil fertility in smallholder crop-livestock farming systems. Although planting peanut at an inter-row spacing of 30 cm was more beneficial than the other spacings, the cost-benefit analysis and pest management implications of the 30 cm inter-row spacing were not evaluated in this study. Future studies on the implications of these in 30 cm inter-row spacing for peanut production in northern Ghana is recommended.
Acknowledgements
The authors acknowledge this work as part of crop-livestock trials conducted in Ghana by Support of Africa Research In Sustainable Intensification for the Next Generation (Africa RISING) project. The Project was coordinated by International Institute of Tropical Agriculture (IITA) with funds from the United State Agency for International Development (USAID).
Literature Cited
Akin, D.E. 1989. Histological and physical factors affecting digestibility of forages. Agron. J. 81: 17– 25.
Ansa, K.O., Antwi, C. Osafo, E.L.K. Enning, S. and Adu-Dapaah. H. 2019. Manure characteristics of small ruminants fed agro by-products in the guinea savannah agro-ecological zone of Ghana. Ghana J. Agric. Sci. 54: 67– 76.
AOAC. 2005. Fiber (acid detergent) and lignin in animal feed. (973.18). Official Methods of Analysis. Association of Official Analytical Chemists (AOAC). 15th edition.
Attandoh, N., Yidana, S.M. Abdul-Samed, A. Sakyi, P.A. Banoeng-Yakubo, B. and Nude. P. 2012. Conceptualization of the hydrogeological system of some sedimentary aquifers in Savelugu–Nanton and surrounding areas, Northern Ghana. Hydrol. Process 27: 1664– 1676.
Ayantunde, A.A., Delfosse, P. Fernandez-Rivera, S. Gerard, B. and Dan-Gomma. A. 2007. Supplementation with groundnut haulms for sheep fattening in the West African Sahel. Trop. Anim. Health Prod. 39: 207– 216.
Bell, M.J., Wright, G.C. Suryantini and Peoples. M.B. 1994. The N2 fixing capacity of peanut cultivars with differing assimilate partitioning characteristics. Aust. J. Agric. Res. 45: 1455– 1468.
Blümmel, M., Vellaikumar, S. Devulapalli, R. Nigam, S.N. Upadhyaya, H.D. and Khan. A. 2005. Preliminary observations on livestock productivity in sheep fed exclusively on haulms from eleven cultivars of groundnut. Int. Crops Res. Institute for the Semi-Arid Tropics online J. 1: 1– 3.
Buxton, D.R., and Fales S.L. 1994. Plant environment and quality. In: Fahey G.C. Collins, M. Mertens, D.R. and Moser L.E. (eds.). National conference on forage quality evaluation, and utilization. American Society of Agronomy, Crop Science Society of America, Soil Science Society of America, Madison, WI. Pages 155– 199.
Carvalho, G.G.P., Reboucas, R.A., Campos, F.S, Santos, E.M, Araujo, G.G.L, Gois, G.C., Oliveira, J.S., Oliveira, R.L. Rufino, L.M. Azevedo, J.A.Gand Cirne. J.G.A. 2017. Intake, digestibility, performance, and feeding behavior of lambs fed diets containing silages of different tropical forage species. Anim. Feed Sci. Technol. 228: 140– 148.
Charray, J., Humbert, J.M., and Levif, J. 1992. Manual of sheep production in the humid tropics of Africa. CAB International. 187 pages
Dapaah, H.K., Mohammed, I. and Awuah. R.T. 2014. Growth yield performance of groundnuts (Arachis hypogaea L. ) in response to plant density. International Journal of Plant and Soil Science 3: 1069– 1082.
Decandia, M., Atzori, A.S. Acciaro, M. Cabiddu, A. Giovanetti, V. Molina Alcaide, E. Carro, M.D. Ranilla, M.J. Molle, G. and Cannas. A. 2011. Nutrition al and animal factors affecting N excretion in sheep and goats. In: Ranilla, M.J. Carro, M.D. Ben Salem, H. and Morand-Fehr P. (ed.). Challenging strategies to promote the sheep and goat sector in the current global context. Zaragoza. CIHEAM / CSIC / Universidad de León / FAO, 2011. p. 201– 209 (Options Méditerranéennes: Série A. Séminaires Méditerranéens; n. 99).
Etela, I., and Dung. D.D. 2011. Utilization of stover from siximproved dual-purpose groundnut (Arachis hypogaea L.) cultivars by west African dwarf sheep. African J. Food Agric. Nutr. Dev. 11: 4538– 4545.
FAO. 2018. Nitrogen inputs to agricultural soils from livestock manure New statistics. Integrated crop management. Food and Agriculture Organization of the United Nations. Rome. Vol. 24 – 2018. 67 pp.
Foster, J.L., Adesogan, A.T. Carter, J.N. Blount, A.R. Myer, R.O. and Phatak. S.C. 2009. Intake, digestibility, and nitrogen retention by sheep supplemented with warm-season legume haylages or soybean meal. J. Anim. Sci. 87: 2899– 2905.
Foster, J.L., Carter, J.N. Sollenberger, L.E. Blount, A.R. Myer, R.O. Maddox, M.K. Phatak, S.C. and Adesogan. A.T. 2011. Nutritive value, fermentation characteristics, and in situ disappearance kinetics of ensiled warm-season legumes and bahiagrass. J. Dairy Sci. 94: 2042– 2050.
Giayetto, O., Cerioni, G.A. and Asnal. W.E. 1998. Effect of sowing spacing on vegetative growth, dry matter production, and peanut pod yield. Peanut Sci. 25: 86– 87.
Horrocks, R.D., and Vallentine. J.F. 1999. Harvested Forages. Academic Press, San Diego, CA. 384 pp.
Jaaffar, Z., and Gardner. F.P. 1988. Canopy development, yield and market quality in peanut as affected by genotype and planting pattern. Crop Ecol. Prod. Management. 28: 299– 305.
Johnson J.C., Butler, J.L. and Williams. E.J. 1979. Composition and nutritive value of whole plant peanuts (Arachis hypogaea L.) ensiled with and without propionic acid-formaldehyde treatment. J. Dairy Sci. 62: 1258– 1263.
Kaps, M., and Lamberson. W.R. 2009. Biostatistics for Animal Science: An Introductory Text. CAB International 2nd Edition. 504 pp.
Khan, M.T., Khan, N.A. Bezabih, M. Qureshi, M.S. and Rahman. A. 2013. The nutritional value of peanut hay (Arachis hypogaea L.) as an alternate forage source for sheep. Trop. Anim. Health Prod. 45: 849– 853.
Kinirya, J.R., Simpson, C.E. Schubert, A.M. and Reed. J.D. 2004. Peanut leaf area index light interception, radiation use efficiency, and harvest index at three sites in Texas. Field Crops Research. 91: 2– 3.
Konlan, S., Sarkodies-Addo, J. Asare, E. and Kombiok. M.J. 2013. Groundnut (Arachis hypogaea L. ) varietal response to spacing in the Guinea Savanna agro-ecological zone of Ghana: Nodulation and nitrogen fixation. Agriculture and Biology Journal of North America 4: 324– 335.
Konlan, S.P., Ayantunde, A.A. Addah, W. Dei, H.K. and Karbo. N. 2018. Emerging feed markets for ruminant production in urban and peri-urban areas of Northern Ghana. Trop. Anim. Health Prod. 50: 169– 176.
Larbi, A., Dung, D.D. Olorunju, P.E. and Adekunle, I.O. 1999. Groundnut (Arachis hypogaea) for food and fodder in crop-livestock systems: Forage and seed yields, chemical composition and rumen degradation of leaf and stem fractions of 38 cultivars. Anim. Feed Sci. Technol. 77: 33– 47.
Manyuchi, B., Deb Hovell, F.D. Ndlovu, L.R. Topps, J.H. and Tigere. A. 1997. The use of peanut hay as a supplement for sheep consuming poor quality natural pasture hay. Anim. Feed Sci. Techol. 69: 17– 26.
Minson, D.J. 1990. Forage in ruminant nutrition. Academic Press Inc. San Diago, California. pp. 85– 161.
National Research Council (NRC). 1985. Nutrient requirements of sheep. Sixth edition. National Academy Press. Washington, D.C.
Oglesby, K.A., and Fownes. J.H. 1992. Effects of chemical composition on nitrogen mineralization from green manures of seven tropical leguminous trees. Plant Soil 143: 127– 132.
Onat, B., Bakal, H. Gulluoglu, L. and Arioglu. H. 2017. The effects of row spacing and plant density on yield and yield components of peanut grown as a double crop in Mediterranean environment in Turkey. Turkish J. Field Crops 22: 71– 80.
Palm, C.A, Gachengo, C.N. Delve, R.J. Cadisch, G. and Giller. K.E. 2001. Organic inputs for soil fertility management in tropical agroecosystems: Application of an organic resource database. Agric. Ecosys. Environ. 83: 27– 42.
Prasad, K.V.S.V., Khan, A.A. Vellaikumar, S. Devulapalli, R. Reddy, R. Ch. Nigam, S.N. and Blümmel. M. 2010. Observations on livestock productivity in sheep fed exclusively on haulms from 10 different genotypes of groundnut. Anim. Nutr. Feed Technol. 10S (Special Issue): 121– 126.
Samireddypalle, A., Boukar, O. Grings, E. Fatokun, C.A. Kodukula, P. Devulapalli, R. Okike, I. and Blümmel, M. 2017. Cowpea and Groundnut haulms fodder trading and its lessons for multidimensional cowpea improvement for mixed crop livestock systems in West Africa. Front. Plant Sci. 8: 30.
Silva, A.G., Wanderley, R.C. Pedroso, A.F. and Ashbell. G. 1997. Ruminal digestion kinetics of citrus peel. Anim. Feed Sci. Technol. 68: 3– 4.
Suttie, J.M. 2000. Hay and straw conservation for small-scale farming and pastoral conditions. FAO Plant Production and Protection Series No. 29. Food and Agriculture Organization of the United Nations, Rome. 303 pages.
Tetteh, F.M., Larbi, A. Nketia, K.A. Senayah, J.K. Hoeschle-Zeledon, I. and Rahman. N. Abdul 2016. Suitability of soils for cereal cropping in Northern Ghana: Evaluation and recommendations. International Institute of Tropical Agriculture (IITA), Ibadan.
Tubbs, R.S., Beasley J.P. Culbreath, A.K. Kemerait, R.C. Smith, N.B. and Smith. A.R. 2011. Row pattern and seeding rate effects on agronomic, disease, and economic factors in large-seeded runner peanut. Peanut Sci. 38: 93– 100.
Van Soest, P.J. 1982. Nutritional ecology of the ruminant. O and B Books Inc. 374 pp.
Wei-gua, L., Meng-lu, R. Ting, L. Yung-li, D. Tao, Z. Xiao-ming, L. Jiang, L. Hussain, S. and Wen-yu. Y. 2014. Effect of shade stress on lignin biosynthesis in soybean stems. J. Integr. Agric. 16: 60345– 60347.
Woyengo, T.A.; Gachuiri, C.K. Wahome, R.G. and Mbugua. P.N.E. 2004. Effect of protein supplementation and urea treatment on utilization of maize stover by red Maasai sheep. S. Afr. J. Anim. Sci. 34: 23– 30.
Yang, C.-M.J. 2005. Proteolysis, fermentation efficiency, and in vitro ruminal digestion of peanut stover ensiled with raw or heated corn. J. Dairy Sci. 88: 2903– 2910.
Notes
- First and second authors: Graduate Student and Associate Professor, Animal Science Dept., Faculty of Agriculture, University for Development Studies, P. O. Box TL 1882 Tamale, Ghana; Third author: Postdoctoral Fellow, Agronomy, International Institute of Tropical Agriculture-Ghana, Tamale Station, P. O. Box TL 06, Tamale; Fourth author: Lethbridge Research and Development Centre, Agriculture and Agri-Food Canada, 5403 1st Avenue South P. O. Box 3000, Lethbridge, Alberta T1J 4B1, Canada [^] *Corresponding author Email: addweseh@yahoo.com
Author Affiliations