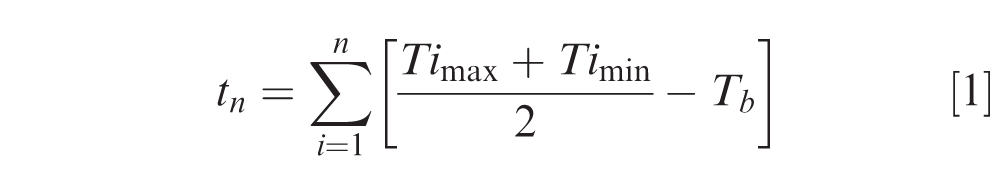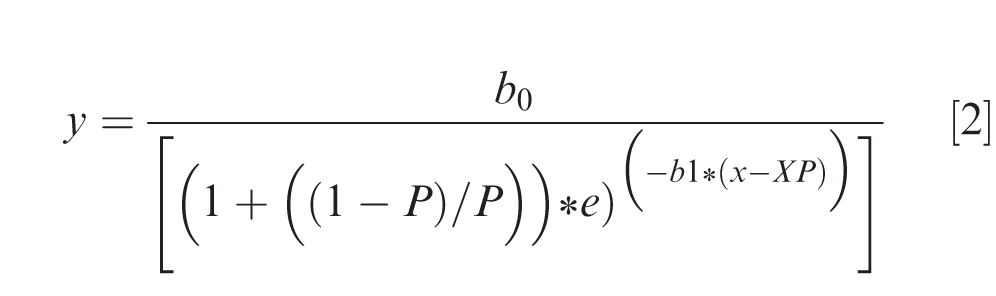Introduction
Cultivated peanut (Arachis hypogaea L.) is an important oil and food crop, grown on approximately 28.5 million hectares worldwide (FAOSTAT, 2018). It is the third major oilseed of the world behind soybean [Glycine max (L.) Merr.] and cotton (Gossypium hirsutum L.). China, India and the United States grow about 70% of the world crop. Peanuts are high in protein, fiber, tocopherols, unsaturated fatty acids, and other nutritionally advantageous compounds (Ros, 2010), and are used for oil extraction, edible food products and confectionary purposes across the globe.
Oil content and composition of peanut seed significantly affects flavor and product shelf-life. Traditionally, peanuts contain approximately 52% oil by weight. Two fatty acids, oleic and linoleic acid, comprise over 80% of the oil content in peanut. Standard peanuts contain approximately 45% oleic acid and 35% linoleic acid, resulting in an oleic to linoleic acid ratio (O/L) of approximately 1.3:1 (Dong et al., 2015). Norden was the first to discover a spontaneous mutant peanut line that was found to contain approximately 80% oleic acid and 2% linoleic acid (Norden et al., 1987). This high oleic line, F435, resulted from a mutation in the gene that codes for the enzyme responsible for the conversion of oleate to linoleate, oleoyl-PC desaturase, or Δ12 fatty acid desaturase. Much work has been done to determine the inheritance of the high oleate trait in the different peanut market types (Chu et al., 2007, 2009; Isleib et al., 1996; Jung et al., 2000a, 2000b; Lopez et al., 2001). Reports have determined that the fatty acid composition of peanut is quantitatively inherited, and investigations proved that two loci, Ol1 and Ol2, control the high O/L ratio in runner and virginia market types (Isleib et al., 2006b; Moore and Knauft, 1989). Lopez et al. (2001) confirmed that these loci also control the high oleate trait in spanish-type peanuts.
In general, saturated fatty acids are less susceptible to oxidative degradation than their less saturated counterparts. High O/L peanuts and peanut products have an increased shelf life and improved flavor after roasting when compared to those with a standard O/L ratio (Davis et al., 2016). Other benefits of the high oleate trait have also been determined. Oils that have high oleic acid content and food products containing these oils have been shown to be nutritionally beneficial. Oleic acid has been shown to be associated with a reduction in blood pressure (Teres et al., 2008) and LDL cholesterol levels (O’Byrne et al., 1997). High-oleic peanuts have health benefits compared to standard peanuts because the linoleic (polyunsaturated fat) and palmitic (saturated fat) fatty acids have been naturally replaced by the healthier oleic fatty acid (monounsaturated fat). High oleic peanuts and peanut oil high in oleic acid have also been shown to have a beneficial effect in type II diabetes and ultimately reverse the negative effects of inflammatory cytokines observed in obesity and non-insulin dependent diabetes mellitus (Vassiliou et al., 2009).
Peanut production in the US is limited to three geographical regions: The Southeast region (Alabama, Arkansas, Florida, Georgia, and Mississippi), the VC region (North Carolina, South Carolina and Virginia) and the Southwest region (New Mexico, Oklahoma, and Texas). Each growing region differs from the others in climate and disease pressure and thus, breeding programs tend to develop peanut cultivars which are specifically adapted and have superior performance in a growing region. The health and extended shelf life benefits of high oleic peanut products has resulted in an increased manufacturer demand for high oleic peanut seed and a growing number of high oleic peanut cultivars have been released. Currently, commercial production of such cultivars occurs in all US growing regions, with the Southwestern US producing 100% high oleic peanuts. Production of only high oleic cultivars in the Southwestern U.S. is largely a result of a reduction in off-flavors found in high oleic cultivars and contract restrictions influenced by market demand. With high oleic peanut production on the rise, studies on the effect of the high oleic trait on seed composition and crop agronomic characteristics have been conducted.
Several studies examining possible differences between standard peanut and high oleic genotypes reported no significant changes in seed composition. Isleib et al. (2006a), examined differences between near-isogenic lines (NILs) differing only in the high oleic trait and found no major impact on chemical composition or sensory quality in high oleic peanuts when compared to standard peanut lines, and reductions in the negative sensory attributes of cardboard and painty were noted for high oleic genotypes. In another report examining differences among the virginia type cultivar Bailey and high oleic backcross derivatives (Isleib et al., 2015), again found no significant effects of the high oleic trait on flavor attributes, disease resistance, or agronomic traits. Similar results have been found for other crops including soybean (Bachleda et al., 2017) and sunflower (Fernández-Martínez et al., 1993).
The high oleic trait has been suggested to negatively affect seed germination, especially at below optimal germination temperatures. Bachleda et al. (2017) reported that a possible reduction in soybean germination was related to high oleic when compared to normal oleic soybean germination in cold soils. Bera et al. (2019), found no significant effects of the high oleic trait on peanut seed with the exception of germination percentage, which was significantly lower for high oleic seed compared to standard peanut seed. Sun (2005) reported that high oleate virginia peanut lines were reduced in field emergence and vigor compared to standard peanut lines. Upon further study, Sun et al. (2014) reported that virginia peanuts high in oleic acid content were reduced in conductivity and vigor under all production temperatures, especially lower temperatures, when compared to peanuts with standard oil chemistry.
Information on the effect of the high oleic trait on peanut seed germination rate across specific peanut market types (runner, spanish, virginia, valencia) is limited. Producers of all crops depend on information provided by state laboratories regarding seed germination rate to determine planting rate and anticipated field emergence. State laboratories use germination testing methods developed using peanut with standard oil chemistry and do not include adjustments made for high oleate peanut seed, resulting in possible inaccurate labeling of germination rate on lots of high oleic peanut seed passed on to producers. In turn, producers may be hesitant to grow high-oleic cultivars due to the uncertainty of the high-oleic effect on agronomic traits, such as seed germination, yield and grade. In this study, we use NILs of all four peanut market types to examine the effect of the high-oleic trait on peanut seed germination and emergence in all major peanut growing regions, as well as on a thermal gradient table.
Materials and Methods
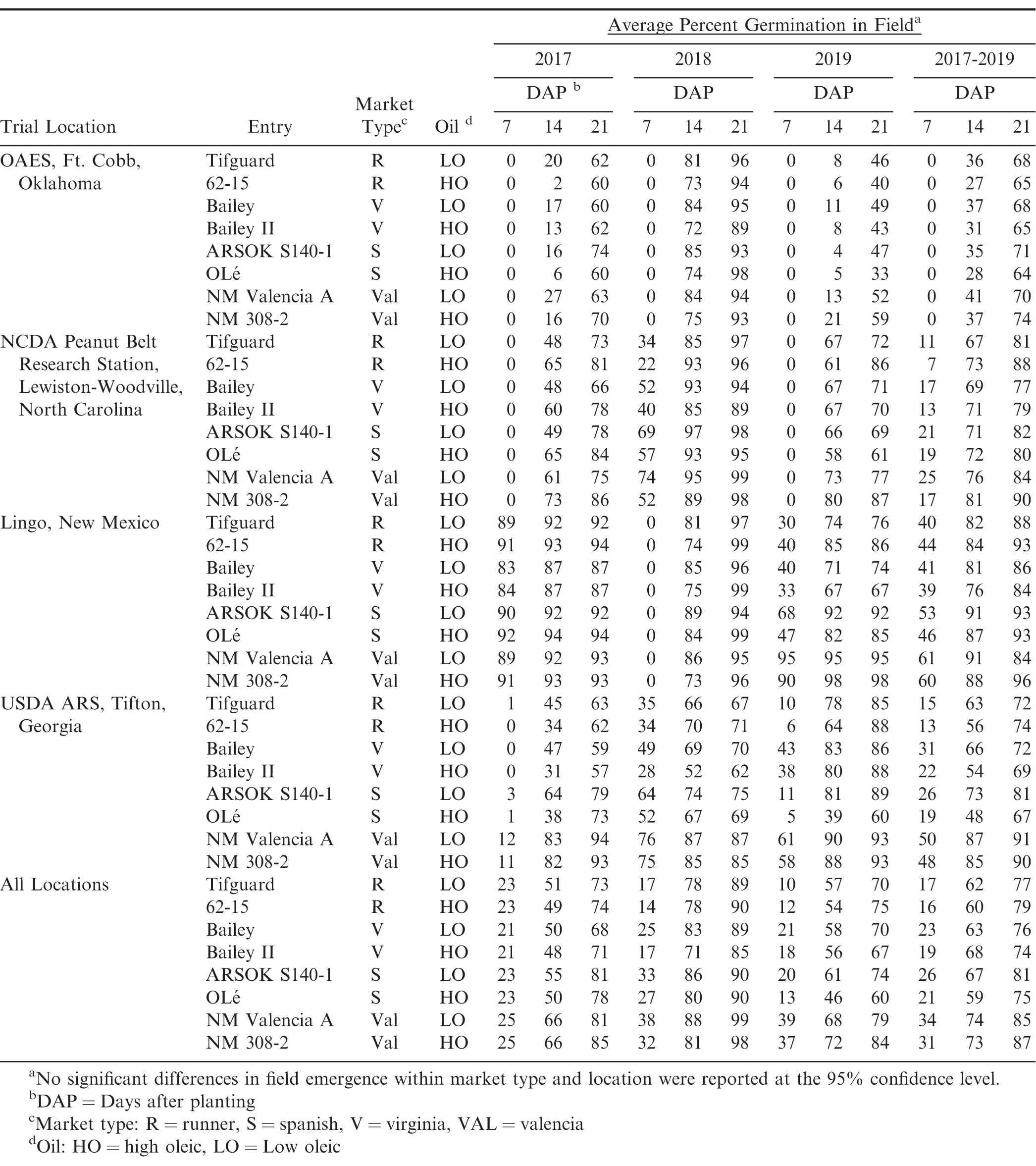
Plant Materials and Field Evaluation
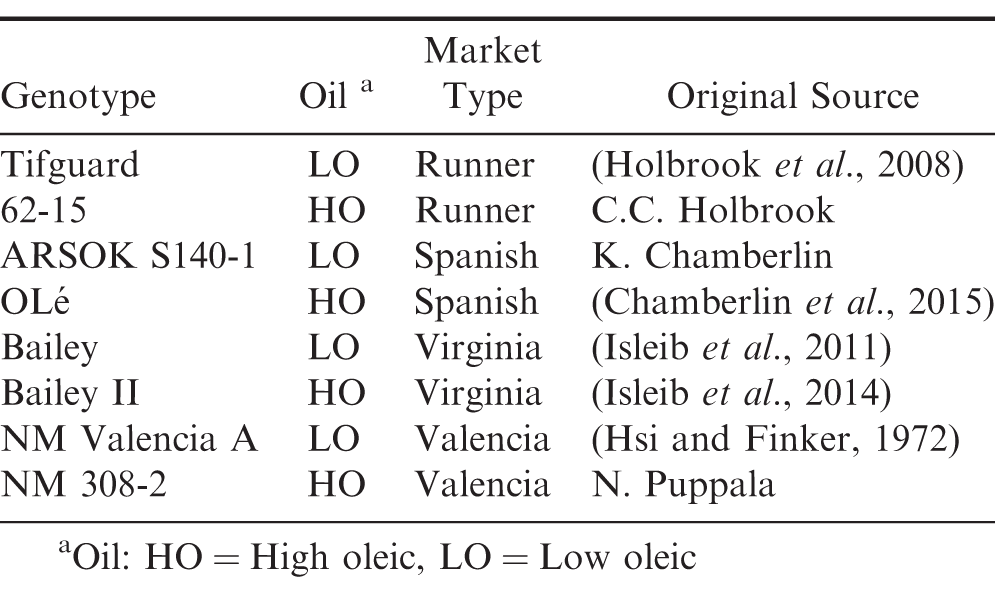
Plant materials included in these studies are listed in Table 1. Lineage of each entry can be found in the registration article of each cultivar or germplasm line with the exception of the high-oleic valencia line NM 308-2, low oleic line ARSOK S140-1, and the high oleic runner line 62-15. The NIL pairs were generated by the traditional method of crossing to incorporate the high oleic trait, followed by subsequent back crossing of progeny to the recurrent parent to the BC6 generation while selecting for the high oleic trait. High oleic line 308-2 resulted from a cross between New Mexico Valencia A and the high oleic cultivar Olin (Simpson et al., 2003). Low oleic line ARSOK S140-1 resulted from a cross between Tamspan 90 (Smith et al., 1991) and F435 (Norden et al., 1987) and was determined to be non-high oleic in nature. ARSOKS140-1 was then used as a recurrent parent in the development of OLé. High oleic line 62-15 resulted from a cross between Tifguard (Holbrook et al., 2008) and cultivar Florida-07 (Gorbet and Tillman, 2009). Seed of each genotype was increased annually in plots at the Caddo Research location of the Oklahoma Agricultural Experiment Station (OAES) located near Ft. Cobb, Oklahoma, and distributed to cooperating locations for planting in replicated field trials. Each year, before planting and after harvesting, seed from each plot was tested for oleic acid content at the USDA ARS Market Quality and Handling Research Unit in Raleigh, NC, to ensure purity (high oleic or low oleic). Oil quality of all seed was as expected at time of planting and at harvest (data not shown). Trial locations and soil types were as follows: Ft. Cobb, OK (Caddo sandy loam); USDA ARS, Tifton, GA (Tifton loamy sand); Lingo, NM (Amarillo loamy fine sand); Lewiston-Woodville, NC (Norfolk sandy loam/Goldsboro sandy loam). At each location, a randomized complete block (RCB) design with 3 replications was used to plant two-row plots that were 4.6 m long planted 90 cm apart, at a rate of 15 seeds/row meter. Stand counts (indicating field emergence) were taken at 7, 14, 21 and 28 days after planting (DAP) to determine germination rate. Spanish and valencia plots were harvested at 120 DAP. Virginia and runner plots were harvested at 145 DAP. Peanuts were placed in a forced air dryer until moisture reached 10 percent. Percent total sound mature kernels (%TSMK or grade) were determined on a 200 g sample from each plot and estimated yield in kg/ha was calculated. Seed from each annual harvest was then sent to the Tifton Campus of the University of Georgia for thermogradient table germination and vigor testing and to the USDA ARS Market Quality and Handling Research Unit in Raleigh, NC, for oil analysis (data not shown).
Statistical analysis
Statistical analysis for field data was performed. Repeated measures analysis of variance was used to assess the effects of the factors (genotype, HO, DAP) on the germination percentage using PROC MIXED (SAS Institute, 2013). The response variable was transformed using an arcsine square root transformation to alleviate issues with normality and heterogeneity of variance. DAP was considered the repeated factor, and simple effect tests of HO given genotype and DAP were assessed with planned contrasts. Means and standard errors were reported but none were significant at a 95% confidence level.
Peanut seed vigor testing on thermal gradient
Field grown seed harvested from individual plots in each state included Bailey II, Bailey (Isleib et al., 2011), OLé (Chamberlin et al., 2015), ARSOK S140-1, 62-15, Tifguard (Holbrook et al., 2008), NM 308-2, and NM VAL A (Hsi and Finker, 1972). Thermal gradient tables used were described by Grey et al. (Grey et al., 2016). Briefly, each table is constructed from solid aluminum blocks measuring 2.4 m long by 0.9 m wide by 7.6 cm thick with a mass of 470 kg. At the end of the aluminum blocks, a 1.0 cm hole was drilled across the side section to allow fluid to be pumped into the table. For one side a chiller was set at 12 C and on the other side a heating unit was set at 36 C and ethylene glycol plus water (1:10 mixture) was pumped at 3.8 L per min to generate the thermal gradient. Solutions were independent and never mixed with each other. Along the length of the thermal gradient, approximately 1.0 C increments occurred every 10 cm, with a constant temperature across the width. This allowed for 24 increments across the length to obtain different temperatures, with nine cells across the width at each temperature (Cardina and Hook, 1989; Chatterton and Kadish, 1969), resulting in 216 total cells.
Duplex insulated PR-T-24 wire (Omega Engineering, Inc. Stamford, CT) was used to make thermocouples that were mounted to the underside of the table from the hot to cold ends. Thermocouples were inserted vertically into a hole on the underside of the table and held in place by a washer and screw. Holes measured 8 mm wide by 7 cm deep to allow the thermocouple to be within 5 mm of the upper table surface at 10 cm intervals along the length of the table. Data indicated a continuous temperature gradient from 12 to 36 C. Temperature was monitored continuously for each thermocouple and recorded at 30 min intervals with a Graphtec data logger (MicroDAQ,com Ltd., Contoocook, NH). All temperature data for each thermocouple was downloaded daily in a spreadsheet format.
Peanut seed for each year (3), cultivar (8), location (4) and replication (3) collected by plot was individually tested for vigor and germination (Table 2). Seeds were treated with azoxystrobin plus fludioxonil plus mefenoxam seed treatment fungicide (Syngenta Crop Protection, Greensboro, NC). For each plot, peanut seed were evenly distributed on germination paper (SDB 86 mm, Anchor Paper Co., St. Paul, MN), which was placed in a 100 by 15 mm sterile plastic Petri dish (Fisher Scientific Education, Hanover Park, IL). Twenty seeds were placed in each Petri dish followed by the addition of 15 to 20 ml of distilled water. A single Petri dish was then placed at each 1.0 C increment every 10 cm along the length of the table for a total of 24 dishes per plot (n = 480 seed per plot). Beginning at 72 hr after seeding, peanut seed germination was counted when the radicle extended more than 5 mm beyond the seed, and then the seed was removed from the dish. Peanut seed with radicles longer than 2 mm from the seed coat are considered germinated (Ketring, 1992), but 5 mm was chosen as it has been used in previous research (Ketring and Morgan, 1969). Tests were run for 7 d with counts taken 5 consecutive days beginning at 72 hrs after planting. All counts were taken in less than one hr each day at approximately the same time, depending upon when an experiment was started on day -0-. Counts were conducted from the cold end working toward the warm end. Seed availability was limited (n = 480 seed per field replication, n = 1440 seed per cultivar each year). Germination data was converted to a percentage by day, and cumulative germination was determined for each Petri dish over the duration of that assay. Temperature data included maximum and minimum (±0.5 °C for each thermocouple), representing the highest and lowest measures, respectively taken during one germination experiment for a specific Petri dish. Maximum and minimum temperatures were then used to determine the thermal time (Cardina and Hook, 1989; Chatterton and Kadish, 1969), or growing degree day (GDD) accumulation for the equation
where tn is the sum of GDD for n days, and Timax and Timin are the daily maximum and minimum temperature C of Day i (Ketring and Wheless, 1989), and Tb is the base temperature for peanut, in this model Tb was set at 15 C (Ketring et al., 1982).
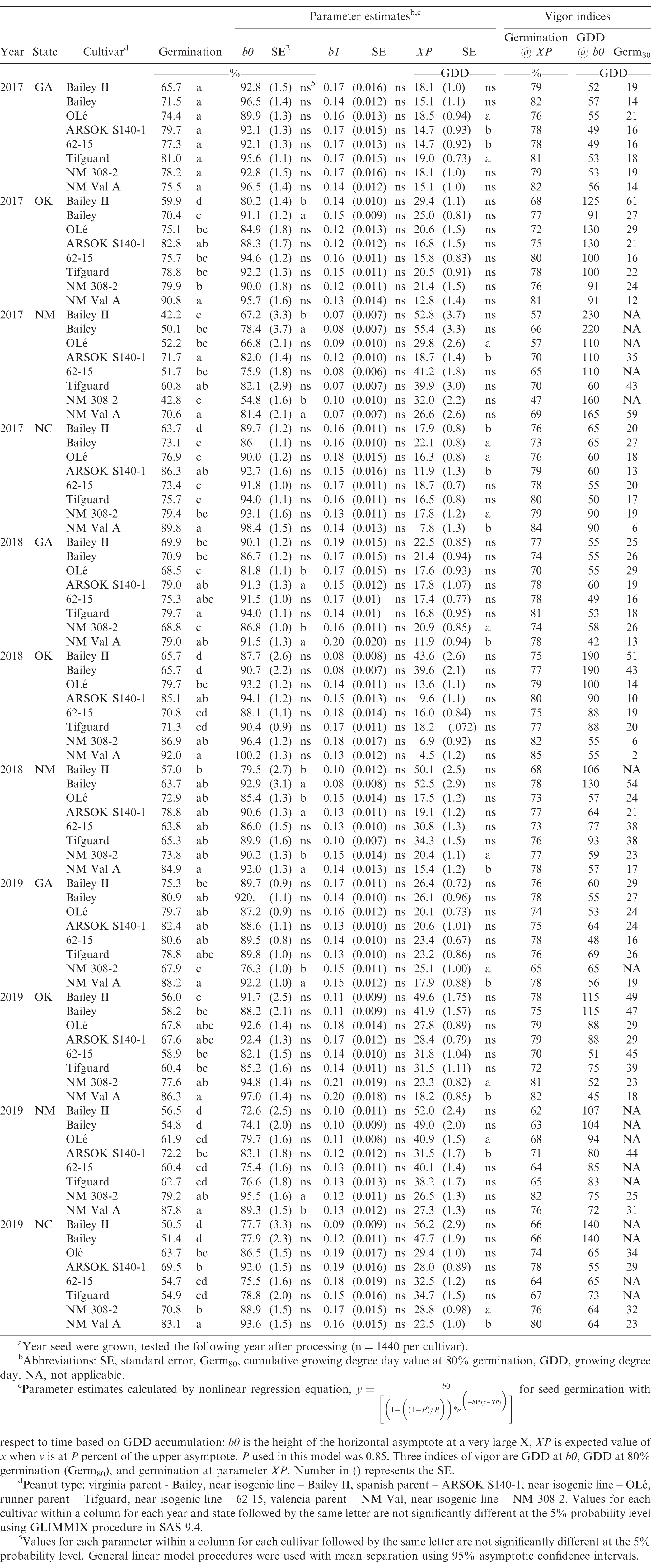
Parent and near isogenic high oleic peanut seed germination, logistic growth parameter estimates, standard error, and vigor indices for runner-, virginia-, valencia-, and spanish-type cultivars using a thermal gradient assay, when simultaneously grown in Georgia, Oklahoma, New Mexico, and North Carolina over three seasonsa from 2017 to 2019.
To model thermal time in GDD (x) required for achieving a certain level of germination (y), nonlinear regression using a three-parameter logistic growth curve (Freund and Littell, 1991) was then employed:
Where b0 is the upper asymptote, b1 is the relative slope, XP is the value of x when y is at P percent of the upper asymptote, P used in this model was 80. Three indices of vigor that were calculated from this equation are 1) growing degree days at b0, 2) growing degree days at 80% germination (Germ80), and 3) germination at XP (Table 2). A separate curve was fit for each cultivar seed set each year, and the 95% confidence limits of the parameters were used to determine differences between them. To determine actual temperature effect on germination, data for cumulative germination at 168 h after planting was fit to the average temperature of the 24 cells using the logistic growth equation [2]. This model indicated the minimum temperature at which germination occurred within a 168 hr (7 d) period (Table 2).
Data for germination percentage from the thermal gradient table was analyzed using PROC MIXED (SAS Institute, 2013). Data for cultivar equation [2] were subjected to ANOVA using the general linear models procedures with mean separation using 95% asymptotic confidence intervals. The 95% confidence limits of three parameters in the equations were used to compare the significant differences for the equation. Nonlinear regressions were used to fit data using SAS NLIN (SAS Institute, 2013) and graphed using SigmaPlot 14.
Results
Field emergence
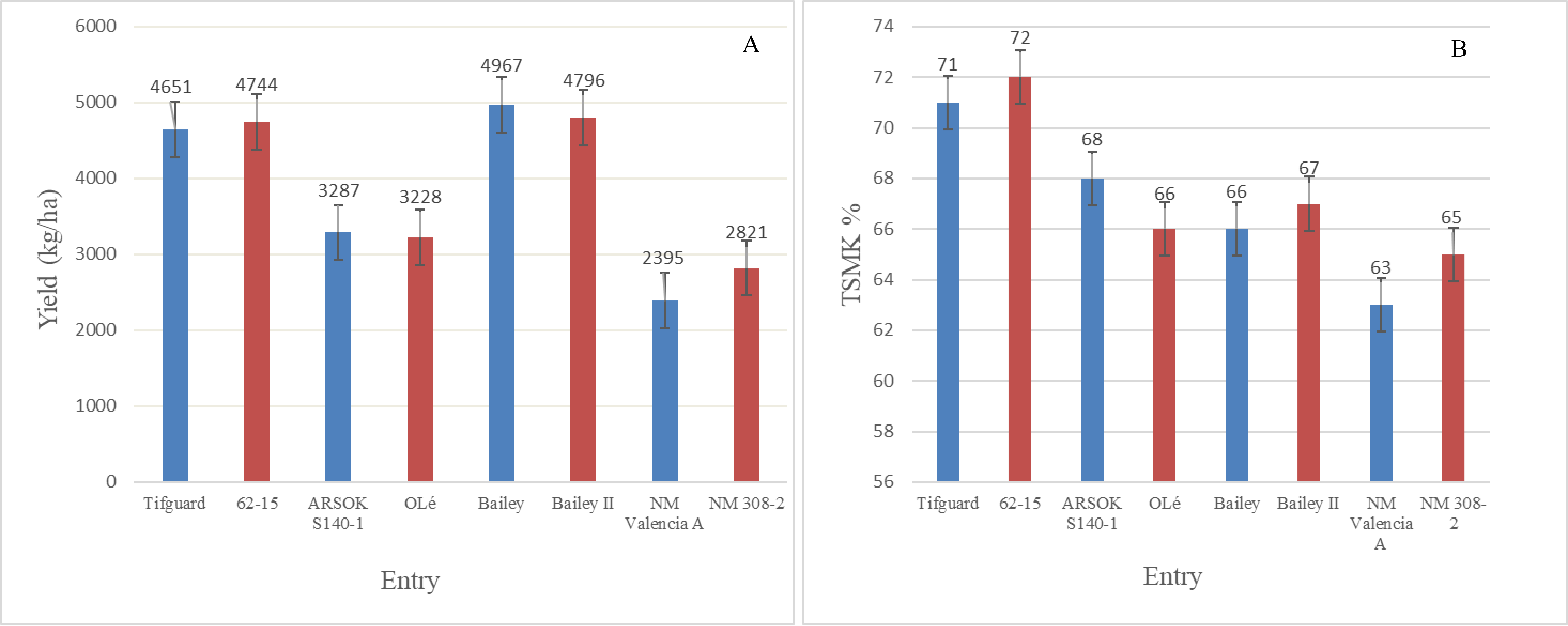
Field emergence observations were taken in three locations for three years (2017-2019) (Table 3). Although there was a notable visual lag in germination seen for most high oleic genotypes when compared to low oleic genotypes at 14 DAP, no statistical differences were found between the high oleic lines and their low oleic counterparts. Also, by 21 DAP no lag in germination was apparent between low oleic genotypes and their high oleic counterparts. Although differences were seen among market types and locations, when data were averaged across all locations and years, no differences were found between low oleic genotypes and their high oleic counterpart (within market type) in percent germination (Table 3, Figure 1), yield or grade at a 95% confidence limit.
The average yield across years and locations for the standard oleic runner type line Tifguard was 4651 kg/ha which was not different than that for the high oleic runner 62-12 which averaged 4744 kg/ha. Grades for this NIL pair were also essentially identical. Similar observations were reported for the other three market types. For the spanish NIL pair, standard oleic line ARSOK 140-1 averaged 3287 kg/ha and 68% TSMK, while high oleic line OLé averaged 3228 kg/ha and 66% TSMK. Standard oleic virginia line Bailey had an average yield of 4967 kg/ha and average grade of 66%TSMK compared to its high oleic counterpart Bailey II which averaged 4796 kg/ha and a grade of 67%TSMK. No differences in average yield and grade were noted for the valencia NIL pair New Mexico Valencia A and NM 308-2 at 2395 kg/ha and 63% TSMK, and 2821 kg/ha and 65% TSMK, respectively.
These results are not unlike those reported in studies by Isleib et al. (2006b) and Sun (2005). However, the visual and numerical lags in germination observed in most cases across locations do cause concern to producers who are rigorous regarding field emergence within the first 10 days after planting. Environmental variables associated with growing conditions were eliminated by subjecting seed from each location to germination testing on a thermal gradient table.
Harvested seed evaluation
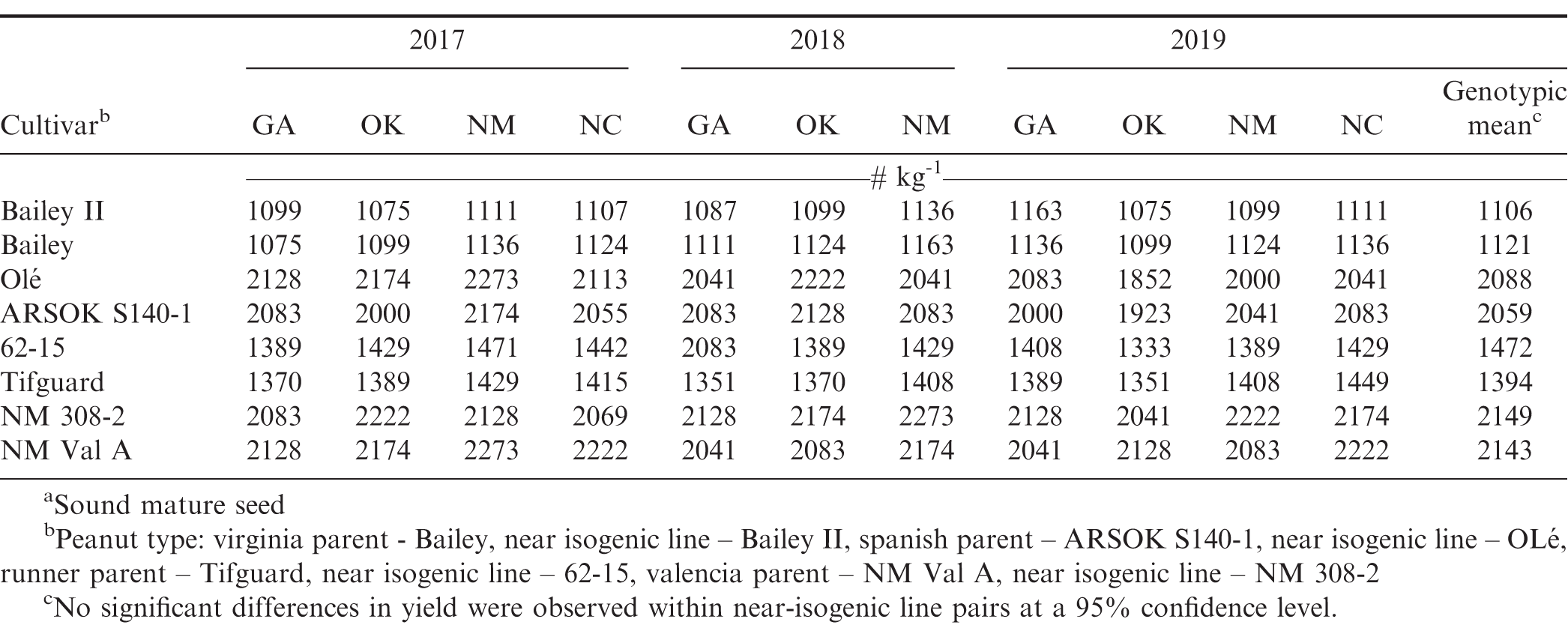
The harvested genotypic mean seed size with respect to seed number/kg was: NM 308-2 (2149) > NM Val A (2143) > OLé (2088) > ARSOK S140-1 (2059) > 62-15 (1472) > Tifguard (1394) > Bailey (1121) > Bailey II (1106) (Table 4). Average individual seed peanut mass for the virginia, runner, valencia, and spanish parent lines were 0.90, 0.72, 0.47, and 0.49 g seed−1 as compared to the near isogenic lines at 0.89, 0.68, 0.47 and 0.48 g seed−1, respectively. Within market-type, seed sizes were consistent across locations and over years, showing no differences (data not shown). These data agree with the information registered for each cultivar (Holbrook et al., 2008; Chamberlin et al., 2015; Isleib et al., 2014).
ANOVA indicated significant differences in germination rate for years, cultivars, locations, and interactions of the factors (n = 126,720 seed for the entire study, n = 31,680 seed per peanut type). Parameter estimates varied by year and significant cultivar-by-location interactions prevented data from being combined across tests. Therefore, data for locations and years were analyzed separately and presented each year by state, parent and near isogenic HO line.
Thermal gradient table germination
Average germination across all temperatures (12 to 36 C) varied by peanut type and cultivar each year (Table 2). Seed germination across all peanut types and temperatures was 71.1% (90,0085 of 126,720 seed). ANOVA indicated significant differences in germination by each state for all cultivars, except for GA in 2017. With respect to peanut type, valencia parent NM Val A consistently had the greatest germination across states and years (7 of 11 experimental sites). In contrast, virginia type HO Bailey II consistently had the lowest germination across states and years (9 of 11 experimental sites). Runner and spanish peanut seed germination rates were between valencia and virginia types across locations and years, with the trend of parents always having higher germination than the near isogenic HO lines. Field studies have previously noted difference in emergence for peanut types as runner > virginia (Balota and Phipps, 2013).
Overall cultivar germination by state in 2017 indicated that the parent line was greater by peanut type with valencia > spanish > runner > virginia: NM Val A (81.7%) > ARSOK S140-1 (80.1%) > Tifguard (74%) > Bailey (66.3%) (data not shown), respectively. This same trend was observed for the HO lines with NM 308-2 (70.1%) > OLé (69.7%) > 62-15 (69.5%) > Bailey II (57.9%), (data not shown) respectively. In 2018, with three states, the same trend was observed for the parent: NM Val A (85.3%) > ARSOKS140-1 (80.9%) > Tifguard (72.1%) > Bailey (66.8%), and HO lines: NM 308-2 (76.5%) > OLé (73.7%) > 62-15 (70%) > Bailey II (64.2%) (data not shown), respectively. It was the same in 2019 across states for the parent lines and HO: NM Val A (86.4%) > ARSOK S140-1 (72.9%) > Tifguard (64.2%) > Bailey (61.3%), NM 308-2 (73.9%) > OLé (68.3%) > 62-15 (63.6%) > Bailey II (59.6%), (data not shown) respectively. What is unique about these germination data for the parent and HO lines was that it was inversely related to genotypic seed size when averaged across all 11 sites: NM Val A < ARSOK S140-1 < Tifguard < Bailey, NM 308-2 < OLé < 62-15 < Bailey II, respectively (Table 4). Genotypically, this indicates that smaller seeded peanut types such as valencia, have greater potential to achieve higher germination for both the parent and HO lines than large seed size type virginia. By state, New Mexico produced seed phenotypically had the lowest seed germination each year (Table 4).
Parameter estimates
Using 95% confidence intervals, the three parameter estimates for equation [2] in the logistic growth curve models were compared within parent and HO cultivars by peanut type by state and year (Table 2). The predicted maximum germination rates (parameter b0), germination as affected by time (b1), and GDD to maximum germination (XP), varied (Table 2).
For maximum germination parameter estimate b0 (the upper asymptote of the line), NM Val A consistently had the highest maximum germination rates (11 of 11 sites) at 70.6 to 92% (Table 2). Of the 44 parent and near isogenic HO line comparisons for b0, 10 were significantly different from each other. Half of those were NM 308-2 compared to NM Val A, three of those were seed produced in New Mexico, and in all instances the parent line had greater vigor than the near isogenic HO line (2017, 2018, 2019). This could be phenotypically related to valencia type seed and environmental growing conditions as this was not observed for GA, OK, or NC. Virginia type peanut seed was the only other comparison that exhibited similar variations in b0 with three instances occurring for Bailey and Bailey II (OK, 2017: NM 2017, 2018). As previously noted, virginia type seed are larger and have shown vigor differences in side by side comparisons using cool germination testing, as near isogenic HO lines had lower germination than the parent (Sun et al., 2014).
There were no differences for the parent as compared to the near isogenic HO cultivars as affected by time to germination represented by parameter b1 for state or year (Table 2). This parameter ranged from 0.07 to 0.20 indicating the consistent germination of these seed (as the number increases, the slope flattens and would indicate no germination at b1=1.0). This is a significant finding in that it quantifies the importance of breeding the near isogenic lines from the parents, incorporating the HO trait, while maintaining genetic stability across 11 experimental site years. Variation in peanut seed vigor has been reported and attributed to phenotypic factors in peanut seed production (Grey et al., 2016; Sun et al., 2014).
Parameter XP establishes how many GDDs are required to reach b0. For this research XP varied widely due to seed production at four states across three years, ranging from 7.8 (NC 2017, NM Val A) to 52.5 GDD (NM 2018, Bailey). For 10 of the 12 significant differences noted, parent lines required less GDD to reach XP than the near isogenic HO (Table 4). This indicates that some near isogenic HO lines will require greater GDDs for field establishment as compared to their parent lines, i.e. longer to emerge from soil, as was noted in the field experiments.
Vigor indices
Two indices can be obtained from the logistic growth equation to elucidate the seed germination performance of each cultivar by solving equation [2]. One is GDD @ b0, which varied for this research across all years and cultivars (42 to 230 GDD). However, looking at GDD @ b0 by year indicated similarities for the parent and near isogenic HO lines for nearly all comparisons. While the near isogenic HO lines may have slower germination, it only required a few extra GDDs to reach the maximum potential as compared to the parent line. This does not imply that germination was the same, only that to reach parameter estimate b0 in GDDs was similar for parent and HO lines, again indicating the genotypic stability that was achieved in breeding. The second indices are the GDD values at Germ80, which is an indication of seed vigor. The lower the Germ80 value the greater the seed vigor. Germ80 did not always reflect the same trends observed with XP, as the near isogenic HO lines were the same, and sometimes required fewer GDD than the parent. This indicates the phenotypic variation in seed production that can occur, but genetic consistency in the near isogenic HO and parent lines will result in similar stand establishment in the field, as was previously noted. Based on Germ80, seed vigor was most consistent for production in GA (2017, 14 to 21 GDD, 2018, 18 to 29 GDD, 2019, 19 to 29 GDD) across cultivars. OK produced seed was similar in vigor to GA, but NC was variable. NM consistently had the lowest vigor levels, with several instances in 2017 and 2019 where seed never achieved 80% germination.
Discussion
Data from this research indicated that with respect to peanut seed size from 11 site-year locations, the order was valencia < spanish < runners < virginia types. Seed from all cultivars exhibited phenotypic vigor variation with respect to the state and year it was produced. However, there was genotypic consistency for the parents as well as the near isogenic HO lines. Comparing data generated from the thermal gradient using these growth curve models gave maximum germination rates and established that parent lines required less GDD (XP) to achieve maximum germination that the near isogenic HO lines. All genotypes exhibited stability over the course of this research for the three years the variables were measured. These data assisted in determining phenotypic and genotypic variation between cultivars when grown under known environmental conditions.
Germination begins as the seed becomes imbibed, triggering biochemical changes that lead to the reinitiating of metabolic processes that induce radical growth and shoot formation (Rajjou et al., 2012). This process involves the conversion of fatty acids (such as oleic and linoleic acid) to acetyl-CoA and ATP which are used to synthesize sugars used by the embryo as a food source. These changes occur through beta-oxidation (Goodwin and Mercer, 1983). One possible explanation of the germination delay in HO peanut seed involves this process. HO seed have a high concentration of oleic acid and low of linoleic acid, and the complete oxidation of oleic acid is slower than that of linoleic acid (Holman and Elmer, 1947; Hiltunen et al., 1986). Therefore, breakdown of the fatty acids and formation of glucose needed for germination takes longer in HO seed compared to LO seed.
One explanation as to why high oleic peanut seed germinate more slowly under cooler conditions than low oleic counterparts may involve cell membrane fluidity. Oleic acid is less saturated than linoleic acid, and thus more viscous (Davis et al., 2016). When reacting to stress such as temperature change, plants regulate their degree of cell membrane lipid unsaturation, increasing the percentage in cool temperatures to maintain function (Upchurch, 2008). Attia et al. (2021) reported that the cell membranes of oil seed crops produced at cool temperatures contain higher levels of polyunsaturated lipids. Because high oleic peanut seeds contain a mutated oleoyl-PC desaturase, or Δ12 fatty acid desaturase gene, the ability to regulate cell membrane fluidity may be diminished, resulting in an increased rigidity of high oleic cell membranes may contribute to lower rates of seed germination, especially at lower temperatures.
However, the process eventually does occur in HO seed, resulting in little effect on agronomic performance of HO cultivars compared to standard cultivars. Several authors have reported that HO seed germination is noticeably delayed in comparison with LO seed at low soil temperatures (Bachleda et al., 2017; Jungman, 2000; Sun et al., 2014), and our thermal gradient table data also reflects those observations. These observations are consistent with thoughts that fatty acid oxidation rate is key in seed germination efficiency. This phenomenon is likely more obvious at lower temperatures because the enzymes involved in beta oxidation have an optimal temperature at which reactions are catalyzed. Since oleic acid requires longer to convert to glucose compared to linoleic acid (Holman and Elmer, 1947), the high concentration of oleic acid found in HO seed would result in a slowed seed germination rate, especially at lower temperatures where the enzymes involved work less efficiently.
In the US, state laboratories perform germination tests on seed lots of various seed classes, annually. These tests determine the capability of a seed lot to produce normal seedlings under favorable controlled conditions. Producers rely on the results of state germination testing to predict field emergence and crop vigor. Standardized testing procedures for the US, Canada and some Central American countries are set forth by the Association of Seed Analysts (AOSA, 2019) and are specific to individual types of seed. For peanut, the same testing protocol is followed regardless of whether the seed is HO or LO (AOSA, 2019). According to the AOSA rules for testing seeds, the first count of germinated seed is taken five days after initiation of the test, with the final count being taken at day 10. Our study indicates that the HO genotypes examined in this study varied by the four peanut market types. The required GDD to obtain maximum HO germination for the near isogenic lines was NM 308-2 (21.9) < OLé (22.9) <62-15 (25.7) < Bailey II (38.1) (data not shown). While the parent lines GDD were NM Val A (16.4) < ARSOKS140-1 (19.7) < Tifguard (26.6) < Bailey (36) (data not shown). Only the runner parent line of Tifguard required more GDD than the HO near isogenic line of 62-15. Germination data for the parent and near isogenic lines by peanut type indicated that GDD’s for valencia < spanish < runner < virginia. The current AOSA protocols for testing peanut germination take the first germination measurement at five days and a final count at seven days (AOSA, 2019). These protocols were developed before the wide-spread production of high oleic peanut cultivars and have not been adapted to consider the early germination lag of high oleic seed, demonstrated by this study, when compared to standard oleic peanuts. Considering the results of this study, modification of the AOSA testing procedure by extending the germination window to 21 days would more accurately evaluate the percent germination of HO peanut cultivars and reflect their viability on bags of registered seed. As a long season crop requiring up to 150 days to maturity, peanut stand establishment is critical in maintaining a profitable crop. The results from this study will assist growers with making HO seed selections and better inform producers on expectations of early field emergence of HO cultivars.
Acknowledgements
This project was funded by USDA ARS CRIS project numbers 3072-21220-008-00-D, 6048-21220-016-00-D, and 6070-43440-013-00-D, New Mexico State University, University of Georgia, and North Carolina State University. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Literature Cited
Attia, Z., Pogoda, C.S., Reinert, S., Kane, N.C., and Hulke. B.S. 2021. Breeding for sustainable oilseed crop yield and quality in a changing climate. Theor. Appl. Genet. doi: 10.1007/s00122-021-03770-w.
Association of Official Seed Analysts (AOSA). 2019. Rules for Testing Seeds 1:6–16 and Principles and Procedures: Seedling Evaluation, 4: 57– 60.
Bachleda, N., Grey, T., and Li. Z. 2017. Effects of high oleic acid soybean on seed yield, protein and oil contents, and seed germination revealed by near-isogenic lines. Plant Breed. 135: 539– 547. doi: 10.1111/pbr.12497
Balota, M. and Phipps. P. 2013. Comparison of virginia- and runner-type peanut cultivars for development, disease, yield potential, and grade factors in eastern Virginia. Peanut Sci. 40: 15– 23.
Bera, S.K., Kamdar, J.H., Kasundra, S.V., Patel, S.V., Jasani, M.D., Maurya, A.K., Chandrashekar, A.B., Rani, K., Manivannan, N., Pasupuleti, J., Pandey, M.D., Vasanthi, R.P., Dobariya, K.L., Radhakrishnan, T., and Varshney. R.K. 2019. Steady expression of high oleic acid in peanut bred by marker-assisted backcrossing for fatty acid desaturase mutant alleles and its effect on seed germination along with other seedling traits. PLoS ONE, 14(12): e0226252. https://doi.org/10.1371/journal.pone.0226252
Cardina, J. and Hook. J.E. 1989. Factors influencing germination and emergence of Florida beggarweed (Desmodium totruosum). Weed Technol. 3: 402– 407.
Chamberlin, K.D., Bennett, R.S., Damicone, J.P., Godsey, C.B., Melouk, H.A., and Keim. K. 2015. Registration of ‘OLé’ peanut. J. Plant Reg. 9: 154– 158 / doi: 10.3198/jpr2014.10.0072crc
Chatterton, N. and A. Kadish, A. 1969. A temperature gradient germinator. Agron. J. 61: 643– 644.
Chu, Y., Holbrook, C.C., and Ozais-Akins. P. 2009. Two alleles of ahFAD2B control the high oleic acid trait in cultivated peanut. Crop Sci. 49: 2029– 2036.
Chu, Y., Ramos, L., Holbrook, C.C., and Ozais-Akins. P. 2007. Frequency of a loss-of-function mutation in oleoyl-PC desaturase (ahFAD2A) in the mini-core of the U.S. peanut germplasm collection. Crop Sci. 47: 2372– 2378.
Davis, J. P., Price, K., Dean, L., Sweigart, D.S., Cottonaro, J., and T.H. Sanders, T.H. 2020. Peanut oil stability and physical properties across a range of industrially relevant oleic acid/linoleic acid ratios. Peanut Sci. 43 (1): 1– 11. ).
Dong, X. Y., Zhong, J., Wei, F., Lv, X., Wu, L., Lei, Y., Liao, B.S., Quek, S.Y., and Chen. H. 2015. Triacylglycerol composition profiling and comparison of high-oleic and normal peanut oils. J. Amer. Oil Chem. Soc. 92: 233– 242.
Fernández-Martínez, J., Muňoz, J., and Gómez-Arnau. J. 1993. Performance of near-isogenic high and low oleic acid hybrids of sunflower. Crop Sci. 33: 1158– 1163.
Food and Agriculture Organization of the United Nations, FAOSTAT. 2018. Available: http://faostat.fao.org accessed on 30-8-2020.
Freund, R.J. and Littell. R. SAS system for regression. 2nd ed. 1991. SAS Institute, Cary NC.
Goodwin, T.W. and Mercer. E.I. 1983. Introduction to Plant Biochemistry, 2nd ed, Pergamon Press, New York, pp. 258– 259.
Gorbet, D.W. and Tillman. B.L. 2009. Registration of ‘Florida-07’ peanut. J. Plant Reg. 3: 14– 18. doi: 10.3198/jpr2008.05.0276crc
Grey, T.L., Branch W., Tubbs R.S., Snider J., Webster T.M., Arnold, J., and Li. X. 2016. The impact of genotype x environment effects on runner-type peanut seed vigor response to temperature. Agron. J. 108: 1424– 1433.
Hiltunen, J.K., Kärki, T., Hassinen, I.K., and Osmundsen. H. 1986. β-Oxidation of polyunsaturated fatty acids by rat liver peroxisomes: A role for 2, 4-dienoyl-coenzyme A reductase in peroxisomal β-oxidation. J. Biol. Chem. 261: 16484– 16493.
Holbrook, C.C., Timper, P., Culbreath, A.K., and Kvien. C.K. 2008. Registration of ‘Tifguard’ peanut. J. Plant Reg. 2: 92– 94. doi: 10.3198/jpr2007.12.0662crc
Holman, R.T. and Elmer. O.C. 1947. The rates of oxidation of unsaturated fatty acids and esters. J. Amer. Oil Chem. Soc. 24: 127– 129.
Hsi, D.C. and Finkner. R.E. 1972. Registration of New Mexico Valencia A peanut. Crop Sci. 12: 256.
Isleib, T.G., Copeland, S.C., Hollowell, J.E., Patee, H.E., and Milla-Lewis. S.R. 2015. Comparison of Bailey Virginia Type Cultivar with High-Oleic Backcross Derivatives. In: Proc. of the 47th annual meeting of the American Peanut Research and Education Society, p. 106.
Isleib, T.G., Day, J.L., Coy A.E., Beasley, J.P.,and Branch. W.D. 2014. Genotype-by-irrigation interaction in the Georgia Official Variety Test. Peanut Sci. 41: 1– 7
Isleib, T.G., Milla-Lewis, S.R., Pattee, H.E., Copeland, S.C., Zuleta, M.C., Shew, B.B., Hollowell, J.E., Sanders, T.H., Dean, L.O., Hendrix, K.W., Balota, M., and J.W. Chapin, J.W. 2011. Registration of ‘Bailey’ peanut. J. Plant Reg. 4: 27– 39. doi: 10.3198/jpr2009.12.0742crc
Isleib, T.C., Pattee, H.E., Sanders, T.H., Hendrix, K.W., and Dean. L. 2006 a. Compositional and sensory comparisons between normal- and high-oleic acid peanuts. J. Ag. Food Chem. 54: 1759– 1763.
Isleib, T.G., Wilson, R.F., and Novitzky. W.P. 2006 b. Partial dominance, pleiotropism, and epistasis in the inheritance of the high-oleate trait in peanut. Crop Sci. 46: 1331– 1335.
Isleib, T.G., Young, C.T., and Knauft. D.A. 1996. Fatty acid genotypes of five Virginia-type peanut cultivars. Crop Sci. 36: 556– 558.
Jung, S., Powell, G., Moore, K., Abbott. A. 2000 a. The high oleate trait in the cultivated peanut [Arachis hypogaea L.]. II. Molecular basis and genetics of the trait. Mol. Gen. Genet. 263: 806– 811.
Jung, S., Swift, D., Sengoku, E., Patel, M., Teule, F., Powell, G., Moore, K., and Abbott. A. 2000 b. The high oleate trait in the cultivated peanut [Arachis hypogaea L.]. I. Isolation and characterization of two genes encoding microsomal oleoyl-PC desaturases. Mol. Gen. Genet. 263: 796– 805.
Jungman, B.S. 2000. The effect of fatty acid profiles on peanut seed germination at low soil temperatures. PhD Thesis. Texas Tech University.
Ketring D.L. Physiology of oil seeds. X. 1992. Seed quality of peanut genotypes as affected by ambient storage temperature. Peanut Sci. 19: 72– 77.
Ketring, D.L., Brown, R.H., Sullivan, G.A., and Johnson. B.B. 1982. Peanut Physiology In: Pattee H.E., Young C.T. eds., Peanut Science and Technology, American Peanut Research and Education Society, Inc. Stillwater, Oklahoma.
Ketring, D.L. and Morgan. P.W. 1969. Ethylene as a component of the emanations from germinating peanut seeds and its effect on dormant Virginia-type seeds. Plant Physiol. 44: 326– 330.
Ketring D.L. and Wheless. T.G. 1989. Thermal time requirements for phonological development of peanut. Agron. J. 81: 910– 9177.
Lopez, Y., Smith, O.D., Senseman, S.A., and Rooney. W.L. 2001. Genetic factors influencing high oleic acid content in Spanish market-type peanut cultivars. Crop Sci. 41: 51– 56.
Moore, K.M. and Knauft. D.A. 1989. The inheritance of high oleic acid in peanut. J. Hered. 80: 252– 253.
Norden, A.J., Gorbet, D.W., Knauft, D.A., and Young. C.T. 1987. Variability in oil quality among peanut genotypes in the Florida breeding program. Peanut Sci. 14: 7– 11.
O’Byrne, D.J., Knauft, D.A., and Shireman. R.B. 1997. Low fat-monosaturated rich diets containing high-oleic peanuts improve serum lipoprotein profiles. Lipids. 32: 687– 695.
Rajjou, L., Duval, M., Gallardo, K., Catusse, J., Bally, J., Job, C., and Job. D. 2012. Seed germination and vigor. Ann. Rev. Plant Biol. 63: 307– 333.
Ros, E. 2010. Health benefits of nut consumption. Nutrients. 2: 652– 682.
SAS Institute. 2013. SAS 9.4, SAS Institute Inc., Cary, N.C.
Simpson, C.E., Baring, M.R., Schubert, A.M., Melouk, H.A., Lopez, Y., and Kirby. J.S. 2003. Registration of ‘OLin’ peanut. Crop Sci. 43: 1880– 1881.
Smith, O.D., Simpson, C.E., Grichar, W.J., and Melouk. H.A. 1991. Registration of ‘Tamspan90’ peanut. Crop Sci. 31: 1710– 1711.
Sun, M.H. 2005. Effect of production environment on seed quality of normal and high-oleate large seeded Virginia-type peanut (Arachis hypogaea L.). In: Seed Quality Issues Associated with High-Oleic Peanut. PhD Thesis, North Carolina State University.
Sun, M., Spears, J.F., Isleib, T.G., Jordan, D.L., Penny, B., Johnson, D., and Copeland. S. 2014. Effect of production environment on seed quality of normal and high oleate large seeded Virginia-type peanut. Peanut Sci. 41: 90– 99.
Teres, S., Barcelo-Coblijn, G., Benet, M., Alvarez, R., Bressani, R., Halver, J.E., and Escriba. P.V. 2008. Oleic acid content is responsible for the reduction in blood pressure induced by olive oil. Proc. Nat. Acad. Sci, USA, 105: 13811– 13816.
Upchurch RG. 2008. Fatty acid unsaturation, mobilization, and regulation in the response of plants to stress. Biotechnol Lett. 30 (6): 967– 77. doi: 10.1007/s10529-008-9639-z. Epub 2008 Jan 29. PMID: 18227974.
Vassiliou, E.K., Gonzalez, A., Garcia, C., Tadros, J.H., Chakraborty, G., and Toney. J.H. 2009. Oleic acid and peanut oil high in oleic acid reverse the inhibitory effect of insulin production of the inflammatory cytokine TNF-α in both in vitro and in vivo systems. Lipids Health Dis. doi: 10.1186/1476-511X-8-25
Notes
- USDA ARS, Wheat, Peanut, and Other Field Crops Research Unit, 1301 N. Western Rd., Stillwater, OK 74074. [^]
- Department of Crop and Soil Sciences, University of Georgia, 2360 Rainwater Road, Tifton, GA, 31793 [^]
- New Mexico State University, 2346 State Road 288, Clovis, NM, 88101 [^]
- USDA ARS, Coastal Plain Experiment Station, P.O. Box 748, Tifton GA, 31793 [^]
- Department of Crop and Soil Sciences, North Carolina State University, Method Road Unit 3, Raleigh, NC, 27695 [^]
- USDA ARS, Food Science and Market Quality Handling Research Unit, 322 Schaub Hall, Raleigh, NC, 27695 [^]
- Department of Statistics, Oklahoma State University, Stillwater, OK 74078 [^] *Corresponding author Email: Kelly.chamberlin@usda.gov
Author Affiliations