Introduction
Leaflets of normal (Arachis hypogaea L.) phenotypes have typically two-pairs of leaflets. However, the size of leaflets and plants may vary depending of genotypes (Ashri, 1970; Mouli and Kale, 1981; Branch and Hammons, 1983; Patil and Mouli, 1984; Essomba et al, 1993).
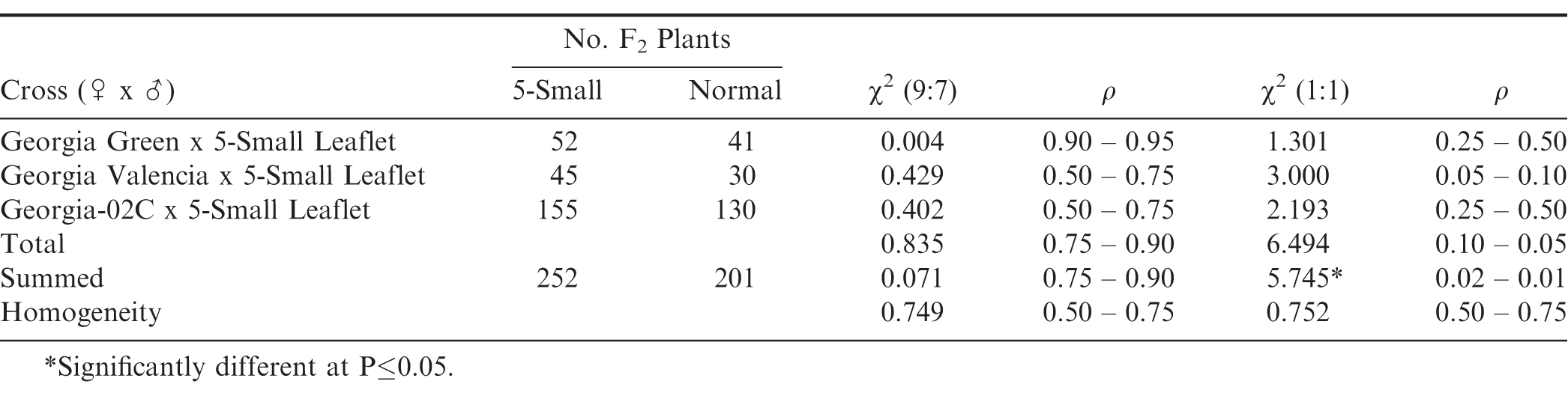
During 2001, an unusual individual mutant plant with 5-Small Leaflets (Fig. 1) was found within the 'Georgia Green' runner-type cultivar (Branch, 1996). Subsequently, continued selfing through the S9 generation of the progeny from this unusual plant did not breed true-to-type for the 5-Small Leaflet characteristics. It has continued to segregate for four-normal leaflets and 5-Small Leaflet plant genotypes.
The objective of our study was to determine the inheritance of the 5-Small Leaflet mutant. Also, another underlying objective was to determine the cause for continuous segregation within this self-pollinated genotype.
Materials and Methods
During the fall and winter of 2003-04, reciprocal crosses were made in the greenhouse between the 5-Small Leaflet Mutant x Georgia Green, 'Georgia Valencia' (Branch, 2001), and 'Georgia-02C' (Branch, 2003). In 2005, F1 seed were space-planted approximately 122-cm apart in one-row plots, 6.1 m long by 1.8 m wide at the UGA Coastal Plain Experiment Station, Gibbs research farms, Tifton, GA. F2, F3, and F4 seed from each cross combination were space-planted each following year (2006, 2007, and 2008, respectively) approximately 30-cm apart in two-row plots with varying length by 1.8 m wide beds. These field genetic nursery plots were in a three-year rotation following corn (Zea mays L.) and cotton (Gossypium hirsutum L.). Each year, the soil-type was a Tifton loamy sand (fine-loamy, siliceous, thermic, Plinthic Kandidult). Recommended cultural practices with irrigation were used throughout each growing season.
In 2011, a S9 generation 5-Small Leaflet Mutant yield test was conducted at the same location. Selfed seed from three individual plant selections were made and increased in 2010. The four entries included: Normal Leaf from normal leaf selection, Normal Leaf from 5-Small Leaflet selection, 5-Small Leaflet Mutant Selection, and the Georgia Green check cultivar. A randomized complete block field design was used with six replications. Each plot consisted of 2-rows, 6.1 m long by 1.8 m wide, and recommended cultural practices with irrigation were used for determining maximum yield potential among the four genotypes. Since segregation still occurred in some entries throughout the growing season, any off-type plants were rogued when apparent. The 5-Small Leaflet trait becomes noticeable about 3-4 weeks after emergence.
During the fall and winter of 2011-12, a full diallel crossing block was made between the three selections from the yield test in 2011 (Normal from normal leaf, Normal from 5-Small Leaflet, and 5-Small Leaflet Mutant). In 2012, F1 seed were space-planted approximately 122-cm apart in one-row plots 6.1 m long by 1.8 m wide at the same location. F2 and F3 seed from each cross combination were also space-planted each year (2013 and 2014, respectively) approximately 30-cm apart in two-row plots with again varying length by 1.8 m wide beds.
Phenotypic classifications were based upon individual plants for leaf characteristics. Segregation data was analyzed by chi-square analysis for goodness-of-fit (P≤0.05) to expected genetic ratios (Strickberger, 1968). Data from the 2011 yield test was subject to analyses of variance. Waller-Duncan's t-Test (k-ratio = 100) was used for mean separation of significant differences (P≤0.05).
Results
It became apparent that none of the crosses using 5-Small Leaflet mutant as a female (♀) parent were successful. Only when 5-Small Leaflet mutant was used as a male (♂) parent were hybrid seed produced. Subsequently, stained pollen from the flowers of the 5-Small Leaflet mutant were indeed found to be normal with plump sound looking pollen grains.
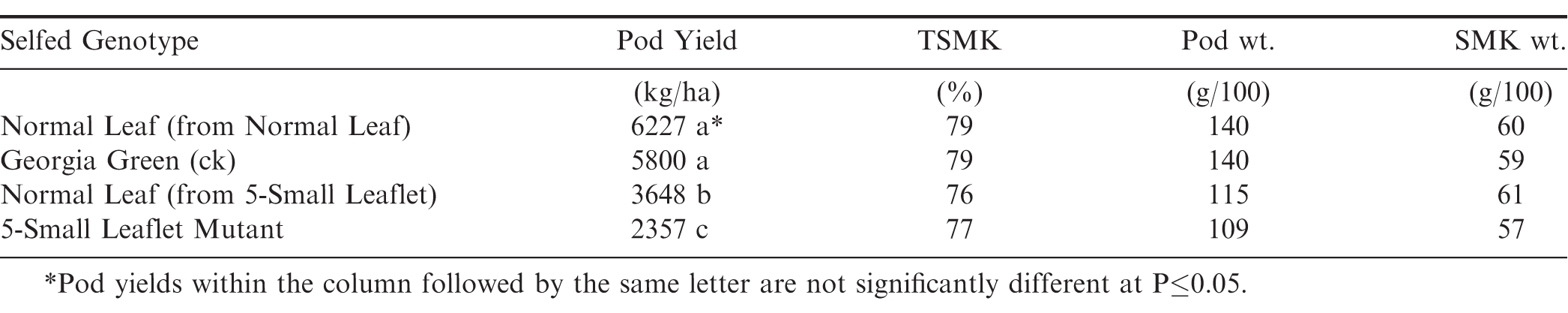
A striking elongated stigma was also noticeable which ranged from 1-15 mm above the keel and was common among about half of the flowers from individual 5-Small Leaflet Mutant plants (Fig. 2). This would reduce self-fertilization and allow for potential cross-pollination; however, after numerous attempts to hand-pollinate the elongated stigma, none were successful.
F1 plants had the 5-Small Leaflet trait among the following crosses: Georgia Green (♀) x 5-Small Leaflet (♂), Georgia Valencia (♀) x 5-Small Leaflet (♂), and Georgia-02C (♀) x 5-Small Leaflet (♂). This suggests that the 5-Small Leaflet trait is dominant to the normal 4 leaf.
The F2 individual plant segregation from these three cross combinations showed an acceptable fit for both a 9 (5-Small Leaflet) to 7 (normal) and 1 (5-Small Leaflet) to 1 (normal) genetic ratios, respectively (Table 1). Total, summed, and homogeneity chi-square values were found acceptable for each of these two expected ratios, except for the summed 𝛘2 value for the 1:1 ratio.
F3 and F4 progeny row segregation among F2:3 normal leaf and F2:4 5-Small Leaflet both showed an acceptable fit for a 1 segregating to 1 non-segregating ratio (Table 2). However, the F2:4 normal leaf showed an acceptable fit to a 1 segregating to 2 non-segregating ratio, and all of the progeny rows from the F2:3 5-Small Leaflet segregated.
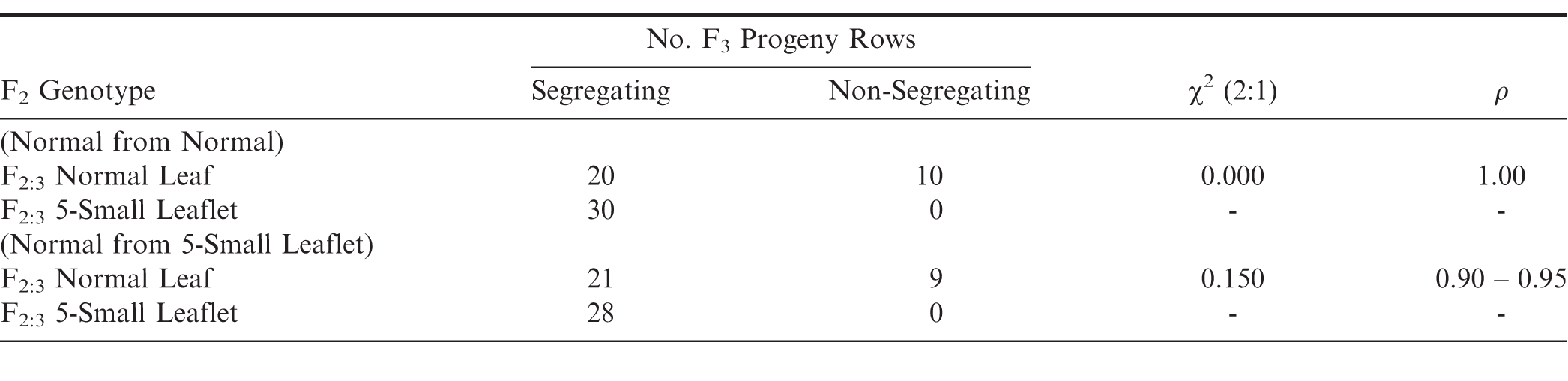
The 2011 yield test resulted in significant differences (P≤0.05) in pod yield among four entries (Table 3). The normal leaf from normal leaf and Georgia Green (Fig. 3) resulted in the two highest yielding entries at 6227 and 5800 kg/ha, respectively. Normal Leaf from 5-Small Leaflet was next highest followed by the lowest yielding entry, the 5-Small Leaflet Mutant (Fig. 4) at 2357 kg/ha which equals approximately 40% yield reduction compared to the Georgia Green cultivar. Likewise, total sound mature kernels (TSMK) percentage, 100 pod and 100 sound mature kernels (SMK) weights were lower for the 5-Small Leaflet compared to Georgia Green.
A full diallel crossing block of these same three selections used in the yield test (Normal from normal, Normal from 5-Small Leaflet, and 5-Small Leaflet) resulted in only F1 seed from (Normal from normal) x (Normal from 5-Small Leaflet) and (Normal from normal) x (5-Small Leaflet) crosses. The other four possible diallel cross combinations did not produce any viable seed, only an occasional immature pod. F1 hybrid plants from these two crosses exhibited the 5-Small Leaflet trait which again suggests that it is dominant to normal leaf.
The F2 individual plant segregation from both cross combinations showed an acceptable fit for a 9 (normal) to 7 (5-Small Leaflet) and a 1 (normal) to 1 (5-Small Leaflet) genetic ratios, respectively (Table 4). The 9 (normal) to 7 (5-Small Leaflet) is the reverse compared to the earlier cross combinations (Table 1), with a 9 (5-Small Leaflet) to 7 (normal). Total, summed, and homogeneity chi-square values were also found acceptable for both of these two expected genetic ratios 9:7 and 1:1 (Table 4).
F3 progeny row segregation among F2:3 normal leaf from both normal from normal and normal from 5-Small Leaflet showed an acceptable fit for a 2 segregating to 1 non-segregating ratio (Table 5). Whereas, the F2:3 5-Small Leaflet progeny rows were all segregating from both normal from normal leaf and normal from 5-Small Leaflet cross combinations.
Discussion
These findings suggest that the 5-Small Leaflet mutant is heterozygous since it continues to segregate upon selfing for numerous generations. The fact that the 5-Small Leaflet mutant can only be used as a male and not a female parent, could be related to its elongated stigma abnormality which was only noticeable on about 50% of the flowers from individual plants during crossing. Many attempts to make crosses with 5-Small Leaflet as a female on both normal and elongated stigma were not successful. It also exhibits pleiotropic effects for several other plant characteristics: smaller canopy size (Figure 4), lower pod yield, reduced pod and seed weight (Table 1), as well as elongated stigmas (Figure 2).
The inheritance of the 5-Small Leaflet mutant trait appears to be quite novel. In certain cross combinations it appears dominant (Table 1) while in other crosses it appears to be recessive (Table 4). Whereas in all F3 progeny rows, 5-Small Leaflet continues to segregate without any non-segregating progeny rows (Tables 2 and 5). However in the F2:4, 5-Small Leaflet progeny rows did show an acceptable fit to a 1:1 ratio of segregating to non-segregating progeny rows, respectively (Table 2). The reason or cause for continuous segregating is not clear, but it could be related to the fact that the 5-Small Leaflet plants cannot be used as a female parent in crosses.
Another unique aspect of the 5-Small Leaflet inheritance and subsequent segregation appears to be related to an acceptable fit for a (1:1) genetic ratio. Modified genetic ratios have previously been reported for another small leaf character in peanut (Patil and Mouli, 1984). This imparipinnate mutant appeared to be recessive compared to normal leaf, and "preferential" segregation in favor of normal leaf was suggested for the modified monohybrid ratio of 6:1 found to fit imparipinnate inheritance.
Branch and Hammons (1983) likewise reported upon a partial dominant micro phenotype found in peanut. The F1 and F2 data suggest monogenic inheritance for the micro phenotype.
The 5-Small Leaflet mutant appears dominant at least in the three wider genetic cross combinations (Table 1), but it has continuous segregation which suggest it is heterozygous and does not breed true-to-type upon selfing. These characteristics might suggest a chromosome deletion, thus pseudo-dominance inheritance.
Genomic mapping of the cultivated allotetraploid peanut was recently completed through an international effort (Bertioli et al., 2019). These authors found that variable deletions were common (i.e. chromosome Arahy .04/Arahy .14) among different A. hypogaea genotypes representing both subspecies, hypogaea and fastigiata. It could be possible that the maternal-sterile outcrossing trait is linked and found in a similar deletion. This could also explain the lack of obtaining a true-breeding 5-Small Leaflet genotype.
Abbreviations
F1, F2, F3, and F4, filial generations; TSMK, total sound mature kernels, ♀, female and ♂, male; 9:7, 1:1, and 2:1, genetic ratios.
Literature Cited
A Ashri, (1970). Inheritance of small leaflets in a wide cross in peanuts, Arachis hypogaea. Oleagineux 25 ((3)): 153- 154.
D. J., Bertioli, J Jenkins, J Clevenger, O Dudchenko, D Gao, G Seijo, S. C. M Leal-Bertioli, L Ren, A. D Farmer, M. K Pandey, S. S Samoluk, B Abernathy, G Agarwal, C Ballén-Taborda, C Cameron, J Campbell, C Chavarro, A Chitikineni, Y Chu, S Dash, M El Baidouri, B Guo, W Huang, K. D Kim, W Korani, S Lanciano, C. G Lui, M Mirouze, M. C Moretzsohn, M Pham, J. H Shin, K, Shirasawa, S Sinharoy, A Sreedasyam, N. T. Weeks, X Zhang, Z Zheng, Z Sun, L Froenicke, E. L. Aiden, R Michelmore, R. K. Varshney, C. C. Holbrook, E. K. S. Cannon, B. E. Scheffler, J. Grimwood, P Ozias-Akins, S. B. Cannon, S. A. Jackson, J. Schmutz (2019). The genome sequence of segmental allotetraploid peanut Arachis hypogaea. Nature Genetics 51: 877- 884.
W. D Branch, (1996). Registration of 'Georgia Green' peanut. Crop Sci 36: 806.
W. D Branch, (2001). Registration of 'Georgia Valencia' peanut. Crop Sci 41: 2002- 2003.
W. D Branch, (2003). Registration of 'Georgia-02C' peanut. Crop Sci 43: 1883- 1884.
W. D Branch, and R. O Hammons (1983). Inheritance of a micro phenotype in peanut. Crop Sci 23: 1045- 1046.
N. B., Essomba, T. A Coffelt, W. D Branch, and S. W Van Scoyoc (1993). Inheritance of leaflet size in peanut (Arachis hypogaea L.). Peanut Sci. 20: 90- 93.
C Mouli, and D. M Kale (1981). Imparipinnate leaf with normal size leaflet in groundnut. Current Sci 50 ((15)): 684- 685.
S. H Patil, and C Mouli (1984). Preferential segregation of two allelic mutations for small leaf character in groundnut. Theor. Appl. Genet 67: 327- 332.
Strickberger, M. W 1968 Chap. 8. Probability and statistical testing Pp 126- 152 In Genetics The Macmillan Co, New York, NY.
Notes
- W. D. Branch, B. M. Schwartz, and W. W. Hanna. Department of Crop & Soil Science, University of Georgia, Tifton, GA; S. P. Tallury, USDA/ARS, Plant Genetic Resources Conservation Unit, Griffin, GA; and J. P. Clevenger, Mars-Wrigley Confectionery, Center for Applied Genetic Technologies, Athens, GA. [^] *Corresponding author email: wdbranch@uga.edu
Author Affiliations