Introduction
Plant growth regulators have been evaluated and utilized intermittently for more than 40 years to manage vegetative growth on fast growing cultivars of virginia and runner market type peanut (Arachis hypogaea L.). There are several key reasons for managing vine growth in peanut. Research has shown that peanut plants produce more vegetative growth than needed to achieve maximum pod yield (Mitchem et al., 1996), excessive vines can be problematic for managing diseases (Henning et al., 1982; Maloy, 1993), and excessive vines can decrease digging and harvesting efficiency (Beam et al., 2002). Excessive vines can also become damaged by tractor tires during mid- and late season pesticide applications potentially causing an increase in disease and yield losses (Wu and Santelman, 1977). Daminozole was an effective plant growth regulator used throughout the 1970’s and the 1980’s. However, daminozole’s registration was discontinued by EPA in 1989 due to safety concerns in food products (Smith, 1989). With growers still in need of managing excessive vine growth to minimize disease issues and harvest difficulties, the chemical industry continues to evaluate potential growth regulators with lower health risks while suppressing vegetative growth similar to daminozole. In the early 1980’s, prohexadione calcium [calcium salt of 3,5-dioxo-4 propionylcyclohexanecarboxylic acid] (Apogee 27.5 WDG, BASF Corp., 26 David Dr., Research Triangle Park, NC 27709 or Kudos 27.5 WDG, Fine-Americas, 1850 Mt. Diablo Blvd., Walnut Creek, CA 94596), a novel plant growth regulator, was discovered (Motojima, et al., 1984). In April of 2000, prohexadione calcium was developed and registered by BASF Corporation for use in the United States (Giles-Parker, 2000).
Prohexadione calcium, is a plant growth regulator used in apple (Malus xdomestica Borkh.), grain sorghum [Sorghum bicolor (L.) Moench], oilseed rape (Brassica napus L.), peanut, rice (Oryza sativa L.), tomato (Solanum lycopersicum L.), and wheat (Triticum aestivum L.) to slow vegetative growth (Yamaji et al., 1991, Nakayama et al., 1992; Grossman et al., 1994, Mitchem et al.1996, Lee et al., 1998; Byers and Yoder, 1999). Prohexadione calcium works by inhibiting the biosynthesis of gibberellin, a natural plant hormone that regulates cell elongation by blocking kaurene oxidase. It also increases the level of abscisic acid and cytokines (Grossman et al., 1994).
Initial research of prohexadione calcium (Mitchem et al., 1996), showed a reduction in main branch length and cotyledon branch length of 32% and 30%, respectively over the non-treated peanut. This reduction in vegetative growth was similar to reductions by daminozole. It was also reported that prohexadione calcium increased pod yield by 8% and increased the percentage of extra-large kernels by 3% but had no effect on the percentage of fancy pods and total sound mature kernels.
Since that time, many studies have been conducted on virginia market type peanut cultivars to learn more about the effect of prohexadione calcium on peanut. Culpepper et al. (1997) showed that yield response was cultivar dependent with yield increases varying among six virginia market type peanut cultivars. Prohexadione calcium increased the yield of cultivar NC 9 by 7% while decreasing the yield of cultivar VA-C 92R by 8%. The plant growth regulator also had no yield response on cultivars NC 10C (Wynne et al., 1991), NC 12C (Isleib et al., 1997), or NC-V 11 (Wynne et al., 1991). In a similar experiment comparing the effect of prohexadione on four virginia market type peanut cultivars, yield response to prohexadione calcium was shown to be greatest in twin rows, as opposed to single rows, regardless of cultivar (Faircloth et al., 2005). Applying prohexadione calcium with urea ammonia nitrate, either alone or with crop oil concentrate, increased the effect more than when prohexadione calcium was applied without the urea ammonium nitrate (Jordan et al., 2000). Beam et al., (2002) reported that applying prohexadione calcium reduced digging losses by as much as 4% regardless of digging date and lifting treatment compared with nontreated peanuts. During the initial time period when prohexadione calcium was registered and sold commercially, runner market type cultivars changed from having an excessive vine growth habit to a moderate growth habit. Research during that time also reported that a tractor guidance system with a Global Positioning systems with Real-Time Kinematics (GPS RTK) was more effective at optimizing yield than prohexadione calcium (Roberson and Jordan, 2014). This reduced growth habit, along with GPS RTK guidance, reduced the cost effectiveness of the growth regulator on runner-type peanut cultivars. Even though the benefits of prohexadione calcium have been established in the literature on virginia market type peanut cultivars, the price (estimated at $148/ha for 2 applications at 140 g/ha – excluding cost of application) of prohexadione calcium can be cost prohibitive to growers (Bullen et al., 2019). However, Jordan et al. (2019) reported that over 50% of growers in North Carolina and Virginia apply prohexadione calcium at least once, demonstrating perceived value at the farm level.
The use of prohexadione calcium at the labeled rate (140 g/ha) on runner market type peanuts has not been recommended over the last 20 years due to the limited vine growth of cultivars like Georgia Green (Branch, 1996) and Georgia-06G (Branch, 2007). This is unlike recent virginia market type peanut cultivars such as NC-V 11, NC 12C, Perry (Isleib et al., 2003), Gregory (Isleib et al., 1999), and Bailey (Isleib et al. 2011), which exhibit a robust growth habit. However, the resurgence since 2012 of runner market type cultivars with excessive vegetative growth, like Georgia-12Y (Branch, 2013), has increased the interest for the growth regulator to manage vine growth in order to allow better inverting and harvesting operations. Therefore, research was conducted to quantify the growth response and economics of using prohexadione calcium on currently available runner and virginia market type peanut cultivars in the Southeast and Carolinas. Since vine growth is different among runner and virginia market type peanut cultivars, research was also conducted to determine the efficacy of the growth regulator at reduced rates on both peanut types.
Materials and Methods
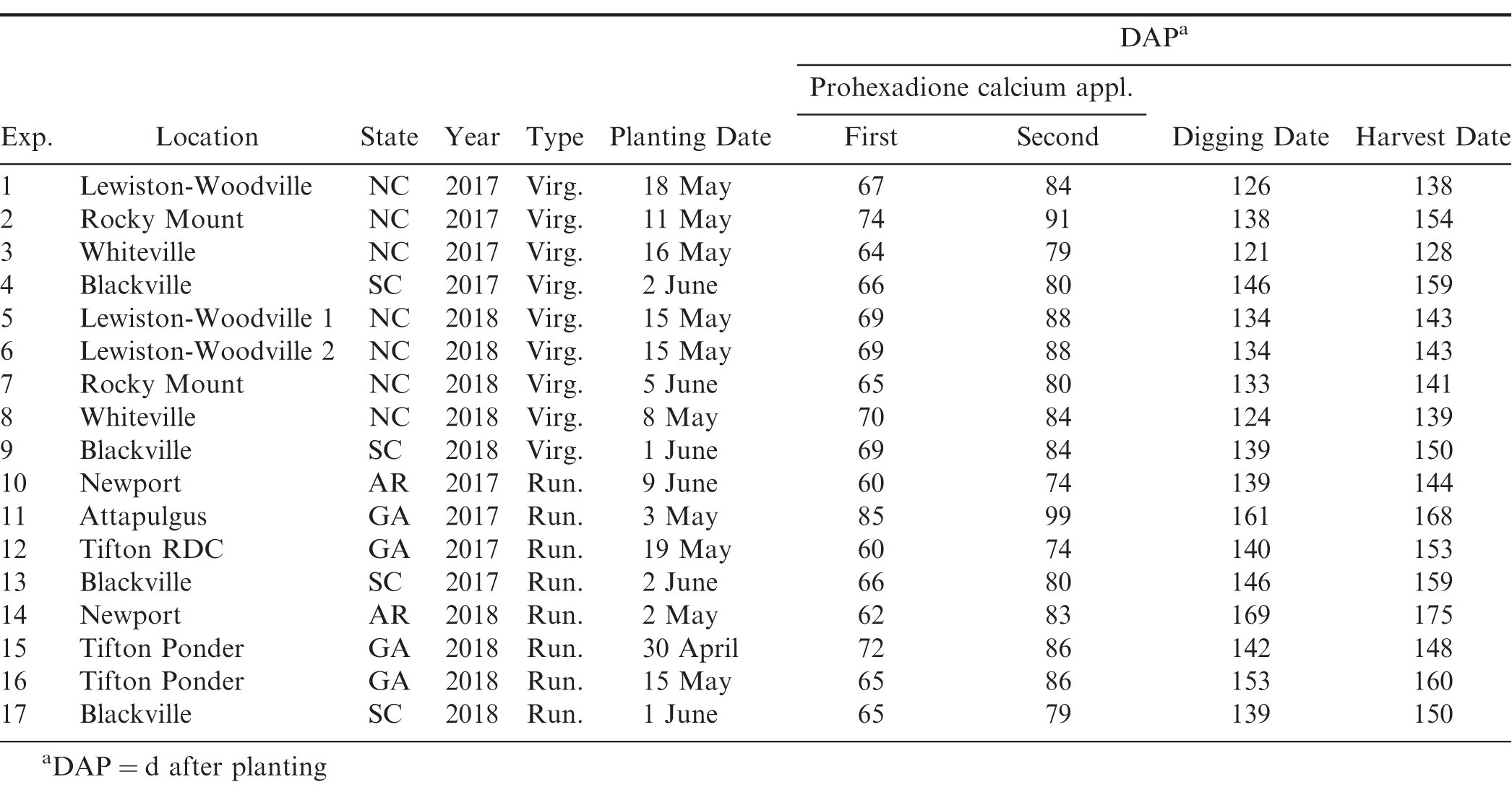
All experiments were conducted in 2017 and 2018 under irrigation in Arkansas, Georgia, Mississippi, North Carolina, and South Carolina to evaluate growth and yield response of peanut based upon prohexadione calcium application rate. Prohexadione calcium treatments evaluated were 1) the manufacturer’s recommended use rate of 140 g a.i /ha (1.0x); 2) 105 g a.i /ha (0.75x); and 3) 70 g a.i /ha (0.5x). A non-treated control was included in all experiments. As per label directions, crop oil concentrate, (Agri-Dex, 83% paraffin based petroleum oil and 17% surfactant, Helena Chemical Co., 5100 Poplar Ave., Memphis, TN ) was applied at 2.3 L/ha and 28% urea ammonium nitrate or 21% ammonium sulfate was applied at 1.2 L/ha with prohexadione calcium applications. Prohexadione calcium was applied in 233 L/ha water using a CO2 -pressurized backpack sprayer equipped with 8002 regular flat fan nozzles (Teejet nozzles, Spraying Systems Co., Wheaton, IL) in all small plot experiments and 140 L/ha of water using commercial large-scale crop sprayers for the large plot on-farm experiments. All treatments were applied independently of any other pesticide applications. Commercial large-scale crop sprayers (type and size) varied by location. In all experiments, prohexadione calcium treatments were initiated when at least 50% lateral vines from adjacent rows were touching. A second application of each treatment was applied 14 d after the first application. Peanut market type, cultivar, and planting date varied by location and are described in more detail in Table 1. Seed was planted at rates to achieve a final in-row plant population of 13.1 to 16.4 plants/m in a single row planting pattern (91.4 cm). Small plot dimensions were 1.8 m wide and 7.6 m to 9.1 m long with two untreated rows between each plot. Large plot trial dimensions ranged from 5.5 m to 11 m wide and 152.4 m to 457.2 m long. All experiments were randomized complete block design with four replicates. Peanut production management decisions were made based on individual state University Cooperative Extension Service recommendations. Main stem height (cm) was measured 2 weeks after the second application of prohexadione calcium was applied to determine plant growth response to the growth regulator. This measurement was taken by measuring the height of the main stem in cm at three random locations in the plot. Peanut plants in each plot were dug and inverted based on maturity profile method (pod mesocarp color) (Williams and Drexler, 1981). Plants were dried for 5 to 7 d depending on weather and harvested mechanically using commercial peanut combines. Pod yield was assessed at harvest and final pod weight was adjusted to 7% moisture. Return on investment for each treatment was calculated based on the gross yield*base loan value – cost of prohexadione calcium. The base loan value for peanut in the United States is $355/farmer stock ton.
Analysis of variance was conducted using the PROC MIXED function within SAS version 9.4 (SAS Institute, Cary, NC). Location, market type (virginia or runner), and plot size (small plot or large plot on-farm) were analyzed separately because of variation in field types and plant growth habit to minimize variability among experiments. Appropriate means were separated with Fisher’s protected least significant difference (LSD) test at P = 0.05.
Virginia market type small plot experiments.
Nine small plot experiments were conducted using virginia market type peanut cultivars on university research stations at Lewiston-Woodville, Rocky Mount, and Whiteville in North Carolina and at Blackville, SC (Table 1). Planting date varied by location ranging from early May to early June (Table 1). All experiments in North Carolina were planted with the cultivar Bailey, while Bailey, Wynne (Isleib et al, 2015), and Sullivan (Isleib et al., 2015) were used in South Carolina. Since cultivars varied among states, data were combined across cultivars for the multi-location analysis. Plant architecture was assessed visually 2 to 4 weeks after final prohexadione calcium application in North Carolina using a 1 to 10 row visibility rating scale developed by Mitchem et al. (1996) where 1 = a flat peanut canopy with an indistinguishable main stem where row definition is unclear and 10 = a peanut canopy with triangular-shaped plants (apex at central mainstem).
Runner market type small plot experiments.
Seven small plot experiments were conducted on university research stations with runner market type peanuts in Arkansas, Georgia, and South Carolina (Table 1). Peanut planting date varied by location and ranged from early May to early June. Runner market type cultivars varied by state; thus, data were combined across cultivars in the multi-location analysis. Runner market type cultivars included Georgia-06G, Georgia-09B (Branch, 2010), Georgia-12Y, and TUFRunner 511 (Tillman and Gorbet, 2017).
Large plot on-farm experiments.
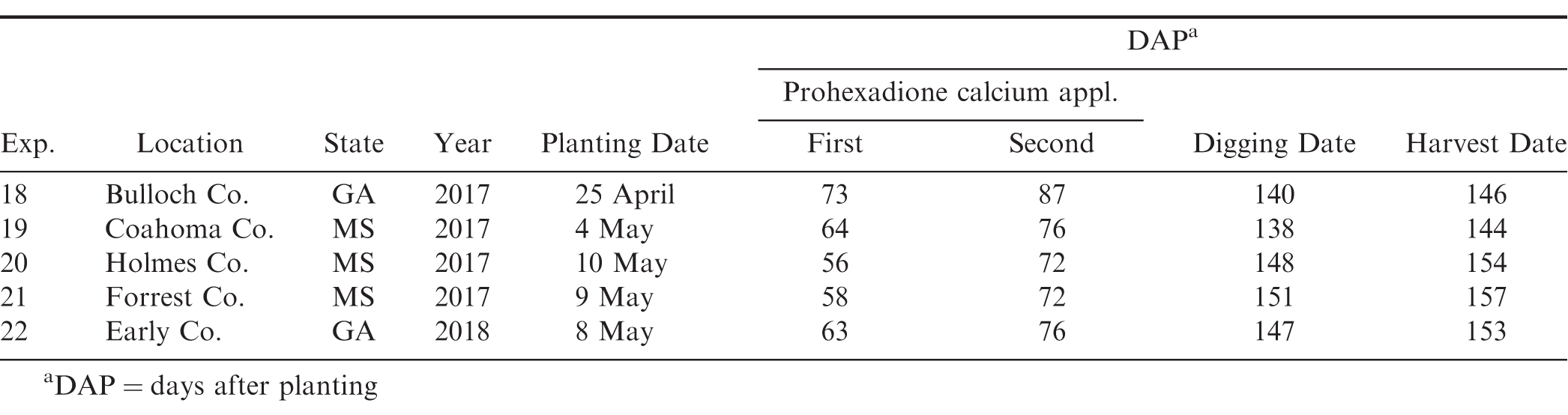
In 2017, a large plot on-farm experiment was conducted in Georgia (Bulloch County) and three in Mississippi (Coahoma, Holmes, and Forrest Counties), and, in 2018, one large plot on-farm experiment was conducted in Georgia (Early County) (Table 2). Peanut planting date varied by location and ranged from early to mid-May. Runner market type cultivars varied by state; thus, data were combined across cultivars in the multi-location analysis. Runner market type cultivars included Georgia-06G, Georgia-12Y, TUFRunner-297 (Tillman, 2017), TUFRunner-511.
Results
Virginia market type small plot experiments.
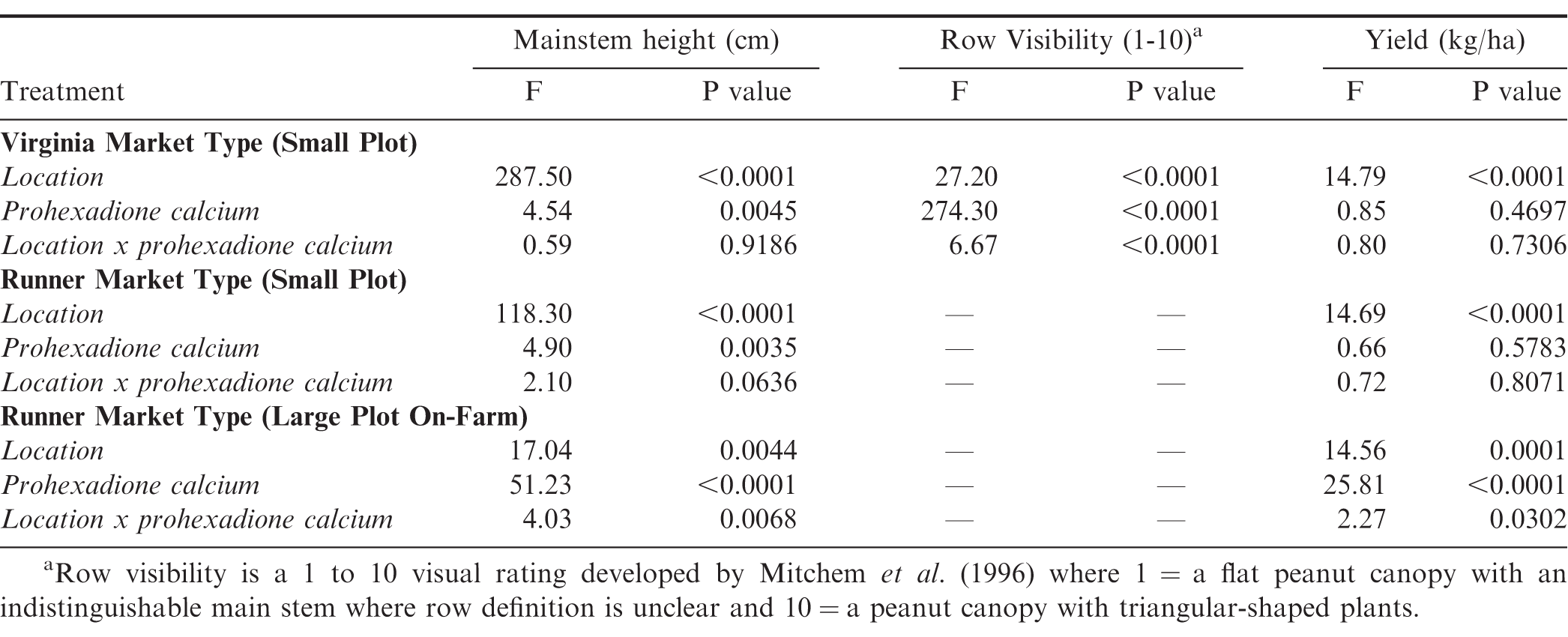
The location by treatment interaction was not significant for mainstem height or pod yield in the virginia market type small plot experiments (Table 3), thus experiments were additionally combined across locations for analysis. Location and prohexadione calcium rate was significant for mainstem height and row visibility at P < 0.0001 and = 0.0045; and P < 0.0001 and < 0.0001, respectively. Location was significant for pod yield (P < 0.0001).
Analysis of variance for peanut crop growth and yield for virginia market type in small plot experiments, runner market type in small plot experiments, and large plot on-farm experiments for Location (Virginia, South Carolina, North Carolina, Georgia, Arkansas, and Mississippi in 2017 and 2018) and prohexadione calcium rates.
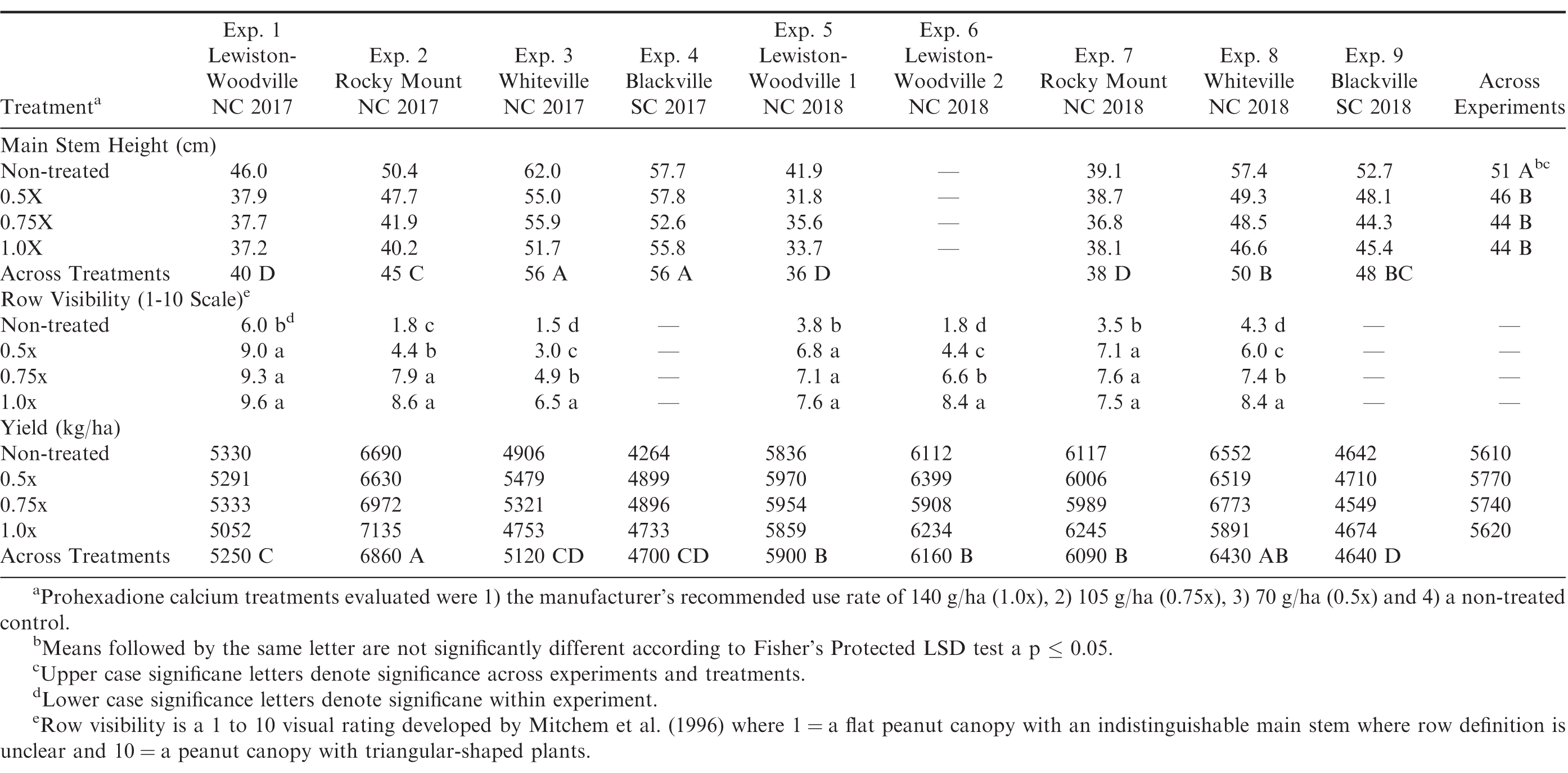
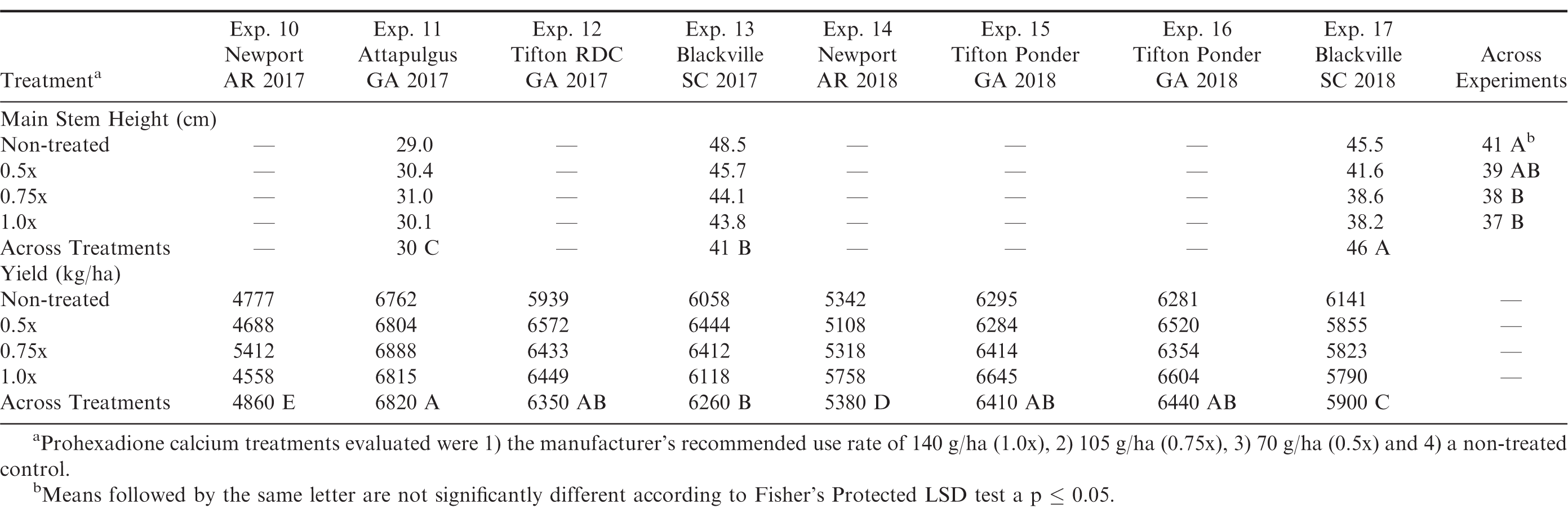
Main stem heights ranged from 36 to 56 cm across locations and treatments (Table 4). Mainstem heights varied among locations which could be due to variations in environmental conditions and their effects on crop growth. Experiments 3 and 4 had the tallest main stem heights, while Experiments 1, 5, and 7 had the shortest average main stem heights. Microclimate conditions including temperature and moisture likely contributed to the variations in canopy growth across locations. In examining the treatment effects of prohexadione calcium, the non-treated plots had the tallest main stem heights of all treatments for all experiments (Table 4). Reduced rates (0.5x and 0.75x) provided similar growth inhibition as the full labeled rate of the growth regulator (Table 4). These results were similar to the runner market type experiments and results from Mitchem et al. (1996) testing the effect of prohexadione calcium on the virginia market type peanut cultivar NC 9.
Row visibility for each location was analyzed separately due to a location by prohexadione calcium interaction. Based on this data row visibility was least for the non-treated control in all experiments, indicating the non-treated control had a flat canopy shape (Table 4). Although the full labeled rate (1.0x) had the greatest row visibility, all prohexadione calcium treatments had significantly better row visibility than the non-treated control at each location. Similar observations were made by Mitchem et al. (1996). Mitchem also noted that increased row visibility can lead to greater disease and insect management due to the condensed canopy. Pod yield for the virginia market type small plot experiments varied across locations and pod yield ranged from 4600 kg/ha to 6900 kg/ha with experiments 2, 5, 6, 7, and 8 having the greatest pod yield (Table 4). When combined across experiments, the yield response from utilizing prohexadione calcium to manage canopy growth was not different from the non-treated control at the P = 0.05 level.
Runner market type small plot experiments.
The location by treatment interaction was not significant for mainstem height or pod yield in the runner market type small plot experiments (Table 3). Location was significant for both mainstem height and pod yield with the treatment effect of prohexadione calcium only being significant for mainstem height (Table 3). Height measurements were taken for only experiments 11, 13, and 17. Experiment 17 had the tallest plant main stem height compared to experiment 11 and 13 (Table 5). When the data were averaged across all three locations, prohexadione calcium applications at the 0.75x and the 1.0x were the only rates that significantly reduced mainstem height compared to the non-treated control (Table 5). Pod yield for the runner market type small plot experiments varied across locations and treatments with pod yield ranging from 4900 kg/ha to 6800 kg/ha with experiments 11, 12, 13, 15, and 16 having the greatest pod yield at P = 0.05 (Table 5). In all runner market type small plot experiments, the use of prohexadione calcium to manage canopy growth did not affect pod yield over that of the non-treated control. When combined across experiments, pod yield is not different when using prohexadione calcium from the non-treated control at the P = 0.05 level.
Large plot on-farm experiments.
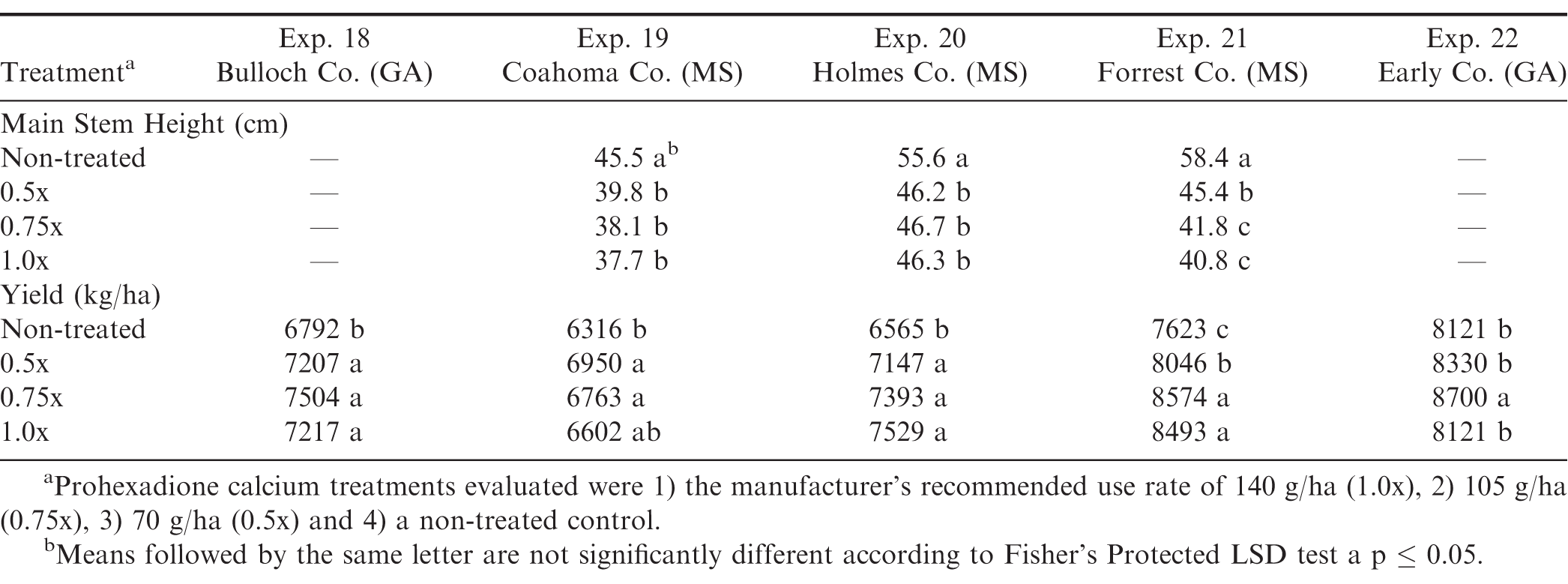
The interaction of location by treatment and the corresponding main effects were significant for mainstem height and pod yield in the large plot on-farm runner type experiments (Table 3). Mainstem height was significantly greater for the non-treated control compared to all prohexadione calcium treatments at all locations (Table 6). When examining the individual experiments, the 0.75x and 1.0x rates provided significant reductions in main stem heights compared to the non-treated control across all locations (Table 6).
Pod yield was significantly greater for 0.75x rates of prohexadione calcium compared to the non-treated control for all of the large plot on-farm experiments. The 0.75x treatment was similar in yield response to the 0.5x and 1.0x rate in experiments 18, 19, and 20 and similar to the 1.0x rate in experiment 21. The 0.75x treatment was significantly better than the 1x treatment in experiment 22 (Table 6). Return on investment for each treatment was calculated based on the gross yield*base loan value – cost of prohexadione calcium. Therefore, net dollar value for prohexadione calcium treatments for the on-farm experiments across all locations were $3,242/ha (b), $3,327/ha (a), and $3,181/ha (bc) for the 0.5x, 0.75x, and 1.0x prohexadione calcium rates, respectively, compared to the non-treated control at $3,117/ha (c) at P = 0.05. The return on investment for the large plot on-farm experiments was greater for the 0.5x and 0.75x prohexadione calcium treatments compared to the non-treated control. Based on these results, growers could increase their revenues $62 (0.5x) to $146 (0.75x)/ha by using a reduced rate of prohexadione calcium compared to the manufacturer’s recommended use rate (1.0x rate) for managing their canopy growth.
Discussion and Summary
The goal of this multistate project was to evaluate the efficacy and economics of prohexadione calcium at reduced and labeled rates on runner and virginia market type peanut cultivars. Results of these experiments supported previous research showing prohexadione calcium is effective at reducing canopy growth at the labeled rate on both runner and virginia market type peanut cultivars (Mitchem et al., 1996). Further, these data showed that reduced rates of prohexadione calcium can be used to reduce mainstem height and increase row visibility similar to the 1.0x rate in both runner and virginia market type peanut cultivars no matter the plot size. This research does support previous findings that at the labeled rate (1.0x) of prohexadione calcium did not provide a consistent increase in yield nor an increase in return on investment (Faircloth et al., 2005). However, reduced rates performing similarly to the labeled rate could potentially save growers an average of $49.40 (0.75x) to $74.10 (0.5x)/ha in growth regulator costs making it more cost effective to manage vine growth in extreme cases.
The evaluation of prohexadione calcium was conducted in both small plot and large on-farm experiments for runner market type peanut in Georgia and Mississippi. The results of the large plot on-farm experiments supported the small plot experiments in which significant reductions in canopy growth were achieved for all rates of the growth regulator. The intriguing part of this research project was the significant yield responses observed as a result of applying the growth regulator in the large on-farm experiments. In every experiment, there was at least one prohexadione calcium treatment that had greater yield than the non-treated control, and the 0.75x rate consistently provided improved yield compared to the non-treated control in all experiments. The labeled rate (1.0x) and the 0.5x rate were inconsistent in providing a yield enhancement compared to the 0.75x rate.
Based on these experiments, it is evident that prohexadione calcium manages canopy growth consistently in both small plot and large plot on-farm experiments at labeled and reduced rates. The differences in pod yield responses in small plot experiments and large plot on-farm experiments needs to be examined further in relationship to prohexadione calcium. There are many factors that cannot be accounted for which could have an impact on pod yield in the large plot on-farm experiments that might not be observed in small plot experiments. For instance, the large plot on-farm experiments may have had larger variation in excessive vine growth due to environmental/soil factors which made the effect of prohexadione calcium greater. Some of these environmental/soil factors may include variations in soil types, soil water holding capacity, fertility and pH differences, as well as elevation. These types of differences tend to be greatly reduced in small plot experiments. Based on previous research, it is also possible that the increased row visibility from prohexadione calcium contributes to the yield increase through increasing digging efficiency and reducing the loss of pods through the digging and inversion process (Jordan, 2008). This could explain some of the differences in small plot versus large on-farm experiments in which the digger losses might be minimized due to the short distance of the plot and reduced speed of the tractor compared to large-scale equipment (Kendal Kirk, personal communication). These types of differences might be a reason to evaluate products like growth regulators under a more grower-oriented environment. Overall, this multistate research project confirms that prohexadione calcium provided consistent management of canopy growth at reduced rates of 70 (0.5x) to 105 (0.75x) g/ha on both runner and virginia market type peanut cultivars, while the reduced rate of 105 g a.i /ha provided a significant yield improvement over the non-treated control. This reduction in rate and the increase in yield provided a return of investment of $210/ha over the cost of the non-treated control.
Acknowledgements
BASF Crop Protection, Fine Americas Inc., The Georgia Peanut Commodity Commission, North Carolina Peanut Growers Association, South Carolina Peanut Board, Mississippi Peanut Promotion Board, and the University of Arkansas provided partial funding for this research. Appreciation is expressed to technical staff at the University of Georgia Tifton Campus; North Carolina Department of Agriculture and Consumer Services Border Belt Tobacco Research Station, Peanut Belt Research Station, and the Upper Coastal Plains Research Station; Clemson University Blackville Research Center; University of Arkansas Lonoke Extension Center; and Mississippi State University for their assistance with this research. Donation of seed provided by the Georgia Seed Development Commission is greatly appreciated. This project was conducted as part of the objectives outlined in Federal Hatch project #GEO00273.
Literature Cited
Beam, J.B., Jordan, D.L. York, A.C. Isleib, T.G. Bailey, J.E. McKemie, T.E. Spears, J.F. and Johnson. P.D. 2002. Influence of prohexadione calcium on pod yield and pod loss of peanut. Agron. J. 94: 331– 336.
Branch, W.D. 2013. Registration of ‘Georgia -12Y’ Peanut. J. Plant Reg. 7: 151– 153. Doi:10.3198/jpr2012.11.0048crc.
Branch, W. D. 2010. Registration of ‘Georgia-09B’ Peanut. J. Plant Reg. 4: 175– 178. doi:10.3198/jpr2009.12.0693crc.
Branch, W.D. 2007 a. Registration of ‘Georgia-06G’ Peanut. J. Plant Reg. 1: 120. doi:10.3198/jpr2006.12.0812crc.
Branch, W.D. 1996. Registration of ‘Georgia Green’ peanut. Crop Sci. 36: 806. doi:10.2135/cropsci1996.0011183X003600030051x.
Bullen, S.G., Washburn, D. and Jordan. D.L. 2019. Peanut production budgets. Pages 2– 15in 2019 Peanut Information. North Carolina Cooperative Extension Service Publication AG-331. 193 pages.
Byers, R.E., and Yoder. K.S. 1999. Prohexadione calcium inhibits apple, but not peach, tree growth, but has little influence on apple fruit thinning or quality. Hort. Science 34: 1205– 1209.
Culpepper, A.S., Jordan, D.L. Batts R.B. and York. A.C. 1997. Peanut response to prohexadione calcium as affected by cultivar and digging date. Peanut Sci. 24: 85– 89.
Faircloth, J.C., Coker, D.L. Swann, C. Mozingo, W. Phipps, P.M. and Jordan. D.L. 2005. Response of Four Virginia-Type Peanut Cultivars to Prohexadione Calcium as Affected by Cultivar and Planting Pattern. Peanut Sci. 32: 42– 47.
Giles-Parker, C. 2000. Prohexadione Calcium Pesticide Fact Sheet. EPA. Retrieved from https://nepis.epa.gov/Exe/ZyPDF.cgi/P100BICE.PDF?Dockey=P100BICE.PDF
Grossman, K., Koenig, S. and Kwaitkowski. J. 1994. ·Phytohormonal changes in intact shoots of wheat and oilseed rape treated with the acylcyclohexanedione growth retardant prohexadione calcium. Physiol. Plant. 90: 139– 143.
Henning, R.J., Allison, A.H. and Tripp. L.D. 1982. Cultural practices, pp. 123– 138. In Pattee H. E. and Young C. T.(eds.) Peanut Science and Technology. Amer. Peanut Res. Educ. Soc, Yoakum, TX.
Isleib, T.G., Pattee, H.E. Tubbs, R.S. Sanders, T.H. Dean, L.O. and Hendrix. K.W. 2015. Intensities of sensory attributes in high-and normal-oleic cultivars in the uniform peanut performance test. Peanut Sci. 42: 83– 91.
Isleib, T.G., Milla-Lewis, S.R. Pattee, H.E. Copeland, S.C. Zuleta, M.C. Shew, B.B. Hollowell, J.E. Sanders, T.H. Dean, L.O. Hendrix, K.W. Balota, M. and Chapin. J.W. 2011. Registration of ‘Bailey’ peanut. J. Plant Reg. 5: 27– 39. doi:10.3198/jpr2009.12.0742crc.
Isleib, T.G., Rice, P.W. Mozingo, R.W. Bailey, J.E. Mozingo, R.W. and Pattee. H.E. 2003. Registration of ‘Perry’ peanut. Crop Sci. 43: 739– 740. doi:10.2135/cropsci2003.0739.
Isleib, T.G., Rice, P.W. Mozingo, R.W. Mozingo, R.W.and Pat-tee. H.E. 1999. Registration of ‘Gregory’ peanut. Crop Sci. 39: 1526.
Isleib, T.G., Rice, P.W. Bailey, J.E. Mozingo, R.W. and Pattee. H.E. 1997. Registration of ‘NC 12C’ peanut. Crop Sci. 37: 1976.
Jordan, D. L., Hare, A.T. Roberson, G.T. Ward, J. Shew, B.B. Bradenburg, R.L. Anco, D. Thomas, J. Balota, M. Mahl, H. and Taylor. S. 2019. Survey of practices by growers in the Virginia-Carolina region relative to digging and harvesting peanut. J. Crop, Forage, and Tufgrass Manag. 5: 1– 4.
Jordan, D. L., Nuti, R.C. Beam, J.B. Lancaster, S.H. Lanier, J.E. Lassiter B.R.,, and Johnson. P.D. 2008. Peanut (Arachis hypogaea L.) Cultivar Response to Prohexadione Calcium. Peanut Sci. 35: 101– 107.
Jordan, D.L., Swann, C.W. Culpepper, A.S. and York. A.C. 2000. Influence of adjuvants of peanut (Arachis hypogaea L.) response to prohexadione calcium. Peanut Sci. 27: 30– 34.
Lee, I.J., Foster, K.R. and Morgan. P.W. 1998. Effect of gibberellin biosynthesis inhibitors on native gibberellin content, growth and floral initiation in Sorghum bicolor. J. Plant Growth Regul. 17: 185– 195.
Maloy, O.C. 1993. Plant Disease Control: Principles and Practices. John Wiley and Sons, New York.
Mitchem. W.E. York, A.C. and Batts. A.C. 1996. Peanut response to prohexadione calcium, a new plant growth regulator. Peanut Sci. 23: 1– 8.
Mozingo, R.W., Wynne, S.C. Porter, D.M. Coffelt, T.A. and Isleib. T.G. 1994. Registration of ‘VA-C 92R’ peanut. Crop Sci. 34: 539– 540.
Motojima, K., Miyazawam T. Toyokawa, Y. Matsuzawa, M. Hokari H, and Kusano, S. 1984. Cyclohexane derivatives and plant growth regulators. Jpn. Kokai Tokkyo Koho. JP 59-196840.
Nakayama, I., Kobayashi, M. Kamiya, Y. Abe, H. and Sakurai. A. 1992. Effects of a plant-growth regulator, prohexadione-calcium (BX 112), on the endogenous levels of gibberellins in rice. Jpn. Soc. Plant Physiol. 33: 59– 62.
Roberson, G.T., and Jordan. D.L. 2014. RTK GPS and automatic steering for peanut digging. Am. Soc. Agricultural and Biological Engineers. 30(3): 405– 409.
SAS Institute Inc. 2015. The Proc MIXED procedure. p. 6048– 6234. In: SAS User’s Guide, Version 14.1 Edition. SAS Institute, Cary, NC.
Smith, W. 1989. Daminozide (Alar, B-Nine) EPA Preliminary Determination to Cancel 6/89. Pesticide Management Education Program. Retrieved from http://pmep.cce.cornell.edu/profiles/herb-growthreg/dalapon-ethephon/daminozide/herb-prof-daminozide-canc.html
Tillman, B.L. 2017. Registration of ‘TUFRunner 297’ Peanut. J. of Plant Reg. 12: 31– 35. doi:10.3198/jpr2017.02.0007crc
Tillman, B.L. and Gorbet. D.W. 2017. Registration of ‘TUFRunner 511’ Peanut. J. of Plant Reg. 11: 235– 239. doi:10.3198/jpr2016.11.0064crc
William, E.J. and Dexler. J.S. 1981. A Non-Destructive Method for Determining Peanut Pod Maturity. Peanut Sci. 8: 134– 141.
Wu, C.H., and Santelmann. P.W. 1977. Influence of six plant growth regulators on Spanish peanuts. Agron. J. 69: 521– 522.
Wynne, J.C., Beute, M.K. Bailey, J. and Mozingo. R.W. 1991. Registration of ‘NC 10C’ peanut. Crop Sci. 31: 484.
Wynne, J.C., Coffelt, T.A. Mozingo, R.W. and Anderson. W.F. 1991. Registration of ‘NC-V11’ peanut. Crop Sci. 31: 484– 485.
Wynne, J.C., Mozingo, R.W. and Emery. D.A. 1986. Registration of ‘NC 9’ peanut. Crop Sci. 26: 197.
Yamaji, H., Katsura, N. Nishijima, T. and Koshioka. M. 1991. Effects of soil-applied uniconazole and prohexadione calcium on the growth and endogenous gibberellin content of Lycopersicon esulentum Mill. seedlings. J. Plant Physiol. 138: 763– 764.
Notes
- Department of Crop and Soil Sciences, University of Georgia, Tifton, GA, 31793; [^]
- Department of Crop and Soil Sciences, North Carolina State University, Raleigh, NC, 27695; [^]
- Department of Plant and Environmental Sciences, Clemson University, Blackville, SC, 29817; [^]
- Indigo Ag, Memphis, TN, 38103; [^]
- Department of Plant and Soil Sciences, Mississippi State University, Starkville, MS, 39762; [^]
- University of Arkansas Division of Agriculture, Lonoke Extension Center, Lonoke, AR, 72086; [^]
- University of Georgia Cooperative Extension, Early County, Blakely, GA, 39823 ; [^]
- University of Georgia Cooperative Extension, Bulloch County, Statesboro, GA, 30458 [^] *Corresponding author Email: smonfort@uga.edu
Author Affiliations