Introduction
Athelia rolfsii (Curzi) C.C. Tu & Kimbr. (= Sclerotium rolfsii Sacc.; Xu et al., 2010) is a cosmopolitan soilborne pathogen distributed primarily in warmer areas such as the southern U.S. (Aycock, 1966; Punja and Rahe, 2001). In addition to infecting more than 500 plant species including tomato, carrot, beet, sweet potatoes, and melon (Aycock, 1966; Jenkins and Averre, 1986), A. rolfsii is considered to be one of the most economically damaging pathogens of peanut in the U.S. (Backman and Brenneman, 1997). In Georgia's 2015 season alone, the value of control costs and yield losses was estimated to be $59.7 million (Kemerait, 2015). Substantial yield losses to A. rolfsii are also experienced in peanut-producing regions of Africa (Subrahmanyam et al., 1997; Cilliers et al., 2000), Asia (Mayee and Datar, 1988), the Middle East (Grinstein et al., 1979), and South America (Marinelli et al., 1998). Cultivars with high resistance to A. rolfsii such as Bailey and Georgia-12Y are available, but most commercial cultivars obtainable today are less resistant (Kemerait et al., 2018).
Despite the need for more resistant cultivars, few laboratory assays have been developed for evaluating resistance to A. rolfsii in peanut and other crops. Akem and Dashiell (1991) found differences in resistance among soybean genotypes by inoculating detached shoots from 6-week-old plants with mycelial plugs. In addition to measuring lesion length over time, they also counted the number of sclerotia and percentage of germinable sclerotia produced by each soybean genotype. Pratt and Rowe (2002) were able to discriminate between resistant and susceptible genotypes of alfalfa and obtained similar results from both excised leaflets and whole plants inoculated with mycelial plugs of A. rolfsii. A greenhouse assay for evaluating resistance in Jerusalem artichoke was developed in Thailand using inoculum produced in sorghum broth and seeds (Sennoi et al., 2010). More recently, Xie et al. (2014) used sclerotia to inoculate tomato, pepper, and peanut (cv. Georgia Green) with 19 isolates of A. rolfsii. However, tomato plants were more susceptible than pepper and peanut plants at the same age (8 weeks after germination), and the assay was adjusted so that tomatoes were inoculated using four sclerotia instead of the ten used for pepper and peanut (Xie et al., 2014). Bera et al. (2016) used sorghum seed colonized by A. rolfsii to screen 25 accessions of wild species in pots for resistance. Using this approach, two resistant accessions with less than 13 and 14% mortality (Arachis pusilla DGR 12047 and Ar. appresipila ICG 8945, respectively), and two moderately resistant accessions with 25 and 26% mortality (Ar. monticola ICG 8135 and Ar. duranensis ICG 8204, respectively), were identified (Bera et al., 2016). Finally, the most extensive work on developing a laboratory assay to evaluate A. rolfsii resistance in peanut was conducted by Shokes and colleagues (1996). Five inoculation methods, varying from mycelium slurries to colonized oat grains, were used to inoculate 7-week-old Florunner plants in the greenhouse and field. The most effective inocula were germinating sclerotia on agar disks and clothespins impregnated with mycelium and potato dextrose broth (PDB)(Shokes et al., 1996). The agar disk technique was later used to screen 11 genotypes in two locations for three years to successfully identify resistant germplasm (Shokes et al., 1998).
Multi-year, replicated field trials are generally the best way to evaluate disease resistance (Brenneman et al., 1990, 2014), but field trials are labor-, time-, and space-intensive. Unfavorable environmental conditions and uneven distribution of inoculum (Shew et al., 1984) may also negatively affect field results. In addition, it may be difficult to procure enough seed of non-standard peanut genotypes such as wild species for replicated field plots (Bera et al., 2016). In contrast, laboratory-based resistance assays cannot accurately reflect field conditions but may be useful for pre-screening material or for screening entries for which seed is limited. Because one of our long-term goals is to evaluate PI accessions of wild Arachis species for disease resistance, our objective was to develop an assay, first using cultivated genotypes, for screening intact peanut plants for resistance to Athelia rolfsii.
Materials and Methods
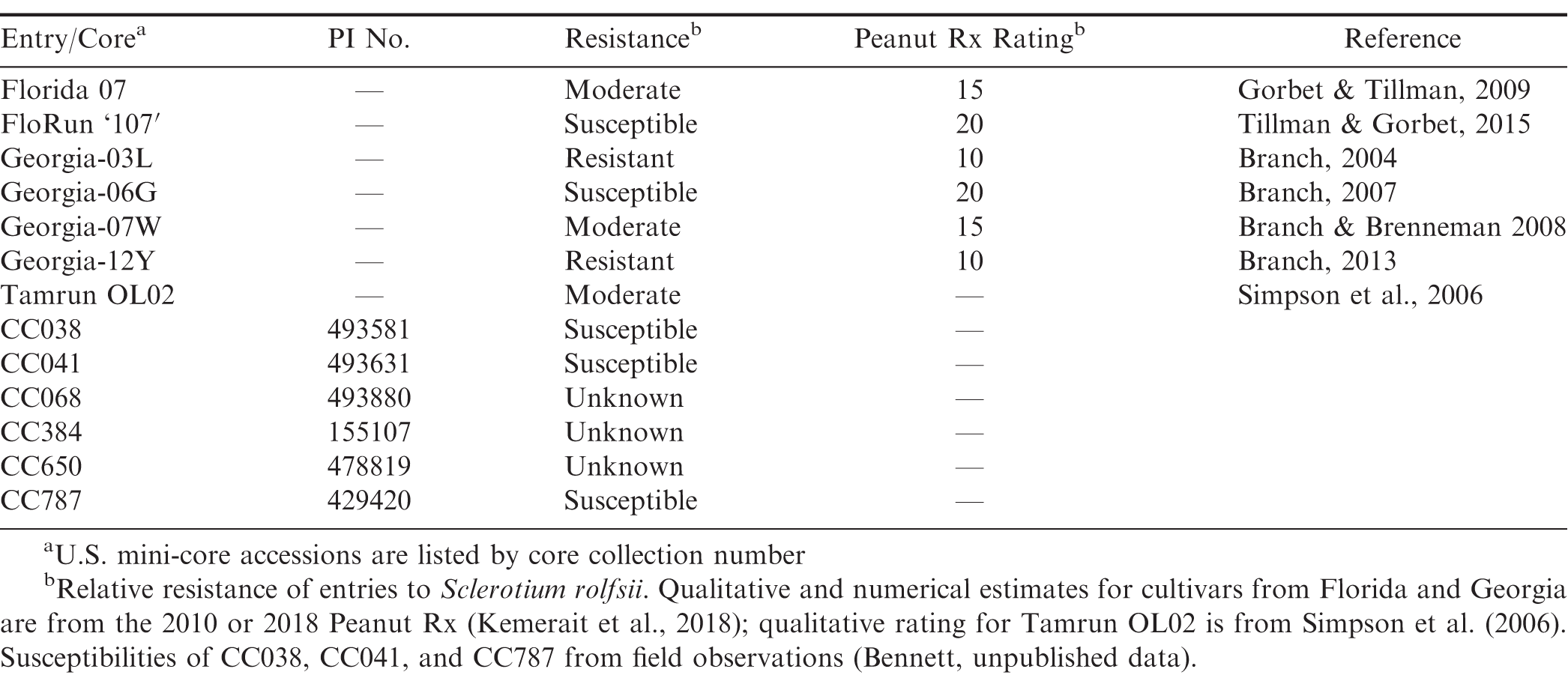
A total of thirteen peanut genotypes were used in the assays (Table 1). Cultivars included the resistant Georgia-03L (Branch, 2004; Woodward et al., 2008; Chapin et al., 2010; Culbreath et al., 2010) and Georgia-12Y (Branch, 2013; Branch and Brenneman, 2015); moderately resistant Florida-07 (Gorbet and Tillman, 2009), and Georgia-07W (Branch and Brenneman, 2008), and Tamrun OL02 (Simpson et al., 2006); and moderately susceptible FloRun '107' (Tillman and Gorbet, 2015) and Georgia-06G (Branch, 2007). Cultivars Georgia-03L, Georgia-12Y, Florida-07, Georgia-07W, FloRun '107', and Georgia-06G are rated 10, 10, 15, 15, 20, and 20, respectively, by Peanut Rx (Kemerait et al., 2018). Susceptible controls also included U.S. mini-core accessions CC038 (CC, core collection number; PI 493581), CC041 (PI 493631), and CC787 (PI 429420); these accessions appeared particularly susceptible to A. rolfsii over multiple years of field experiments (Bennett, unpublished data). Lastly, the following three mini-core accessions with unknown susceptibilities to A. rolfsii were also included: CC068 (PI 493880), CC384 (PI 155107), and CC650 (PI 478819). CC068 and CC650 are resistant to Sclerotinia minor in the field (Bennett et al., 2018).
Plants were grown in a greenhouse maintained at 22 to 32°C. Three seeds of each entry were planted in 11-cm-diam. pots filled with Metro-Mix 350 (Sun Gro Horticulture). Two weeks after planting, all but one seedling was culled. Plants were fertilized at 5 and 7 weeks after planting with 30 mL of 0.2% ammonium nitrate. At 8 weeks after planting, plants were inoculated with a virulent isolate of A. rolfsii (Ar-15-1A), which was originally collected in 2015 from a diseased plant in Fort Cobb, Oklahoma. The fungus was grown on 90-cm Petri plates filled with 15 mL of full-strength potato dextrose agar dispensed with a peristaltic pump (UniSpense, Wheaton). Cultures were incubated in the dark at 28.5°C (near optimum temperatures for A. rolfsii; Aycock, 1966) and 2-d-old cultures were used for inoculations.
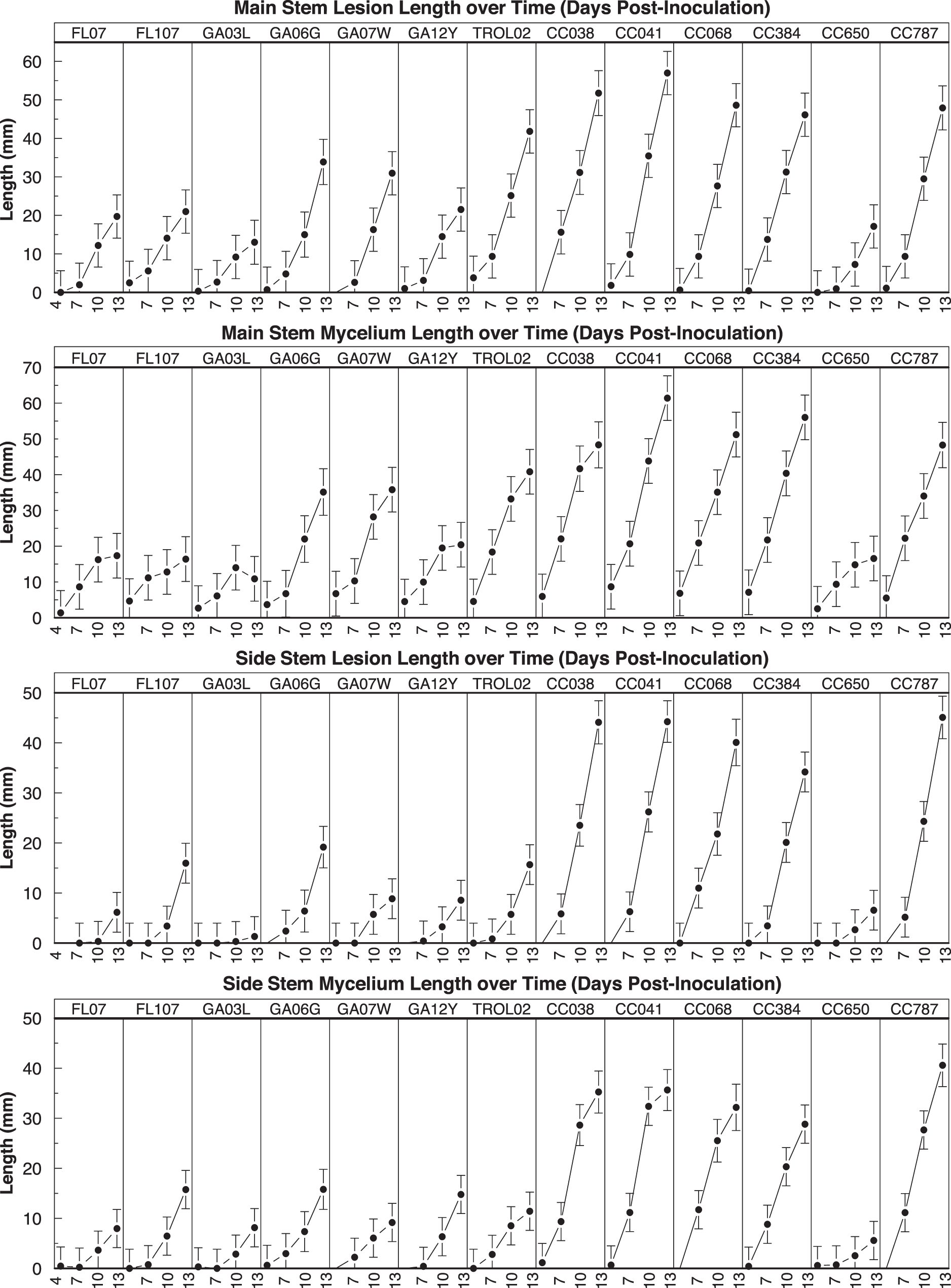
New or old flowers present on the plants were removed prior to inoculation to reduce sporulation of other fungi. Plants were inoculated at the base of a side stem arising from the second node. A sterile, half-circle of a cotton cosmetic pad was soaked with sterile water. A 9-mm-diam. agar plug was taken from the margin of a colony and placed mycelium-side up on center of the cotton pad. The pad was placed below the side branch and wrapped loosely but securely, so that the mycelium and agar plug were in direct contact with the stem. Inoculated plants were misted with reverse osmosis water, were placed on bath towels saturated with water, and covered with large clear plastic storage bins to maintain high humidity. Pots were arranged in a randomized complete block design with three replications in a growth chamber set at 28.5°C and 14-h photophase with fluorescent lights. Towels were wetted daily. To monitor temperature and relative humidity, two HOBO sensors were placed inside the growth chamber. Data were collected at 4, 7, 10, and 13 days after inoculation. Lesion length was measured on the main stem, as well as on the side branches arising from the first node, using digital calipers (Mitutoyo America). In preliminary experiments, mycelial growth appeared to be greater in susceptible genotypes, so length of mycelium on the main stem and first node side stems was also measured. If the plant or branch died before the end of the experiment, the measurements were recorded as missing data. The experiment (trial) was conducted four times between January and March 2016.
All analyses were conducted in SAS Version 9.4 (SAS Institute, Cary, NC). For analyses of the side branch data, the mean of the two branches were used. Differences among entries in disease progression (lesion length) and mycelial growth were analyzed using repeated measures ANOVA in PROC MIXED with TOEP covariance structure. Trial and block(trial) were used as random variables in the model. The SLICE option was used to examine differences among and within entries at 4, 7, 10, and 13 days after inoculation. In addition, the area under the disease progress curve (AUDPC) for lesion length and mycelial growth was estimated using the formula of Shaner and Finney (1977). To examine consistency among trials in addition to differences among entries, AUDPC means were compared with PROC GLIMMIX using a split-plot design with trial as the whole plot and entry as the subplot. The SLICE option was used to check for differences among trials and entries. All pairwise comparisons were adjusted for Type I error with the ADJUST = SIMULATE option at 𝜶 = 0.05. Correlation analysis between lesion and mycelium lengths were conducted using PROC CORR.
Results and Discussion
A significant interaction was present between entry and time (P < 0.01) in all repeated measures analyses of main and side stems, indicating that differences among peanut entries in lesion length and mycelial growth were dependent on the d the measurements were taken. In addition, there were no significant differences among entries at days 4 or 7 after inoculation, regardless of stem type or response variable (P = 0.11 to 1) when interaction was examined by d (data not shown).
Main Stem Lesions and Mycelia
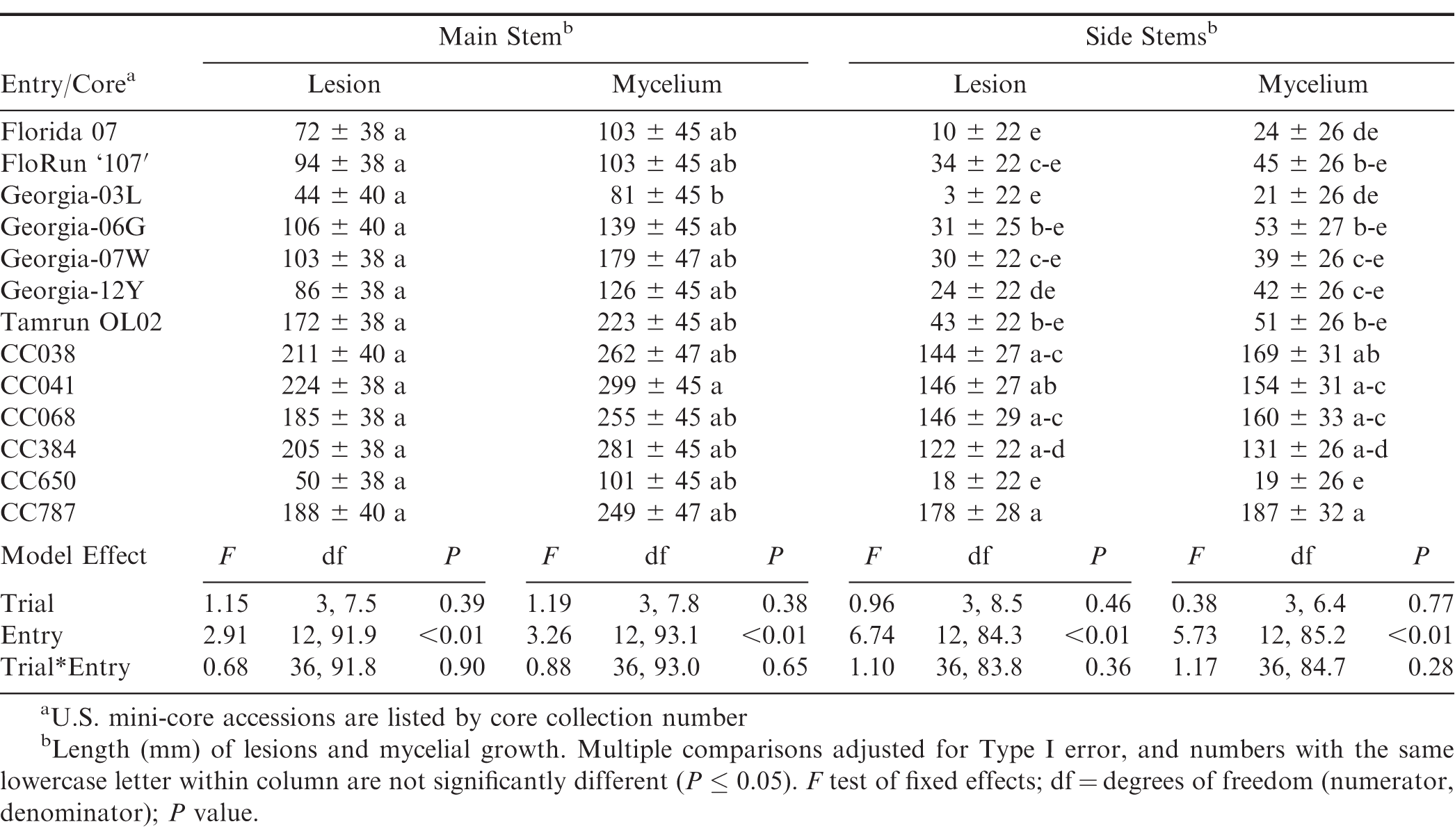
When the interaction between entry and d was examined by entry for main stem lesions, all genotypes had significantly different responses over the duration of the experiment except Georgia-03L (P = 0.32) and FloRun '107' (P = 0.06). In these two cultivars, lesion length at d 4 was not significantly different from days 7 to 13 when pairwise comparisons were corrected for Type I error. Mini-core entry CC650 was almost non-significant (P = 0.05). When the interaction was sliced by d, CC041 had the numerically longest lesions on d 10, which differed significantly from CC650 and Georga-03L (Table 2). By d 13, CC041, in addition to CC038, CC068, and CC787, had longer lesions than Florida-07, FloRun '107', Georgia-03L, Gerogia-12Y, and CC650.
Changes in main stem mycelium length were non-significant over time within the following five cultivars: Georgia-12Y (P = 0.13), Florida-07 (P = 0.34), Georgia-03L (P = 0.19), CC650 (P = 0.30), and FloRun '107' (P = 0.36). Among entries within d 10, mycelium length of CC041 was significantly greater than CC650, Georgia-03L, and FloRun '107' (Table 2). On d 13, the most mycelium was found in CC041, CC384, and CC068, and these entries differed from CC650 and all other cultivars except Georgia-06G, Georgia-07W, and Tamrun OL02. The correlation between main stem lesion and mycelium length over all measurement days was high (r = 0.92; P < 0.01).
Side Stem Lesions and Mycelia
Changes in mean lesion length for first-node side stems were not significant over time within the following cultivars: Florida-07 (P = 0.24), Georgia-07W (P = 0.25), Georgia-12Y (P = 0.29), CC650 (P = 0.54), and Georgia-03L (P = 0.99). These results, sliced by entry, best reflected expectations from PeanutRx points for A. rolfsii (Kemerait et al., 2018). When the interaction was examined by d, side stem lesion length in the mini-core accessions CC041 and CC787 at d 10 were significantly longer than in all cultivars except Georgia-06G (Table 2). At d 13, lesion length in CC384 did not differ from Georgia-06G or FloRun '107'
Patterns of mycelial growth among entries in side stems were similar to lesion length, except that Georgia-12Y was numerically, albeit not statistically, among the more susceptible cultivars at d 13. Mean mycelial growth in side stems did not differ by d within Tamrun OL02 (P = 0.20), Georgia-03L (P = 0.39), Georgia-07W (P = 0.40), Florida-07 (P = 0.46), and CC650 (P = 0.77). At 10 days after inoculation, CC068 and CC384 were not significantly different than Tamrun OL02, Georgia-06G, and FloRun '107' (Table 2). By d 13, CC384 and C068 did not differ from Georgia-06G, Florun '107', and Georgia-12Y. There was a high correlation between side stem lesion and mycelium length over all measurement days (r = 0.88; P < 0.01).
AUDPC
In the AUDPC analyses of main and side stem lesions and mycelium, the main effect of trial and the interaction between trial*entry were not significant, but the effect of entry was significant (Table 3). Despite large numerical differences, main stem lesion lengths were not statistically different among entries after adjusting for multiple comparisons. For main stem mycelium growth, CC041 had the greatest AUDPC and was significant larger than Georgia-03L.
AUDPC analyses of side stem data resulted in better separation of entries (Table 3). While none of the cultivars and CC650 differed significantly from each other, Tamrun OL02, FloRun '107' and Georgia-06G also did not differ from any of the susceptible mini-core accessions except CC787. In addition, AUDPC in Georgia-07W and Georgia-12Y were not significantly different from that of CC038, CC068, and CC384. Similar results were obtained from AUDPC analyses of mycelial growth.
These results clearly indicate that laboratory assays can identify peanut genotypes that are extremely susceptible physiologically to A. rolfsii. The most susceptible entries tested (CC038, CC041, CC068, CC384, and CC787) had the longest lesions and greatest mycelial growth regardless of stem type (Figure 1). However, physiological susceptibility does not correlate perfectly with field resistance because other characteristics such as open canopy structure can create microclimates unfavorable for disease (Blad et al., 1978; Dow et al., 1988; Bailey and Brune, 1997). For example, the cultivar Southwest Runner (Damicone et al., 2010) and several mini-core accessions including CC038, CC068, and CC787 (Bennett et al., 2018) exhibit resistance to Sclerotinia minor in the field but are susceptible when inoculated in the laboratory. While severe outbreaks of A. rolfsii are rare in current peanut production areas of Oklahoma, an inoculated field study of the U.S. mini-core that included three cultivars used here indicated that the A. rolfsii assays may correlate with field results. In the most favorable year for disease, resistant genotypes CC650 and Georgia-03L had 4% and 13% disease incidence, respectively. Susceptible genotypes Georgia-06G, CC038, CC041, CC068, and CC787 had 48%, 61%, 42%, 31%, and 42%, respectively (Bennett, in press). Disease incidence in Georgia-07W was numerically lower at 7% than in Georgia-03L but the two cultivars did not differ statistically.
More work is required to determine if the assays tested here can be improved to better identify resistant genotypes. Resistant entries Georgia-03L and CC650 consistently had shorter lesions and less mycelial growth in both stem types, but the assays were less able to identify resistance in Georgia-12Y, one of the most resistant cultivars available today (Kemerait et al., 2018). In addition, the assays were generally unable to discriminate among entries with intermediate resistances (relative to the highly susceptible mini-core accessions), which included most of the commercial cultivars tested. Georgia-06G and FloRun '107' are considered by Peanut Rx to be susceptible to A. rolfsii, but neither were statistically distinct from Florida-07, Georgia-07W, and Georgia-12Y, cultivars with better resistance scores. More susceptible commercial cultivars such as NC-V11 or AT-215 (30 points in the 2013 Peanut Rx, Kemerait et al., 2013) may have provided more separation, but these cultivars were not included in this study. Tamrun OL02 reportedly has some resistance (Simpson et al., 2006), but published field evaluations of this cultivar for A. rolfsii resistance are few and inconclusive (Grichar et al., 2010a). Anecdotally, Grichar and colleagues (2010b) observed that Tamrun OL02 is susceptible and less resistant to A. rolfsii than Tamrun 96, its recurrent parent. Results from these laboratory assays only indicate that Tamrun OL02 is neither extremely susceptible or resistant to A. rolfsii.
Despite limitations, these assays may be useful for pre-screening germplasm to identify accessions with high levels of physiological resistance. It also appears that the assays may be simplified without greatly compromising results. First, fewer days of data may be collected since there were no differences among entries at days 4 and 7 after inoculation. Second, the high correlation between lesion and mycelium length indicates that measuring either mycelia or lesions may be sufficient. Lesions caused by A. rolfsii were generally obscured by mycelia, particularly on days 4 and 7 after inoculation, and it would have been more efficient to measure only mycelium. Third, data collected from the side stems appeared demonstrate differences among entries better than data from the main stem (Tables 2 and 3), perhaps due to additional barriers for the pathogen. To infect the first node side stems, the fungus had to infect the second node side stem, move into the main stem, and finally move down and into first node. More work is also needed to confirm that lesion length in side stems best correlates with resistance in the field. Others have observed that field ratings taken after digging better correlated with yield (Rideout et al., 2002), and it is unknown how well laboratory assays relate to root/crown ratings. Ample evidence suggests phenotypic (e.g. pathogenicity and fungicide resistance) and genotypic diversity in A. rolfsii (Xie et al., 2014; Khatri et al., 2017), so a simple assay that could more easily accommodate the added complexity of multiple isolates would be helpful.
Acknowledgements
The author thanks Angela Harting and Courtney Peterson for technical assistance, as well as anonymous reviewers for helpful editorial suggestions. This research was supported by USDA-ARS CRIS Project No. 3072-21220-008-00D. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Literature Cited
C.N., Akem, and K.E Dashiell (1991). Detached shoot technique to evaluate the reaction of soybean cultivars to Sclerotium rolfsii. Crop Prot 10: 325- 327.
Aycock, R 1966 Stem rot and other diseases caused by Sclerotium rolfsii: or the status of Rolfs' fungus after 70 years N. C. Agric. Exp. Stn. Tech. Bull. 174.
Backman, P.A., and T.B Brenneman 1997 Stem rot Pages 36- 37 In Compendium of Peanut Diseases American Phytopathological Society, St. Paul, MN.
J.E., Bailey, and P.D Brune (1997). Effect of crop pruning on Sclerotinia blight of peanut. Plant Dis 81: 990- 995.
R.S., Bennett, K.D Chamberlin, and J.P Damicone (2018). Sclerotinia blight resistance in the US peanut mini-core collection. Crop Sci 58: 1306- 1317.
S.K., Bera, J.H Kamdar, S.V Kasundra, and P.P Thirumalaisami (2016). Identification of groundnut genotypes and wild species resistant to stem rot using an efficient field screening technique. Electron. J. Plant Breed 7: 61- 70.
B.L., Blad, J.R Steadman, and A Weiss (1978). Canopy structure and irrigation influence white mold disease and microclimate of dry edible beans. Phytopathology 68: 1431- 1437.
W.D Branch, (2004). Registration of 'Georgia-03L' peanut. Crop Sci 44: 1485- 1486.
W.D Branch, (2007). Registration of 'Georgia-06G' peanut. J. Plant Regist 1: 120.
W.D Branch, (2013). Registration of 'Georgia-12Y' peanut. J. Plant Regist 7: 151- 153.
W.D., Branch, and T.B Brenneman (2008). Registration of 'Georgia-07W' peanut. J. Plant Regist 2: 88- 91.
W.D., Branch, and T.B Brenneman (2015). Stem rot (white mold) and tomato spotted wilt resistance among peanut genotypes. Peanut Sci 42: 18- 22.
T.B., Brenneman, W.D Branch, and A.S Csinos (1990). Partial resistance of Southern Runner, Arachis hypogaea, to stem rot caused by Sclerotium rolfsii. Peanut Sci 17: 65- 67.
Brenneman, T.B., B.L Tillman, and N Dufault 2014 Phenotyping peanut diseases caused by soilborne pathogens Proc. Amer. Peanut Res. Educ. Soc. 46: 16- 17 (abstr.).
J.W., Chapin, J.S Thomas, T.G Isleib, F.M Shokes, W.D Branch, et al (2010). Field evaluation of Virginia-type peanut cultivars for resistance to tomato spotted wilt virus, late leaf spot, and stem rot. Peanut Sci 37: 63- 69.
A.J., Cilliers, L Herselman, and Z.A Pretorius (2000). Genetic variability within and among mycelial compatibility groups of Sclerotium rolfsii in South Africa. Phytopathology 90: 1026- 1031.
A., Culbreath, J Beasley, R Kemerait, E Prostko, T Brenneman, et al (2010). Peanut Rx: Minimizing diseases of peanut in the southeastern United States, the 2010 version of the peanut disease risk index. Athens, GA: Univ. of Georgia Coop. Ext. Serv.. , .
J.P., Damicone, C.C Holbrook, D.L Smith, H.A Melouk, and K.D Chamberlin (2010). Reaction of the core collection of peanut germplasm to Sclerotinia blight and pepper spot. Peanut Sci 37: 1- 11.
R.L., Dow, N.L Powell, and D.M Porter (1988). Effects of modification of the plant canopy environment on Sclerotinia blight of peanut. Peanut Sci 15: 1- 5.
D.W., Gorbet, and B.L Tillman (2009). Registration of 'Florida-07'peanut. J. Plant Regist 3: 14- 18.
Grichar, W.J., A.J Jaks, and M.R Baring 2010 a Peanut cultivar response to fungicide spray programs under light to moderate disease pressure Online. Crop Manag. doi: 10.1094/CM-2010-0706-01-RS.
Grichar, W.J., A.J Jaks, and J Woodward 2010 b Using prothioconazole plus tebuconazole for foliar and soilborne disease control in Texas peanut Online. Crop Manag. doi: 10.1094/CM-2010-0405-02-RS.
A., Grinstein, J Katan, A Abdul-Razik, O Zeidan, and Y Elad (1979). Control of Sclerotium rolfsii and weeds in peanuts by solar heating of soil. Plant Dis. Report 63: 1056- 1059.
S.F., Jenkins, and C.W Averre (1986). Problems and progress in integrated control of southern blight of vegetables. Plant Dis 70: 614- 620.
Kemerait, R.C 2015 Peanut Page 13 In Williams-Woodward, J., editor, Georgia Plant Disease Loss Estimates Univ. of Georgia, Coop. Ext. Serv.
Kemerait, R., A Culbreath, J Beasley, E Prostko, T Brenneman, et al 2013 Peanut Rx: Minimizing diseases of peanut in the southeastern United States, the 2018 version of the peanut disease risk index Univ. of Georgia Coop. Ext. Serv, Athens, GA.
Kemerait, R., A Culbreath, E Prostko, T Brenneman, S Tubbs, et al 2018 Peanut Rx: Minimizing diseases of peanut in the southeastern United States, the 2018 version of the peanut disease risk index Univ. of Georgia Coop. Ext. Serv, Athens, GA.
K., Khatri, S Kunwar, R.L Barocco, and N.S Dufault (2017). Monitoring fungicide sensitivity levels and mycelial compatibility groupings of Sclerotium rolfsii isolates from Florida peanut fields. Peanut Sci 44: 83- 92.
A., Marinelli, G.J March, A Rago, and J Giuggia (1998). Assessment of crop loss in peanut caused by Sclerotinia sclerotiorum, S. minor, and Sclerotium rolfsii in Argentina. Int. J. Pest Manag 44 ((4)): 251- 254.
C.D., Mayee, and V.V Datar (1988). Diseases of groundnut in the tropics. Rev. Trop. Plant Pathol 5: 85- 118.
R.G., Pratt, and D.E Rowe (2002). Enhanced resistance to Sclerotium rolfsii in populations of alfalfa selected for quantitative resistance to Sclerotinia trifoliorum. Phytopathology 92: 204- 209.
Punja, Z.K., and J.E Rahe 2001 Sclerotium Pages 166- 170 In Singleton, L.L., Mihail, J.D., and Rush, C.M., editors, Methods for Research on Soilborne Phytopathogenic Fungi American Phytopathological Society, St. Paul, MN.
Rideout, S.L., T.B Brenneman, and K.L Stevenson 2002 A comparison of disease assessment methods for southern stem rot of peanut Peanut Sci 29: 66- 71 doi: 10.3146/pnut.29.1.0012.
R., Sennoi, S Jogloy, W Saksirirat, and A Patanothai (2010). Pathogenicity test of Sclerotium rolfsii, a causal agent of Jerusalem artichoke (Helianthus tuberosus L.) stem rot. Asian J. Plant Sci 9: 281- 284.
G., Shaner, and R.E Finney (1977). The effect of nitrogen fertilization on the expression of slow-mildewing resistance in Knox wheat. Phytopathology 67: 1051- 1056.
B.B., Shew, M.K Beute, and C.L Campbell (1984). Spatial pattern of southern stem rot caused by Sclerotium rolfsii. Phytopathology 74: 730- 735.
F.M., Shokes, K Róáalski, D.W Gorbet, T.B Brenneman, and D.A Berger (1996). Techniques for inoculation of peanut with Sclerotium rolfsii in the greenhouse and field. Peanut Sci 23: 124- 128.
F.M., Shokes, Z Weber, D.W Gorbet, H.A Pudelko, and M Taczanowski (1998). Evaluation of peanut genotypes for resistance to southern stem rot using an agar disk technique. Peanut Sci 25: 12- 17.
C.E., Simpson, M.R Baring, A.M Schubert, M.C Black, H.A Melouk, et al (2006). Registration of 'Tamrun OL 02' peanut. Crop Sci 46: 1813.
P., Subrahmanyam, P.S Van Wyk, C.T Kisyombe, D.L Cole, G.L Hildebrand, et al (1997). Diseases of groundnut in the Southern African Development Community (SADC) region and their management. Int. J. Pest Manag 43: 261- 273.
B.L., Tillman, and D.W Gorbet (2015). Registration of 'Florun '107'' peanut. J. Plant Regist 9: 162- 167.
J., Woodward, T Brenneman, R. C Kemerait, N. B Smith, A Culbreath, et al (2008). Use of resistant cultivars and reduced fungicide programs to manage peanut diseases in irrigated and nonirrigated fields. Plant Dis 92: 896- 902.
C., Xie, C.-H Huang, and G.E Vallad (2014). Mycelial compatibility and pathogenic diversity among Sclerotium rolfsii isolates in the southern United States. Plant Dis 98: 1685- 1694.
Z., Xu, T.C Harrington, M.L Gleason, and J.C Batzer (2010). Phylogenetic placement of plant pathogenic Sclerotium species among teleomorph genera. Mycologia 102: 337- 346.
Notes
- Research Plant Pathologist, USDA-ARS, Wheat, Peanuts and Other Field Crops Research Unit, Stillwater, OK 74075. [^] *Author's E-mail: rebecca.bennett@usda.gov
Author Affiliations