Introduction
Hermetic or airtight storage of grain has been used for thousands of years to preserve quality and protect against destruction by insect pests (Murdock and Baoua, 2014). Villers et al. discussed the development of hermetic storage particularly for developing countries to preserve quality and minimize losses to insects and spoilage. Murdock and Baoua describe the development and successful implementation of a triple-bag storage system for storing cowpea (Vigna Savi) in many parts of Africa by researchers from Purdue University through the Bean/Cowpea Collaborative Research Support Program (CRSP). The Purdue Improved Crop Storage (PICS) system uses three plastic bags: two 80-μm high-density polyethylene (HDPE) bags, one inside the second; both are enclosed by a third bag made of woven polypropylene to provide mechanical protection from abrasion to the internal HDPE bags. This low-cost system reduces losses of cowpea (Freitas et al., 2016; Murdock et al., 2012) and corn (Zea mays L.) (Walker et al., 2014; Williams et al., 2014). A commercial system for hermetic storage has been developed similar to the PICS bags by GrainPro® (Washington, DC) as larger scale hermetic storage solutions for many commodities. Research is available that compares hermetic storage in the PICS and GrainPro® products (Baoua et al., 2013). Use of PICS bags has become widespread in Africa for storing grain commodities through extensive education and outreach efforts (Ibro et al., 2014; Moussa et al., 2014).
Research related to hermetically storing peanuts has been conducted with mixed results. Slay et al. stored shelled Florunner (Norden et al., 1969) peanut seed in conventional and hermetically-sealed bags for seven months to determine the effect of low oxygen storage on germination, moisture content, and market grade factors. Three of the six seed lots showed a significant difference in post-storage percent germination due to storage method. Peanuts stored in low oxygen had a higher germination rate in one of the three seed lots, while two of the seed lots stored conventionally had a higher germination. Hermetic storage of farmers’ stock (in-shell) peanuts has been attempted on numerous occasions. Butts et al. (2007) attempted flushing commercial and 1/10th-scale monolithic concrete domes used to store farmers’ stock peanuts with nitrogen reducing the oxygen levels to less than 5%. They were unable to maintain the low oxygen atmosphere in the 1/10th-scale domes but were able to reduce and maintain the low oxygen atmosphere in the commercial storage. However, the commercial facility was opened in early spring for inspection and resealed. The facility was not aerated sufficiently to equilibrate the peanut temperature with the warmer, more humid air prior to flushing with nitrogen allowing mold to grow on the top of the peanut mass. In smaller scale studies flexible intermediate bulk containers (totes) were stored unsuccessfully in the GrainPro® Cocoon (Butts et al., 2008). Similarly, a test in which dry farmers’ stock and shelled oilstock peanuts were placed in grain bags. After 40 t of farmers’ stock peanuts were stored for 30 d, sufficient moisture migration and condensation in the top of the bag had occurred to render the peanuts unsuitable for consumption . Others have successfully used PICS bags on shelled peanut to minimize postharvest losses due to insects and mold (Baributsa et al., 2017; Sudini et al., 2017). However, these studies did not investigate the final seed quality nor flavor. Abudulai et al. (2020) had stored intact peanut pods in hermetically sealed bags (Grain Pro, Inc., Washington, DC). Their results showed that aflatoxin concentration increased about 4 μg/kg during storage in both woven polypropylene bags and hermetically sealed bags.
By funding collaborative research efforts, USAID has been instrumental in improving peanut production methods to increase yields so that a commercial enterprise can begin to use locally sourced peanuts in countries such as Guyana, Haiti, and Malawi (Peanut & Mycotoxin Innovation Lab, 2018). A level of success in production had been reached to require research and education efforts on postharvest drying and storage to maintain edible peanut quality related to aflatoxin contamination and future seed viability. Therefore, the objective of this study was to determine the effect of storing shelled peanuts in PICS bags on aflatoxin contamination, seed germination, and oil chemistry.
Materials and Methods
Runner market type peanut was produced in research plots on farms within a 20-km radius of Dawson, GA using conventional irrigated production practices and harvested at optimum maturity (Williams and Drexler, 1981). Both high oleic and normal oleic peanut cultivars used in the study were excess peanut harvested on the research farms according to conventional practice, and dried at the National Peanut Research Laboratory in Dawson, GA. The high oleic peanut was a mixture of Georgia-09B (Branch, 2010) and Georgia-13M (Branch, 2014) intentionally commingled during harvest and the normal oleic cultivar was Georgia-06G (Branch, 2007). Peanut was placed in four different drying wagons, maintaining the integrity of the high oleic and normal oleic segregation. Two wagons, one high oleic and one normal oleic, were dried so that the moisture content after shelling would be approximately 8% wet basis, and the other two dried to reach a target of 7%. After drying, peanut was cleaned (Model 403-404 Cleaner, Hobbs Engineering Co, Suffolk, VA) and shelled using pilot-scale commercial shelling equipment (LMC Manufacturing, Donalsonville, GA) located at the National Peanut Research Laboratory in Dawson, Georgia. After shelling the peanuts were passed over a 6.3-mm screen to remove the small and split kernels leaving a composite of Jumbo-, Medium-, and Number 1-sized kernels (USDA, 2004) for the storage study. Peanut was placed in either burlap bags (Model S-11053, ULine, Pleasant Prairie, WI) or the Purdue Improved Crop Storage (PICS) II bags. The PICS II system consists of two 80-μm high-density polyethylene (HDPE) bags, one placed inside the other, then placed inside a woven polypropylene sandbag (Model S-16504, ULine, Pleasant Prairie, WI). Treatments for the experiment are summarized as follows:
-
Two Fatty Acid Profiles
-
High Oleic
-
Normal Oleic
-
-
Two Initial Moisture Contents
-
ca 7%
-
ca 8%
-
-
Four Storage Treatments
-
Burlap
-
PICS: PICS bags with standard protocol
-
PICS + VAC: PICS bags with air extracted using a shop vac
-
PICS + ClO2: PICS bags with 20-g sachet containing timed-release chlorine dioxide fumigant.
-
There were three replications of each combination of fatty acid profile, moisture, and storage treatment resulting in a total of 48 samples. Burlap bags were filled with approximately 23 kg of shelled peanuts, labelled, and sewn closed and placed in a small monolithic concrete dome (Butts et al., 2007). Approximately 27 kg of shelled peanuts were placed in each of the PICS bags. The standard PICS treatment (storage treatment b) consisted of filling and sealing the bags following the protocol outlined in the PICS training video . In the PICS + VAC storage treatment (treatment c), the air was evacuated from the PICS bags using a wet-dry vacuum (Model CMXEVBE17584, Craftsman, Towson, MD). The PICS + ClO2 was the same as treatment b with the addition of a small sachet containing the dry fumigant, chlorine dioxide (ICA TriNova, LLC Forest Park, GA) in the inner HDPE bag prior to evacuating and sealing.
All bags were placed in a monolithic concrete dome (Butts et al., 2007) with only four points of entry: a removable door for personnel access, a 61-cm diameter capped vent in the top, a 8-cm diameter duct through the sidewall for aeration/ventilation, and a 4-cm diameter conduit through the sidewall with instrumentation cabling. Rodent bait stations were placed around the exterior perimeter of the dome and monitored monthly by a commercial pest control contractor.
The temperature and relative humidity in the dome were monitored using 13 thermistors and a single capacitive relative humidity sensor connected to a data logger (Model CR23X, Campbell Scientific, Logan, UT). The data logger monitored the temperature and humidity and controlled a small space heater to maintain the temperature between 18 and 27 C. A Bluetooth datalogger (HOBO MX1102A, Onset Computer Corporation, Bourne, MA) was placed inside one replication of the normal oleic, 8% moisture content treatments for each of the storage treatments to record temperature, relative humidity, and CO2 concentration. Temperature, humidity, and CO2 data were retrieved from inside the bags without opening the bags using the loggers’ Bluetooth communications.
Periodically, all bags were removed from the dome, weighed, opened, and a 2 to 3-kg sample removed from each bag. If a CO2 logger was in the bag, the CO2 sensor was re-calibrated using the logger’s self-calibration protocol, then replaced in the same bag. The bag was then resealed in the same manner as when originally filled. Approximately 100 g of peanuts were subsampled from the original sample for aflatoxin analysis and 100 g subsampled for germination. The aflatoxin sample was ground into small particles in a food blender. Then 10 g were analyzed using an ELISA test strip system (AccuScan Gold, Neogen, Lansing, MI). The germination sample was treated with Vitavax® PC seed treatment (Bayer CropScience, Research Triangle Park, NC) and sent to the Georgia Department of Agriculture’s Laboratory in Tifton, GA for germination analysis.
The remainder of the sample was used in a non-destructive moisture analysis (GAC 2100, Dickey-john, Auburn, IL) and forwarded to collaborators at USDA, ARS Market Quality and Handling Research Unit (MQHRU) in Raleigh, NC. The sample was analysed by MQHRU for percent free fatty acid and peroxide values. If the aflatoxin was below detectable limits and the samples were deemed edible by MQHRU staff, the sample was roasted, ground, and evaluated by a trained sensory panel using a 0-10 scale to rate the intensity of various sensory attributes .
Storage tests were begun 13 October 2017. Intermediate sampling occurred after 67 d (19 Dec 2017), 159 d (21 Mar 2018), and 249 d (19 Jun 2018) in storage. Tests were terminated after 301 d in storage on 10 Aug 2018.
Results and Discussion
Table 1 shows the overall analysis of variance for the dependent variables of moisture content, germination, aflatoxin, percent free fatty acid, and peroxide value as affected by fatty acid profile (Hi/Normal Oleic), initial moisture content, storage duration, and storage type.
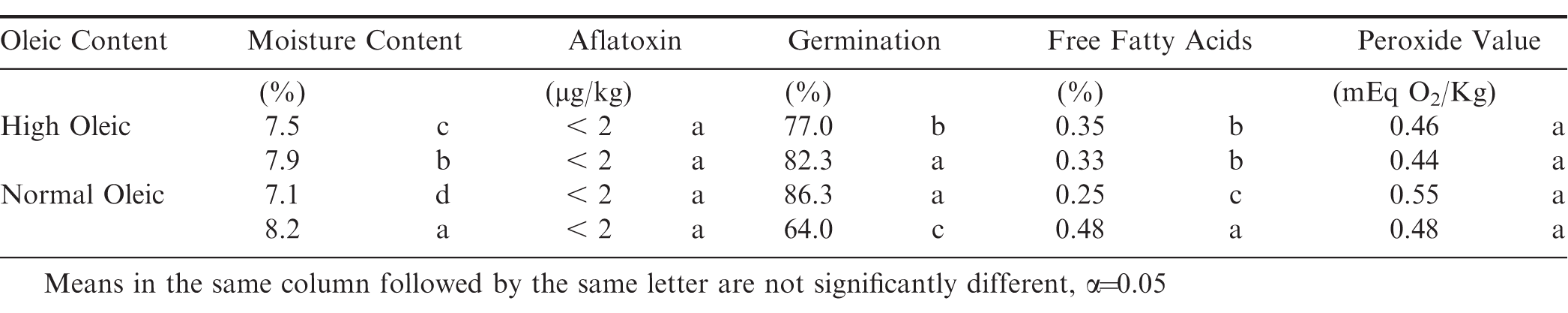
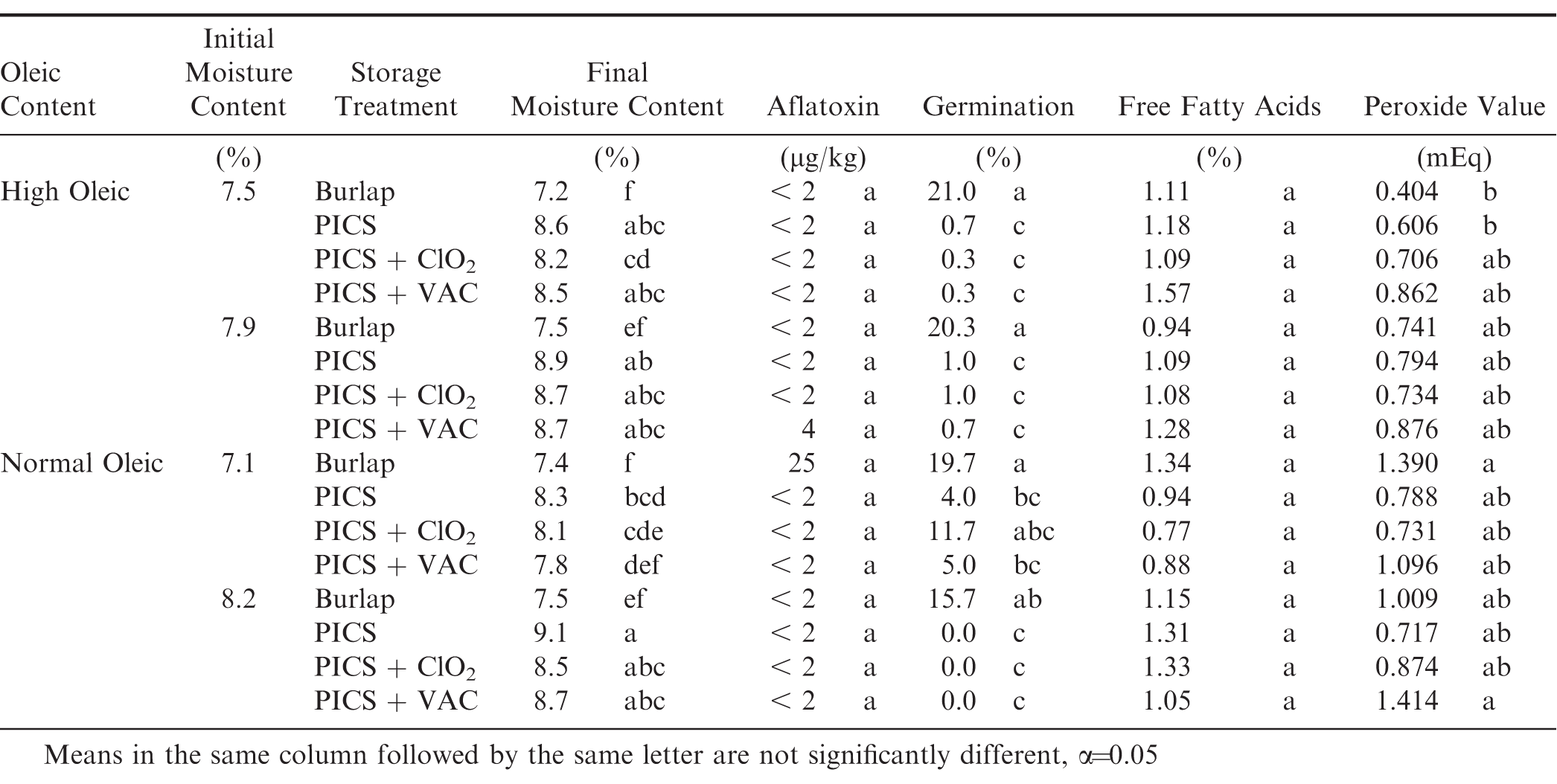
Fatty acid profile had no significant effect on any of the dependent variables tested. The initial conditions (Days in Storage = 0) of the peanuts when placed in the various storage bags are shown in Table 2. The significant difference in initial moisture content shows that having differing initial moisture contents was achieved. There was no detectable aflatoxin or peroxide values in the peanuts when initially placed in storage. There were significant differences in the initial germination and percent free fatty acid, therefore subsequent samples will be compared to the initial values for each initial moisture content. Initial percent free fatty acid was similarly affected by initial moisture content.
Storage Bag Integrity
Entry points into the monolithic domes were protected against intrusion by rodents and other pests. Both ends of the conduit containing the instrumentation wires were filled with 000 steel wool. The 10-cm fan outlet was covered with 6-mm mesh hardware cloth. Rodent bait stations were placed around the periphery of the dome and checked monthly by a professional pest control service. The dome was opened biweekly to download data from the four temperature/humidity dataloggers in the bags and the bags near the walls inspected. In spite of efforts to exclude rodents from the structure, at least one rodent apparently successfully gained entry and took up residence in the aeration duct on the floor. When the bags were removed from storage after 60 d for sampling, there was no evidence of physical damage to any of the bags. Both burlap and all PICS bags average 0.1 kg loss in mass during the first 60 d of storage. When the bags were taken from storage after 159 d of storage, the PICS bags had lost no additional weight, but the burlap bags had lost an additional 1.4 kg/bag. Approximately, 0.5 kg/bag can be attributed to moisture loss. There were two burlap bags with apparent rodent damage, and they lost 2.3 and 1.3 kg each. The remaining burlap bags’ weight loss was within the 0.5 kg loss expected due to moisture loss. After 249 d in storage, additional rodent damage was observed primarily in the burlap bags and four out of twelve bags had a spillage of more the 2 kg of peanuts due to rodent damage. Two out of 36 of the PICS bags had rodent damage losing 1.3 and 1.7 kg of each. After 301-d study, one PICS bag lost an additional 1 kg of peanuts due to rodent damage. No other bags suffered additional damage. At the completion of the study, the aeration ducts were removed and inspected. A single nest approximately 15-cm in length was found inside the aeration duct. The nesting material was predominantly strings of burlap with some of the white polypropylene from the outer liner of the PICS bags. None of the inner polyethylene bag material was found in the nest.
Temperature and Relative Humidity
A small space heater was controlled automatically to turn on when the temperature fell below 18 C and turn off when the temperature exceeded 27 C. The average temperature in the storage facility during the test was 23.9 C with a maximum of 33.7 C and a minimum of 9.2 C. Temperature remained within the desired 18 – 27 C range 56% of storage period. The temperature fell below the desired minimum of 18 C 7% of the storage period. It exceeded the desired maximum of 27 C 37% of the storage period and occurred from 11 May 2018 until the end of the test in August (Fig. 1). After approximately 210 d in storage, the ambient temperature gradually increased. Cooling was not available to prevent excessive temperatures.
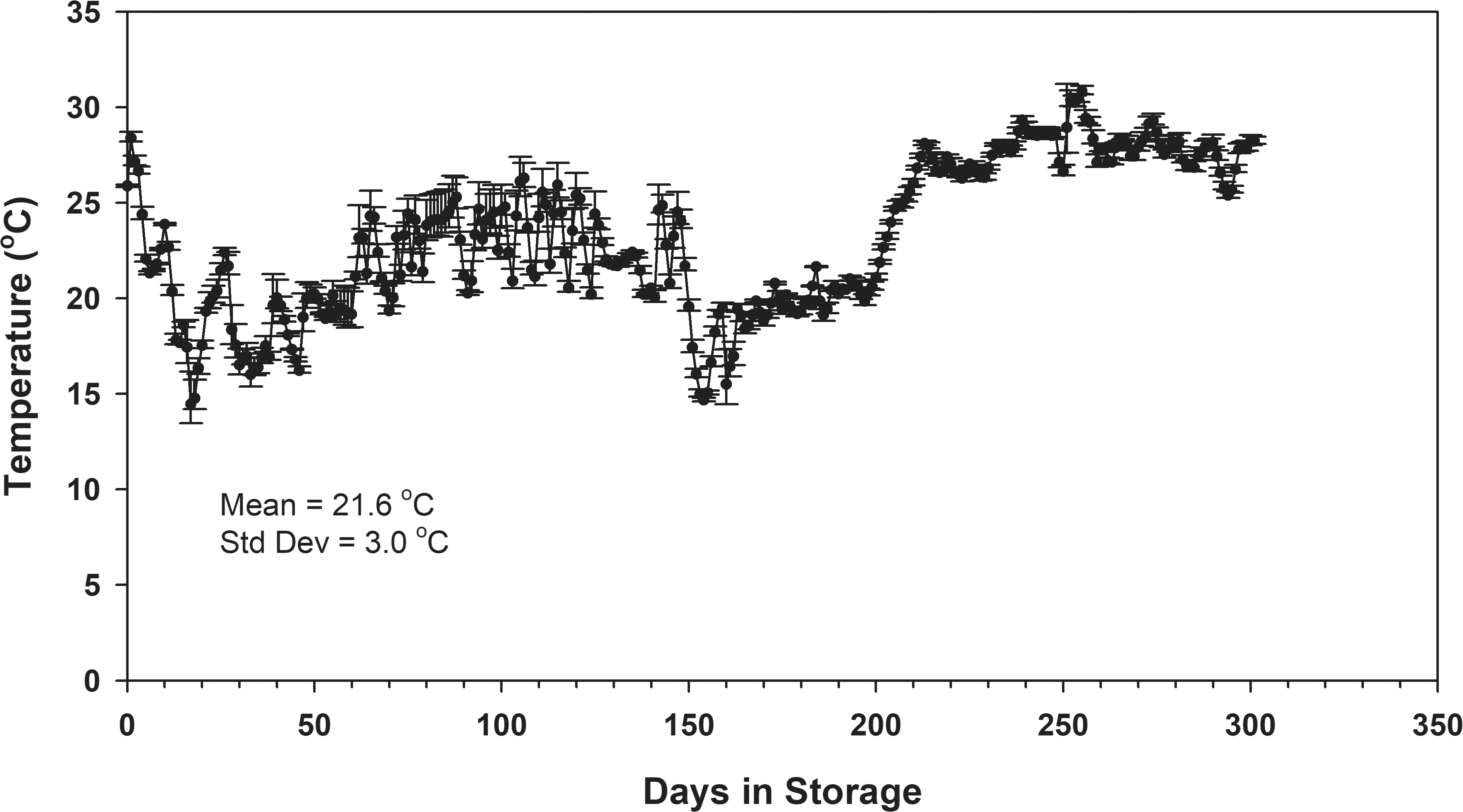
Maintaining an average relative humidity of 65% results in an equilibrium moisture content of 7.5% wet basis (ASABE, 2007). The relative humidity in the storage structure remained between 55 and 80% approximately 44% of the time (Fig. 2). The maximum desired relative humidity of 80% was exceeded approximately 4% of the time but was below the desired 55% minimum 53% of the storage period. The relative humidity in the storage structure averaged 49%. The relative humidity measured inside the three PICS storage treatments (PICS, PICS+VAC, PICS+ClO2) were not significantly different from one another and averaged 76%. The relative humidity recorded in the top of the burlap bag averaged 60% and was significantly different than that observed in the PICS treatments. The relative humidity in the burlap bag tended to follow the daily average relative humidity of the headspace in the storage structure. The minimum observed relative humidity in the burlap was 35% compared to 68% in the PICS treatments. The maximum observed relative humidity in the burlap and the PICS treatments was 82%.
Carbon Dioxide Concentration
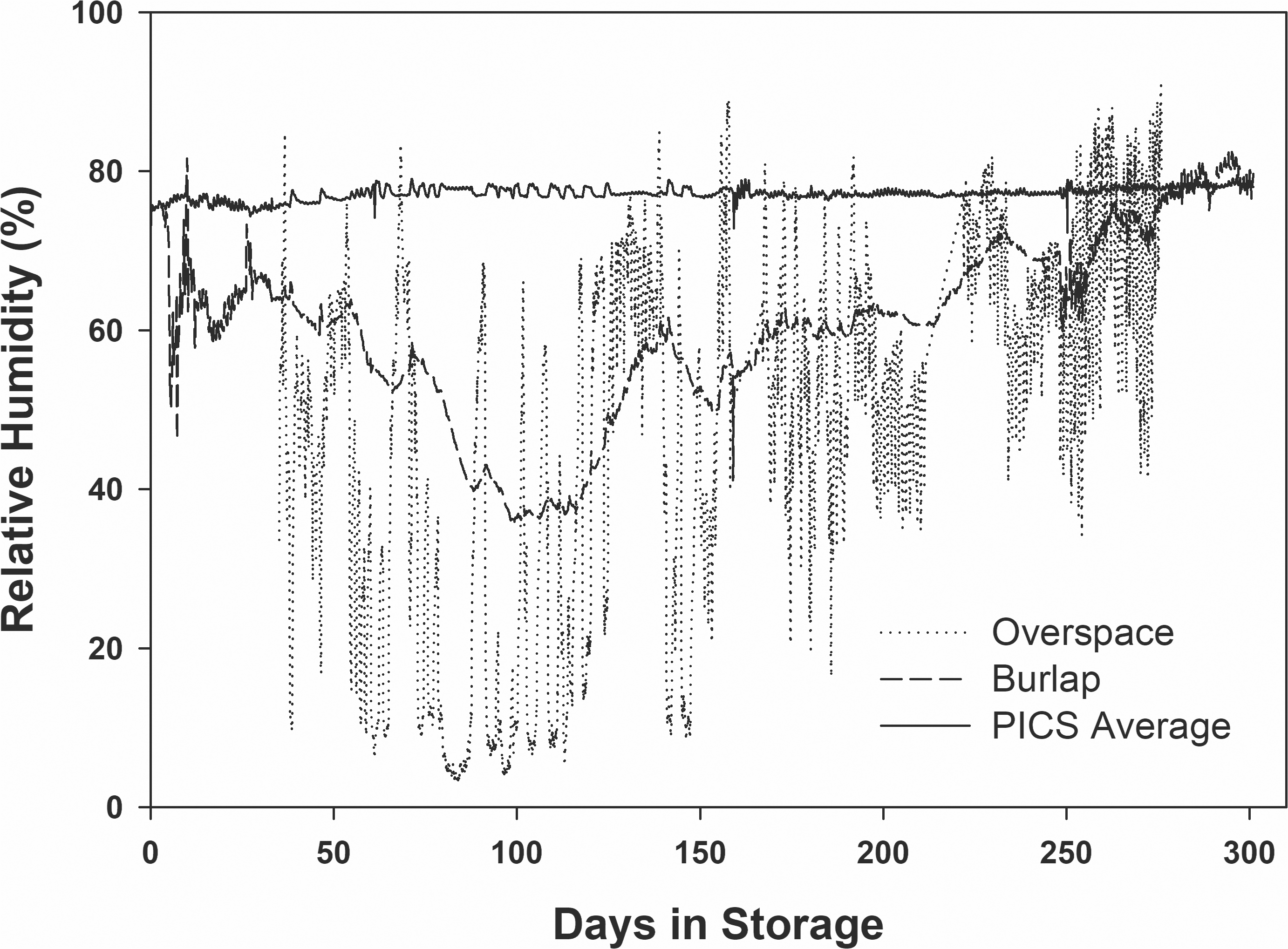
The premise behind the small-scale hermetic storage is that the respiration of the stored product, insects, and microflora reduces the oxygen concentration and increases carbon dioxide (CO2) causing asphyxiation of the insects and microflora. Fig. 3 shows the CO2 concentration (ppm) during the first ten days of storage. The CO2 concentration in the burlap bag remained at or slightly above the ambient CO2 of 400 ppm. The CO2 concentration in the three storage treatments using the PICS bags increased to maximum level measurable by the datalogger (5000 ppm) in five to six days and remained at that level throughout the study. The PICS + ClO2 took slightly more than 6 days to reach 5000 ppm compared to 5 days for the standard PICS storage treatment. This could have been due to the fumigant, ClO2, killing some of the insects, and reducing the overall respiration rate of the contents of the bags. However, this cannot be confirmed without replicated loggers to record CO2 concentrations. Another explanation is that degradation of ClO2 produces chloride and chlorate ions, but ultimately forms oxygen. Thus, presence of extra oxygen could delay the increase in the accumulation of CO2 (https://www.clordisys.com/faq.php).
In all PICS treatments, the CO2 levels remained near the 5000 ppm levels throughout the storage period. When the bags were opened for sampling, the dataloggers were removed and re-calibrated according to the self-calibration procedures prescribed in the owner’s manual, then replaced in the bag where the measured CO2 levels quickly returned to 5000 ppm. No live insects were found in any of the PICS storage treatments.
In comparison, the burlap bag had CO2 levels at or slightly above 400 ppm. This was expected due to the highly breathable characteristic of the burlap material. No quantitative data were recorded regarding the insect populations, however, adult Indian meal moth [Plodia interpunctella (Hübner)] were observed in apparently increasing numbers over time in the headspace of the storage facility. Unidentified insect larvae were observed in apparently increasing numbers over time in the peanuts stored in burlap bags.
Moisture Content
The moisture content of the peanuts within each oleic treatment was initially at two different levels when placed in the bags for storage (Table 2). The normal oleic and the high oleic peanuts had a high moisture level of 8.2 and 7.9%, respectively. The lower level initial moisture content in the high oleic peanuts was 7.5% compared to 7.1% in the normal oleic peanuts. The average initial moisture content for all storage treatments was 7.5%.
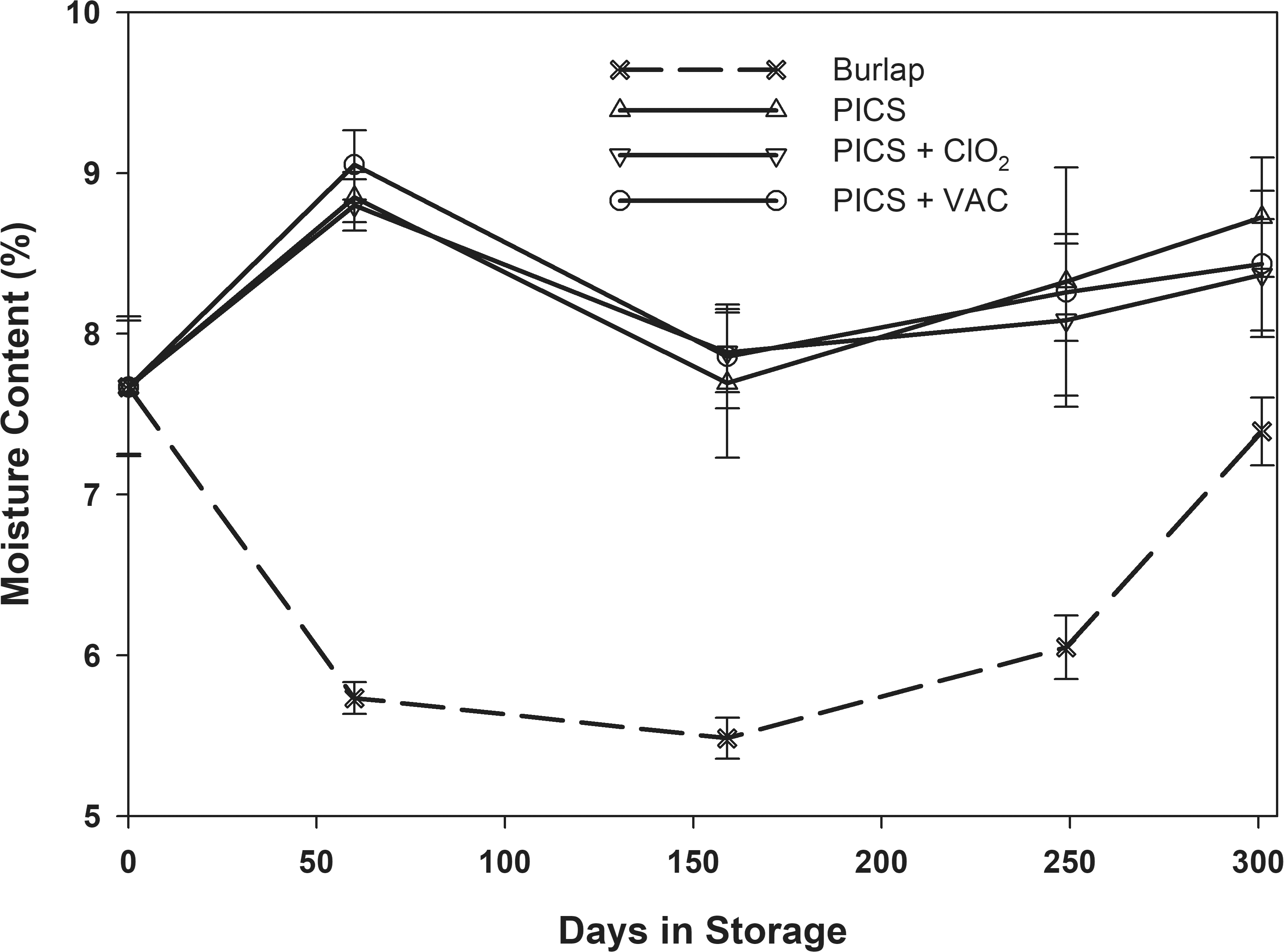
The average moisture content responded to the average relative humidity in the storage container. During the first 60-d of storage, the moisture content in the PICS bags increased to an average of 8.7% in response to the 75-80% relative humidity (Fig 2) and equilibrated to between 7.9 and 8.5% for the remainder of the storage tests (Fig 4). The peanuts in the burlap were exposed to the daily fluctuations in relative humidity in the storage structure which decreased to about 40% after 90-d storage (Fig 2). The moisture content in the seed decreased to approximately 5.5%. As the relative humidity increased, the moisture content followed eventually returning to approximately 7% at the end of the study. At the end of the study, there was no significant difference in the moisture content of the peanuts in the burlap bags due to initial moisture content (Table 3). In contrast, the peanuts stored in all PICS bag treatments increased from the average initial 7.7% to a final average moisture content more than 8% due to the elevated relative humidity maintained in the hermetic containers (Fig 4). There were differences among the final moisture contents in the PICS bag treatments with varying degrees of significance related to the initial moisture content of the peanuts when placed in the bags (Table 3). There was no difference in the final moisture content among the PICS bag treatments within the same oleic acid content and initial moisture content combination. For instance, in the Hi Oleic peanuts with initial moisture content of 7.5%, the final moisture contents of the PICS, PICS+ClO2, and PICS+VAC were 8.6, 8.2, and 8.5%, respectively. All were significantly higher than the 7.2% final moisture content of the peanuts stored in burlap for the oleic acid/initial moisture content combination.
After 159 d in storage, a powdery coating was observed on the peanuts stored in one (1) of the PICS bags and two (2) of the PICS + Vac bags (Fig 5). The powdery substance was identified as a mold (Arias et al, unpublished data; 2020) which propagates very slowly at relatively low water activity in vitro and apparently tolerates the hermetic conditions in the PICS storage bags. Mold was first observed in bags filled with higher initial moisture (8.2%) normal oleic peanuts. However, by the end of the 301-d storage period, the mold was observed on about half of the peanuts stored in PICS bags and seemed to occur at about the same rate in the normal and high oleic, both in high and low moisture peanuts. This mold was not observed on the peanuts stored in the burlap bags.
Peanut Sensory
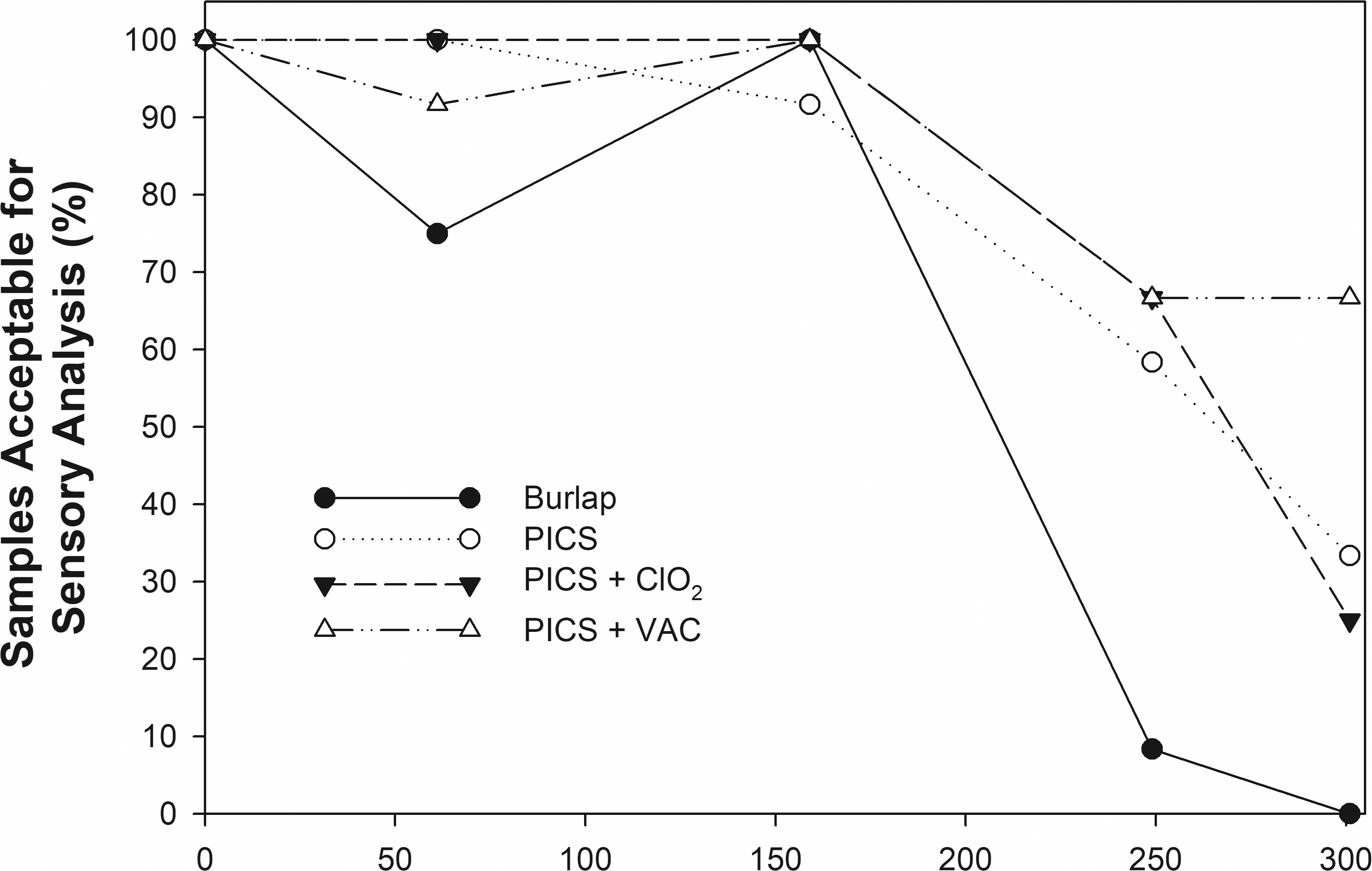
Each raw sample was inspected by the sensory panel administrator for excessive insect damage or discoloration. If the sensory panel administrator determined that the kernel condition was unacceptable for sensory analysis due to insect/rodent damage or mold, the sample was not presented to the sensory panel for analysis. Each sample was roasted for sensory analysis. After roasting, kernels unacceptable for sensory analysis were separated from the sample (Fig. 6). Fig. 7 shows the percentage of samples at each sampling date that had enough acceptable peanuts for sensory analysis. In general, at least 90% of the samples from any of the PICS storage treatments were acceptable for sensory analysis through the 159-d sampling date. Approximately 75% of the samples retrieved after 60-d storage in burlap were acceptable. Those rejected samples were due to advice from NPRL personnel that the samples had come from bags with rodent damage. After 159 d in storage, all the samples retrieved from burlap bags were acceptable for sensory analysis. At 250 d of storage, less than 10% of the peanuts in burlap were acceptable for sensory analysis due to insect infestation, and none of the burlap samples were acceptable for sensory analysis at the end of the study. The primary source of the damage in the burlap samples was insect damage.
After 301 d in storage, 42% of all the samples from the PICS bag treatments were acceptable for sensory analysis. The percent of acceptable samples ranged from 67% from the PICS+VAC storage treatment to 33% from the standard PICS treatment. The primary source of damage in the PICS treatments was discoloration after roasting due to mold growth described earlier.
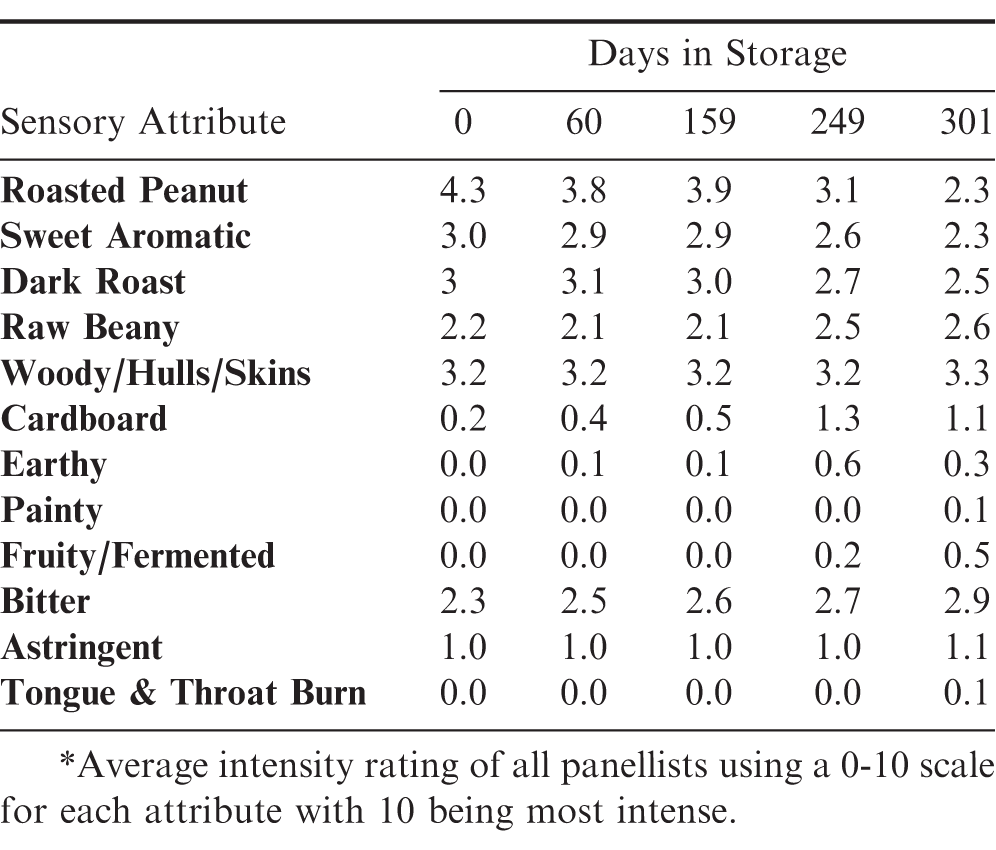
There were no significant changes in the intensity of the Roasted Peanut (RP) attribute in the samples tested due to oleic acid content or storage method. Changes were primarily due days in storage with a marginal effect due to initial moisture content. The average RP intensity was 4.2 initially and continually declined to an average intensity of 2.3 (Fig 8). Similar declines occurred in Sweet Aromatic and Dark Roast attributes (Table 4). There was a slight increase over time in the off-flavor attribute of Cardboard (Table 4). All other attributes remained relatively stable throughout the storage period.
Seed Germination
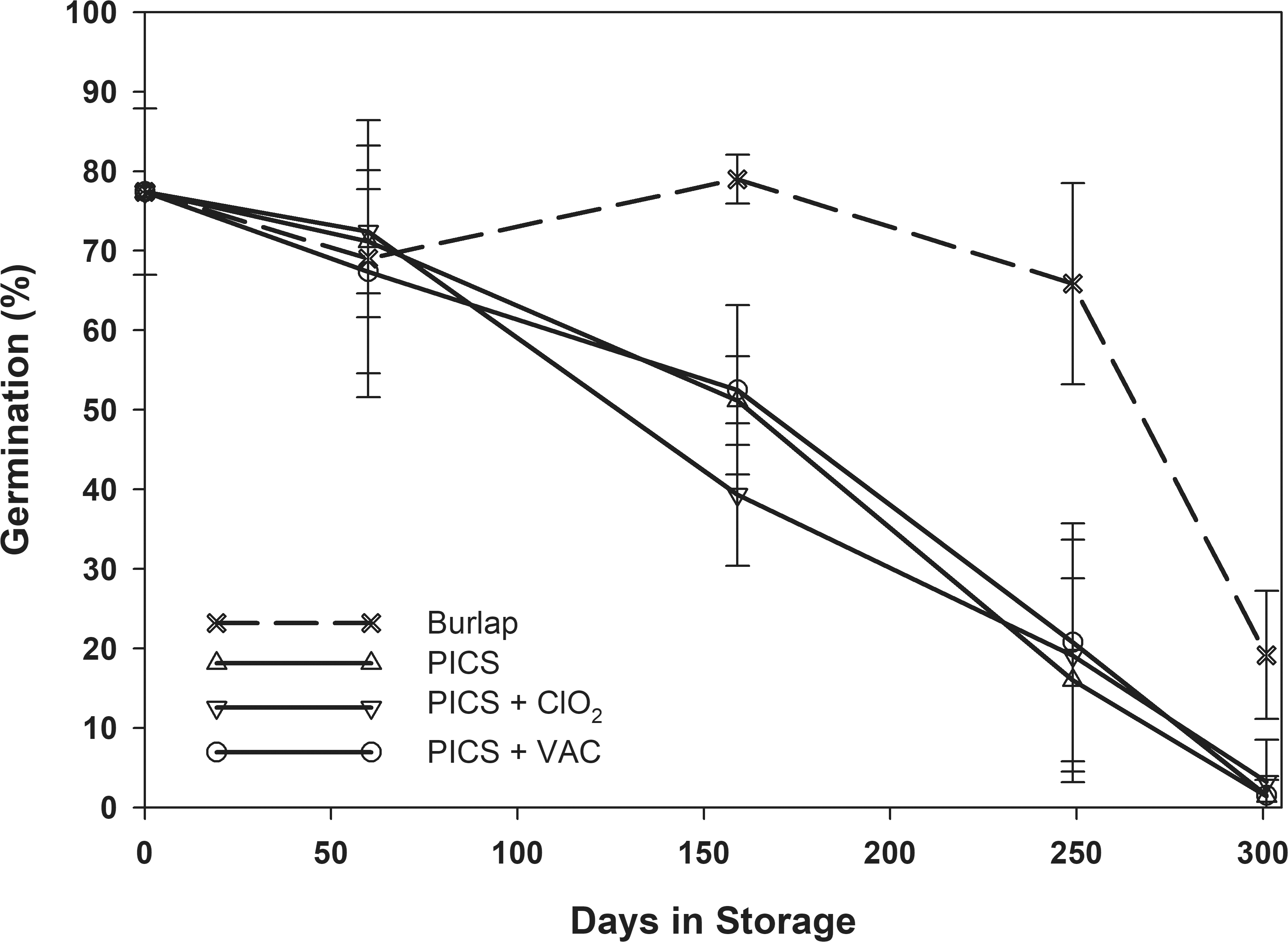
The initial germination rates varied from 64 to 86% (Table 2). The normal oleic, low moisture averaged 86% germination rate and was similar to the high oleic, high moisture peanuts at 82%. The high oleic, low moisture peanuts had a lower initial germination of 77% and the germination of the normal oleic high moisture was lower still at 64%. The analysis of variance showed that the initial moisture content, type of storage, and the time in storage all had significant effects on the seed germination (Table 1). After storing the peanuts for 301 d, the germination for all treatments decreased significantly compared to the initial condition (Table 3). All peanuts stored in burlap had final germination rates that were statistically similar regardless of initial moisture content or oleic acid content and ranged from 16 to 21%.
Averaging the germination rates for all initial moisture contents for each storage treatment and comparing to the germination rate of peanuts over time shows that the germination rate of peanuts stored in all PICS treatments continuously declined to negligible germination rates at the end of the 301-d storage period (Fig 9). This was caused by the continuous exposure of the peanuts to elevated relative humidity inside the PICS bags (Fig 2). However, the peanuts stored in the burlap had an average germination of at least 70% through 250 d of storage. After 250 d in storage, the temperature within the burlap bags exceeded 25 C for the remainder of the storage period and the average germination decreased to an average of 19% at the end of the study.
Aflatoxin Levels
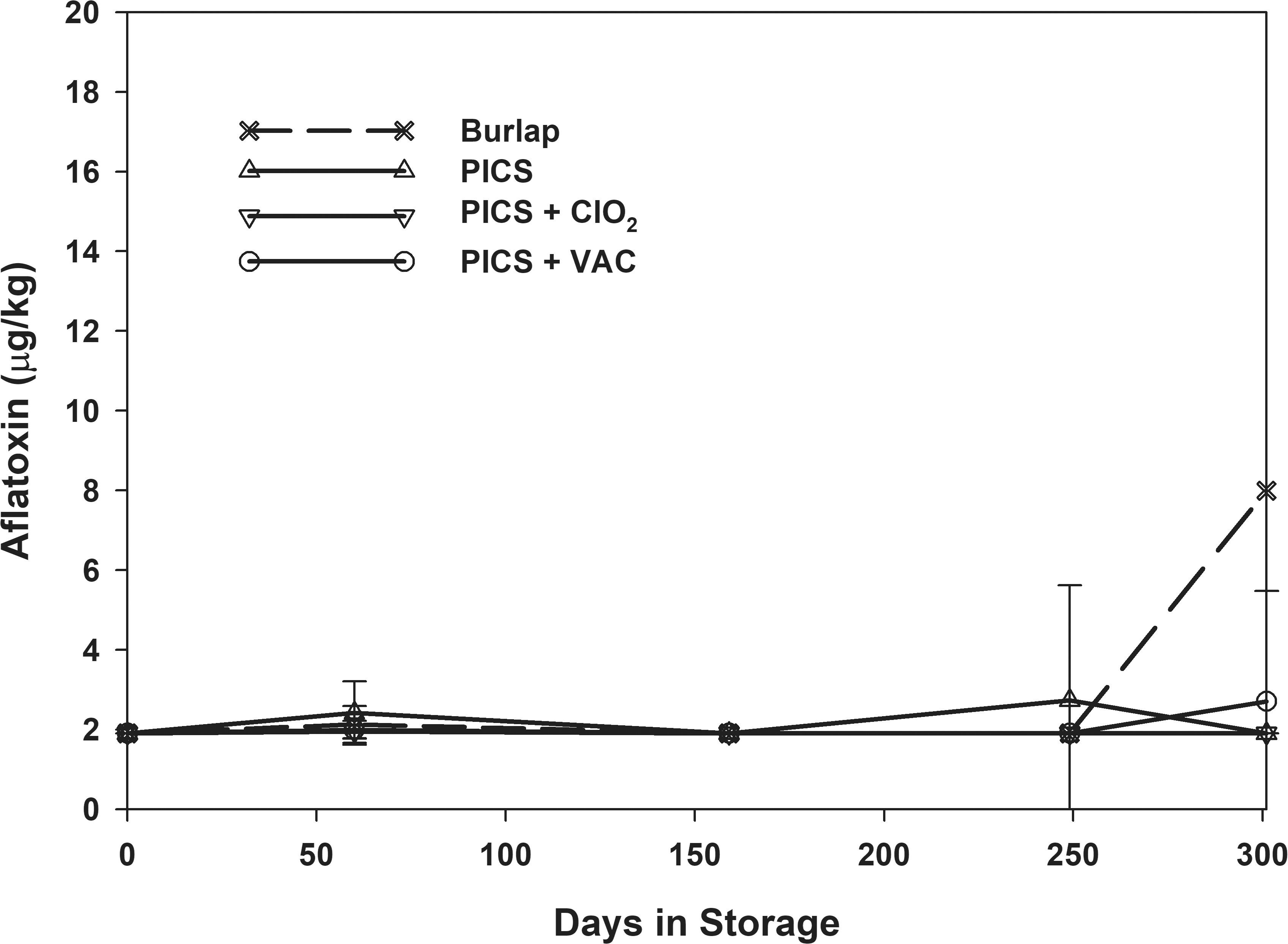
Aflatoxin levels were initially below detectable limits (< 2 μg/kg) in all samples (Table 2). Aflatoxin remained below detectable limits regardless of oleic acid content, initial moisture content and storage method through 250 d of storage. There were no significant differences in the final aflatoxin levels due to oleic content, initial moisture content, or storage method. However, the peanuts stored in burlap had an average aflatoxin level of 25 μg/kg; the high moisture, high oleic peanuts stored in the PICS + VAC treatment averaged 4 μg/kg; all other treatments had aflatoxin levels below detectable limits (Table 3). Fig. 9 shows the average aflatoxin contamination over time, and all samples remained below detectable limits until 250 d of storage. Between 250-d and the final sampling, the aflatoxin levels in the burlap increased to an average of 11 μg/kg (Fig. 10).
Peroxide Values
There were no significant differences in the initial values of peroxide values due to oleic acid content or initial moisture content (Table 2) and averaged 0.48 mEq O2/Kg. The peroxide value for the high oleic, low moisture peanuts stored in the burlap bag remained about the same and were 0.40 mEq after storage (Table 3). The normal oleic peanuts with the higher initial moisture content stored in the PICS + VAC had the highest final peroxide value of 1.41 mEq. The peroxide values for all other treatment combinations were between 0.4 and 1.4 mEq with no clear pattern due to oleic acid, initial moisture content, nor storage method (Table 3). There were significant differences in the mean percent free fatty acids due to oleic acid content and initial moisture content. The mean percent free fatty acids in the high oleic peanuts averaged 0.34% and no difference was observed due to initial moisture content. The mean percent fatty acids in the normal oleic peanuts averaged 0.25% in the low moisture sample compared to 0.48% in the high moisture samples. At the end of the study the percent free fatty acids ranged from 0.77% to 1.57%, but no significant difference in the final percent free fatty acids were found due to oleic acid content, initial moisture content or storage method.
Summary and Conclusions
Peanuts stored in the burlap bags suffered considerable loss due to insect and rodent damage, but maintained seed germination rates no lower than 70% for approximately 250 d. Moisture content equilibrated to the ambient conditions, decreased to approximately 5% during low humidity periods, and rehydrated to approximately 7% as relative humidity increased toward the end of the storage period. Aflatoxin remained low until the final sample date.
There were minimal differences among the hermetic storage treatments (PICS, PICS + ClO2, PICS + VAC). There were no losses due to insect infestation in any of the PICS treatments. There was some rodent damage to the PICS bags, but primarily to the outer protective woven polypropylene bag and not the inner polyethylene liners. Seed germination continuously decreased throughout the storage period with no seed germinating by the end of the study. Moisture content remained between 7 and 8% in all PICS treatments with the constant elevated relative humidity contributing to the decline in germination. A white, powdery mold was observed on peanuts in the PICS bags. Sensory decline was similar in both the PICS and burlap bags and no significant differences in attribute intensities was observed between the high and normal oleic peanuts in this study.
There was no benefit to using a small vacuum to evacuate the air from the PICS bags compared to manually squeezing the air from the inner liners. Nor was there any benefit to adding the sachets of chlorine dioxide to the PICS bags.
The primary conclusions from this study include:
-
Storing shelled peanuts in PICS bags or similar hermetically sealed containers may be beneficial to small stakeholder farmers if a primary concern for storage is to prevent losses due to insects and rodents.
-
Moisture content should be no higher than 7% for safe storage.
-
The PICS bags may not be suitable storage for the shelled peanuts if they are to be used for seed due to the serious decline in germination.
Acknowledgements
The authors gratefully acknowledge the provision of the PICS bags by the Feed the Future Peanut Innovation Lab and the motivation for conducting the study. We acknowledge the assistance provided by John Gardner, Terence Moon, and Dan Todd in filling and sampling the bags, and Valerie Orner for her assistance in the culture and identification of the mold found at the end of the study.
Literature Cited
Abudulai, M., Mahama, G, Dzomeku, I., Seidu, A., Sugri, I, Nboyine, J.A., Opoku, N., Alhassan, M.H., Appaw, W., Ellis, W.O., Akromah, R., Mochiah, M.B., Dankyi, A., Jordan, D.L., Brandenburg, R.L., Bravo-Ureta, B.E., Jelliffe, J., Boote, K., MacDonald, G., Chen, J., Phillips, R.D., Mallikarjunan, K., Balota, M., Hoisington, D., and Rhoads, J. (2020). Evaluation of Agricultural Practices to Increase Yield and Financial Return and Minimize Aflatoxin Contamination in Peanut in Northern Ghana. Peanut Sci. 47 (3): 156– 162. doi: 10.3146/PS20-15.1
ASABE. (2007). Moisture Relationships of Plant-based Agricultural Products. ASABE Standard D245.6.
Baoua, I. B., Amadou, L., Lowenberg-DeBoer, J. D., & Murdock, L. L. (2013). Side by Side Comparison of GrainPro and PICS Bags for Postharvest Preservation of Cowpea Grain in Niger. J. Stored Products Res. 54, 13– 16.
Baributsa, D., B, B. I., Bakoye, O. N., Amadou, L., & Murdock, L. L. (2017). PICS Bags Safely Store Unshelled and Shelled Groundnuts in Niger. J. Stored Products Res. 72, 54– 58.
Branch, W. (2007). Registration of ’Georgia-06G’ Peanut. J. Plant Registrations, 1 (2), 120– 120.
Branch, W. (2010). Registration of ’Georgia-09B’ Peanut. J. Plant Registrations, 4 (3), 175– 178.
Branch, W. (2014). Registration of ’Georgia-13M’ Peanut. J. Plant Registrations, 8 (3), 253– 256.
Butts, C., & Ward, J. (2016). Storing Peanuts in Grain Bags. 2016 Annual International Meeting of the ASABE. Paper No. 162456481, p. 16 pp. Orlando, FL: ASABE. doi:10.13031/aim.20162456481
Butts, C., Dorner, J., Lamb, M., & Sanders, T. (2007). Low Oxygen Storage of Farmer Stock Peanuts. 2007 Annual International Meeting of the ASABE. Paper No 076220, p. 12 pp. Minneapolis, MN: ASABE. doi:10.13031/2013.23530
Butts, C., Dorner, J., Lamb, M., Sorensen, R., & Sanders, T. (2008). Storing Peanuts in Flexible Hermetically Sealed Containers. 2008 Annual International Meeting of the ASABE. Paper No. 084198, p. 11 pp. Providence, RI: ASABE. doi:10.13031/2013.24766
Freitas, R. S., Faroni, L. R., & Sousa, A. H. (2016). Hermetic Storage for Control of Common Bean Weevil, Acanthoscelides Obetectus (Say). J. Stored Products Res. 66, 1– 5.
Ibro, G., Sorgho, M. C., Idris, A. A., Moussa, B., Baributsa, D., & Lowenber-DeBoer, J. (2014). Adoption of Cowpea Hermetic Storage by Women in Nigeria, Niger, and Burkina Faso. J. Stored Products Res. 58, 87– 96.
Johnsen, P. B., Civille, G. V., Vercellotti, J. R., Sanders, T. H., & Dus, C. A. (1988). Development of a Lexicon for the Description of Peanut Flavor. J. Sensory Studies, 3, 9– 17. doi:10.1111/j.1745-459X.1988.tb00426.x
Moussa, B., Addoulaye, T., Coulibaly, O., Baributsa, D., & Lowenberg-DeBoer, J. (2014). Adoption of On-Farm Hermetic Storage for Cowpea in West and Central Africa in 2012. J. Stored Products Res. 58, 77– 86.
Murdock, L. L., & Baoua, I. B. (2014). On Purdue Improved Cowpea Storage (PICS) Technology: Background, Mode of Action, Future Prospects. J. Stored Products Res. 58, 3– 11.
Murdock, L., Margam, V., Baoua, I., Balfe, S., & Shade, R. (2012). Death by Dessication:Effects of Hermetic Storage on Cowpea Bruchids. J. Stored Prod. Res. 49, 166– 170.
Norden, A. J., Lipscomb, R. W., & Carver, W. A. (1969). Registration of Florunner Peanuts (Reg. No. 2). Crop Sci, 9, 850. doi:10.2135/cropsci1969.0011183X000900060070x
Peanut & Mycotoxin Innovation Lab. (2018). Feed the Future Innovation Lab for Collaborative Research on Peanut Productivity and Mycotoxin Control: Final Report (31 July 2012 – 30 September 2017). Athens, GA: PMIL Management Entity, University of Georgia.
Purdue University. (2015). Purdue Improved Crop Storage: Videos - PICS Bag Demonstration. Retrieved Feb 07, 2020, from Purdue Improved Crop Storage: https://picsnetwork.org/resources/?tab_id=Videos
Slay, W. O., Ferguson, W. G., & Pomplin, J. A. (1985). Some Effects of Conventional and Low-Oxygen Atmosphere Storage and Processing Methods on Florunner Peanut Seed. Peanut Sci. 12 (1), 8– 11. doi:10.3146/pnut.12.1.0003
Sudini, H., Ranga Rao, G. V., Gowda, C. L., Chandrika, R., Margam, V., Rathore, A., & Murdock, L. L. (2015). Purdue Improved Crop Storage (PICS) Bags for Safe Storage of Groundnuts. J. Stored Products Res. 64, 133– 138.
USDA (2004). Milled Peanuts (Shelled Peanuts and Cleaned Virginia Type in the Shell) Inspection Instructions. USDA, Agricultural Marketing Service, Fruit and Vegetable Division. Washington, DC.
Villers, P., de Bruin, T., & Navarro, S. (2006). Development and Applications of the Hermetic Storage Technology. Alternative Methods to Chemical Control. PS 7-8-6119, pp. 719– 729. Brazil: 9th International Working Conference on Stored Product Protection. Retrieved from https://pdfs.semanticscholar.org/b255/2771179795b63b77796000d15d2a256f76df.pdf
Walker, S., Jaime, R., Kagot, V., & Probst, C. (2018). Comparative effects of hermetic and traditional Storage Devices on Maize Grain: Mycotoxin Development, Insect Infestation, and Grain Quality. J. Stored Products Res. 77, 34– 44.
Williams, E. J. & Drexler, J. S. (1981). A Non-Destructive Method for Determining Peanut Pod Maturity. Peanut Sci. 8 (2): 134– 141. doi:10.3146/i0095-3679-8-2-15
Williams, S. B., Baributsa, D., & Woloshuk, C. (2014). Assessing Purdue Improved Crop Storage (PICS) Bags to Mitigate Fungal Growth and Aflatoxin Contamination. J. Stored Products Res. 59, 190– 196.
Notes
-
1Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer
Author Affiliations
2First, fourth, fifth and sixth authors are Research Agricultural Engineer, Research Plant Pathologist, Research Agronomist, Supervisory Research Food Technologist, USDA ARS National Peanut Research Laboratory, Dawson, GA 39842; Second and third authors are Food Technologists, USDA ARS Food Science and Market Quality and Handling Research Unit, Raleigh, NC 27695 *Corresponding author Email: Chris.Butts@usda.gov