Introduction
Peanut (Arachis hypogaea L.) production in the mid-south of the US has gradually increased in recent years. Peanut provides a complimentary crop rotation species for cotton (Gossypium hirsutum L.) on more coarse textured soils in place of other crops with lower commodity value (Johnson III et al. 2001). Tobacco thrips [Frankliniella fusca (Hinds)] is a common early season pest of peanut. However, no treatment thresholds for foliar sprays have been established for this pest and those listed are generally vague. As an example, foliar sprays should be considered in Virginia when 25% of leaves are damaged prior to mid-June and live thrips are present (Linker et al. 2020). Historically, foliar insecticide applications for thrips management have shown conflicting results and generally have not been considered economical (Tappan and Gorbet 1981). While suppressing thrips populations with insecticides may not be necessary under normal conditions, it may be important when additional stress occurs from other sources such as herbicide injury (Murdock et al. 1986; Brecke et al. 1996).
At-planting soil insecticides are the primary control option for managing thrips in peanut. Previous research has shown that in-furrow treatments can reduce first generation thrips larvae by 70-95%, resulting in less feeding injury (Smith and Sams 1977; Tappan and Gorbet 1981; Lynch et al. 1984; Chamberlin et al. 1992). Thrips are also known to transmit tomato spotted wilt virus (TSWV), an important disease of peanut. At least eight species of thrips have been found to transmit this disease (Mound 2002; Herbert et al. 2007) with tobacco thrips being the most predominant species because it reproduces more efficiently on peanut foliage (Todd et al. 1994, 1995, 1996; Shrestha et al. 2013). Use of in-furrow insecticides can reduce the incidence of TSWV (Herbert et al. 2007). Also, foliar applications of acephate reduced plant injury from thrips and incidence of TSWV in that study.
Herbicide resistant weeds are an increasing problem in all crops. Currently, 23 different weed species in the mid-south US are resistant to certain classes of herbicides (Heap 2020). An effective weed management program is crucial due to the growth habit of peanut. Peanut produces a low spreading canopy that grows slowly outward resulting in a longer period for canopy closure to occur. This delay in canopy closure allows weeds to continually germinate, emerge, and grow during the early part of the growing season. Therefore, implementing a preemergence herbicide program with residual herbicides is critical for managing herbicide resistant weeds. Flumioxazin (Valor® SX, Valent U.S.A. Corporation, Walnut Creek, CA) is a popular preemergence herbicide used in peanut production with less documented herbicide resistant species than other preemergence herbicides. Flumioxazin is an herbicide that inhibits protoporphyrinogen oxidase in susceptible plant species (Anonymous 1988; Cranmer et al. 2000; Price et al. 2004), but can cause phytotoxicity to tolerant plants when applied at the labeled rate under certain conditions. These conditions inlcude high soil moisture or when heavy rainfall splashes treated soil onto emerged plant tissues (Taylor-Lovell et al. 2002, Main et al. 2003). An example of this injury is bronzing and burning of leaves, which is more severe after a heavy rainfall event following a preemergence application. This type of herbicide injury can slow seedling development and increase plant susceptibility to other stresses, such as injury from thrips and other early season pests. The combination of thrips injury, herbicide injury, and moist soils has been observed to delay pod development and reduce yield in peanut (Brecke et al. 1996; Funderburk et al. 1998; Drake et al. 2009). The current experiment was conducted to evaluate the impact of at-planting insecticides and foliar applications of acephate for thrips management on flumioxazin injured peanut and uninjured peanut.
Materials and methods
An experiment was conducted at the Delta Research and Extension Center in Stoneville, MS in 2016 and 2017. The soil at this location consisted of a Beulah very fine sandy loam (Coarse-loamy, mixed, active thermic Typic Dystrudepts, USDA-NRCS, 2019) that consisted of 64% sand, 26% silt, and 10% clay. The pH was 6.3 with 1.0% organic matter. A similar, but separate experiment was conducted at the R.R. Foil Plant Science Research Center in Starkville, MS during those same years. The soil at this location consisted of a Marietta fine sandy loam that consisted of 71% sand, 17% silt, and 11% clay (Fine-loamy, siliceous, active, thermic Fluvaquentic Eutrudepts, USDA-NRCS 2000). The pH was 6.6 with 0.8% organic matter.
Stoneville, MS Experimental Design
Treatments were arranged as a split-split-plot within a randomized complete block design with four replications. The main-plot, sub-plot, and sub-sub-plot factors included flood irrigation at two levels, herbicide application at two levels, and thrips management at four levels, respectively. The main plot factor of flooding included flooded and not flooded. The flooding factor was included to simulate a heavy rainfall event that would cause water ponding and maximize herbicide injury. Flooding treatments were imposed within two d after peanut emergence. Levees were erected around the exterior of the flooded plots with an implement commonly used in rice production (levee plow). Smaller levees were also constructed in the alleys between each replication to ensure sufficient flooding. Surface water was pumped from a nearby creek through 30.5-cm diam. poly-ethylene tubing. Water was held on plots in each replication for 30 minutes to maximize injury. Replications were flooded individually before breaching the levee between replications. This allowed water to flow into plots of the next replication until all were flooded separately. One week after flooding, levees were removed. The sub-plot factor of herbicide included a preemergence application of flumioxazin at a rate of 0.163 kg ai/ha or no flumioxazin. Flumioxazin (Valor® EZ, Valent U.S.A., LLC, Walnut Creek, CA) was applied with a tractor mounted sprayer with a compressed air system calibrated to deliver 140 L ha−1 at 296 kPa through 8004 flat fan nozzles immediately after planting. The sub-sub-plot factor of thrips management included imidacloprid (Admire® Pro, Bayer CropScience, Raleigh, NC) sprayed into the open seed furrow at 0.36 g ai ha−1 at-planting, one foliar application of acephate (Acephate 90 WDG, Loveland Products, Loveland, CO) at seven d after flooding, two foliar applications of acephate at 7 and 14 d after flooding, and an untreated control. The in-furrow imidacloprid treatment was applied with a nozzle attached to the planter positioned over each seed furrow immediately in front of the closing wheels. The application volume was 46.7 L ha−1 at 138 kPa through an 8002 flat fan nozzle. The flat fan nozzle was oriented parallel to the open seed furrow. This resulted in the entire spray pattern being directed into the open seed furrow to insure optimum coverage of the seed furrow and the seed. All foliar applications of acephate were made at a rate of 0.55 kg ai/ha. Foliar applications were made with a self-propelled sprayer (MudMasterTM, Bowman Manufacturing, Newport, AR) with a compressed air system calibrated to deliver 93.3 L ha−1 through TX-6 hollow cone nozzles at 241 kPa.
The size of each sub-sub-plot was four rows (102-cm row spacing) that were 12.2-m in length separated by 3.05-m unplanted alleys. Plots were planted on 06 May, 2016 and 10 May, 2017 at a seeding rate of 21 seeds per row m. The cultivar used for this experiment was Georgia-06G (Birdsong Peanuts, Suffolk, VA). A peanut inoculant (Bradyrhizobium sp., Primo Power CL, Verdesian Life Sciences, Cary, NC) was applied in-furrow to all plots at the time of planting at a rate of 0.95 L product ha−1. All plots received preemergence applications of S-metolachlor (Dual Magnum, Syngenta Crop Protection, Greensboro, NC) and diclosulam (Strongarm, Dow AgroSciences, Indianapolis, IN) at rates of 1.37 kg ai ha−1 and 26.4 g ai ha−1, respectively, to minimize weed infestations.
Starkville, MS Experimental Design
Treatments for the experiment in Starkville were similar to Stoneville, except they were arranged as a split-plot within a randomized complete block design with four replications. The flooding factor was not included at this location because a levee plow was not available. The main plot factor was herbicide application at two levels. This included an application of flumioxazin at a rate of 0.163 kg ai ha−1 or no flumioxazin. Because the flooding factor was not included in this study, flumioxazin was applied when plants began to emerge instead of preemergence in order to maximize herbicide injury. The sub-plot factor was thrips management at four levels identical to the treatments of the Stoneville experiment. The size of each sub-plot was four rows (96.5 cm row spacing) 9.14 m in length separated by 3.05 m unplanted alleys. Plots were planted 06 May 2016 and 11 May 2017 with the same cultivar and seeding rate used in the Stoneville experiment.
Experimental Measurements at Both Locations
Thrips populations were sampled 10 and 17 d after plant emergence (8 and 15 d after flooding in Stoneville) by cutting five random plants at the soil surface from each plot and placing them into 0.95-L self-sealing plastic bags (Ziploc®, S. C. Johnson & Son, Inc., Racine, WI). These timings corresponded to one day after the acephate foliar applications made at 7 and 14 d after flooding, respectively. Plants were washed using a whole plant method developed for cotton (Burris et al. 1990). Plants were soaked in a bleach and soapy water solution in the bags. Plants were placed on a 2.0-mm sieve set atop a funnel with a smaller 45.0-μm sieve underneath. Plants were washed with water to remove insects from plants onto the smaller sieve. The contents of the smaller sieve were then transferred onto a 9.0-cm diam ruled filter paper. Remaining residue from the sieve was washed with a 70% ethanol solution onto the filter paper. Excess ethanol was drained from the filter paper using a Buchner funnel and vacuum pump. The filter paper was then examined under a microscope for number of adult and immature thrips.
Thrips injury was estimated two d after the second insecticide application by visually examining the center two rows of each plot. These ratings were adapted from thrips injury ratings used on cotton (North et al. 2019) and based on a scale from one to five with one being no injury to the plants and five being severely injured plants with almost complete necrosis of plant tissue. At the Stoneville location, herbicide injury was estimated based on chlorosis, necrosis, and stunting at 30 d after planting in 2016 and 25 d after planting in 2017 to determine the effect of treatments on plant growth. These ratings were based on a scale from 1 to 5 with 1 being no injury and 5 being severe injury with dead plants. Herbicide injury was not rated in the trial in Starkville.
Plant biomass was recorded 34 d after planting by randomly removing five plants from each plot and placing them in paper bags. Paper bags were placed in a greenhouse and allowed to air dry for two weeks. Upon drying, plants were weighed on an analytical laboratory balance (Mettler-Toledo AL54, Mettler-Toledo, LLC, Columbus, OH). Width between plant canopies was measured 45 and 60 d after planting. This was defined as the width between the outermost vines of plants on rows two and three. Five locations per plot were measured by placing a m stick between the vines on these two rows and then averaged.
Digging dates were determined by the hull scape maturity profile method to determine plant maturity (Williams and Drexler 1981). At maturity, plants were inverted with a two-row KMC digger-shaker-inverter (Kelley Manufacturing, Tifton, GA) and harvested with a two-row KMC peanut combine with a bagging attachment. In Stoneville, plots were harvested 153 d after planting in 2016 and 139 d after planting in 2017. In Starkville, plots were harvested 145 d after planting in 2016 and 147 d after planting in 2017. Bags were weighed in pounds using a digital hanging scale (Model CS200, Intercomp, Medina, MN).
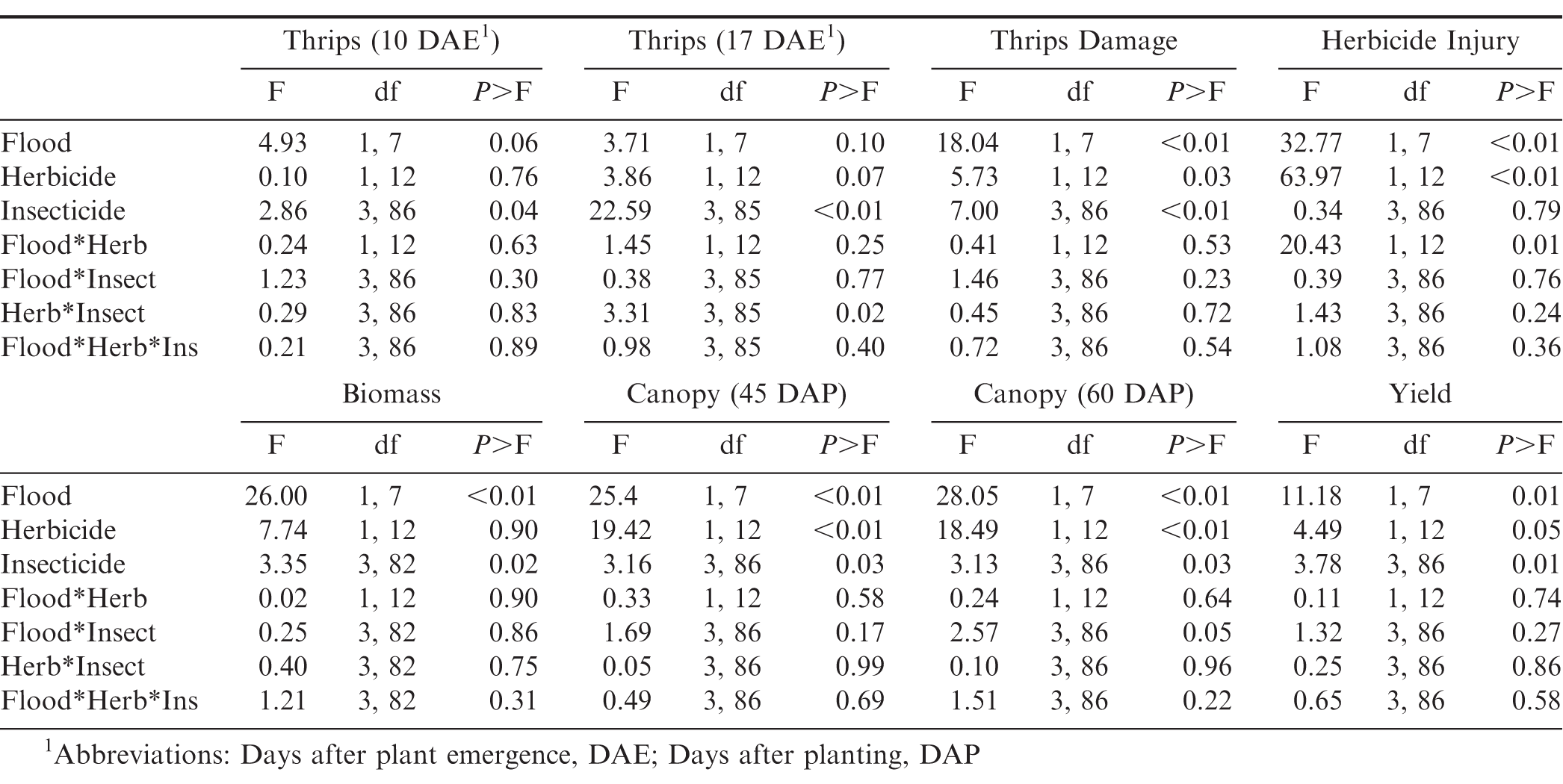
Combined thrips counts (adults plus immatures), thrips injury ratings, herbicide injury ratings, plant biomass, width between canopies, and yield data were analyzed with a mixed model analysis of variance (PROC GLIMMIX, SAS 9.4, SAS Institute Inc. Cary, NC). Flooding (Stoneville only), herbicide, insecticide, and their interactions were considered fixed effects in the model. Locations were analyzed separately because the experimental arrangement was different at each location. Year, replication nested in year, and replication by herbicide nested in year were considered random effects in the model. Degrees of freedom were calculated using the Kenward-Roger method. Means and standard errors were calculated with PROC MEANS. Means were separated using LSMEANS and adjusted using the Tukey method for separation. Differences were considered significant at α=0.05.
Results
Stoneville, MS Experiment
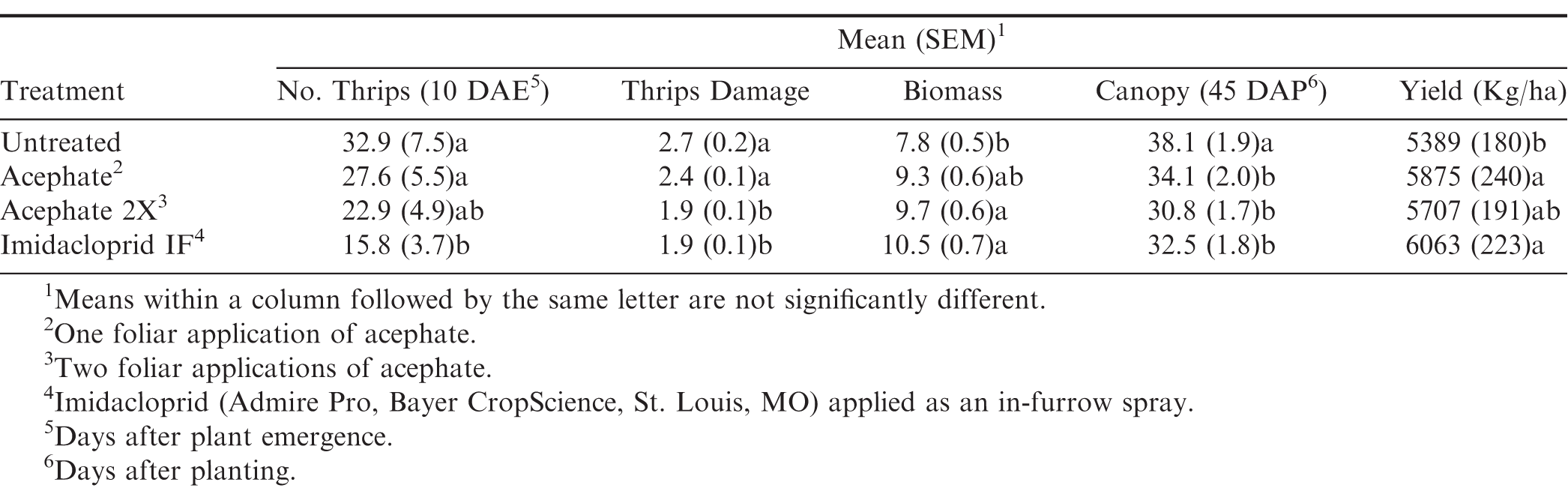
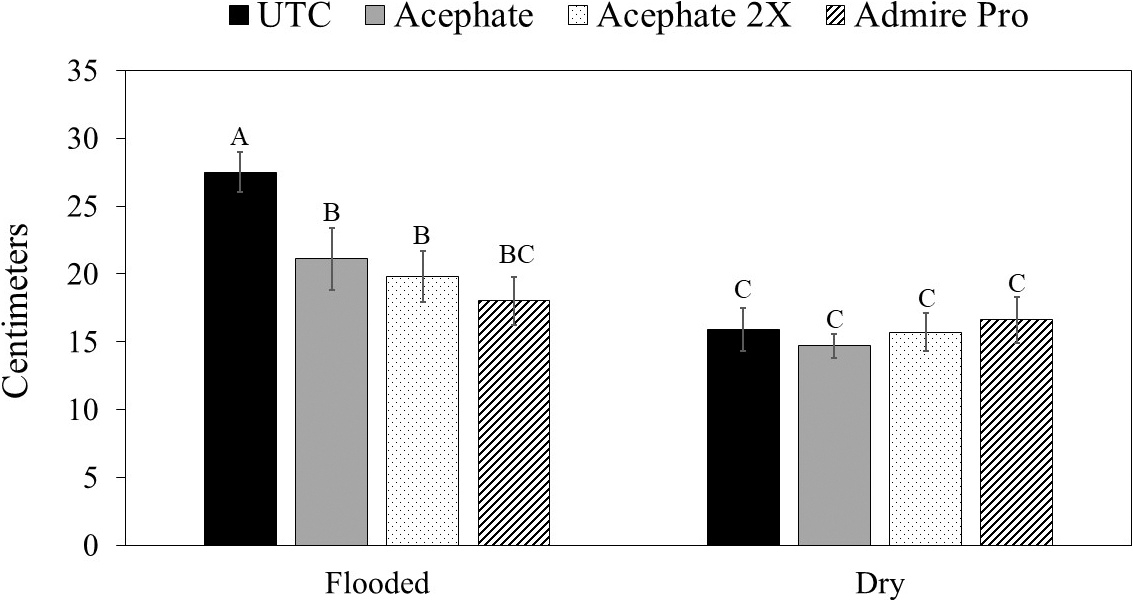
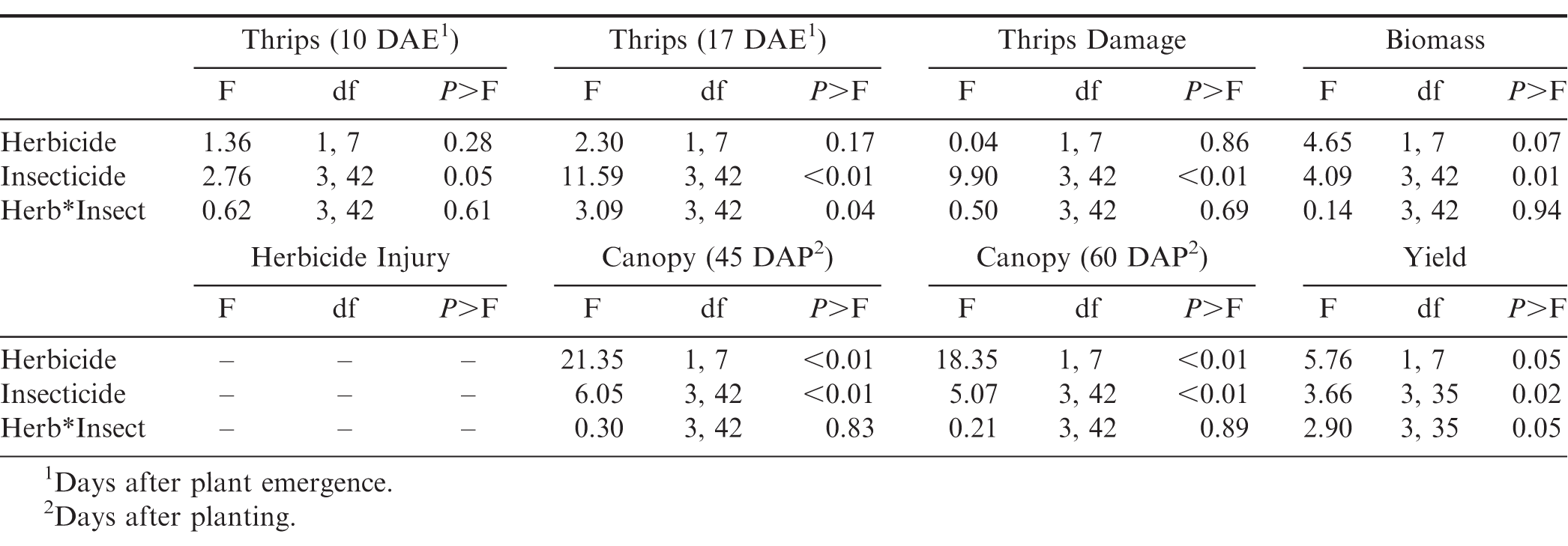
There was no interaction between flooding, herbicide, and insecticide for any of the variables measured (Table 1). For thrips numbers at the first sample date, the only factor that impacted thrips numbers was the main effect of insecticide (Table 1). Imidacloprid applied in the seed furrow at-planting was the only treatment that reduced thrips numbers below that in the untreated control (Table 2). For the second sample date, there was an herbicide by insecticide interaction for thrips numbers (Table 1). The untreated control (no insecticide) where flumioxazin was not applied at planting had more thrips than all other treatment combinations including the untreated control where flumioxazin was applied (Fig. 1). All of the insecticide treatments reduced thrips numbers below that in the untreated control where flumioxazin was not applied at planting. In contrast, only the treatments where two foliar applications of acephate were made or where imidacloprid was applied in-furrow reduced thrips numbers below that in the untreated control (Fig. 1). For thrips injury, there were main effects for flood, herbicide, and insecticide (Table 1). Peanut that experienced temporary flooded conditions (2.55 ± 0.10) had a higher thrips injury rating than peanut that did not experience flooded conditions (1.95 ± 0.10). Thrips injury ratings were also greater for peanut where flumioxazin was applied preemerge (2.39 ± 0.11) than where peanut was grown without the use of flumioxazin (2.11 ± 0.11). Thrips injury ratings were lower on peanut where two foliar applications of acephate were made or where imidacloprid was applied in-furrow compared to peanut that received one foliar application of acephate or the untreated control (Table 2). Herbicide injury was impacted by the interaction between flood and herbicide (Table 1). Peanut that was grown under flooded conditions and where flumioxazin was applied preemergent (3.00 ± 0.15) was greater than all other treatments. Peanut that was grown under dry conditions and where flumioxazin was applied preemergent (1.61 ± 0.11) had more herbicide injury than where peanut was grown under dry conditions and flumioxazin was not applied (1.21 ± 0.12), but not where peanut was grown under flooded conditions and flumioxazin was not applied (1.50 ± 0.09).
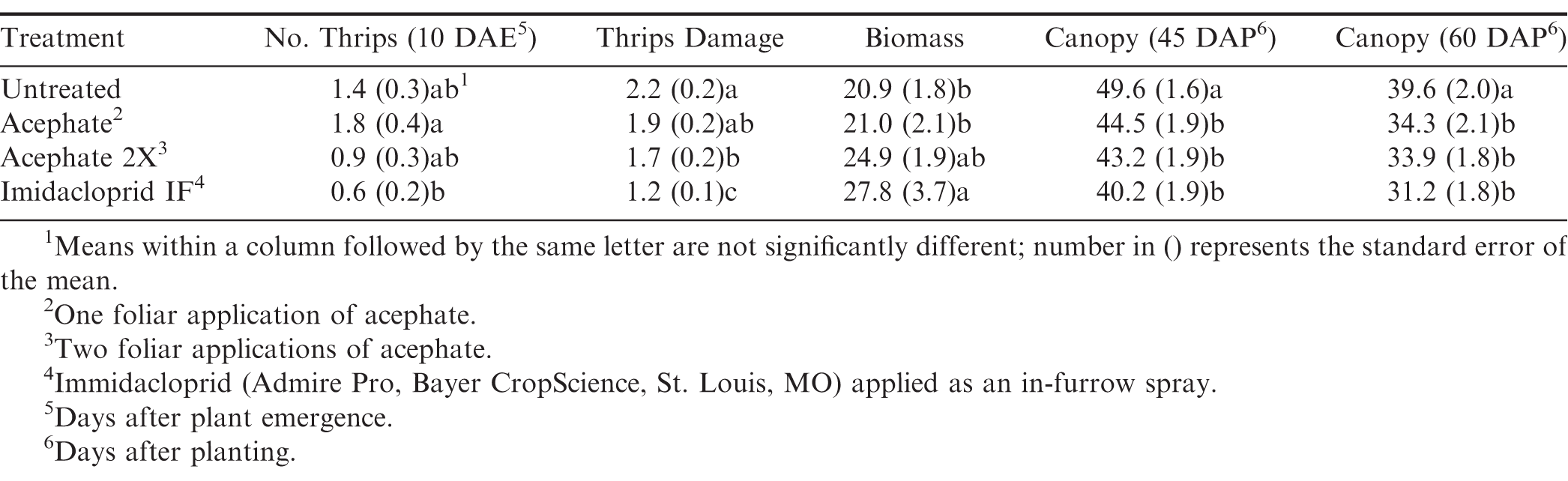
In terms of plant growth, flood and insecticide had an impact on plant biomass (Table 1). Peanut grown under temporary flood conditions (7.91 ± 0.33 g) had less biomass than peanut grown under dry conditions (10.84 ± 0.46 g). Biomass of peanut was greater where two foliar applications of acephate were made or where imidacloprid was applied in the seed furrow at-planting compared to the untreated control (Table 2). Flood, herbicide, and insecticide each had an effect on distance between vines on adjacent rows at 45 d after planting (Table 1). The distance between vines was greater for peanut grown under temporary flood conditions (38.3 ± 1.4 cm) than peanut grown under dry conditions (29.5 ± 1.1 cm). Similarly, the distance between vines for peanut grown where flumioxazin was applied preemergent (36.0 ± 1.3 cm) was greater than where flumioxazin was not applied (31.8 ± 1.4 cm). All of the insecticide treatments resulted in greater peanut growth with shorter distances between vines at 45 d after planting compared to the untreated control (Table 2). At 60 d after planting, there was a significant effect of herbicide and a flood by insecticide interaction for distance between vines on adjacent rows (Table 1). Similar to at 45 d after planting, the distance between vines for peanut grown where flumioxazin was applied preemerge (20.7 ± 0.9 cm) was greater than where flumioxazin was not applied (17.0 ± 1.0 cm). Under flooded conditions, the distance between vines on adjacent rows was greater for the untreated control than all insecticide treatments (Fig. 2). In contrast, no differences were observed for distance between vines for any of the insecticide treatments or the untreated control under dry conditions. The distance between vines was greater for both acephate treatments under temporary flood conditions compared to both acephate treatments under dry conditions, respectively (Fig. 2). In contrast, the distance between vines was similar for peanut where imidacloprid was applied in the seed furrow at planting for both temporary flood and dry conditions.
There was a significant effect of flood, herbicide, and insecticide on peanut pod yields (Table 1). Pod yield was greater when grown under dry conditions (5812 ± 132 kg ha−1) in comparison to temporary flood conditions (5728 ± 175 kg ha−1). In terms of herbicide effect, pod yield was reduced where flumioxazin was used preemerge (5664 ± 140 kg ha−1) compared to where flumioxazin was not used (5888 ± 166 kg ha−1). For the insecticide effects, plants not treated for thrips (nntreated control) had lower pod yield than plants sprayed one time with acephate or plants grown with imidacloprid applied in the seed furrow at planting (Table 2).
Starkville, MS
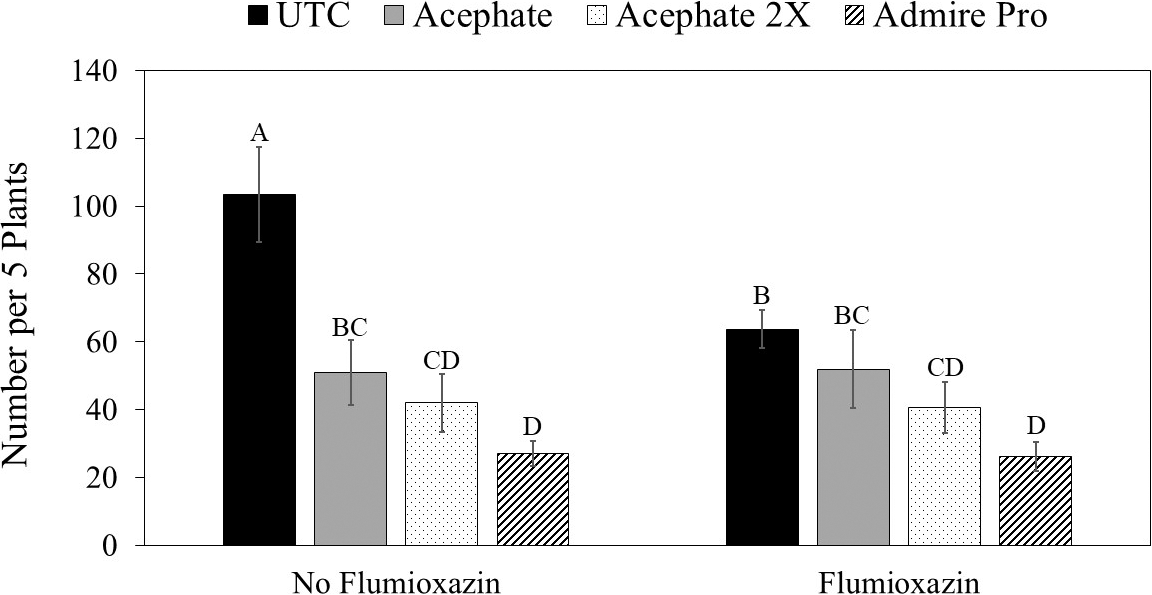
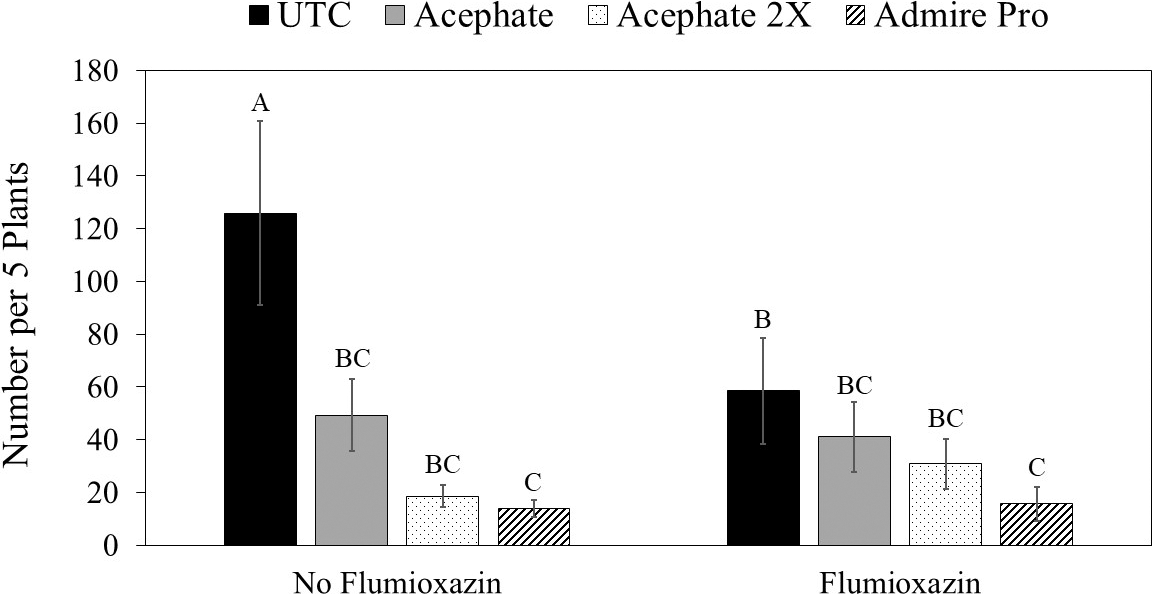
Insecticide was the only factor that impacted thrips populations at the first sample date (Table 3). Although peanuts with imidacloprid applied in the seed furrow at planting had fewer thrips than peanuts that received one foliar application of acephate, none of the insecticide treatments reduced thrips numbers below that in the untreated control (Table 4). There was an interaction between herbicide and insecticide for thrips numbers at the second evaluation timing (Table 3). Where flumioxazin was not used, all of the insecticide treatments reduced thrips numbers below that in the untreated control (Fig. 3). In contrast, imidacloprid applied in the seed furrow at planting was the only treatment that reduced thrips numbers below the untreated control where flumioxazin was applied. Thrips numbers were greater in untreated peanuts where flumioxazin was not applied compared to untreated peanuts where flumioxazin was applied at the second sample date (Fig. 3). There was an effect of insecticide on thrips injury (Table 4). Untreated peanuts had a greater thrips injury rating than peanuts treated with two foliar applications of acephate or peanuts treated with imidacloprid applied in the seed furrow at planting. Peanuts with imidacloprid applied in the seed furrow at planting had less thrips injury compared to all other treatments (Table 4). Herbicide injury was not evaluated in this study because the flumioxazin application was made on emerging peanuts and injury was minor in those treatments (Table 4).
For peanut growth, there was an effect of herbicide and insecticide on canopy coverage at both rating periods, and an effect of insecticide on biomass (Table 3). Peanuts with imidacloprid applied in the seed furrow at planting had greater biomass than untreated peanuts and peanuts with one foliar application of acephate (Table 4). The distance between peanut vines on adjacent rows were greater for untreated peanuts than for all of the insecticide treatments at 45 and 60 d after planting (Table 4). No differences were observed between insecticide treatments at either timing.
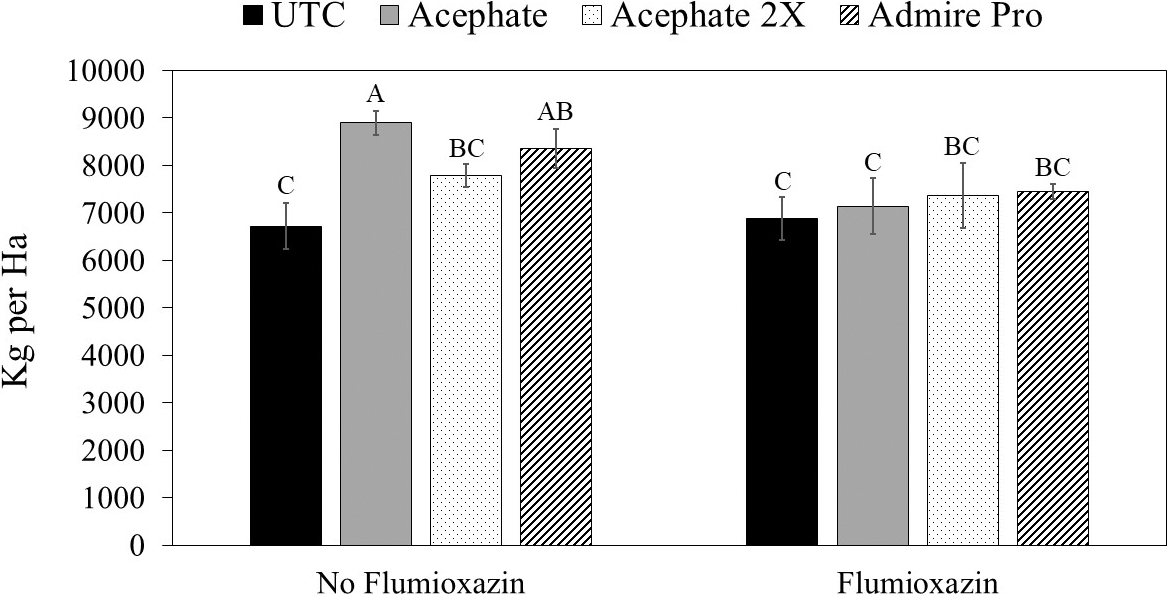
For peanut pod yields, there was an herbicide by insecticide interaction. Pod yield of peanut plants grown with one foliar application of acephate and plants grown with imidacloprid applied in the seed furrow at planting was greater than pod yield for untreated peanuts where flumioxazin was not applied (Fig. 4). In contrast, no differences in pod yield was observed between insecticide treatments and the untreated control where flumioxazin was applied.
Discussion
Early season biotic and abiotic stressors can limit yield in multiple cropping systems. Injury from the use of flumioxazin for preemergence weed control reduced pod yield at the Stoneville location. At the Starkville location, flumioxazin was applied at the cracking stage instead of preemergence and little injury was observed, but pod yield was still lower than where flumioxazin was not used. Previous research suggests that flumioxazin rarely causes yield losses in peanut when applied preemerge (Scott et al. 2001, Grichar et al. 2004, Grichar and Dotray 2013). Additionally, temporary flooding within two d after seedling emergence reduced yields of peanut at the Stoneville location, further supporting the impact of early season stress impacting pod yield.
In general, use of an insecticide for thrips management resulted in fewer thrips, less thrips injury, and increased plant vigor than where no insecticides were used. This was expected as similar results were observed in previous research (Smith and Sams 1977; Tappan and Gorbet 1981; Lynch et al. 1984; Chamberlin et al. 1992). In Stoneville, thrips populations were lower in plots that had flumioxazin applications compared to no flumioxazin and no insecticides. This could be the result of plants being less attractive to thrips for feeding due to herbicide injury and may be an artifact of small plot research where thrips adults were able to choose non-injured plants for feeding and oviposition. The same might not be observed in a large scale production setting where all of the plants in the field have flumioxazin injury.
At the Stoneville location, the flumioxazin-treated plots had more injury from thrips than plots with no herbicide. This was not observed in the Starkville experiment where flumioxazin applied at cracking resulted less injury than that observed in Stoneville. The additional stress from flooding resulted in plants incurring more injury in the flooded plots than the unflooded plots which may have also had an effect on thrips injury. Flooding, which moved the flumioxazin onto the plants from the soil surface increased herbicide injury and further delayed seedling development, which may have rendered plants more susceptible to thrips injury.
Use of an insecticide resulted in more vigorous plants compared to those in plots with no insecticide application across all years at both the Stoneville and Starkville locations. Flooding and flumioxazin use had negative effects as plants were not able to grow as vigorously as uninjured plants. In a non-flooded environment, there was no difference in vigor whether there was a flumioxazin application or not.
In general, plants with an insecticide treatment had greater biomass and faster canopy closure compared to the untreated control. Flumioxazin use created adverse growing conditions for plants, which in turn resulted in less biomass and slower canopy closure. Like flumioxazin and thrips injury, flooding also had a negative effect on biomass and canopy closure in Stoneville. Canopy closure is important in peanut production for weed control. The narrower the width between canopies of adjacent rows, the more likely weeds are to be shaded out and not able to become established. Similarly, the incidence of tomato spotted wilt virus has been shown to be reduced with thicker plant canopies and in twin-row production systems (Brown et al. 1996).
In regards to yield, significant differences were only observed in the experiment in Stoneville. Any insecticide application resulted in a significant yield increase compared to the untreated control. Flumioxazin caused a yield decrease in the Stoneville experiment, however, this what not observed in Starkville. Flooding during the seedling stage also resulted in lower yields of peanut. Previous research has shown thrips injury, herbicide injury, and saturated soils can have adverse effects on yield (Brecke et al. 1996; Funderburk et al. 1998; Drake et al. 2009).
No interaction between flumioxazin use, flooding, and insecticide use, was observed. However, each factor individually effected peanut growth and yield. With herbicide resistant weeds becoming more of a concern, choosing not to make a preemergence application of flumioxazin could result in yield reduction due to competition from weeds. Insecticide applications early in the growing season could result in greater yields. Insecticide applied at-planting are recommended to manage thrips, preserve plant vigor, expedite canopy closure, and preserve yield. Inoculant is already being applied in-furrow at the time of planting, so co-applying imidacloprid is a less expensive option than making one or two foliar applications of acephate after plant emergence. Foliar application of acephate is an option if additional stress from thrips needs to be alleviated. Additionally, more research is needed to determine the best herbicide options for a preemergence program that maximize weed control and minimize plant injury.
Acknowledgements
The authors thank the Mississippi Peanut Promotion Board, Mississippi Peanut Growers Association, and Cotton Incorporated for partial funding of this project. This material is based upon work that is supported by the National Institute of Food and Agriculture, U.S. Department of Agriculture, Hatch project under accession number 1003452.
Literature Cited
Anonymous. 1988. Flumioxazin. Pages 29– 31 In Hatzios K. K. (ed.) WSSA Herbicide Handbook—Supplement to 7th Edition. Allen Press, Lawrence, KS.
Brecke, J. B., Funderburk, J. E. Teare, I. D. and Gorbet. D. W. 1996. Interaction of early-season herbicide injury, tobacco thrips injury, and cultivar on peanut. Agron. J. 88: 14– 18.
Brown, S. L., Todd, J. W. and Culbreath. A. K. 1996. Effect of selected cultural practices on incidence of tomato spotted wilt virus and populations of thrips vectors in peanuts. Acta Hortic. 431: 491– 498.
Burris, E. A. M. Pavloff, B. Leonard, R. Graves, J. B. and Church. G. 1990. Evaluation of two procedures for monitoring populations of early season insect pests (Thysanoptera: Thripidae and Homoptera: Aphididae) in cotton under selected management strategies. J. Econ. Entomol. 83: 1064– 1068.
Chamberlin, J.R., Todd, J.W. Farrow, J.M. and Mullinix B.G. 1992. Aldicarb residue persistence in leaf terminals of ‘florunner’ peanut. J. Econ. Entomol. 85: 1072– 1078.
Cranmer, J. R., Altom, J. V. Braun, J. C. and Pawlak. J. A. 2000. Valor™ herbicide: a new herbicide for weed control in cotton, peanuts, soybeans, and sugarcane. Proc. South. Weed Sci. Soc. 53: 158.
Drake, W. L., Jordan, D. L. Lassiter, B. R. Johnson, P. D. Brandenburg, R. L. and Royals. B. M. 2009. Peanut cultivar response to injury from tobacco thrips and paraquat. Agron. J. 101: 1388– 1393.
Funderburk, J. E., Gorbet, D. W. Tearre, I. D. and Stavisky. J. 1998. Thrips injury can reduce peanut yield and quality under conditions of multiple stress. Agron. J. 90: 563– 566.
Grichar, W. J., and Dotray. P. A. 2013. Smellmellon control and peanut response to flumioxazin and paraquat alone and in combination. Peanut Sci. 40: 135– 141.
Grichar, W. J., Besler, B. A. Dotray, P. A. Johnson, W. C.and Prostko. E. P. 2004. Interaction of flumioxazin with dimethenamid or metolachlor in peanut (Arachis hypogaea L.). Peanut Sci. 12-16.
Heap, I. 2020. The international survey of herbicide resistant weeds. Online. Internet. Friday, November 13, 2020. Available www.weedscience.org.
Herbert, D. A. Malone, S. Aref, S. Brandenburg, R. L. Jordan, D. L. Royals, B. M. and Johnson. P. D. 2007. Role of insecticides in reducing thrips injury to plants and incidence of tomato spotted wilt virus in Virginia market-type peanut. J. Econ. Entomol. 100: 1241– 1247.
Johnson W. C., Brenneman, T. B. Baker, S. H. Johnson, A. W. Sumner, D. R. and Mullinix, B. G. 2001. Tillage and pest management considerations in a peanut-cotton rotation in the southeastern coastal plain. Agron. J. 93: 570– 576.
Linker, H. M., Jordan, D. Brandenburg, R. Bailey, J. Herbert, D. A. Phipps, P. M. and Swann. C. W. 2020. Integrated pest management scouting guide. Virginia Cooperative Extension Pub. 444-146, Blacksburg, VA. https://www.pubs.ext.vt.edu/content/dam/pubs_ext_vt_edu/444/444-126/444-126_pdf.pdf.
Lynch, R. E., Garner, J. W. and Morgan. L. W. 1984. Influence of systemic insecticides on thrips damage and yield of florunner peanuts in Georgia. J. Agric. Entomol. 1: 33– 42.
Main, C. L., Ducar, J. T. Whitty, E. B. and MacDonald. G. E. 2003. Response of three runner-type peanut cultivars to flumioxazin. Weed Technol. 17: 89– 93.
Mound, L. A. 2002. So many thrips—so few tospoviruses. In proc 2001 7th International Symposium on Thysanoptera. Australian National Insect Collection, Canberra, Australia. (http:// www.ento.csiro.au/thysanoptera/symposium.html).
Murdock, E. C., Alden, J. A. and Toler. J. E. 1986. Interactive effects of tobacco thrips control and herbicides on competition between large crabgrass (Digitaria sanguinalis) and peanuts (Arachis hypogaea). Weed Sci. 34: 896– 900.
North, J. H., Gore, J. Catchot, A. L. Cook, D. R. Dodds, D. M. and Musser. F. R. 2019. Quantifying the impact of excluding insecticide classes from cotton integrated pest management programs in the U.S. mid-south. J. Econ. Entomol. 112: 341– 348.
Price, A. J., Wilcut, J. W. and Cranmer. J.R. 2004. Physiological behavior of root-absorbed flumioxazin in peanut, ivyleaf morningglory (Ipomoea hederacea), and sicklepod (Senna obtusifolia). Weed Sci. 52: 718– 724.
Scott, G. H., Askew, S. D. and Wilcut. J. W. 2001. Economic evaluation of diclosulam and flumioxazin systems in peanut (Arachis hypoaea). Weed Technol. 15: 360– 364.
Shrestha, A., Srinivasan, R. Sundaraj, S. Culbreath, A.K. and Riley. D.G. 2013. Second generation peanut genotypes resistant to thrips-transmitted tomato spotted wilt virus exhibit tolerance rather than true resistance and differentially affect thrips fitness. J. Econ. Entomol. 106: 587– 596.
Smith, J. W. and Sams. R. L. 1977. Economics of thrips control on peanuts in Texas. Southwest Entomol. 2: 149– 154.
Tappan, W. B. and Gorbet. D. W. 1981. Economics of tobacco thrips control with systemic pesticides on florunner peanuts in Florida. J. Econ. Entomol. 74: 283– 286.
Taylor-Lovell, S., Wax, L.M. and Bollero. G. 2002. Preemergence flumioxazin and pendimethalin and postemergence herbicide systems for soybean (Glycine max). Weed Technol. 16: 502– 511.
Todd, J. W., Culbreath, A. K. Roger, D. and Demski. J. W. 1994. Contraindications of insecticide use relative to vector control and spotted wilt disease progress in peanut. Proc. Am. Peanut Res. Ed. Soc. 26: 24.
Todd, J. W., Culbreath, A. K. Chamberlin, J. R. Beshear, R. J. and Mullinix. B. G. 1995. Colonization and population dynamics of thrips in peanuts in the southern United States, pp. 453– 460. In Parker, B. Skinner, M. and Lewis T. (eds.), Thrips Biology and Management, vol. 276. Plenum Press, New York.
Todd, J. W., Culbreath, A. K. and Brown. M. R. 1996. Dynamics of vector populations progress of spotted wilt disease relative to insecticide use in peanuts. Acta Hortic. 431: 483– 490.
USDA-NRCS. 2000. Official soil series descriptions. [Online].USDA Nat. Res. Cons. Serv., Washington, D.C. Available at https://soilseries.sc.egov.usda.gov/OSD_Docs/M/MARIETTA.html (Accessed 10 Nov 2020).
USDA-NRCS. 2019. Official soil series descriptions. [Online].USDA Nat. Res. Cons. Serv., Washington, D.C. Available at https://soilseries.sc.egov.usda.gov/OSD_Docs/B/BEULAH.html (Accessed 10 Nov 2020).
Williams, E. J., and Drexler. J. S. 1981. A non-destructive method for determining peanut pod maturity. Peanut Sci. 8: 134– 141.
Notes
- First, second, fourth, and fifth authors: Graduate Student, Research Professor, Associate Research Professor, Assistant Extension Professor, Delta Res. & Ext. Center, Mississippi State Univ. Stoneville, MS 38776; Third and eighth author: Extension Professor and Graduate student, Dept. of Biochemistry, Molecular Biology, Entomology, & Plant Pathology, Mississippi State University, Mississippi State, MS 39762. Sixth, seventh (formerly), and ninth authors, Dept. of Plant & Soil Sciences, Mississippi State Univ., Mississippi State, MS 39762 [^] *Corresponding author Email: jgore@drec.msstate.edu
Author Affiliations