Introduction
Georgia is the leading producer of peanut (Arachis hypogaea L.) in the United States (USDA-NASS, 2019). Weed control is an integral part for ensuring that Georgia peanut growers produce yields that ensure profitability. Estimated herbicide costs for growers can be up to $135 per hectare (Rabinowitz and Smith, 2017). Timely and accurate herbicide applications help reduce additional input costs.
The critical period of weed control for peanut is from 3 to 8 wk after planting. This makes postemergence (POST) herbicide applications important and necessary to avoid irreversible yield loss (Everman et al., 2008). Paraquat (1, 1'-dimethyl-4,4'-bipyridinium) became a staple POST herbicide after the loss of dinoseb (2-[1-methylpropyl]-4,6-dinitophenol) in 1986 (Buchanan et al., 1982; Shaner, 2014; Wilcut et al., 1989). Paraquat is a non-selective, contact herbicide used to control most annual broadleaf and grass weed species (Wehtje et al., 1986). Paraquat must be applied no later than 28 d after emergence (DAE) to avoid significant foliar damage to peanut (Wilcut and Swann, 1990). While paraquat can cause injury to peanut even when applied correctly, this damage does not correlate to yield loss (Wilcut et al., 1989). Paraquat is typically tank-mixed with herbicides having multiple modes of action to broaden the weed control spectrum, provide longer weed control, and reduce crop injury (Wilcut et al., 1995). Producers can reduce injury to peanut while also increasing the flexibility of the application window by tank-mixing bentazon (3-91-methylethyl)-1H-2,1,3- benzothiadiazin-4(3H)-one 2,2-dioxide) with paraquat (Shaner, 2014; Wehtje et al., 1992).
Bentazon acts as a safener by reducing both paraquat injury on peanut and efficacy on susceptible weed species (Wehtje et al., 1992). Wehtje et al. (1992) described fluctuating control of multiple weed species when bentazon was tank-mixed with paraquat. Smallflower morningglory (Jacquemontia tamnifolia L.) is known to be susceptible to bentazon and tolerant to paraquat. Bentazon in combination with other herbicides, such as fluazifop, reduced control of annual grass weed species because of the herbicide's antagonistic nature (Grichar and Boswell, 1987). However, paraquat must be applied effectively and timely in order to obtain adequate control of broadleaf weed species, such as sicklepod (Senna obtusifolia [L.]) or Florida beggarweed (Desmodium tortuosum [S.w.]) (Wilcut et al., 1995).
Peanut producers in Georgia have become interested in using a liquid fertilizer replacement for bentazon in their paraquat POST tank-mixtures. Specifically, Ele-Max® Nutrient Concentrate (inorganic liquid nutrient [ILN]) (Helena Chemical Company, Colliervile, TN, 38017), is an 11-8-5 (N-P2O5- K2O) with ethylenediaminetetraacetic acid (EDTA) chelated micronutrients (B, Fe, Mn, Cu, Zn, Co, and Mo) (Anonymous, 2017). No information is available on the impact and viability of ILN as a safener for paraquat in POST tank-mixtures on runner-type peanut cultivars. The main objective of the field study was to evaluate the safening potential of ILN when applied alone or in combination with other herbicides on peanut vegetation, pod yield, and grade. The main objective of the greenhouse study was to evaluate the phytotoxic effects and safening potential of ILN when applied alone or in combination with other herbicides on broadleaf and grass weed species. Therefore, studies were conducted to evaluate the phytotoxic effects and safening potential of ILN when applied alone or in combination with paraquat tank-mixtures on peanut and weed species.
Materials and Methods
Peanut Field Study
Two separate experiments were conducted, one managed with supplemental irrigation and one rainfed. The first location was the University of Georgia (UGA) Southwest Georgia Research and Education Center (SWREC) in Plains, GA (32.0468, -84.3662, which had a Greenville sandy loam (fine, kaolinitic, thermic Rhodic Kandiudult) soil with 3.8% organic matter (OM), 60% sand, 10% silt, and 30% clay. The second location was the UGA Attapulgus Research and Education Center in Attapulgus, GA (30.7608, -84.4870, which had an Orangeburg loamy sand (fine-loamy, kaolinitic, thermic Typic Kandiudult) soil with 1.5% OM, 86% sand, 6% silt, and 8% clay. Soil pH was 6.0 and 5.6, respectively. In 2016, only the irrigated field experiment was conducted at the UGA Attapulgus Research and Education Center, while both the irrigated and non-irrigated field experiments were conducted at the UGA SWREC. In 2017, the irrigated and non-irrigated field experiments were conducted at the UGA SWREC only.
All trial sites were prepared by disc harrowing, moldboard plowing (30 cm deep), followed by rotary-tillage. Beds were 1.8 m wide (2 rows per bed). Plot length varied by site and year due to differing field dimensions for the given site-year. In Attapulgus plot length was 7.6 m. At the SWREC, plot length in 2016 was 12.2 m while it was 9.1 m in 2017. Peanuts were planted in two single rows (90 cm spacing) on 2 May 2016 in Attapulgus, and 16 May 2016 and 2 May 2017 at the SWREC. Planting was done using a two row Monosem air planter (Monosem-Inc., Edwardsville, KS) at 19 seeds/m of row to a depth of 5 cm. Georgia-06G (Branch, 2007) was planted for all field experiment site-years. Fertilizer applications were applied according to a pre-plant soil test recommendation for peanut at each site and followed UGA Extension recommendations (Harris, 2018). Protective fungicide applications based on the high-risk management program from the Peanut Rx were followed (Kemerait et al., 2017). Fungicides were initiated at the R1 growth stage (Boote, 1982) and continued on 14 d intervals. All plots were maintained weed-free. Irrigation was applied in compliance with the UGA Peanut Production Guide Checkbook method (Porter, 2017; Stansell and Pallas, 1985; Stansell et al., 1976).
The trial was arranged as a 4 by 2 factorial (four levels of herbicide treatments and two levels of ILN treatments) in a randomized complete block design (RCB) with four replications. The herbicide treatments were paraquat (0.21 kg ai/ha) plus nonionic surfactant (0.25% v/v), paraquat (0.21 kg ai/ha) plus S-metolachlor (1.06 kg ai/ha), paraquat (0.21 kg ai/ha) plus S-metolachlor (1.06 kg ai/ha) plus acifluorfen (0.28 kg ai/ha) plus bentazon (0.56 kg ai/ha), and a non-treated control (NTC). The ILN treatments were none (-) and with (+) ILN (2.75 kg plant nutrients/L product). All herbicide treatment plots received a preemergence (PRE) application of flumioxazin (0.11 kg ai/ha) plus pendimethalin (0.9 kg ai/ha) at planting. Irrigated and non-irrigated PRE applications were made immediately after planting and incorporated with 1.3 cm of irrigation for activation. Non-irrigated trials remained as rainfed for the remainder of the season. POST herbicide treatments were applied 28 d after planting.
Data collection included visual injury and stunting ratings of 0 (none) to 100% (complete necrosis/death), vegetative biomass (g/plant), peanut pod biomass (g/plant), peanut pod yield (kg/ha), and grade (total sound mature kernels % [TSMK]). Visual estimates of foliar injury (chlorosis/necrosis) were evaluated at 3, 7, 11, and 14 d after treatment (DAT). Visual stunting was measured at 3, 7, 11, 14, 21 and 28 DAT. Peanut plant biomass data was collected at the V8, R2, and R7 to R8 growth stages (Boote, 1982). Three plants were randomly sampled from each plot and dried in a forced-air dryer for 72 hr. Pods and vegetation were separated by hand and weighed.
Peanut maturity was determined by the hull scrape method (Williams and Drexler, 1981). Peanut digging and inversion were conducted using a 2-row digger in Attapulgus and a 6-row digger in Plains (Kelley Mfg. Co., Tifton, GA). Pods were allowed to dry to approximately 10 to 15% moisture before harvest with a 2-row KMC harvester (Kelley Mfg. Co., Tifton, GA) in Attapulgus and a Columbo harvester (Columbo North America, Adel, GA) in Plains. Yields were then adjusted to 7% moisture for uniformity. Grade was determined following the USDA-AMS grading standards by the USDA Federal-State Inspection Service in Tifton, GA (USDA-AMS, 1997).
Analysis of variance (ANOVA) was conducted for all response variables using PROC MIXED in SAS 9.4 (SAS Institute Inc., Cary, NC, 27513). Preliminary analyses were performed on all response variables to measure the effects of site-year as a fixed effect. Independent variables were site-year, herbicide, and ILN. Significant interactions were detected between site-year and treatments for response variables and were the result of magnitude of differences among treatments but with similar trends of response across site-year. Subsequent analyses were done for all data combined across site-year. Herbicide, ILN, and their interactions were considered fixed effects, while site-year and replication were considered random effects. Irrigated and non-irrigated experiments were analyzed separately. Pairwise comparison of least square means for all response variables were made using the Tukey's honestly significant difference test (𝜶=0.05) (Tukey, 1949).
Weed Species Greenhouse Study
Greenhouse trials were conducted at the UGA Tifton Campus-Coastal Plains Experiment Station in Tifton, Georgia. This experiment evaluated the phytotoxic effects and efficacy of ILN in POST tank-mixtures with paraquat on multiple weed species. Large crabgrass (Digitaria sanguinalis [L.]), Florida beggarweed, bristly starbur (Acanthospermum hispidum), Palmer amaranth (Amaranthus palmeri [S.w.]), prickly sida (Sida spinosa [L.]), pitted morningglory (Ipomoea lacunosa [L.]), smallflower morningglory, and sicklepod seed were used. Each species was planted into two adjacent cells in eighteen cell plastic flats measuring 51 × 26 cm with Miracle-Gro® Potting Mix (Scotts Miracle-Gro, Marysville, OH, 43040). After germination, plants were thinned to two per cell. Plants were fertilized biweekly using 24-8-16 Miracle-Gro® liquid fertilizer (Scotts Miracle-Gro, Marysville, OH, 43040) watered twice daily, and managed under supplemental growth lights (500 μmol/m2/s1) for the duration of the study.
This experiment was conducted as a RCB design with a split-plot restriction on randomization with four replications. The experiment was repeated twice in time during 2017. Herbicide treatments (flats) were the whole plot factor while weed species (cells) were the subplots. Herbicide treatments included paraquat (0.21 kg ai/ha) plus nonionic surfactant (0.25% v/v), paraquat (0.21 kg ai/ha) plus S-metolachlor (1.06 kg ai/ha), paraquat (0.21 kg ai/ha) plus S-metolachlor (1.06 kg ai/ha) plus acifluorfen (0.28 kg ai/ha) plus bentazon (0.56 kg ai/ha), ILN (2.75 kg plant nutrients/L product), paraquat (0.21 kg ai/ha) plus nonionic surfactant (0.25% v/v) plus ILN (2.75 kg plant nutrients/L product), paraquat (0.21 kg ai/ha) plus S-metolachlor (1.06 kg ai/ha) plus ILN (2.75 kg plant nutrients/L product), paraquat (0.21 kg ai/ha) plus S-metolachlor (1.06 kg ai/ha) plus acifluorfen (0.28 kg ai/ha) plus bentazon (0.56 kg ai/ha) plus ILN (2.75 kg plant nutrients/L product), and a NTC.
Weeds were treated at the 2-3 true leaf stage. All applications were made using a moving belt sprayer calibrated to spray 187 L/ha at 3 kph. Visual estimates of injury (same scale previously described) for chlorosis/necrosis were evaluated at 3 and 7 DAT. Above ground biomass (% of the control) was measured at 7 DAT after the visual injury ratings were recorded. Above ground re-growth biomass (% of the control) was collected 14 d after the initial biomass harvest. ANOVA was conducted for all response variables using PROC MIXED in SAS 9.4 (SAS Institute Inc., Cary, NC, 27513). Replication was treated as a random effect while weed species and herbicide treatment were treated as fixed effects. Data were combined over iteration. Pairwise comparison of least square means for all response variables were made using the Tukey's honestly significant difference test (𝜶=0.05) (Tukey, 1949).
Results and Discussion
Peanut Field Study
Irrigated Peanut
Herbicide by ILN interactions were detected for leaf burn at 3 DAT and stunting at 3 and 28 DAT (Table 1). Despite interactions occurring, when the data were separated according to the interactions there was no clear trend. Because of this, data were not separated by the interaction for leaf burn and are presented for the individual effects. Herbicide treatment was significant for all ratings of leaf burn and stunting (Table 1). ILN treatment was significant for leaf burn at 3 DAT and all ratings of stunting (Table 1). Vegetative biomass (V8, R2, and R7-R8 stage), pod biomass (R2, and R7-R8 stage), yield, and grade (Table 2) were not different.
ANOVA for the effect of inorganic liquid nutrients (ILN) in paraquat tank-mixtures on irrigated peanut in Attapulgus (2016) and Plains (2016 and 2017) and non-irrigated peanut in Plains (2016 and 2017). Data were combined for analysis across location (Attapulgus and Plains) and year (2016 and 2017).a
ANOVA for the effect of inorganic liquid nutrients (ILN) in paraquat tank-mixtures on irrigated peanut in Attapulgus (2016) and Plains (2016 and 2017) and non-irrigated peanut in Plains (2016 and 2017). Data were combined for analysis across location (Attapulgus and Plains) and year (2016 and 2017).a
For leaf burn, herbicide injury was greatest at 3 DAT. However, there were no differences among herbicides at 7 DAT (Table 3). Paraquat plus S-metolachlor treated peanut foliage had the greatest amount of injury at 3 DAT (30%). Including acifluorfen plus bentazon with S-metolachlor significantly reduced injury levels (16%). From 14 DAT, there were no differences between herbicide tank-mixtures with all treatments showing greater injury than the NTC. At 3 DAT, including ILN (12%) showed a reduction in injury when compared to excluding ILN (20%) in the mixture.
Across all ratings, paraquat plus S-metolachlor showed the greatest amount of stunting (25 and 15% at 3 and 7 DAT respectively) (Table 4). There were no differences between paraquat plus NIS and paraquat plus S-metolachlor plus acifluorfen plus bentazon treatments at any rating. Including ILN reduced peanut stunting injury between 3-7% across all ratings compared to when it was not included in the tank-mix (Table 5). While ILN did reduce peanut foliar injury and stunting, it had no effect on biomass, yield, or grade (Table 2). The herbicide tank-mixtures also did not influence yield or grade (Table 2), which is noted in previous studies (Everman et al., 2008; Wilcut et al., 1989).
Non-Irrigated Peanut
Herbicide by ILN interactions were present for leaf burn at 3 DAT and stunting at 3 and 14 DAT (Table 1). Despite interactions occurring when the data were separated accordingly, there was no clear trend. Because of this, data were not separated by the interaction for leaf burn and are presented for the individual effects. Herbicide treatment was significant for leaf burn and stunting at all ratings, except 28 DAT. ILN treatment was significant for leaf burn (3 and 7 DAT) and stunting (3, 7, and 11 DAT) (Table 1). No effect was significant for vegetative biomass (V8, R2, and R7-R8 stage), pod biomass (R2, and R7-R8 stage), yield, and grade (Table 2).
At 3 DAT, there were no differences among herbicide treatments for peanut leaf burn ranging from 20 to 25%, but all were greater than the NTC (Table 3). By 7 DAT, foliar injury from paraquat plus S-metolachlor was not different than paraquat plus S-metolachlor plus acifluorfen plus bentazon, however injury was greater than paraquat alone indicating peanut had slower recovery from S-metolachlor than from paraquat in non-irrigated conditions. By 7 DAT, all foliar injury levels were 10% or less. Overall, not including ILN resulted in greater leaf burn at 3 and 7 DAT (Table 6).
For stunting, paraquat plus S-metolachlor showed the greatest amount of injury across all ratings (25 and 15% at 3 and 7 DAT respectively). Including acifluorfen plus bentazon in tank-mix reduced stunting but it did not differ from paraquat plus NIS (Table 4). Including ILN reduced stunting between 5-10% for 3 and 7 DAT (Table 5). ILN had no effect on biomass, yield, or grade (Table 2). The herbicide tank-mixtures also did not influence yield or grade (data not reported), which is noted in previous studies (Everman et al., 2008; Wilcut et al., 1989).
Weed Species Greenhouse Study
Herbicide by weed species interaction was significant for all parameters except for regrowth biomass (Table 7). Herbicide and weed species had significant effects on leaf burn at 3 DAT, 7 DAT, and biomass.
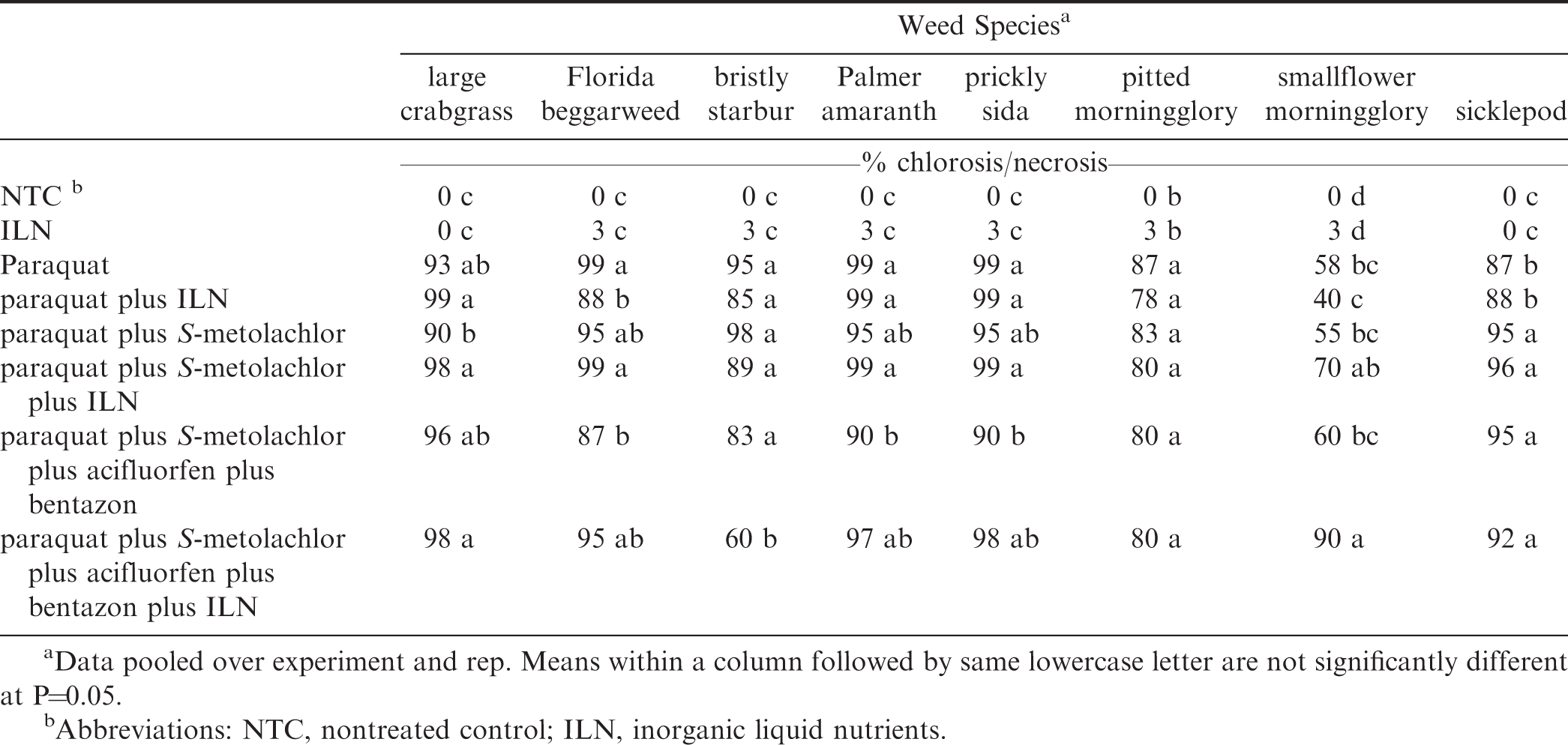
ILN alone did not cause any significant leaf burn across all weed species when compared to the NTC at 7 DAT (Table 8). Large crabgrass leaf burn was above 90% chlorosis/necrosis across herbicide treatments. Paraquat plus S-metolachlor was significantly lower than paraquat plus S-metolachlor plus ILN. Florida beggarweed had greater injury with paraquat than when ILN was added. Bristly starbur showed no differences between herbicide treatments except for paraquat plus S- metolachlor plus acifluorfen plus bentazon plus ILN which caused a reduction in leaf burn. For Palmer amaranth and prickly sida, paraquat plus S-metolachlor plus acifluorfen plus bentazon caused less injury than paraquat alone, paraquat plus ILN, and paraquat plus S-metolachlor plus ILN. For pitted morningglory, there were no differences between herbicide treatments. Smallflower morningglory injury was greatest from paraquat plus S-metolachlor plus ILN and paraquat plus S-metolachlor plus acifluorfen plus bentazon plus ILN. Sicklepod injury was greatest for any treatment that included S-metolachlor. These results showed significantly different trends for ILN in tank-mixture with paraquat and other POST herbicides.
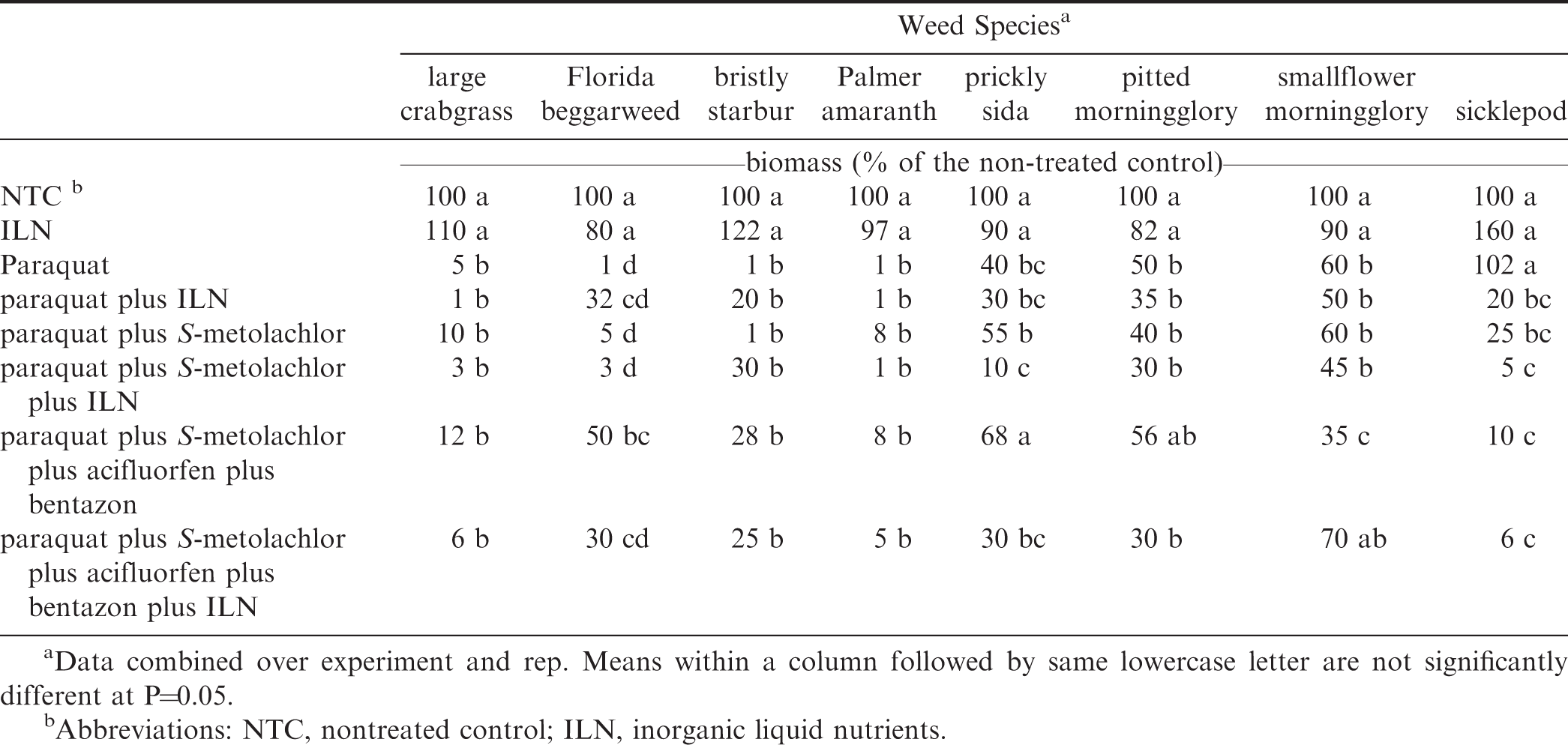
For biomass, ILN alone was not significantly different from the NTC across all weed species (Table 9). Large crabgrass, bristly starbur, Palmer amaranth, and pitted morningglory displayed similar trends for above ground biomass weights. There were no significant differences among herbicide treatments for these weed species, but they were all significantly lower than the NTC. However, the one exception was paraquat plus S-metolachlor plus acifluorfen plus bentazon which showed no difference from the NTC for pitted morningglory. Sicklepod biomass was not affected by paraquat alone. Paraquat plus S- metolachlor plus ILN, paraquat plus S-metolachlor plus acifluorfen plus bentazon, and paraquat plus S-metolachlor plus acifluorfen plus bentazon plus ILN all caused a reduction in biomass when compared to paraquat, ILN, and the NTC for sicklepod. Florida beggarweed and prickly sida had similar injury trends. For both weed species, paraquat plus S- metolachlor plus ILN resulted in lower biomass percentages compared to the standard tank- mixture of paraquat plus S-metolachlor plus acifluorfen plus bentazon. For these two weed species, the addition of ILN to paraquat plus S-metolachlor plus acifluorfen plus bentazon caused no changes in biomass. Previous research shows that while including bentazon in tank-mixture is done to prevent injury to peanut, it can also reduce injury on weed species, resulting in lower control percentages (Wehtje et al. 1992). Smallflower morningglory responded differently than the other weed species examined. There were no differences in the herbicide treatments that did not contain acifluorfen plus bentazon. The paraquat plus S-metolachlor plus acifluorfen plus bentazon treatment reduced biomass when compared to paraquat plus S-metolachlor plus ILN and paraquat plus S-metolachlor plus acifluorfen plus bentazon plus ILN. Typically, bentazon is used in tank-mix with paraquat to control smallflower morningglory (Prostko 2018).
Our results indicated that there were similar trends in chlorosis/necrosis (%) among weed species. All herbicide treatments without acifluorfen plus bentazon were not different for large crabgrass, Florida beggarweed, pitted morningglory, bristly starbur, and sicklepod. At 7 DAT, adding S-metolachlor in the tank-mixture caused more injury, with or without ILN on sicklepod. Adding acifluorfen plus bentazon in the tank-mixture caused a reduction in leaf burn for bristly starbur. However, ILN had no influence on the injury on bristly starbur. Large crabgrass, Florida beggarweed, Palmer amaranth, pitted morningglory, smallflower morningglory, and sicklepod were injured the most by paraquat plus S-metolachlor plus acifluorfen plus bentazon plus ILN.
To determine if ILN could be used as a replacement for bentazon in tank-mixture with paraquat, we compared the paraquat plus S-metolachlor plus ILN treatment and paraquat plus S-metolachlor plus acifluorfen plus bentazon treatment. For large crabgrass, bristly starbur, Palmer amaranth, pitted morningglory, and sicklepod there were no differences in leaf burn and biomass for the two treatments. For Florida beggarweed and prickly sida, including ILN resulted in greater injury (more leaf burn and reduced biomass). However, for Florida beggarweed, paraquat plus S-metolachlor and paraquat plus S-metolachlor plus ILN were not different. This determined that ILN is not the cause of the increased injury and bentazon actually caused a decrease in injury. Smallflower morningglory was the only weed species evaluated that had greater biomass when ILN was included in the tank-mixture instead of bentazon plus acifluorfen.
Summary and Conclusions
The irrigated and non-irrigated studies showed similar trends in injury (leaf burn and stunting) levels. Paraquat plus S-metolachlor caused the most damage on irrigated and non-irrigated peanut. However, in the irrigated study the addition of bentazon plus acifluorfen had more of an impact on reducing injury. Initially, ILN treatments had reduced leaf burn and stunting injury shortly after application on both irrigated and non-irrigated peanut. For the irrigated study, there were no differences in foliar injury among herbicide treatments by 7 DAT while it took until 14 DAT for foliar injury to show no differences among herbicide treatments for the non-irrigated study. Neither herbicide treatment nor use of ILN had an effect on vegetative biomass, pod biomass, yield, or grade. While injury was greatest directly following application, peanut was able to recover with no yield or grade loss, similar to previous studies (Everman et al., 2008; Wilcut et al., 1989).
Overall, including ILN in tank-mixture only slightly reduced peanut injury up to 7 DAT but had no impact on injury after one week. ILN also had no effect on yield or grade for both irrigated and non-irrigated peanut. Additions of ILN improved the control of sicklepod and prickly sida but did not improve the control of large crabgrass, Florida beggarweed, bristly starbur, Palmer amaranth, and pitted morningglory.
While the addition of ILN to the various paraquat tank-mixtures initially reduced injury, it did not correspond to increases in yield or grade. The variability in weed control, transient injury mitigation, and no yield increase indicates that Georgia peanut growers will receive no benefit for including ILN in their paraquat tank-mixtures but if needed to improve crop nutrition, ILN will not reduce weed control.
Acknowledgements
This research was supported by the National Peanut Board and the Georgia Peanut Commission. The authors would like to thank Wen Carter, Kristen Pegues, Sidney Cromer, and Nick Hurdle for technical assistance. Seed was donated by the Georgia Seed Development Commission. This project was also supported as part of Federal Hatch project #GEO00273.
Literature Cited
Anonymous 2017 Ele-Max® nutrient concentrate label. Helena® Chemical Company. 225 Schilling BLVD, Collierville, TN. 1-3.
K. J Boote, (1982). Growth stages of peanut (Arachis hypogaea L.). Peanut Sci 9: 35- 40.
B Branch, (2007). Registration of 'Georgia-06G' peanut. J. Plant Reg 1: 120.
Buchanan, G. A., D. S Murray, and E. W Hauser 1982 Weeds and their control in peanuts In: H. E Patteeand C. T Young, eds. Peanut Sci. and Technol. American Peanut Research Educ. Soc. 206-249.
W. J., Everman, S. B Clewis, W. E Thomas, I. C Burke, and J. W Wilcut (2008). Critical period of weed interference in peanut. Weed Technol 22: 63- 67.
W. J., Grichar, and T. E Boswell (1987). Herbicide combinations in peanut. Weed Technol 1: 290- 293.
G Harris, (2018). Peanut fertility checklist. In UGA Peanut Production Quick Reference Guide. Georgia Coop. Ext. Serv., Athens, GA. 3.,
R., Kemerait, A Culbreath, J Beasley, E Prostko, T Brenneman, S Tubbs, R Srinivasan, M Abney, S Monfort, A Rabinowitz, G. B Tillman, N Dufault, D Rowland, Mulvaney, ().
Hagan, J Sarver, D Anco and N Smith 2017 Peanut Rx, minimizing diseases of peanut in the southeastern United States Univ. of Georgia Coop. Ext. Serv., Athens, GA.
Porter W.M 2017 Peanut irrigation update pp. 19- 24 In: W.S Monfort (ed.) 2017 Peanut Update Spec. Publ. CSS-17-0118. Univ. of Georgia, Athens, GA.
Prostko, E. P 2018 Peanut weed control In: Georgia pest management handbook. UGA Ext. Special Bulletin 28, Athens, GA 215-234.
Rabinowitz, A. N and A. R Smith 2017 Peanut outlook and cost analysis In: W.S Monfort (ed.) 2017 Peanut Update Spec. Publ. CSS-17-0118. Univ. of Georgia, Athens, GA.
D. L Shaner, (2014). Herbicide Handbook. Weed Sci. Soc. Amer, 71- 337.
J. R., Stansell, and J. E Pallas (1985). Yield and quality of Florunner peanut to applied drought at several growth stages. Peanut Sci 12: 64- 70.
J. R., Stansell, J. L Shepherd, J. E Pallas, R. R Bruce, N. A Minton, D. K Bell, and L. W Morgan (1976). Peanut responses to soil water variables in the Southeast. Peanut Sci 3: 44- 48.
J. W Tukey, (1949). Comparing individual means in the analysis of variance. Biometrics 5: 99- 114.
USDA-AMS 1997 United States standards for grades of shelled runner type peanuts Available at http://www.ams.usda.gov/AMSv1.0/getfile?dDocNameSTELPRDC5050496 (verified 20 April 2018) USDA Agric. Marketing Serv., Washington, D.C.
USDA-NASS 2019 Survey of peanut acres harvested in the United States USDA National Agricultural Statistics Service quick stats https://quickstats.nass.usda.gov/results/4D5FEDBA-6C1D-3D70BAE1-6EA8AA74C437 (accessed 1 June 2020).
G., Wehtje, J. A McGuire, R. H Walker, and M. G Patterson (1986). Texas panicum (Panicum texanum) control in peanuts (Arachis hypogaea) with paraquat. Weed Sci 34: 308- 311.
G. R., Wehtje, J. W Wilcut, and J. A McGuire (1992). Influence of bentazon on the phytotoxicity of paraquat to peanuts (Arachis hypogaea L.) and associated weeds. Weed Sci 40: 90- 95.
J. W., Wilcut, and C. W Swann (1990). Timing of paraquat applications for weed control in virginia-type peanuts. Weed Sci 38: 558- 562.
J. W., Wilcut, G. R Wehtje, T. A Cole, V Hicks, and J. A McGuire (1989). Postemergence weed control systems without dinoseb for peanuts. Weed Technol 37: 385- 391.
Wilcut, J. W., A. C York, W. J Grichar, and G. R Wehtje 1995 The biology and management of weeds in peanut In: H. E Patteeand C. T Young, eds Peanut Sci. and Technol. American Peanut Research Educ. Soc., Yoakum, TX 207-244.
E.J., Williams, and J. S Drexler (1981). A non-destructive method for determining pod maturity.. Peanut Sci. 8: 134- 141.
Notes
- Graduate Research Associate, Professor, Professor and Professor, respectively. Department of Crop & Soil Sciences, The University of Georgia, 2360 Rainwater Drive, Tifton, GA 31793; [^]
- Assitant Professor, Crop, Soil and Environmental Sciences, Auburn University, Funchess Hall, Auburn, AL 36345 [^] *Corresponding author Email: kayla.eason25@uga.edu
Author Affiliations