Introduction
Haiti is a country of extreme poverty with a long history of environmental degradation, political instability and natural disasters (CIA, 2017). Out of approximately 10.6 million inhabitants (CIA, 2017), 56% are estimated to live on less than $2.41 per day, 25% live on less than $1.23 a day (World Bank, 2014) and 38.1% depend solely on small-scale farming for subsistence (CIA, 2017). Under nutrition is particularly widespread in Haitian children of whom 29.7% are classified as moderately malnourished and 18.9% as severely malnourished (UNICEF, 2016). In Haiti, one of the primary standards for treating acute malnutrition is with Ready-to-use Therapeutic Food (RUTF) (Iannotti et al., 2015; WHO, 2017).
Medika Mamba, Haitian Creole for "peanut butter medicine", is a peanut-based RUTF that has been produced and distributed in Haiti by the non-profit organization Meds and Food for Kids (MFK) since the early 2000s (MFK, 2017). MFK seeks to positively impact the Haitian economy by providing jobs at the RUTF factory located near Cap Haitien, Haiti, but also aims to provide further stimulus by sourcing 100% of their peanuts from local farmers. However, local purchasing is limited due to inflated market prices resulting from poor yields and low quality peanuts. As a result, MFK's strategy has been to improve the quality and quantity of peanuts produced in Haiti such that the market price would drop sufficiently for the factory to afford local purchase, and simultaneously increase grower profits by allowing significantly greater yields to be sold at a lower purchase price.
Therefore, in 2007 MFK linked up with U.S.-based University peanut specialists in order to obtain the technical expertise needed to improve local production. Since that time, research and extension efforts have been conducted in Haiti as part of a multi-disciplinary team consisting of MFK and other in-country partners and researchers at the University of Georgia and the University of Florida. This research has been funded by the Peanut and Mycotoxin Innovation Lab (PMIL) - part of the Feed the Future program, the US government's global hunger and food security initiative led by USAID (PMIL, 2017), whose overall objective is to improve livelihoods and health in developing countries through advances in peanut research in production, processing and markets.
Currently peanut is grown in Haiti primarily as a cash crop and is consumed by many as popular snack food (Nelson et al., 2003). Locally produced peanut butter is available in many small shops, and small packets of roasted peanuts are commonly sold by street vendors. As a minor crop, peanut is cultivated on approximately three percent of the total arable land in Haiti (FAOSTAT, 2016). Small-scale peanut production (<1 ha) occurs at the greatest frequency in the Northeast and the Central Plateau, but is also scattered widely across Haiti's diverse landscape. Planting, weeding, and harvest depends on manual labor, and production is generally organized by an elected community leader. The primary varieties grown in Haiti include two local landraces - one a runner market-type and the other a Valencia peanut market-type. Seed for planting is either saved by the grower or purchased on the market. The main production period occurs in the rainy season (typically late spring and early summer). An additional crop is planted in the late fall/early winter by growers in the Northern regions of Haiti where a second rainy season usually occurs during this period (G. Kostandini, personal communication).
Average pod yield in Haiti is exceptionally poor (870.9 kg/ha) (FAOSTAT, 2016) and aflatoxin contamination is common (Schwartzbord and Brown, 2015). There are a number of challenges to increasing peanut yield in Haiti including alkaline soils (Bargout and Raizada, 2013), low soil fertility (Nelson et al., 2003), poor seed quality (Nelson et al., 2003), inconsistent planting methods (our observation) and foliar diseases (Fulmer et al., 2014). Late leaf spot (Cercosporidium personatum) (Shokes and Culbreath, 1997) and peanut rust (Puccinia arachidis) (Subrahmanyam, 1997) are the two most destructive diseases of peanut in Haiti (Fulmer et al., 2014). These diseases are not typically managed in Haiti due to economic constraints and limited access to the appropriate resources. Furthermore, growers often mistake defoliation caused by foliar diseases with natural senescence. Management of leaf spot and rust in other developing countries consists of planting resistant cultivars, treatment with fungicide, crop rotation, destruction of plant residue and removing volunteer plants (Fulmer et al., 2016; Waliyar et al., 2000). Previous studies have documented the potential to significantly reduce foliar disease severity and increase yield in Haiti with resistant varieties developed by the International Crops Research Institute for the Semi-Arid-Tropics (ICRISAT) (Fulmer et al., 2012). However, negative roasting and quality issues did not favor wide-scale introduction resulting in the continued large-scale use of the local Haitian varieties.
One of the primary objectives of the PMIL project has been to identify a high yielding, disease resistant Valencia variety suitable for Haiti. Therefore, the purpose of this study was to evaluate the response of five advanced Valencia breeding lines compared to the local Haitian Valencia to foliar disease and yield performance and to determine whether there was an interaction with fungicide. A brief overview of this research has previously been published (Fulmer, 2018).
Materials and Methods
Research Field Sites
Field trials were conducted at research sites belonging to our in-country partners, Meds and Food for Kids (MFK) and Acceso Peanut Enterprise. The Acceso research farm is located in the Central Plateau (CP), near the town of Mirebalais, Haiti (18°50'21.05"N latitude, 72°3'29.33"W longitude). The research plots at the MFK factory are located in Quartier Morin which is near Cap Haitien, Haiti (19°41'32.17"N latitude, 72°9'16.91"W longitude). Based on results from soil samples submitted to the University of Georgia's Soil, Plant, and Water Lab, the soil type in fields used at MFK was most often classified as a sandy clay loam and the fields used at the Acceso research farm as a clay soil type. Calcium levels at both locations were consistently over 3,000 kg/ha and both had a pH (based on CaCl22 method) greater than 7.1 (PMIL, unpublished data). At MFK, all fields had a previous history of peanut. In most cases, peanuts were planted behind peanut, with a 3 to 6 month fallow period between crops. In some cases, peanut followed a rotation of sorghum. At the Acceso research farm, peanut followed several years of common bean and/or sorghum production.
Experimental Design
Five trials were conducted from 2015 to 2017: four trials at the MFK research facility and one at the Acceso research farm. Planting and harvest dates for the trials reported here were as follows: MFK-1, 13 Nov. 2015 - 25 Feb. 2016; MFK-2, 23 Mar. 2016 - 21 Jun. 2016; MFK-3, 24 Aug. 2016 - 29 Nov. 2016; MFK-4, 27 Jan. 2017 - 3 May. 2017; CP-5, 19 Jan. 2017 - 4 May. 2017. A split plot design with 4 to 6 replications was used in all trials. Six Valencia varieties were the main plot treatments and fungicide (3 applications or none) were the sub-plot treatments. The six varieties included the local Haitian Valencia and five advanced breeding Valencia lines developed by the New Mexico Agricultural State University Experiment Station: 309 Red, 309 Tan, M2, M3, and SCGV0801. Fungicide treatments consisted of a combination of tebuconazole (0.23 kg/ha) plus chlorothalonil (0.84 a.i. kg/ha) (Muscle® ADV, Sipcam Agro USA, Inc.) sprayed at 188 L/ha at 45, 60 and 75 days after planting (DAP). Fungicides were applied mid-morning with a hand pumped backpack sprayer equipped with a two nozzle (8002VS flat tip nozzles from Teejet Technologies, Springfield, IL) boom spaced 30.4 cm apart.
Prior to planting, fields were disked at least once, fertilized with 20-20-20 or diammonium phosphate at a rate of 45 or 67.2 kg/ha and then rototilled. Each plot consisted of two rows of peanuts, planted by hand at a rate of 3 seed/30.5 cm in the first trial at MFK and the trial in the Central Plateau (CP); in all other trials, peanuts were planted at a rate of 6 seed/30.5 cm. Blocks were separated by a 1.5 m alley and there was no border row between sub-plots. At planting, seeds were treated with azoxystrobin, fludioxonil and mefenoxam (Dynasty® PD, Syngenta Crop Protection, Inc.) at a rate of 85 g of product per 45.4 kg of seed. Plots were weeded by hand at least three times, beginning 3-4 wk after planting; no herbicides or insecticides were applied. Depending on rainfall, plots were irrigated at both locations when needed. At MFK, plots were generally irrigated twice a week with approximately 1.3 cm of water using with a rotary sprinkler system. At the Acceso farm, plots were irrigated with portable, hand-held watering cans. A Decagon ECRN-50 rain gauge (Decagon Devices, Inc., Pullman, WA) was used to record rainfall at MFK.
Data Collection
Stand counts were conducted at approximately three wk after planting to confirm even distribution of seed germination for each variety. For three of the trials at MFK (2-4), incidence of late leaf spot and rust was assessed by arbitrarily sampling five leaves from the lower canopy of each plot and rating for the presence or absence of each disease. Incidence was calculated as the number of leaflets with at least one late leaf spot lesion or one rust pustule divided by the total number of leaflets sampled. Plots were sampled at approximately 45, 60, 75 and 90 DAP. The day prior to digging the peanuts, plots were rated for final leaf spot and rust severity. Leaf spot severity was rated with the Florida 1 to 10 scale, where 1= no leaf spot and 10 = plants completely defoliated and killed by leafspot (Chiteka et al., 1988). Rust severity was rated with a modified 1 to 9 scale where 1 = no disease and 9 = almost all leaves withered; bare stems seen; 81-100% severity (Subrahmanyam et al., 1995).
Plots were harvested between 90 and 105 days after planting by uprooting each plant by hand and manually picking the pods from the vines. The soil from each plot was then sifted through to recover pods that snapped off the vines in the process of being uprooted from the ground. The total pods from each plot were then placed in mesh bags and washed to remove the remaining soil. Bags were placed in the sun on a concrete pad with the pods spread flat/evenly during day time hours and placed under a shelter during the night time hours. This drying process continued at least 3 days and up to a 2 weeks. Bags were weighed after achieving an acceptable moisture level. Afterwards, 100 pods were sampled from each bag to determine final kernel moisture and %SMK (sound mature kernels - the weight of the total number of sound mature kernels divided by the weight of the unshelled 100 pod sample). Final yield estimates for each plot were standardized to 10% kernel moisture.
Statistical analysis
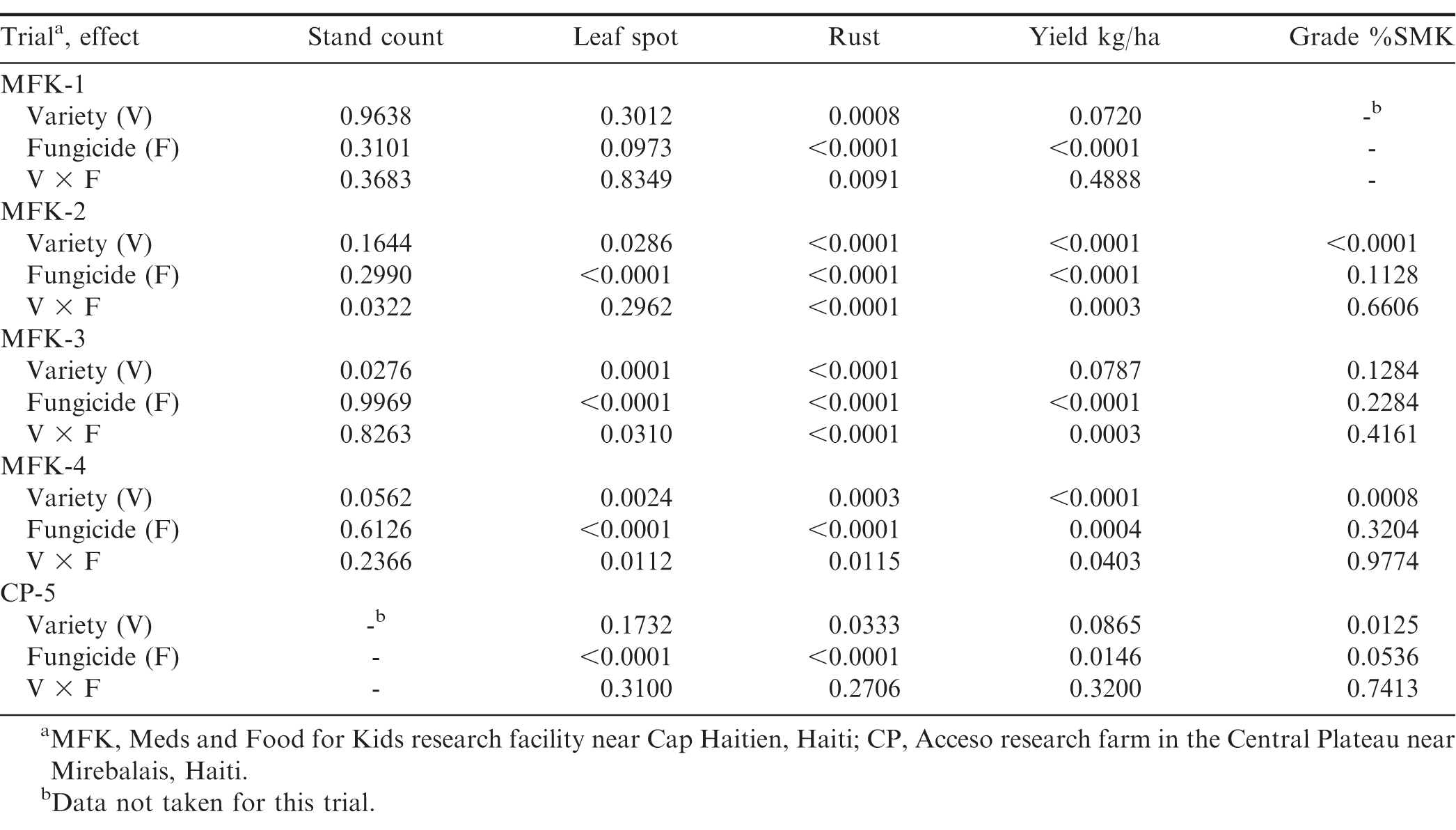
Stand count, leaf spot and rust severity, yield and %SMK were subjected to analysis of variance with PROC GLIMMIX (SAS 9.4 Institute, Cary, NC). Because Haiti has a wide range of environmental conditions throughout the year, the effect of each variety/fungicide combination was analyzed separately for each trial. The model was a split plot design with variety, fungicide and variety by fungicide considered as fixed effects with replication and replication by variety as random effects. In the case of at least one trial with a significant interaction for a given response variable, the SLICE option was used to determine the simple effects for each trial. In all analyses the Kenward-Roger option was used to adjust the degrees of freedom, and differences in the least square means were tested by Tukey's multiple comparisons test. In order to meet assumptions of normality, the arcsin square root and the natural log transformations were used for %SMK and yield, respectively. Transformed variables were back-transformed for presentation in the results section.
Results and Discussion
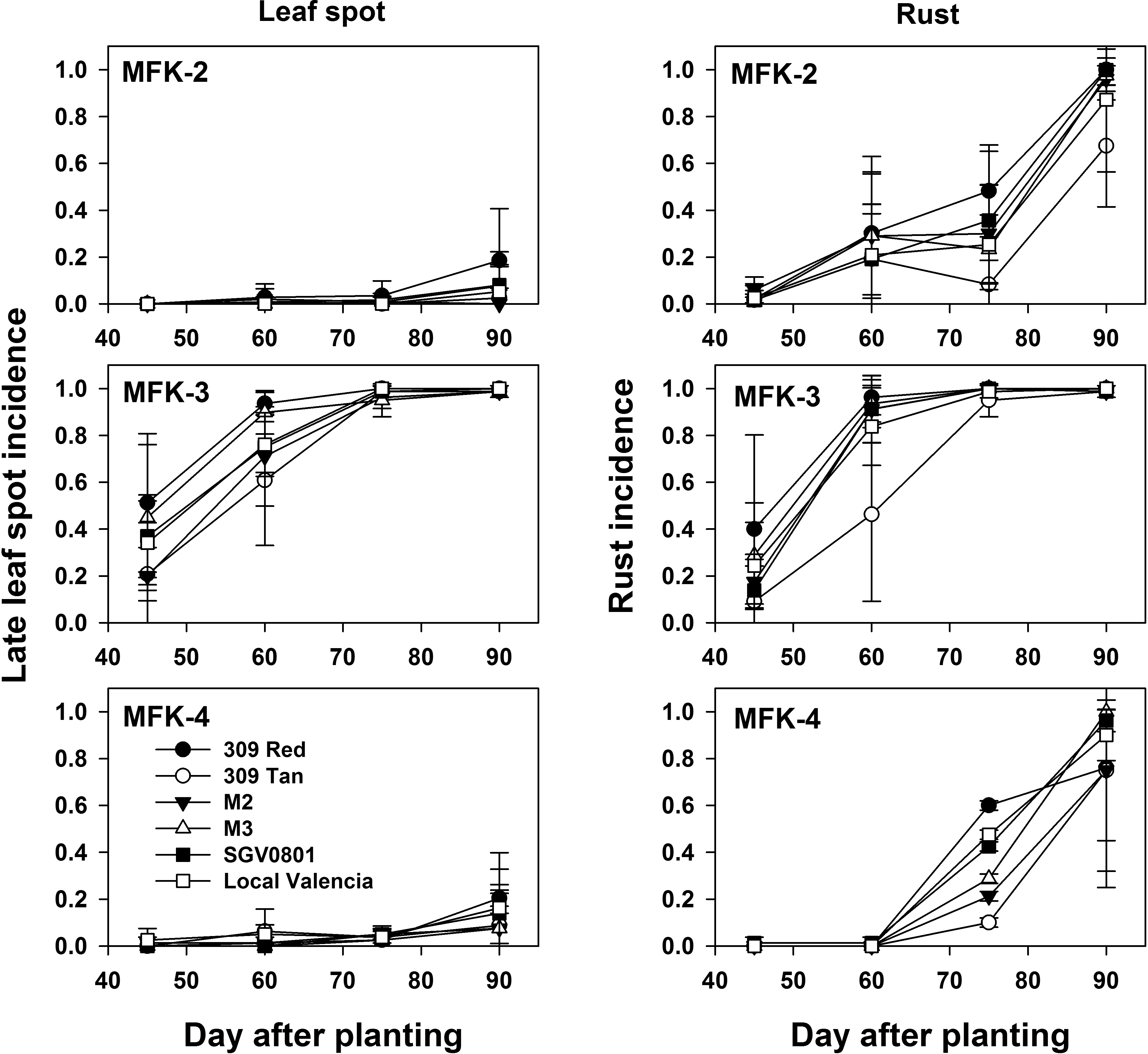
Late leaf spot and rust were both observed in these trials, but the latter was the most prevalent disease. The onset of both diseases (Fig. 1) was generally earliest in trials with the greatest amount of rainfall in the first 30 days after planting (Table 1). Final severity of both diseases differed between trials, and the highest disease pressure was observed in the first and third trial (Fig. 2). Although these trials were irrigated, overall disease severity (Fig. 2) was greatest in trials that had the greatest amount of rainfall (Table 1). However, there was no apparent association between the time of year the trial was conducted and final disease severity. It is therefore probable that differences in rainfall likely contribute more to the varying levels of foliar disease pressure than the time of year a trial is conducted in Haiti.
Mean leaf spot incidence (A), peanut rust incidence (B) and yield (C) across varieties and fungicide treatments for three trials conducted at the Meds and Food for Kids (MFK) research facility near Cap Haitien, Haiti. Planting dates for each trial were as follows: MFK-2, 23 Mar. 2016; MFK-3, 24 Aug. 2016; MFK-4, 27 Jan. 2017. Error bars indicate the standard error of the mean (n = 4).
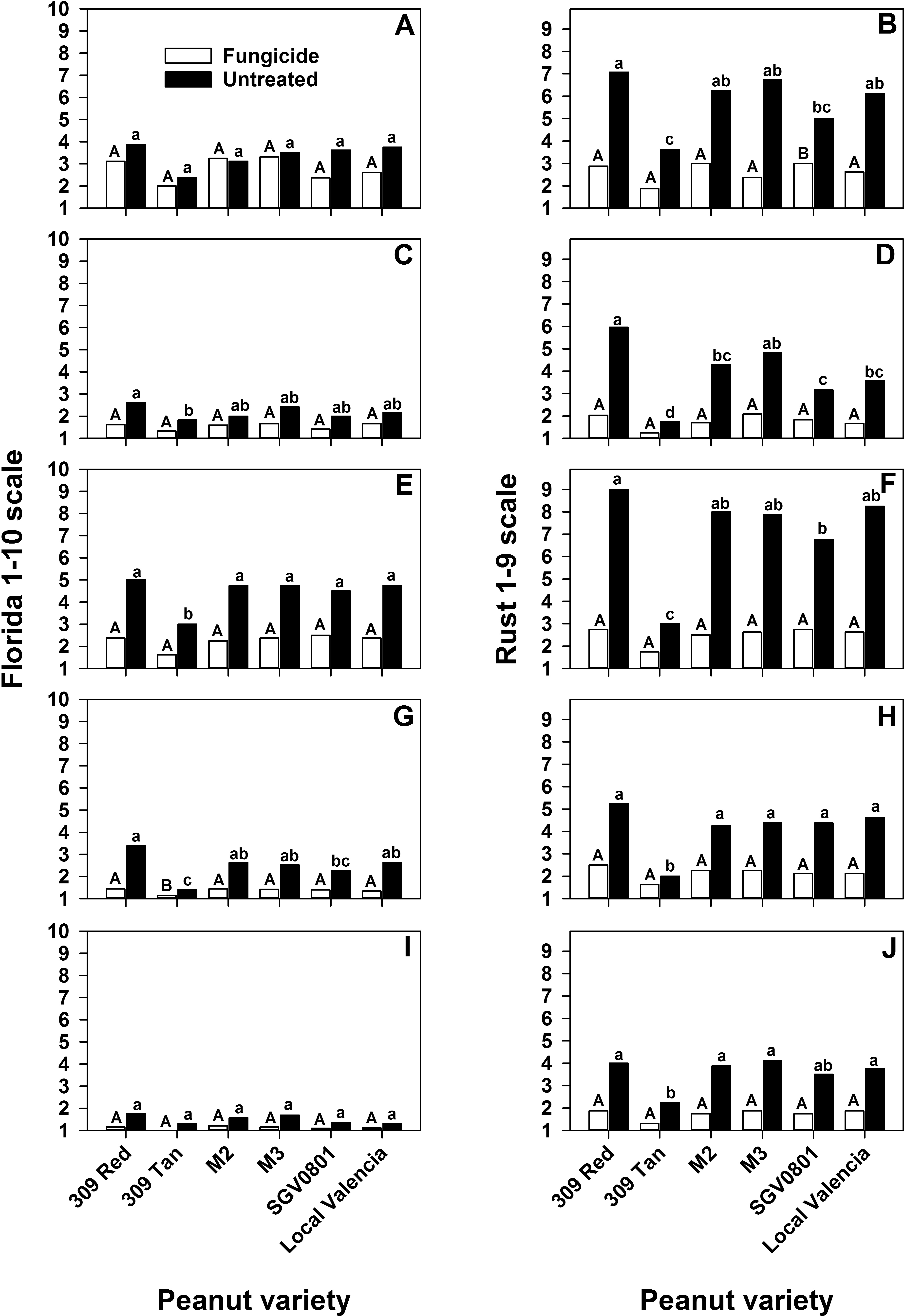
Effect of variety on leaf spot (A, C, E, G and I) and peanut rust (B, D, F, H, and J) severity in five trials conducted in Haiti. A and B, first trial at Meds and Food for Kids (MFK) research facility near Cap Haitien, Haiti, C and D, second trial at MFK, E and F, third trial at MFK, G and H, fourth trial at MFK, I and J, fifth trial, conducted in the Central Plateau (CP) near Mirebalais, Haiti. Analyses were conducted separately for plots treated with and without fungicide and mean separations are indicated by upper and lower case letters, respectively. Bars with different letters are significantly different based upon Tukey's honestly significant difference test.
Disease severity was significantly affected by fungicide in almost every trial (Table 2). Variety had a significant effect on rust severity in every trial (Table 2). However, there was a significant variety × fungicide interaction in two of the trials for leaf spot and four of the trials for rust (Table 2). Based on the analysis of each simple effect with the SLICE option, it was clear that the main interaction was caused by the variety 309 Tan, which consistently had the lowest leaf spot and rust severity regardless of fungicide treatment (Fig. 2). All other varieties had a reduction in foliar disease severity with an application of fungicide (Fig. 2). Apart from 309 Tan, SGV0801 generally had lower levels of rust severity, but all other varieties were comparable to the local Haitian Valencia (Fig. 2).
Final stand counts ranged from 69.6 to 97.4 percent plant emergence of total seeds planted and was significantly different in two of the five trials (data not shown). In the MFK-2 untreated plots, 309 Tan was significantly lower (73.2) compared to 309 Red (82.2), M2 (90.0), M3 (85.4), SGV0801 (83.3) and the local Haitian Valencia (83.0). In the MFK-4 plots treated with fungicide, the local Haitian Valencia (69.6) had significantly lower final plant stands compared to 309 Red (83.1), 309 Tan (76.1), M2 (85.2), M3 (87.8) and SGV0801 (88.1).
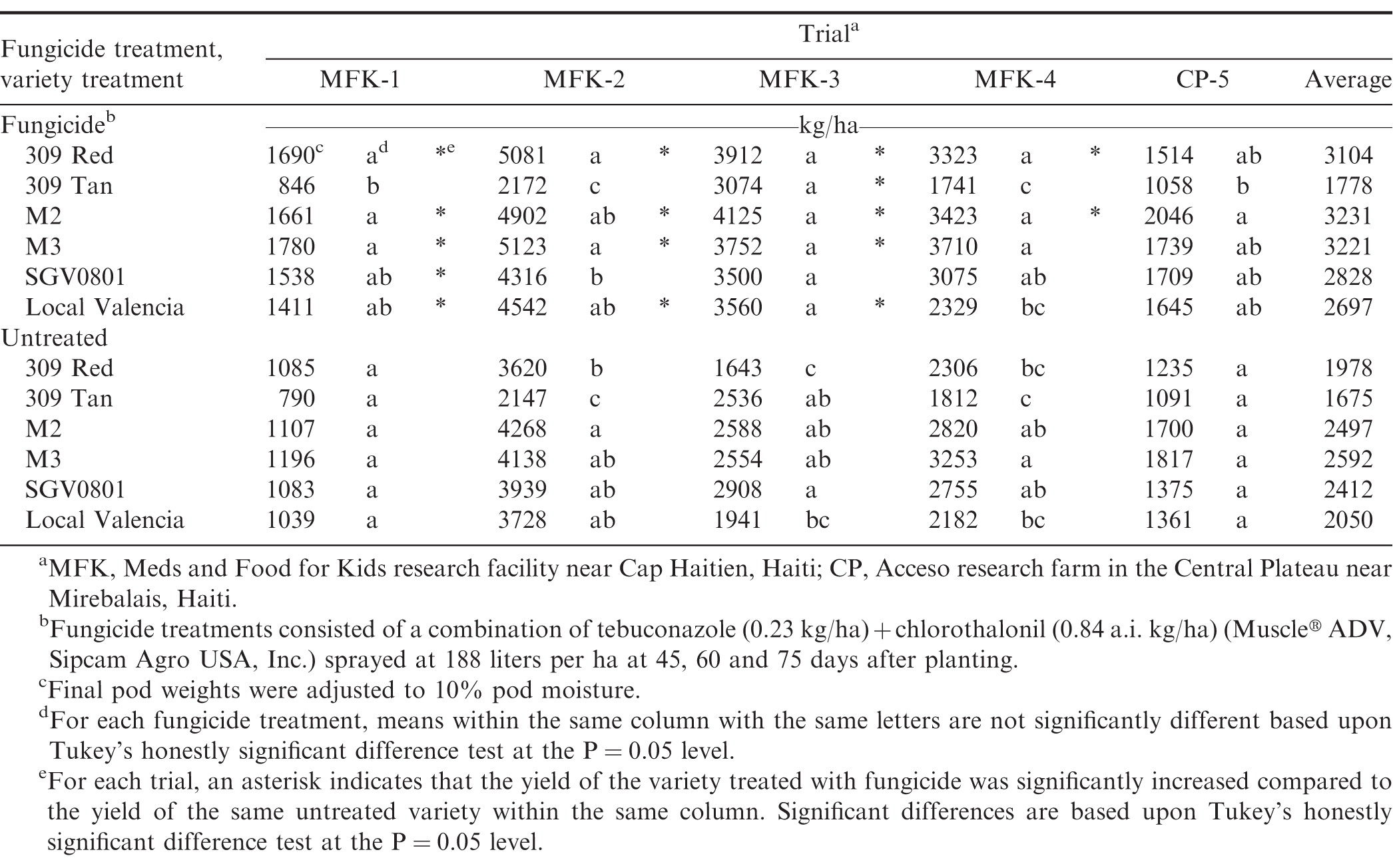
Yield varied by trial (Table 3) and differences did not appear to be associated with rainfall or final stand count. Differences in yield between trials were associated with differences in the seeding rate used for each trial. The lowest yielding trials, MFK-1 and CP-1, were planted at 3 seed/30.5 cm, but all other trials were planted at 6 seed/30.5 cm. Although not the primary objective of this study, these results corroborate other studies that have shown that higher planting densities tend to result in higher yields for bunch-type cultivars (Henning et al., 1982). Similar positive linear correlations between planting density and pod yield have been observed with the local Haitian Valencia (PMIL, unpublished data).
Pod yield was significantly affected by fungicide in every trial, but variety was less consistent (Table 2); however, there was a significant variety × fungicide interaction in three of the trials (Table 2). Generally, there was not a statistical yield increase for any of the N.M. Valencia varieties compared to the local Haitian variety (Table 3). The variety 309 Tan consistently had the lowest numerical yields, but differences were not always significant (Table 3). The single exception was in the untreated plots in trial MFK-3, where rainfall and disease pressure was greatest (Table 1 and Table 3). Similarly, yield did not significantly increase in plots treated with fungicide except for trial MFK-3, but all other varieties (except SGV0801) had a significant yield increase in 3 to 4 of the five trials (Table 3). Fungicide did not have a significant effect in trial CP-5, likely due to the low disease pressure (Fig. 2). These results further validate that low-input fungicide regimes are extremely effective for reducing disease and increasing yield in Haiti. We have recently demonstrated that 3 to 4 fungicide applications can significantly improve pod yield for both runner and Valencia market-types by > 50 and 30%, respectively (Fulmer et al., 2016). Similar results of the efficacy of low-input fungicide programs have been reported in West Africa (Waliyar et al., 2000), Ghana (Naab et al., 2009) and Malawi (Subrahmanyam and Hildebrand, 1997).
The low yield results for 309 Tan were somewhat surprising because this variety has been reported to have excellent yield potential in Africa (N. Puppala, personal communication). It may be that disease pressure was not severe enough in Haiti to capture the full benefit of the high resistance levels in 309 Tan. Lower yields for 309 Tan may also be partially due to linkage drag which often occurs when breeding for resistance (Stalker et al., 2016).
Percent SMK was not significantly affected by fungicide (Table 2), but was significantly affected by variety in three trials (Table 2 and Table 4). 309 Tan was significantly lower than all the other varieties in the second trial, and numerically or statistically lowest in all other trials (Table 4). Overall, % SMK levels are comparable with other studies in Malawi (Subrahmanyam and Hildebrand, 1997), and were similar to % SMK values obtained in other trials conducted in Haiti (PMIL, unpublished data).
Summary and Conclusions
The consistent yield response of susceptible varieties to fungicide applications observed in these studies emphasize the importance of managing foliar diseases in Haiti. However, as fungicide inputs are not widely used in Haiti due to practical and economic constraints, there is need to identify a high yielding, disease resistant varieties for smallholder farmers. In this study we demonstrated that 309 Tan has excellent resistance to late leaf spot and rust, but that this resistance did not result in a positive increase in yield and grade when compared to susceptible varieties. Similarly, while other varieties from the NMSU breeding program did provide higher yields compared to the local Haitian Valencia, the increase does not appear to be significant enough to justify the introduction of these varieties into Haiti at the present time. A major research objective in Haiti will be to develop a long-term strategy of incorporating improved traits of varieties like 309 Tan into the existing adapted varieties through plant breeding.
Acknowledgements
We would like to express our appreciation to the personnel at Meds and Food for Kids and Acceso Peanut Enterprise for providing research facilities and technical support. We also thank the following student interns from the Université Roi Henri Christophe and the Université Chrétienne du Nord d'Haiti for their valuable contributions to this research: Rodnie Valmy, Rodemane Saint Louis, Lainé Dorinvil, Frisnel Pierre and Jean-Baptiste Fontilus. Lastly, we thank the Peanut Mycotoxin Innovation Lab for funding this project.
Literature Cited
R.N., Bargout, and M.N Raizada (2013). Soil nutrient management in Haiti, pre-Columbus to the present day: lessons for future agricultural interventions. Agric. Food Secur 2: 11.
Z., Chiteka, D Gorbet, F Shokes, T Kucharek, and D Knauft (1988). Components of resistance to late leafspot in peanut. I. Levels and variability-implications for selection. Peanut Sci 15: 25- 30.
CIA 2017 The world factbook: Central America and the Caribbean, Haiti The Central Intelligence Agency Online at https://www.cia.gov/library/publications/the-world-factbook/geos/ha.html (accessed 17 Nov 2017) .
FAOSTAT 2016 Food and Agriculture Organization of the United Nations Statistics Division Online at http://www.fao.org/faostat (accessed 17 Nov 2017).
Fulmer, A., ed. 2018 Haiti Peanut Research Report. Coop. Ext. Ser. Bull. 1499, University of Georgia, Athens, GA.
Fulmer, A.M., R.C Kemerait, and T.B Brenneman 2014 Mountains beyond mountains: Challenges and opportunities for managing peanut diseases in Haiti. Phytopathology 104 (Suppl.)S:155.
Fulmer, A.M., R.C Kemerait, and T.B Brenneman 2016 Effect of fungicide timing and frequency on reducing foliar diseases and yield loss on runner and valencia type peanuts in Haiti. Phytopathology 106 (Suppl.)S:66.
Fulmer, A.M., R.C Kemerait, J.L Sherwood, D.L Jordan, J Rhoads, and T.B Brenneman 2012 Evaluation of ICRISAT varieties for resistance to foliar peanut diseases in Haiti. Phytopathology 102 (Suppl.)S2:4.
Henning, R.J., A.H Allison, and L.D Tripp 1982 Cultural Practices Pages 123-138 in: Peanut Science and Technology H Patteeand C Young, eds. American Peanut Research and Education Society, Yoakum, TX.
L.L., Iannotti, N.M Henretty, J.R Delnatus, W Previl, T Stehl, S Vorkoper, J Bodden, A Maust, R Smidt, and M.L Nash (2015). Ready-to-use supplementary food increases fat mass and BMI in haitian school-aged children. J. Nutr 145: 813- 822.
MFK 2017 Meds and Food for Kids Online at https://mfkhaiti.org (accessed 17 Nov 2017).
J., Naab, P Prasad, K Boote, and J Jones (2009). Response of peanut to fungicide and phosphorus in on-station and on-farm tests in Ghana. Peanut Sci 36: 157- 164.
R., Nelson, C Jolly, M Hinds, Y Donis, and E Prophete (2003). Consumer preferences for peanut butter (mamba) products in Haiti: A conjoint analysis. Peanut Sci 30: 99- 103.
PMIL 2017 Peanut Mycotoxin Innovation Lab. Online at http://www.caes.uga.edu/global/feed-the-future-innovation-labs/peanut-mycotoxin-innovation-lab.html (accessed 17 Nov 2017) .
J.R., Schwartzbord, and D.L Brown (2015). Aflatoxin contamination in Haitian peanut products and maize and the safety of oil processed from contaminated peanuts. Food Control 56: 114- 118.
Shokes, F., and A.K Culbreath 1997 Early and late leaf spots Pages 17-20 in: Compendium of Peanut Diseases, 2nd Ed. N Kokalis-Burelle, D Porter, R Rodriguez-Kabana, D Smithand P Subrahmanyam, eds. American Phytopathological Society Press, St. Paul, MN.
Stalker, H.T., S.P Tallury, G.R Seijo, and S.C Leal-Bertioli 2016 Biology, Speciation, and Utilization of Peanut Species Pages 27-66 in: Peanuts: Genetics, Processing, and Utilization. H. T Stalkerand R. F Wilson, eds. Press, AOCS Urbana, IL.
Subrahmanyam, P 1997 Rust Pages 31-33 in: Compendium of Peanut Diseases, 2nd Ed. N Kokalis-Burelle, D Porter, R Rodriguez-Kabana, D Smithand P Subrahmanyam, eds. American Phytopathological Society Press, St. Paul, MN.
P., Subrahmanyam, and G Hildebrand (1997). Responses of peanut genotypes to fungicidal control of early leaf spot in Malawi. Peanut Sci 24: 73- 77.
Subrahmanyam, P., D McDonald, F Waliyar, L Reddy, S Nigam, R Gibbons, V.R Rao, A Singh, S Pande, and P Reddy 1995 Screening methods and sources of resistance to rust and late leaf spot of groundnut. Information Bulletin no. 47. International Crops Research Institute for the Semi-Arid Tropics.
UNICEF 2016 The State of the World's Children 2016: A fair chance for every child United Nations Children's Fund, New York.
F., Waliyar, M Adamou, and A Traoré (2000). Rational use of fungicide applications to maximize peanut yield under foliar disease pressure in West Africa. Plant Dis 84: 1203- 1211.
WHO 2017 Malnutrition World Health Organization On line at http://www.who.int/maternal_child_adolescent/topics/child/malnutrition/en/ (accessed 17 Nov 2017).
World Bank 2014 Investing in People to Fight Poverty in Haiti : Reflections for Evidence-based Policy Making World Bank https://openknowledge.worldbank.org/handle/10986/21519 (accessed 17 Nov 2017) .
Notes
- First, second and third authors: Former Graduate Research Assistant, Professor and Professor, respectively, Department of Plant Pathology, The University of Georgia, Tifton, GA 31793; Fourth, fifth, sixth and seventh authors: Former agronomists, Meds and Food for Kids, Quatier-Morin, Haiti; Eighth author: Agronomist, Acceso Peanut Enterprise, Mirebalais, Haiti; Ninth author: Assistant director of the Peanut Innovation Lab, Department of Crop and Soil Science, Athens, GA 30602; Tenth author: Professor, Agronomy Department, The University of Florida, Gainesville, FL 32611. [^] *Corresponding author's E-mail: afulmer.uga@outlook.com
Author Affiliations