Introduction
Spotted wilt of peanut (SWP, syn. tomato spotted wilt) is a disease caused by Tomato spotted wilt virus (TSWV) (Culbreath et al.,, 2003). The virus has been documented to infect over 1000 plant species and is vectored by thrips (Thysanoptera: Thripidae) (Pappu et al., 2009; Parrella et al., 2003). Western flower thrips [Frankliniella occidentalis (Hinds)] and tobacco thrips [F. fusca (Pergande)] constitute the primary vectors in the United States with the latter being arguably more economically important in the southeast (Culbreath et al., 2003; Todd et al., 1995). Since its first detection in a Texas peanut field in 1971 and subsequent easterly spread, SWP has caused substantial yield losses for peanut producing regions of the U.S. (Culbreath and Srinivasan 2011; Halliwell and Philley 1974; Little et al., 2016; Srinivasan et al., 2017). Symptoms range from asymptomatic to complete plant death (Culbreath et al., 1992), with above-ground symptoms including yellow ring spots, chlorotic mottling or streaking on leaves and stunting of the overall plant. Deformed pods, pegs, and kernels are common underground symptoms along with a reddish discoloration of testa and root necrosis (Costa 1941; Culbreath et al., 1992; Culbreath et al., 2003; Halliwell and Philley 1974; McKinney and Tillman 2017; Srinivasan et al., 2017).
Broad-spectrum insecticides such as phorate, acephate, and aldicarb have been widely used to manage thrips in peanut fields (Srinivasan et al., 2017). However, most insecticides provide effective management of thrips larvae, not adults, and lower thrips larvae populations do not always lead to significant reductions in tomato spotted wilt (Culbreath and Srinivasan 2011). Imidacloprid use has increased among producers due to their effectiveness in suppressing thrips injury and convenient liquid formulation. Despite this, imidacloprid can produce a 20 to 200% increase in SWP incidence in susceptible cultivars (Anco et al., 2018; Culbreath and Srinivasan 2011; Marasigan et al., 2016; Marasigan et al., 2018; Srinivasan et al., 2017). Phorate, which has been documented to reduce incidence of SWP beyond thrips control alone, causes a phytotoxic reaction commonly called "phorate-burn". Symptoms include marginal chlorosis on leaves that turns into necrotic lesions. Phorate-burn has been associated with promotion of oxidative stress in peanut, relating to an enhanced defense response to SWP (Culbreath et al., 2008; Jain et al., 2015), and it has seldom been associated with adverse effects on yield (Culbreath and Srinivasan 2011; Marasigan et al., 2016; Marois and Wright 2003).
Cultural practices such as conservation tillage, twin-row planting, and later planting dates (e.g. mid-to-late May) have been associated with reduced SWP intensity (Anco et al., 2018; Brandenburg et al., 1998; Kemerait et al., 2018). These actions target thrips biological and ecological habits, but implementation in some cases may be limited for producers pending the time required to plant large total farm hectarage and potentially adverse weather conditions during planting (Cantonwine et al., 2006; Culbreath et al., 2010; Culbreath and Srinivasan 2011; McKinney and Tillman 2017; Monfort et al., 2017; Tillman et al., 2006). Cultivar selection is often considered to be the most important individual tactic against SWP (Culbreath et al., 2003; Culbreath et al., 2008; Culbreath and Srinivasan 2011; Srinivasan et al., 2017). Following earlier breeding efforts to develop effective resistance, many recently released cultivars exhibit substantial resistance to SWP, such as cv. TifNV-High O/L (Holbrook et al., 2017). However, if disease incidence is intense enough, even cultivars with moderate resistance can still be adversely affected (Culbreath et al., 2003; Srinivasan et al., 2017). An integrated approach that utilizes chemical, cultural, and biological methods has been successfully shown to reduce losses in SWP-infected peanut (Brown et al., 1996; Culbreath et al., 2003; Culbreath and Srinivasan 2011; Marois and Wright 2003; Sundaraj et al., 2014; Tubbs et al., 2013).
Superabsorbent polymers (SAP) are currently used as soil conditioners and encapsulation devices for delivery of some pesticides (de Barros et al., 2017; Campos et al., 2015). Examples of SAP include cross-linked hydrophilic polymers; SAP utilize monomers of acrylamide, acrylic acid, and their salts potassium acrylate or sodium acrylate to absorb large quantities of liquid, 10 to 1000 times their original weight or volume, in a short period of time (Ahmed 2015; de Barros et al., 2017; Horie et al., 2004; Zohuriaan-Mehr and Kabiri 2008). Examples of beneficial uses of SAP in agriculture include enhancing soil permeability and infiltration rates, soil aeration, water consumption, water use efficiency, microbial activity, and overall plant performance by reducing leaching and promoting root development (de Barros et al., 2017; Campos et al., 2015; Dehkordi 2016; Woodhouse and Johnson 1991). In relation to water use, SAP also have potential to reduce irrigation frequency and associated costs by 50%, as well as erosion and runoff (de Barros et al., 2017; Campos et al., 2015; Dehkordi 2016; Woodhouse and Johnson 1991).
Because current recommended practices for integrated management of SWP are limited to the early part of the growing season (Anco et al., 2018), studies were conducted to determine if SAP could be used to augment efficacy of in-furrow insecticides (i.e., through enhanced product retention) to improve management of SWP later in the season. The main objective of these studies was to investigate the effects, application rate, and economics of the use of SAP in combination with standard in-furrow insecticides and cultivars of varying susceptibility. Effects, rates, and economic value of SAP presence in peanut production were hence investigated to determine if SAP could be used to augment efficacy of in-furrow insecticides across cultivar susceptibilities to assist in management of spotted wilt of peanut.
Materials and Methods
Experiments were conducted in 2017 and 2018 in different fields at Clemson University's Edisto Research and Education Center (EREC; 33.364°N, -81.329°E) in Blackville, SC on a Barnwell loamy sand (fine-loamy, kaolinitic, thermic Typic Kanhapludults), at Pee Dee Research and Education Center (PREC; 34.289°N, -79.738°E) in Florence, SC on a Norfolk loamy sand (fine-loamy, kaolinitic, thermic Typic Kandiudults), and at University of Georgia's Lang and Rigdon Farms at the Coastal Plain Experimental Station (CPES; 31.480°N, -83.522°E) in Tifton, GA on a Tifton loamy sand (fine-loamy, kaolinitic, thermic Plinthic Kandiudults). Treatments were applied according to a randomized complete block design and replicated four times at EREC and PREC and six times at CPES. Dimensions of plots at EREC and PREC were four rows on 96-cm centers by 12-m in length. Plots were further separated into two yield rows and two traffic rows. Agrochemical maintenance applications were applied parallel to plots while driving over traffic rows whereas yield rows were used for data collection except where specified. Where possible, considerations for replicate placement incorporated uniformity of soil electrical conductivity (Johnson et al., 2005). At CPES, plots consisted of two 9-m rows on 91-cm centers.
Peanut cultivars were selected based on susceptibility to SWP: susceptible (S), moderately susceptible (M), and resistant (R) (Anco et al., 2018; Kemerait et al., 2018). Cultivar selections for SC in 2017 consisted of TUFRunner 511 (S), Georgia 06G (M), and Sullivan (R); EREC and PREC were planted on 21 April and 3 May, respectively. Seed were treated (Dynasty PD, 3.2% azoxystrobin, 2% fludioxonil, 0.4% mefenoxam, Syngenta Crop Protection, Greensboro, NC) and planted at a rate of 19 seed/m with inoculant applied at the standard rate of 1 L/ha (Optimize Liquid Peanut, 2 × 109 cfu/ml Bradyrhizobium sp., 1 × 10-7% w/w lipo-chitooligosaccharide, Monsanto Company, St. Louis, MO). Insecticides were selected based on industry standards and included phorate (Thimet 20-G, 20% phorate, AMVAC Chemical Corporation, Los Angeles, CA) applied with a SmartBox calibrated to deliver 51 g a.i./100 m and imidacloprid (Admire Pro, 42.8% imidacloprid, Bayer CropScience, Research Triangle Park, NC) applied at 401.5 g a.i./ha with a D2 orifice set to deliver 81 L/ha at 221 kpa into the seed furrow. In-furrow granular SAP (Aquasorb 3005KM, SNF Floeger, Andrézieux, France) was applied with a Microsem box calibrated to deliver a product rate of 2.24 kg/ha. Cultivars examined at CPES in 2017 included FloRun 157 (S), Georgia 06G, and TifNV-High O/L (R) and were planted on 27 April at a rate of 19 seed/m. For experiments in 2018, all three locations planted TUFRunner 511, Georgia 06G, and TifNV-High O/L on the following dates: 27 April (CPES), 30 April (EREC), and 11 May (PREC). Standard field management practices were performed throughout the growing seasons (Anco et al., 2018; Monfort et al., 2017). Detailed descriptions of treatments are listed in Table 1.
Stand emergence approximately 14 d after planting (DAP) was determined by counting total emerged peanut plants from two arbitrary 1.22-m sections of individual plot yield rows. Total counts were then converted to plants per m. Thrips injury observations were made at EREC at 24 (15 May) and 35 DAP (26 May), PREC at 29 (1 June) and 36 DAP (8 June), and CPES at 27 DAP (24 May) in 2017. Observations in 2018 were performed 28 (28 May) and 35 DAP (4 June) at EREC, 28 (8 June) and 34 DAP (14 June) at PREC, and 35 DAP (1 June) at CPES. Thrips injury was rated using a visual scale (0 = no damage, 1 = 10% injured leaves, 2 = 20% injured leaves, 3 = 30% injured leaves, 4 = 40% injured leaves, 5 = 50% injured leaves, 6 = 50% injured leaves + 5% terminal buds injured, 7 = 50% injured leaves + 25% terminal buds injured, 8 = 50% injured leaves + 50% terminal buds injured, 9 = 50% injured leaves + ≥ 90% terminal buds injured, 10 = complete plant death) (Brandenburg et al., 1998) with interval continuity assumed. The first and last 0.3 m of each plot row was excluded from rating to reduce edge effects (Brown et al., 1996; Culbreath and Srinivasan 2011). Phytotoxicity was rated concurrently with thrips injury observations, except at CPES where phytotoxicity was not rated, using a 0 to 100% scale based on severity.
SWP incidence was evaluated at 56 (16 June), 70 (30 June), and 126 DAP (25 August) at EREC, 68 (10 July) and 121 DAP (1 September) at PREC, and 62 (28 June), 83 (19 July), 114 (19 August), and 133 DAP (7 September) at CPES in 2017. In 2018, incidence was rated 56 (25 June), 70 (9 July), and 119 DAP (27 August) at EREC, 55 (5 July), 70 (20 July), and 119 DAP (7 September) at PREC, and 82 (18 July), 115 (20 August), 125 (30 August), and 137 DAP (11 September) at CPES. Incidence per plot was quantified by summing non-overlapping discrete 0.3-m sections of peanut plants expressing symptoms of spotted wilt and converted to a proportion by dividing by the total ratable row-m per plot (i.e., excluding the ends of plots to avoid edge effects). Confirmation of the presence of Tomato spotted wilt virus was performed 27 August (119 DAP, EREC), 31 August (125 DAP, CPES), and 7 September (119 DAP, PREC) 2018 by using corresponding ImmunoStrip Kits (model ISK 39300, Agdia Inc., Elkhart, IN) on two arbitrarily selected symptomatic leaves from each plot containing untreated TUFRunner 511 peanuts.
Plots were inverted on 12 September (EREC), 29 September (PREC), and 22 September (CPES) in 2017 and 20 September (EREC), 26 October (PREC), and 17 September (CPES) in 2018. Yield data from 2017 were collected 20 September (EREC), 2 November (PREC), and 26 September (CPES) using combines fitted with a load cell basket. In 2018, plots were combined 24 September (EREC), 31 October (PREC), and 24 September (CPES). Peanuts were graded and tested for aflatoxin by the South Carolina Department of Agriculture and Golden Peanut and Tree Nuts (Albany, GA) in 2017 and Carolina Peanut (Cameron, SC) and Palmetto Peanut (Cameron, SC) in 2018. Only EREC and CPES peanuts were graded. Economic value was calculated as treatment net loan value × treatment yield - treatment cost. To determine treatment net loan value, 500-g pod samples per plot were graded by the SC Department of Agriculture Peanut Inspection Service using USDA grading standards (USDA FSA 2018a). Net loan values per 1000 kg for segregation I runner and Virginia types were calculated using the following formulae (Cantonwine et al., 2006; USDA FSA 2018a):
where %TSMK is the percent total sound mature kernels, %OK is the percent of other kernels, and %ELK is the percent of extra-large kernels. Loan rates for 2018 were $4.806 (%TSMK for runner market types), $4.902 (%TSMK for Virginia market types), $1.40 (%OK), and $0.35 (%ELK) (USDA FSA 2018b). Deductions were $0.80 for every percent of sound splits over 4%. If peanuts graded as segregation II or III, corresponding net loan values were $0.137/kg (runner types) and $0.139/kg (Virginia types). Local treatment costs per hectare for phorate, imidacloprid and SAP were respectively estimated at $37.07, $17.05 and $39.54. For the purposes of this study, differences in potential labor, time and equipment depreciation values were not factored into treatment ha value calculations since those factors were relatively constant across all treatments. Actual drying and hauling costs, excluded here for simplicity, would be adjusted based on yield.
The GLIMMIX procedure of SAS 9.4 (SAS Institute, Cary, NC) was used to analyze treatment effects for stand emergence, thrips injury, phytotoxicity, SWP incidence, AUDPC, yield, and acre value according to generalized linear mixed modeling. Laplace approximation was used to improve standard error estimation (Pinheiro and Chao 2006; Stroup 2013). Factors considered fixed effects included cultivar susceptibility, insecticide, presence of SAP, and corresponding interactions. Random effects included replication (individual location-year models) or replication by location by year + location by year + year + location by year by treatment (models over multiple locations and years). Where random effects were estimated to be zero or did not improve fit of the model (i.e., via lower corrected Akaike's information criterion; Stroup 2013), they were excluded. In general, responses were modeled according to the Gaussian distribution. In cases where the Gaussian distribution produced inadequate fits (Stroup 2013), data were modeled according to alternative distributions (e.g., yield data was modeled using the negative binomial distribution). Fisher's protected least significant difference (LSD) was used for pairwise comparisons at 𝜶 = 0.05. For select pairwise comparisons, Fisher's unprotected LSD (𝜶 = 0.05) was utilized to comment on P values greater than 0.05 but less than 0.1.
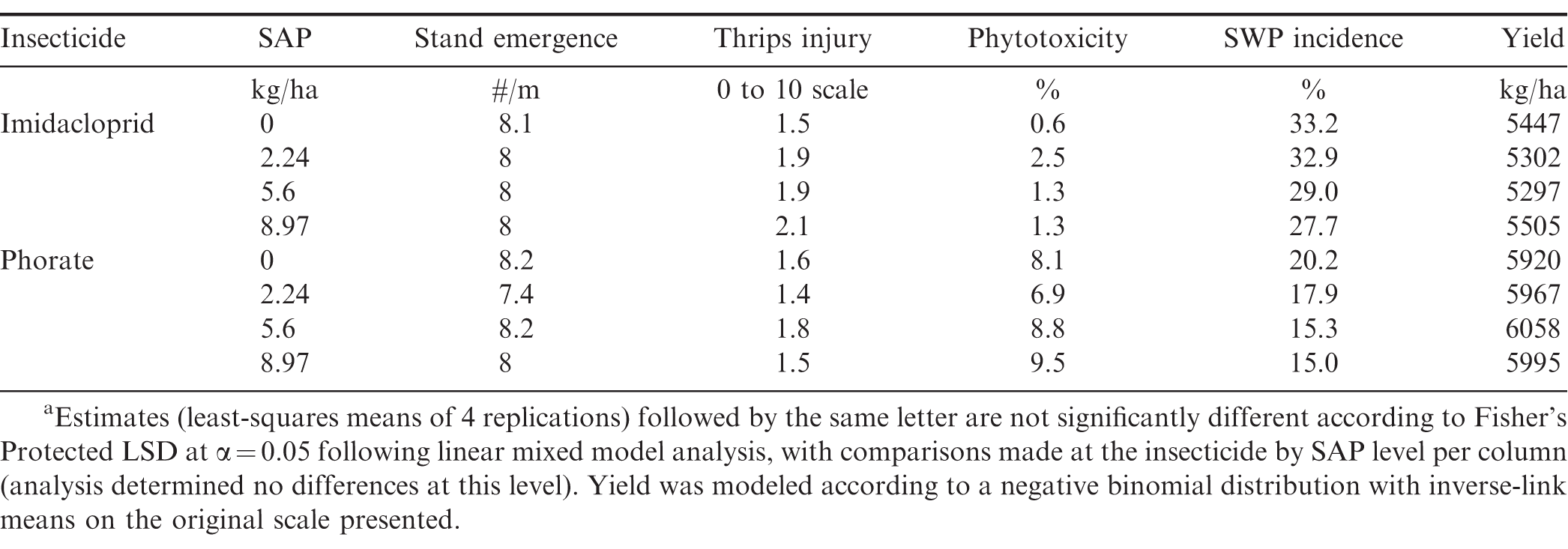
Additional field experiments were conducted in 2017 and 2018 at EREC (Barnwell loamy sand) to compare effects of varying in-furrow SAP rates (0, 2.24, 5.6, and 8.97 kg/ha) in combination with imidacloprid or phorate in TUFRunner 511 peanuts. Experimental design, conduct, data collection, and analysis were as previously described. Trials were planted on 26 May 2017 and 2018. Thrips injury was rated 30 DAP (26 May) in 2017 and 34 (30 May) DAP in 2018, with phytotoxicity rated at the same timings. SWP incidence was evaluated 121 DAP (8 August) in 2017 and 119 DAP (23 August) in 2018. Plots were inverted on 15 September (2017) and 20 September (2018) and harvested 20 September (2017) and 24 September (2018). In addition to the previously described analysis, the ROBUSTREG procedure (SAS 9.4, Cary, NC) was utilized to conduct regression analysis of estimated SWP incidence as a function of the rate of SAP used per insecticide and year.
Results and Discussion
SAP has been used in agricultural applications before (Bai et al., 2013; de Barros et al., 2017; Bhardwaj et al., 2007; Busscher et al., 2009; Campos et al., 2015; Dehkordi 2016; Ekebafe et al., 2011; Hayat and Ali 2004; Pouci et al., 2008; Sojka and Lentz 1997), but to our knowledge, this was the first study to examine its utility in the peanut - Tomato spotted wilt virus system. Spotted wilt incidence in different cultivars and treatment regimens (in the absence of SAP) has been the subject of several prior studies (Branch et al., 2003; Culbreath et al., 2000; Culbreath et al., 2016; Drake et al., 2009; Herbert Jr. et al., 1991; Lynch et al., 1984; Marasigan et al., 2018). As such, while the present study sought to examine treatment effects across cultivars with varying susceptibility, cultivar susceptibility-level comparisons were not the focus of this work.
Emergence
Collectively, SAP increased peanut emergence compared to treatments without SAP in 2017 at EREC (P = 0.0085) by 0.9 plants/m, but this was not observed the subsequent year where SAP tended to weakly decrease emergence by 0.3 plants/m (P = 0.0854) (Table 2). The difference in emergence may have been associated with soil conditions when comparing the two years. Weather conditions were relatively dryer between planting and emergence counts in April and May in 2018 than in 2017 at EREC (19 mm in 2017 and 0 mm in 2018), whereas rainfall during that time in 2018 at PREC was increased by 5.3 mm compared to 2017 (Weather Underground 2019). Lack of precipitation at EREC during the emergence period would plausibly prevent SAP's water-holding capacity from providing those peanut seeds with greater stored moisture compared to peanut seed planted without SAP, and so it is not unexpected that emergence under those conditions did not differ among SAP treatments. The only significant interaction of SAP for emergence was at PREC in 2018 where its interaction with insecticide was at P = 0.0329. From this interaction, imidacloprid without SAP had greater emergence (by 1.4 plant/m) than imidacloprid with SAP, whereas phorate with SAP did not increase emergence compared to phorate without SAP (Table 2).
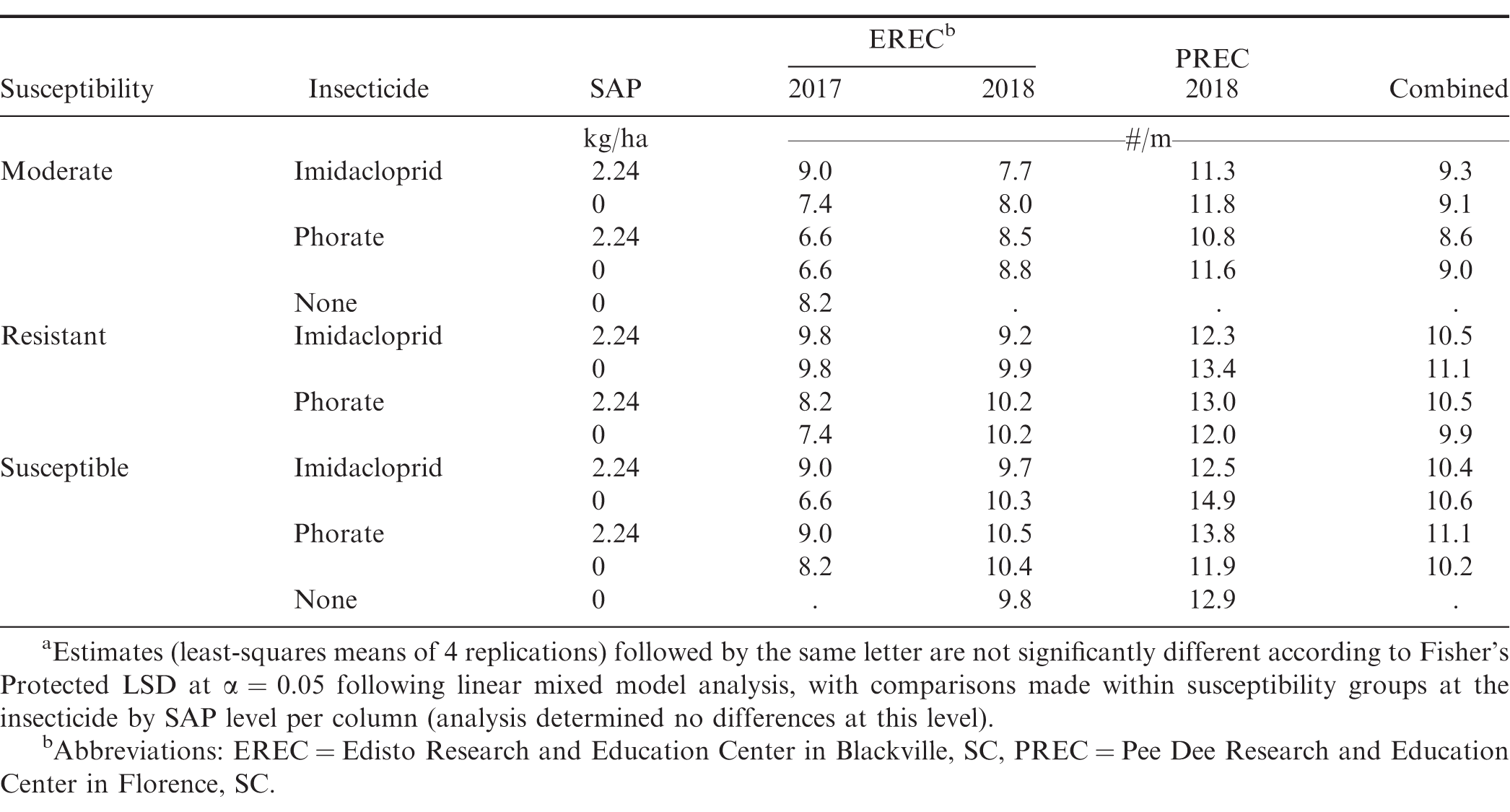
Cultivar susceptibility affected stand counts across the locations and years examined (P < 0.007), with the moderately susceptible cultivar (Georgia 06G) having less emerged plants than resistant and susceptible cultivars. Insecticide (P = 0.0242) and its interaction with cultivar susceptibility (P = 0.003) affected stand counts in 2017 at EREC. Greater counts were observed in moderately susceptible and resistant cultivars treated with imidacloprid compared to phorate, whereas susceptible peanuts did not exhibit a difference between the two insecticides overall (Table 2). At EREC in 2017, imidacloprid-treated plots had more emerged peanuts than phorate-treated plots, but this was reversed in 2018 at EREC (P = 0.0041) and not significant at PREC or in the pooled analysis (P > 0.22).
Phytotoxicity
For simplicity, only phytotoxicity results from 24 to 29 DAP are presented. Across both years and locations (EREC and PREC) and consistent with previous research demonstrating that phorate can injure peanut seedlings (Culbreath et al., 2003; Culbreath et al., 2016; Culbreath and Srinivasan 2011; Herbert et al., 2007; Hurt et al., 2005; Tubbs et al., 2015), peanut treated with phorate generally expressed higher levels of phytotoxicity than those treated with imidacloprid or not treated with insecticide (Table 3). Insecticide was consistently significant at P < 0.01. Over both locations in 2017 as well as in the combined analysis, phytotoxicity varied among cultivar susceptibility (P < 0.001), where Georgia 06G, the only moderate-susceptibility cultivar in the study, treated with phorate with or without SAP either exhibited the greatest phytotoxicity or was within 2% of the greatest phytotoxicity observed (Table 3). While the mechanistic elucidation of this general observation was outside the scope of this study, it is plausible the increased phytotoxicity observed in Georgia 06G was related to how this cultivar was noted to grow slower overall compared to the other examined cultivars (Grey et al., 2011; Holbrook et al., 2017; Tillman and Gorbet 2017). This possibility is corroborated by the (P < 0.0001) lower emergence observed from the combined analysis (9 plants/m compared to 10.5 and 10.6 for resistant and susceptible cultivars, respectively), though formal canopy measurements were not conducted in this study. Slower growth rates would correspond to less total foliage available to absorb or metabolize the same applied rate of phorate (Wehtje et al., 1991), which reasonably could have contributed to a greater proportion of apparent phytotoxicity. The effect of susceptibility by insecticide was also significant (P < 0.02) across all datasets and the combined data, with the exception of PREC in 2018, with observed results generally following the observations noted with Georgia 06G. In addition, imidacloprid was not generally associated with phytotoxicity across cultivars. While neither SAP alone nor its interactions affected phytotoxicity in most cases, a significant interaction with insecticide (P = 0.033) was observed in PREC in 2017. In that location-year, phorate treated with SAP had 4.2% less phytotoxicity than phorate without SAP (P = 0.0005). Phytotoxicity was not affected by SAP rate in either year (Table 4).
Thrips injury
Insecticide treatment affected thrips injury (P < 0.0001, Table 5) across locations and years. The interaction of susceptibility and insecticide was significant (P = 0.0004) in 2017 at CPES, where untreated susceptible peanut (FloRun 157) had more thrips injury than other cultivars (5.6 compared to approximately 4.8, Table 5), as well as in the combined analysis (P = 0.0253). Marasigan et al., (2016, 2018) has previously reported imidacloprid to be as effective as phorate in managing thrips injury, which was reflected in this study. Research from other studies indicate that insecticide treatments tend to provide good management of larval feeding damage (Chamberlin et al., 1992b; Chamberlin et al., 1993; Culbreath and Srinivasan 2011) but were ineffective in killing adults that migrate from outside the field (Chamberlin et al., 1992a; Funderburk et al., 1998; Herbert Jr. et al., 1991; Hurt et al., 2005). In this study, injury was not categorized by the stage of thrips causing the injury. Regardless, both insecticides beneficially contributed to overall thrips management. With the two following exceptions, thrips injury was not affected by SAP or its interactions. The insecticide × SAP interaction was significant in 2017 at PREC (P = 0.0028). Treatments with SAP generally decreased injury compared to those without SAP in both insecticide groups, with this difference being significant for imidacloprid (-0.7, P = 0.0143) but not for phorate (-0.3, P = 0.3429). Treatment with SAP also affected thrips injury (P = 0.0092) at CPES in 2017, where treatments with SAP had less injury, albeit by a small margin: 3.1 compared to 3.4. In 2018, cultivar susceptibility affected thrips injury at EREC and at CPES (P < 0.03). Thrips injury was not affected by SAP rate by itself or its interaction with insecticide (P > 0.5).
Spotted wilt of peanut incidence
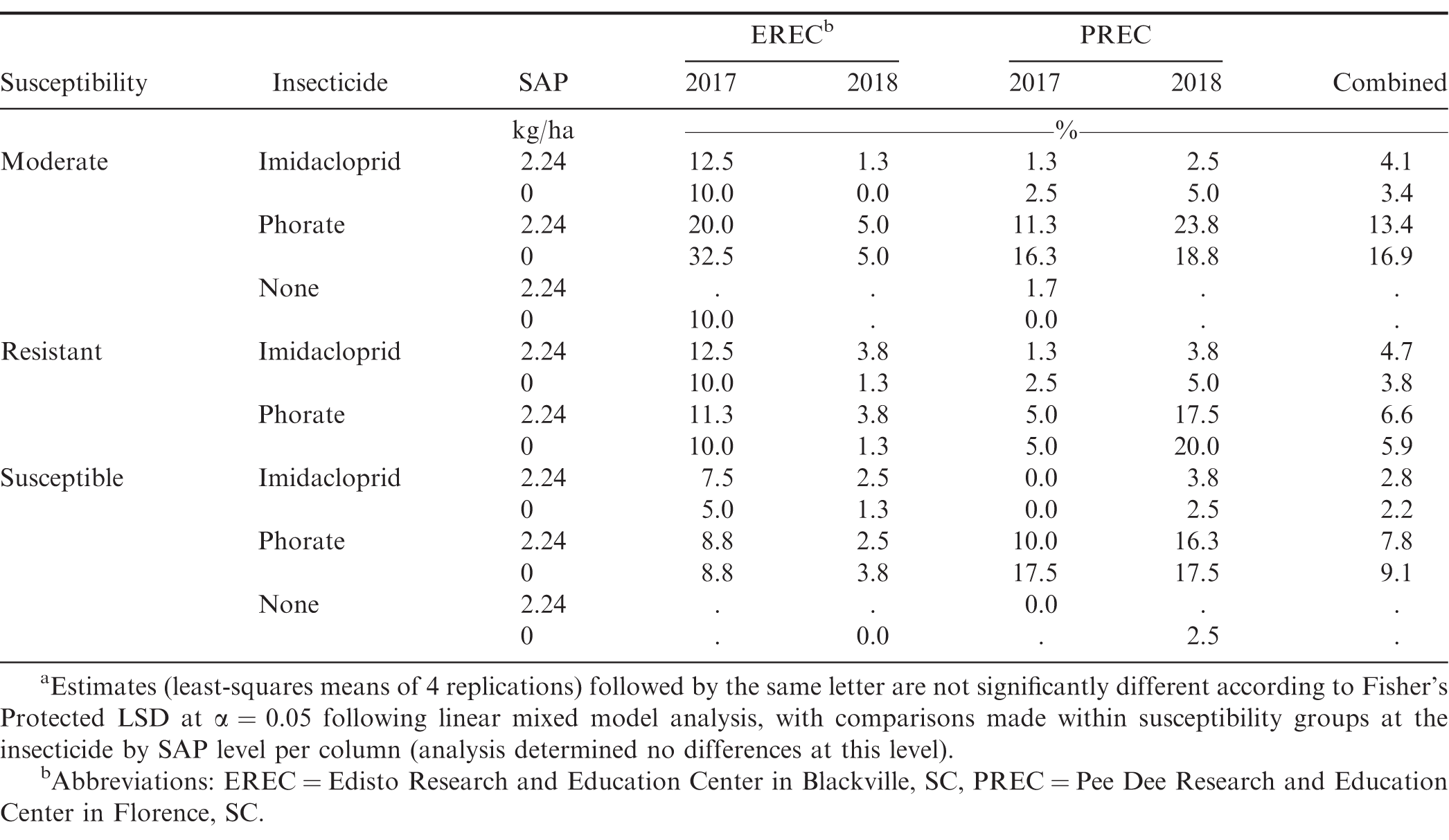
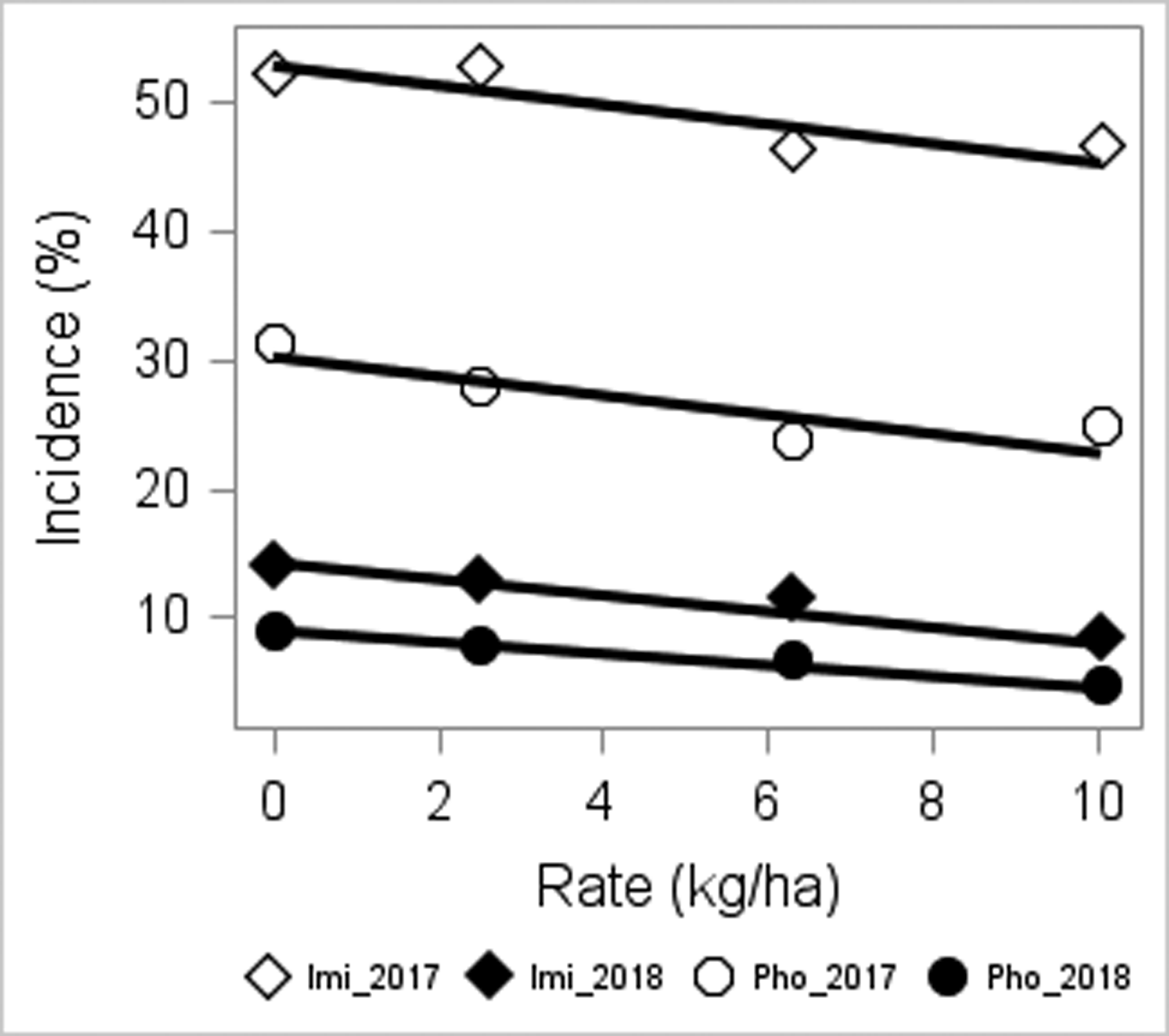
ImmunoStrip Kits used in 2018 confirmed presence of Tomato spotted wilt virus at all testing sites. Cultivar susceptibility affected SWP incidence (P < 0.01), and characteristically, resistant cultivars had mostly lower incidence throughout the study compared to other cultivars (Table 6). Insecticide treatment was also significant (P < 0.02), with the exception of PREC in 2017 where insecticide was marginally significant at P = 0.091. In combined data, peanut with phorate had statistically lower incidence compared to imidacloprid (Table 6) (9.9 vs. 16.5%, respectively), which reflected previous studies (Culbreath et al., 2003; Culbreath et al., 2008; Culbreath et al., 2016; Culbreath and Srinivasan 2011; Hurt et al., 2005). Susceptibility × insecticide affected incidence of SWP at EREC and CPES in 2017, as well as at PREC and CPES in 2018, and this interaction was moderately significant in the combined data (P = 0.0547). Overall, the difference in SWP incidence that developed following imidacloprid compared to phorate treatment was significant for moderate and susceptible cultivars but was not for resistant cultivars (7.4 at P < 0.0001, 9.4 at P < 0.0001, and 3.1% at P = 0.0710, respectively). This corroborates previous reports describing cultivar phenotypes during spotted wilt epidemics (Anco et al., 2018; Branch 2007; Kemerait et al., 2018; McKinney and Tillman 2017; Tillman et al., 2006) and that phorate is more effective in managing this disease. With a few localized exceptions, SAP-related effects generally did not (P > 0.07) affect SWP incidence across location-years. In 2017 at PREC the interaction of cultivar susceptibility by insecticide by SAP was significant at P = 0.0542, within which SWP varied for the susceptible cultivar (P = 0.0397) but not for the moderate or resistant cultivars (P = 0.29). However, in addition to the lack of this observation being significant in other location-years, it is further interesting to note that at PREC in 2017 the susceptible cultivar treated with phorate in the absence of SAP uncharacteristically exhibited higher SWP incidence compared to the corresponding imidacloprid treatment (18.0 compared to 12.7%, respectively). As a result, the phorate + SAP treatment (7.8%) had less SWP incidence than phorate treatment without SAP. Accordingly, it is unsurprising that this three-way interaction was not observed across location-years (i.e., SAP effects elsewhere and overall were marginal, whereas SWP incidence more frequently varied according to the interaction of susceptibility by insecticide). At CPES in 2018 treatment with SAP was associated with a 2.3% decrease in SWP incidence at a significance level of P = 0.0687, but again, this was not observed in other location-years nor in the combined analysis. At PREC in 2018, the effect of SAP was also marginal at P = 0.0889, with a comparable magnitude, 2.0%, observed compared to the 2018 CPES data. When estimated SWP incidence from the SAP rate trial was regressed against SAP rates, significant relationships were observed. Parameter estimates are listed in Figure 1. Corresponding estimated parameter P-values were as such: imidacloprid 2017 intercept P <0.0001, SAP rate slope P = 0.0233; imidacloprid 2018 intercept P <0.0001, SAP rate slope P <0.0001; phorate 2017 intercept P = <0.0001, SAP rate slope P = 0.0314; phorate 2018 intercept P = <0.0001, SAP rate P <0.0001. In 2018 for both insecticide models, 5.6 kg/ha was identified as a substantial outlier and excluded from the robust models (standardized robust residuals > 11, cutoff = 3). The removal of these outliers reduced both applicable model R2 values from ∼0.97 to ∼0.72. Combined-year models per insecticide were examined (data not shown) but did not produce appropriate fits given the variability among years and available data points. From these regression models, a decrease in SWP incidence with increasing SAP rate was observed in both years of the study for both insecticides. When the 2017 - 2018 data were examined within the mixed model, SAP rate was significant overall at P = 0.0641, and SWP incidence began to become different (lower) than treatments without SAP as the rate of SAP increased past 2.24 to 5.6 and 8.96 kg/ha: P = 0.5729, 0.0507, and 0.0213, respectively, with corresponding estimated differences of 1.3, 4.5 and 5.4%. Differences between higher rates of SAP, e.g., 2.24 and 8.96 kg/ha, were significant (P > 0.075).
Spotted wilt incidence as a function of increasing superabsorbent polymer (SAP) rate for imidacloprid and phorate treatments in 2017 and 2018. Estimated regression equation parameters and R2 values for each insecticide-year (Imi = imidacloprid, Pho = phorate) were as follows: Imi_2017, Y = 52.780 - 0.843 × SAP rate, R2 = 0.742; Imi_2018, Y = 14.172 - 0.704 × SAP rate, R2 = 0.718; Pho_2017, Y = 30.160 - 0.834 × SAP rate, R2 = 0.701; Pho_2018, Y = 8.897 - 0.497 × SAP rate, R2 = 0.718.
Results from the susceptibility × insecticide treatment × SAP trial indicated that SAP presence did not have a consistently substantial effect on spotted wilt incidence. Despite this, certain phenomena of cultivar - treatment combinations were observed. Over the combined data, susceptible cultivars treated with phorate and SAP mostly exhibited lower SWP incidence (by 4.8%) than their counterparts without SAP. Previous reports have shown that phorate can lower SWP incidence through induction of plant defense responses (Jain et al., 2015). It may be possible that this combination with SAP promoted a stronger effect compared to phorate alone. Increased spotted wilt was generally observed in TUFRunner 511 and FloRun 157, the susceptible cultivars, and those left as untreated checks or treated with imidacloprid. Previous studies have indicated that imidacloprid can cause an uptick in spotted wilt incidence by promoting activity in thrips movement (Culbreath et al., 2003; Culbreath and Srinivasan 2011).
Yield
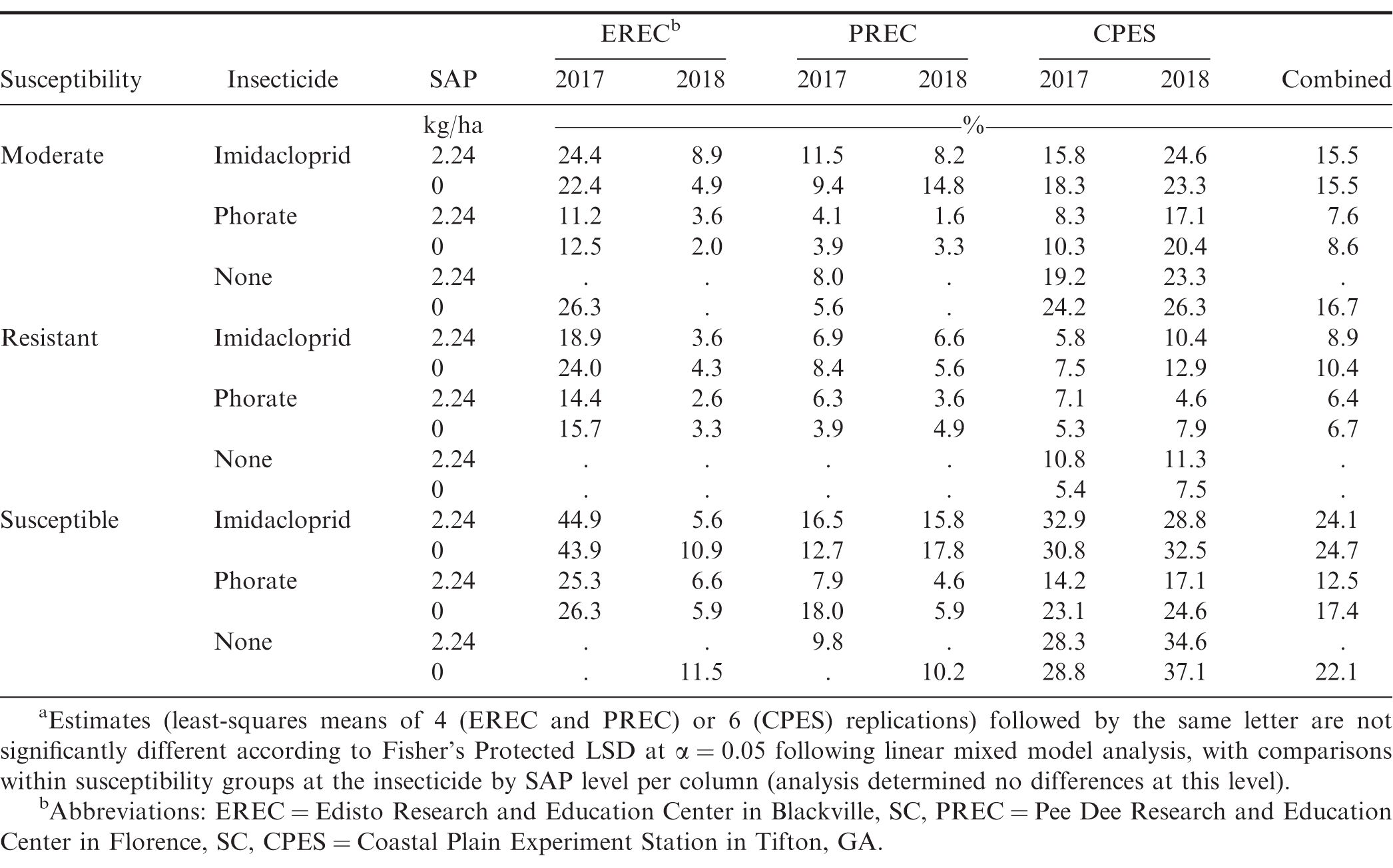
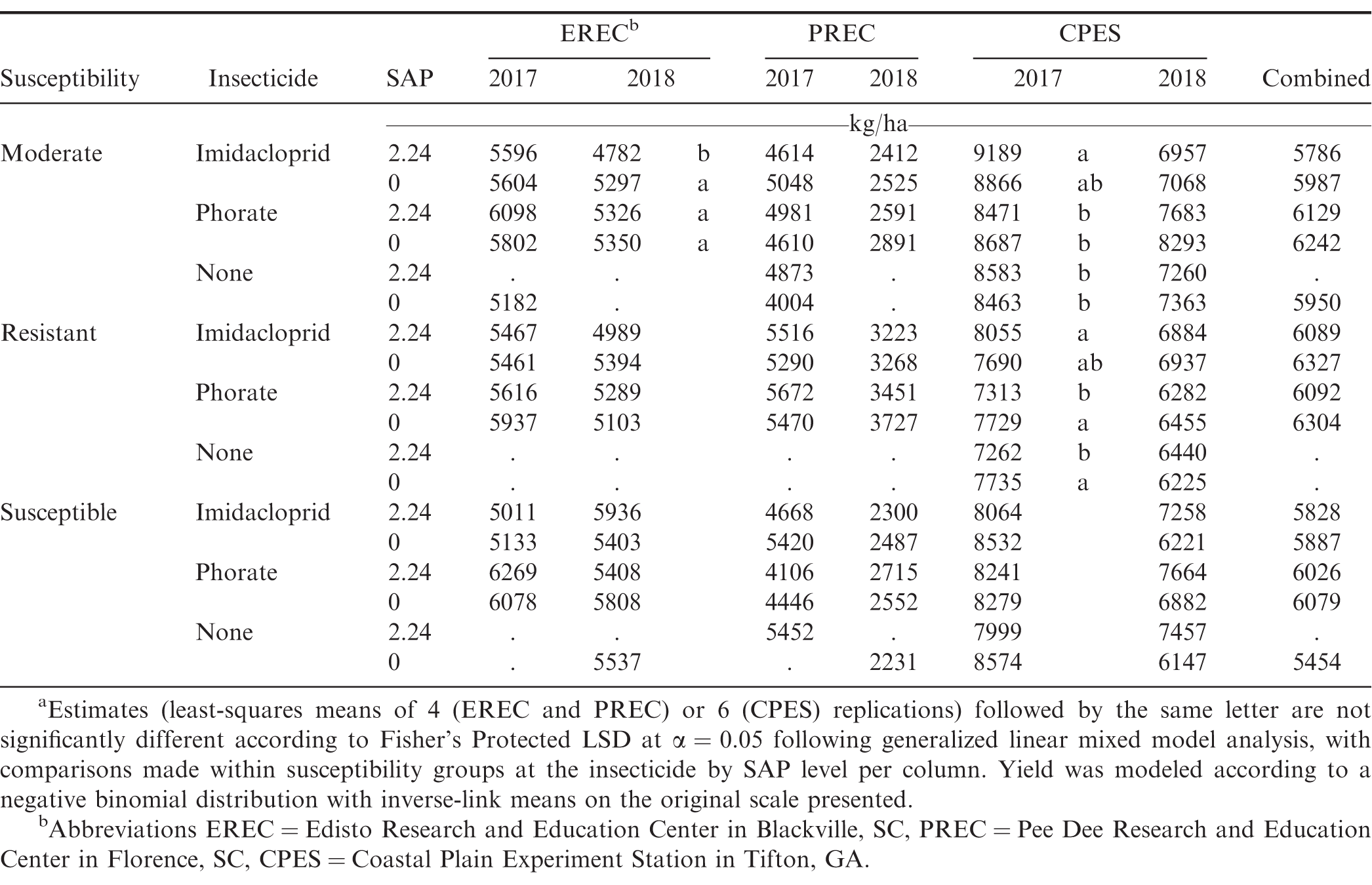
In the combined analysis, yield was not affected by any of the examined factors or interactions (P > 0.21); accordingly, further yield results discussion will pertain to the location-year data. However, it should be noted that while the following yield results and discussion reference cultivar susceptibility groups out of convenience as well as out of a result of the experimental design, actual yield (and subsequent economic value) differences could very well have been due to other genetic factors (beyond susceptibility to SWP) harbored by the specific genotypes examined in these experiments. Over each of the location-years with the exception of EREC in 2017, yield was affected by cultivar susceptibility (P < 0.02). At PREC, resistant cultivars had statistically higher yields compared to moderately susceptible and susceptible cultivars, while at CPES, the moderately susceptible cultivar had greater yields over susceptible and resistant cultivars. Susceptible cultivars also had increased yield compared to resistant cultivars at CPES. Susceptibility was often a significant factor for yield and appeared to have localized effects, plausibly a result of genotype × environment interactions that were not the focus of this study. Cultivars that performed numerically best at EREC were susceptible (TUFRunner 511), PREC were resistant (Sullivan and TifNV-High O/L), with the moderately susceptible cultivar (Georgia 06G) performing best at CPES (Table 7). Insecticide treatment was also significant at EREC and CPES in 2017 (P < 0.009). At EREC, phorate-treated plots had increased yields over those treated with imidacloprid (5960 compared to 5370 kg/ha), whereas at CPES imidacloprid-treated plots had increased yields (8380 compared to 8110 kg/ha). The effect of susceptibility × insecticide was also a significant factor at EREC (P = 0.0006) in 2017, where phorate-treated plots had greater yields with moderately susceptible and susceptible cultivars, whereas phorate only numerically increased yields in resistant cultivars. Yield was not affected (P > 0.05) by SAP-related effects at EREC (except where specified later for susceptibility × insecticide × SAP in 2018) or PREC. While treatments with SAP overall had lower (P = 0.0435) yields at CPES in 2017 (8110 compared to 8270 kg/ha), this was not observed in other location-years. While the interaction of susceptibility × SAP was consistently significant at CPES in 2017 and 2018 (P = 0.0821 and 0.0833, respectively), the subsequent related results were inconsistent between the two years. In 2017, addition of SAP was associated with reduced yield for the susceptible cultivar (8100 compared to 8460 kg/ha), whereas in 2018 it was associated with greater yield for the susceptible cultivar (7460 compared to 6410 kg/ha); in both years, yield of moderate and resistant cultivars were not affected. In 2018, the effect of susceptibility × insecticide × SAP affected yields (P = 0.0023) at EREC (Table 7), resulting in a difference for treatments in the moderately susceptible group, where imidacloprid with SAP had less yield compared to remaining treatments (Table 7). Differences within the resistant and susceptible groups were not significant. At CPES in 2017, the overall susceptibility × insecticide × SAP interaction was significant at P = 0.0573, and this interaction was more significant when examined among the partitioned (sliced) moderate and resistant susceptibility groups: P = 0.0432 and 0.0128, respectively. For the moderate cultivar, yield from imidacloprid + SAP was not different from imidacloprid without SAP but was greater than all other treatments. From the resistant cultivar data, addition of SAP numerically increased yield for imidacloprid treatment but significantly reduced yield for phorate and untreated treatments (Table 7). Among insecticide treatments, phorate did not consistently result in statistically greater yields in combined analysis of years and locations (Table 7). Previous reports from Herbert et al., (2007) and Tubbs (2013) have indicated yield to be increased following phorate application. However, it has also been reported that phorate has an inconsistent effect on peanut yields (Culbreath et al., 2008; Marois and Wright 2003). Hayat and Ali (2004) saw an increase of yield in tomato plants when including SAP (12.5 g/kg) mixed into the soil, however other reports did not find any significant or consistent differences in other crops (Busscher et al., 2009; Hou et al., 2017).
Economic value
Treatment economic value was not affected by SAP or its interactions with other examined factors (P > 0.23). Insecticide treatment alone did not affect economic value (P = 0.1029), but insecticide did interact with susceptibility (P = 0.0244). While no significant differences were observed for moderate and resistant cultivars, susceptible cultivars treated with phorate had greater economic value ($2,637/ha) compared to susceptible cultivars treated with imidacloprid ($2,417/ha) (Table 8). Treatments also differed (P = 0.0008) with cultivar susceptibility, with moderately susceptible ($2,634/ha) and susceptible ($2,523/ha) cultivars having better economic value compared to resistant cultivars ($2,417/ha), P = 0.0002 and 0.0503, respectively; the difference between moderate and susceptible cultivars was also was fairly significantly (P = 0.0534). Regarding aflatoxin and grade segregations, two of the 244 samples analyzed from yields taken at EREC and CPES in 2017 and 2018 were classified segregation II, and two more samples had segregation III classifications; of the corresponding treatments, none included SAP. Of the two segregation II samples, one was from an untreated susceptible treatment ($784/ha) and the other was from an imidacloprid treated susceptible cultivar ($712/ha). Both of the segregation III samples ($578 and $749/ha) were from no-insecticide moderately susceptible treatments.
Spotted wilt disease of peanut was not conclusively mitigated using superabsorbent polymer in these studies. Still, decreases in SWP incidence were frequently observed for susceptible cultivars paired with phorate and polymer compared to phorate alone across location-years, with similar albeit more mitigated effects observed from the moderately susceptible cultivar. Further research should be performed to clarify the mechanisms behind this phenomenon. Superabsorbent polymer could be useful in situations where as much additional protection against SWP as possible is warranted. However, in taking into consideration the variability of the results presented from these studies combined with its current estimated material costs, use of SAP in commercial peanut production at this time does not appear to be cost effective and as such is not recommended at this time.
Acknowledgements
Support for this work was provided by the South Carolina Peanut Board, National Peanut Board, USDA NIFA under project number SC-1700532, AMVAC Chemical, and Georgia Foundation Seed. The authors thank James Thomas, Justin Hiers, Carlysle Coker, staff at Pee Dee Research and Education Center, and staff at Coastal Plain Experimental Station for their vital assistance. Appreciation is given to three anonymous reviewers for providing constructive comments that improved this manuscript. Technical Contribution No. 6769 of the Clemson University Experiment Station.
Literature Cited
E Ahmed, (2015). Hydrogel: preparation, characterization, and applications: A review. J. Adv. Res 6: 105- 121.
D., Anco, J Thomas, M Marshall, N Smith, and K Kirk (2018). Peanut Money Maker - 2018 Production Guide. Clemson Cooperative Extension, .
W., Bai, J Song, and H Zhang (2013). Repeated water absorbency of super-absorbent polymers in agricultural field applications: A simulation study. Acta Agric. Scand. B 63: 433- 441.
A., de Barros, L Pimentel, E Araujo, L de Macedo, H Martinez, V Batista, and M da Paixão (2017). Super absorbent polymer application in seeds and planting furrow: It will be a new opportunity for rainfed agriculture. Semina: Ciências Agrárias 38: 1703- 1714.
A., Bhardwaj, I Shainberg, D Goldstein, D Warrington, and G Levy (2007). Water retention and hydraulic conductivity of cross-linked polyacrylamides in sandy soils. Soil Sci. Am. J 71: 406- 412.
W Branch, (2007). Registration of 'Georgia-06G' Peanut. J. Plant Regist 1: 120.
W., Branch, T Brenneman, and A Culbreath (2003). Tomato spotted wilt virus resistance among high and normal O/L ratio peanut cultivars with and without irrigation. Crop Prot 22: 141- 145.
R., Brandenburg, D Herbert G Sullivan, G Naderman, and S Wright (1998). The impact of tillage practices on thrips injury of peanut in North Carolina and Virginia. Peanut Sci 25: 27- 31.
S., Brown, J Todd, and A Culbreath (1996). Effect of selected cultural practices on incidence of Tomato spotted wilt virus and populations of thrips vectors in peanuts. Acta Hortic 431: 491- 498.
W., Busscher, D Bjorneberg, and R Sojka (2009). Field application of PAM as an amendment in deep-tilled US southeastern coastal plain soils. Soil Tillage Res 104: 215- 220.
E., Campos, J de Oliveira, L Fraceto, and B Singh (2015). Polysaccharides as safer release systems for agrochemicals. Agron. Sustain. Dev 35: 47- 66.
E., Cantonwine, A Culbreath, K Stevenson, R Kemerait, T Brenneman, N Smith, and B Mullinix, (2006). Integrated disease management of leaf spot and spotted wilt of peanut. Plant Dis 90: 493- 500.
Chamberlin, J., J Todd, R Beshear, A Culbreath, and J Demski 1992 a Overwintering hosts and wingform of thrips, Frankliniella spp., in Georgia (Thysanoptera: Thripidae): Implications for management of spotted wilt disease Environ. Entomo 21: 121- 128.
J., Chamberlin, J Todd, A Culbreath, W Johnson, and J Demski (1993). Post-harvest management of tobacco thrips (Thysanoptera, Thripidae) overwintering in peanut fiends. J. Entomol. Sci 28: 433- 446.
Chamberlin, J., J Todd, J Farrow, and B Mullinix 1992 b Aldicarb residue persistance in leaf terminals of 'Florunner' peanut J. Econ. Entomol 85: 1072- 1078.
A Costa, (1941). Una molestia de virus do amendoim (Arachis hypogaea L.). A mancha anular. Biologico 7: 249- 251.
A., Culbreath, A Selph, B Williams, R Kemerait, R Srinivasan, M Abney, B Tillman, C Holbrook, and W Branch (2016). Effects of new field resistant cultivars and in-furrow applications of phorate insecticide on tomato spotted wilt of peanut. Crop Prot 81: 70- 75.
A., Culbreath, and R Srinivasan (2011). Epidemiology of spotted wilt disease of peanut caused by Tomato spotted wilt virus in the southeastern U.S. Virus Res 159: 101- 109.
A., Culbreath, B Tillman, D Gorbet, C Holbrook, and C Nischwitz (2008). Response of new field-resistant peanut cultivars to twin-row pattern or in-furrow applications of phorate for management of spotted wilt. Plant Dis 92: 1307- 1312.
A., Culbreath, B Tillman, R Tubbs, J Beasley, R Kemerait, and T Brenneman (2010). Interactive effects of planting date and cultivar on tomato spotted wilt of peanut. Plant Dis 94: 898- 904.
A., Culbreath, J Todd, and S Brown (2003). Epidemiology and management of tomato spotted wilt in peanut. Annu. Rev. Phytopathol 41: 53- 75.
A., Culbreath, J Todd, J Demski, and J Chamberlin (1992). Disease progress of spotted wilt in peanut cultivars Florunner and Southern Runner. Phytopathology 82: 766- 771.
A., Culbreath, J Todd, D Gorbet, S Brown, J Baldwin, H Pappu, and F Shokes (2000). Reaction of peanut cultivars to spotted wilt. Peanut Sci 27: 35- 39.
D Dehkordi, (2016). The effects of superabsorbent polymers on soils and plants. Pertanika J. Trop. Agric. Sci 39: 267- 298.
W., Drake, D Jordan, B Lassiter, P Johnson, R Brandenburg, and B Royals (2009). Peanut cultivar response to damage from tobacco thrips and paraquat. Agron. J 101: 1388- 1393.
L., Ekebafe, D Ogbeifun, and F Okieimen (2011). Polymer applications in agriculture. Biokemistri 23: 81- 89.
J., Funderburk, D Gorbet, I Teare, and J Stavisky (1998). Thrips injury can reduce peanut yield and quality under conditions of multiple stress. Agron. J 90: 563- 566.
Grey, T., J Beasley, Jr., T Webster, and C Chen 2011 Peanut seed vigor evaluation using a thermal gradient. Int. J. Agron. 2011.
R., Halliwell, and G Philley (1974). Spotted wilt of peanut in Texas. Plant Dis. Rep 58: 23- 25.
R., Hayat, and S Ali (2004). Water absorption by synthetic polymer (Aquasorb) and its effect on soil properties and tomato yield. Int. J. Agri. Biol 6: 998- 1002.
D., Herbert, S Malone, S Aref, R Brandenburg, D Jordan, B Royals, and P.D Johnson (2007). Role of insecticides in reducing thrips injury to plants and incidence of Tomato spotted wilt virus in Virginia market-type peanut. J. Econ. Entomol 100: 1241- 1247.
D., Herbert J Wilcut, and C Swann (1991). Effects of various postemergence herbicide treatments and tobacco thrips (Frankliniella fusca) injury on peanut yields in Virginia. Peanut Sci 18: 91- 94.
C., Holbrook, P Ozias-Akins, Y Chu, A Culbreath, C Kvien, and T Brenneman (2017). Registration of 'TifNV-High O/L' peanut. J. Plant Regist 11: 228- 230.
K., Horie, M Barón, R Fox, J He, M Hess, J Kahovec, T Kitayama, P Kubisa, E Maréhal, W Mormann, R Stepto, D Tabak, J Vohlídal, E Wilks, and W Work (2004). Definitions of terms relating to reactions of polymers and to functional polymeric materials. Pure Appl. Chem 76: 889- 906.
X., Hou, R Li, W He, X Dai, K Ma, and Y Liang (2017). Superabsorbent polymers influence soil physical properties and increase potato tuber yield in a dry-farming region. J. Soils Sediments 2: 1- 11.
C., Hurt, R Brandenburg, D Jordan, G Kennedy, and J Bailey (2005). Management of spotted wilt vectored by Frankliniella fusca (Thysanoptera: Thripidae) in Virginia market-type peanut. J. Econ. Entomol 98: 1435- 1440.
M., Jain, M Gallo, K Chengalrayan, N Shaikh, G MacDonald, and J Davis (2015). Phorate-induced host defence responses condition acquired resistance to tomato spotted wilt in cultivated peanut (Arachis hypogaea L.). J. Phytopathol 163: 853- 866.
C., Johnson, K Eskridge, and D Corwin (2005). Apparent soil electrical conductivity: applications for designing and evaluting field-scale experiments. Comput. Electron. Agr 46: 181- 202.
Kemerait, R., A Culbreath, E Prostko, T Brenneman, S Tubbs, R Srinivasan, M Abney, S Monfort, A Rabinowitz, B Tillman, N Dufault, D Rowland, M Mulvaney, I Small, A Hagan, J Sarver, D Anco, and N Smith 2018 Peanut Rx - Minimizing Diseases of Peanut in the Southeastern United States. The 2018 Version of the Peanut Disease Risk Index University of Georgia Extension Available at: http://www.caes.uga.edu/content/dam/caes-website/extension-outreach/commodities/peanut-team/docs/2018/2018-Peanut-Rx-Disease-Risk-Index.pdf.
Little, E., P Brannen, J Brock, B Dutta, G Jagdale, A Jogi, B Kemerait, A Martinez-Espinoza, and J Williams-Woodward 2016 Georgia plant disease loss estimates - 2014 University of Georgia Available at: http://extension.uga.edu/publications/detail.html?number=AP102-7.
R., Lynch, J Garner, and L Morgan (1984). Influence of systemic insecticides on thrips damage and yield of Florunner peanuts in Georgia. J. Agric. Entomology 1: 33- 42.
K., Marasigan, M Toews, R Kemerait, M Abney, A Culbreath, and R Srinivasan (2018). Evaluation of alternatives to an organophosphate insecticide with selected cultural practices: effects on thrips, Frankliniella fusca, and incidence of spotted wilt in peanut farmscapes. J. Econ. Entomol 111: 1030- 1041.
K., Marasigan, M Toews, R Kemerait, M Abney, A Culbreath, and R Srinivasan (2016). Evaluation of alternatives to carbamate and organophosphate insecticides against thrips and Tomato spotted wilt virus in peanut production. J. Econ. Entomol 109: 544- 557.
J., Marois, and D Wright (2003). Effect of tillage system, phorate, and cultivar on tomato spotted wilt of peanut. Agron. J 95: 386- 389.
J., McKinney, and B Tillman (2017). Spotted wilt in peanut as impacted by genotype resistance, planting date, and plant population. Crop Sci 57: 130- 136.
Monfort, W., P Knox, W Branch, R Tubbs, E Prostko, M Abney, W Porter, B Kemerait, T Brenneman, A Culbreath, and A Rabinowitz 2017 2017 Peanut Update University of Georgia Available at: http://peanuts.caes.uga.edu/content/dam/caes-website/extension-outreach/commodities/peanut-team/docs/2017-peanut-update-rev-1.30.17.pdf.
H., Pappu, R Jones, and R Jain (2009). Global status of tospovirus epidemics in diverse cropping systems: Successes achieved and challenges ahead. Virus Res 141: 219- 236.
G., Parrella, P Gognalons, K Gebre-Selassiè, C Vovlas, and G Marchoux (2003). An update of the host range of Tomato spotted wilt virus. J. Plant Pathol 85: 227- 264.
J., Pinheiro, and E Chao (2006). Efficient Laplacian and adaptive Gaussian quadrature algorithms for multilevel generalized linear mixed models. J. Comput. Graph. Stat 15: 58- 81.
F., Pouci, F Iemma, U Spizzirri, G Cirillo, M Curcio, and N Picci (2008). Polymer in agriculture: a review. American Journal of Agricultural and Biological Sciences 3: 299- 314.
R., Sojka, and R Lentz (1997). Reducing furrow irrigation erosion with polyacrylamide (PAM). J. Prod. Agric 10: 47- 52.
R., Srinivasan, M Abney, A Culbreath, R Kemerait, R Tubbs, W Monfort, and H.R Pappu (2017). Three decades of managing Tomato spotted wilt virus in peanut in southeastern United States. Virus Res 241: 203- 212.
Stroup, W 2013 Generalized Linear Mixed Models: Modern Concepts, Methods and Applications CRC Press. Boca Raton, FL.
S., Sundaraj, R Srinivasan, A Culbreath, D Riley, and H Pappu (2014). Host plant resistance against Tomato spotted wilt virus in peanut (Arachis hypogaea) and its impact on susceptibility to the virus, virus population genetics, and vector feeding behavior and survival. Phytopathology 104: 202- 210.
B., Tillman, and D Gorbet (2017). Registration of 'TUFRunner 511' peanut. J. Plant Regist 11: 235- 239.
Tillman, B., D Gorbet, A Culbreath, and J Todd, 2006 Response of peanut cultivars to seeding density and row patterns Crop Management 5 Available at: https://dl.sciencesocieties.org/publications/cm/abstracts/5/1/CM-2006-0711-01-RS.
J., Todd, A Culbreath, J Chamberlin, R Beshear, and B Mullinix (1995). Colonization and population dynamics of thrips in peanuts in the southern United States. NATO Adv. Sci. Inst. Se 276: 453- 460.
Tubbs, R., K Balkcom, M Toews, and R Srinivasan 2013 Effects of fertilization, tillage, and phorate on thrips and TSWV incidence in early planted peanuts (Abstr.) In Proceedings of the Southern Conservation Agricultural System Conference, Norman, Oklahoma Available at: http://www.ag.auburn.edu/auxiliary/nsdl/scasc/Proceedings/2013/proc2013.html.
R., Tubbs, R Kemerait, B Williams, and J Sarver (2015). Effect of Bradyrhizobia inoculant formulation with phorate in new peanut fields. Peanut Sci 42: 138- 144.
USDA FSA 2018 a Peanut Buyers and Handlers Program Guidelines, 1-118.
USDA FSA 2018 b. Peanut Premiums and Discounts for 2018 Crop Year Available at: https://www.fsa.usda.gov/Assets/USDA-FSA-Public/usdafiles/Price-Support/pdf/2018/2018_peanuts.pdf.
Weather Underground 2019 Weather Underground Available at: https://www.wunderground.com/ [Accessed February 12, 2019] .
G., Wehtje, J Wilcut, J McGuire, and T Hicks (1991). Foliar penetration and phytotoxicity of paraquat as influenced by peanut cultivar. Peanut Sci 18: 67- 71.
J., Woodhouse, and M Johnson (1991). Effect of superabsorbent polymers on surivial and growth of crop seedlings. Agr. Water Manage 20: 63- 70.
M., Zohuriaan-Mehr, and K Kabiri (2008). Superabsorbent polymer materials: a review. Iran. Polym. J 17: 451- 477.
Notes
- Edisto Research and Education Center, Clemson University, Blackville, SC 29817; [^]
- Sandhill Research and Education Center, Clemson University, Columbia, SC 29229; [^]
- Department of Plant Pathology, University of Georgia, Tifton, GA 31793. [^] *Corresponding author email: danco@clemson.edu
Author Affiliations