Introduction
The Albino-Virescent leaf mutant has a very distinct leaf color trait. Seedlings have an almost albino appearance after emergence, except for some light greenish color along the midrib of the leaflets. Few such leaf color mutants are readily apparent at an early seedling stage, except for albinos which die shortly after emergence due to lack of chlorophyll (Branch and Kvien, 1992). However, the Albino-Virescent leaf mutant develops to full maturity under normal field conditions with direct sunlight as it progressively changes to green leaves with maturity.
Hammons (1973) as well as Murthy and Reddy (1993) have previously reported several chlorophyll-deficient leaf mutants in peanut (albina, xantha, chlorina, virescent, lutescent, aureus, and variegated). Albinos and xantha appear white and are controlled by one to three recessive genes. Lack of chlorophyll is lethal, and plants die shortly after emergence, presumably after energy reserves in the seed have been exhausted. Controlled by two recessive genes (au1 and au2), the leaves of aureus mutants turn a yellowish color toward the end of the growing season (Tai et al., 1977). However, the Lutescent-Leaf mutation is also controlled by two recessive genes (lut1 and lut2) and is characterized by yellow mid-rib and leaf margins surrounding a very light green, yellowish leaf which becomes greener with maturity (Branch, 2001).
Previous descriptions of virescent mutants in peanut mention that young leaves are a yellowish light green with pale rachis and midrib (Patil and Bora, 1963). These virescent mutants have been identified in X-ray mutagen populations (Patil and Bora, 1963) and in a plant introduction (Tripp, 1968). After a brief lag period, older leaves darken as chlorophyll begins to accumulate in the leaves (Benedict and Ketring, 1972), eventually leading to normal looking plants with green leaves by the end of the growing season. This description varies from the chlorophyll deficient phenotype, designated Albino-Virescent leaf, which was recently found in the F10 generation of the Georgia breeding line, GA 082524. Leaves of the Albino-Virescent leaf mutant plants are distinctly albino in appearance initially except for very pale green mid-ribs followed by the green darkening with age similar to the previously described virescent phenotype. GA 082524 originated from the three-way cross combination 'Georgia-02C' (Branch, 2003) x ['Georgia-01R' (Branch, 2002) x 'COAN' (Simpson and Starr, 2001)]. GA 082524 is a small-seeded runner market type (ssp. hypogaea var. hypogaea) with a flat decumbent runner growth habit, a tan testa color, and medium-late maturity (ca. 150-160 DAP) in south Georgia. It is a high-oleic (HO) peanut with a very high level of root-knot nematode (RKN) resistance which was confirmed with the SCAR 197/909 and SSR GM565 molecular markers (Branch et al., 2014). The Albino-Virescent mutant is similar to GA 082524, especially during the latter part of the growing season except smaller, however it still has the same two desirable agronomic traits, HO and RKN resistance, respectively.
Many of the previously reported chlorophyll-leaf mutants are not currently available or difficult to maintain. The objective of this study was to determine the inheritance of the newly identified Albino-Virescent leaf mutant identified in a breeding line derived from a diverse genetic background.
Materials and Methods
Reciprocal crosses were made in the greenhouse during the fall and winter months of 2014-15 between plants of Albino-Virescent mutant and GA 082524. In 2015, F1 seed were space-planted approximately 122-cm apart in one-row plots, 6.1m long x 1.8 m wide at the UGA Coastal Plain Experiment Station agronomy research farm, Tifton, GA. F2 seed from each cross were space-planted the following year approximately 30-cm apart in two-row plots 30.5 m long x 1.8 m wide.
The field nursery plots were in a three-year rotation following corn (Zea mays L.) and cotton (Gossypium hirsutum L). Each year, the soil-type was a Tifton loamy sand (fine-loamy, siliceous, thermic, Plinthic Kandidult). Recommended cultural practices with irrigation were used throughout each growing season.
Phenotypic classifications were based upon individual plants which were flagged on a regular basis to identify leaf characteristics. Segregation data was analyzed by chi-square analysis for goodness-of-fit (P≤0.05) to expected genetic ratios (Strickberger, 1968).
Results and Discussion
F1 plants from reciprocal crosses were classified as normal green leaves. This indicated that the Albino-Virescent leaf trait is recessive without any maternal or cytoplasmic effects.
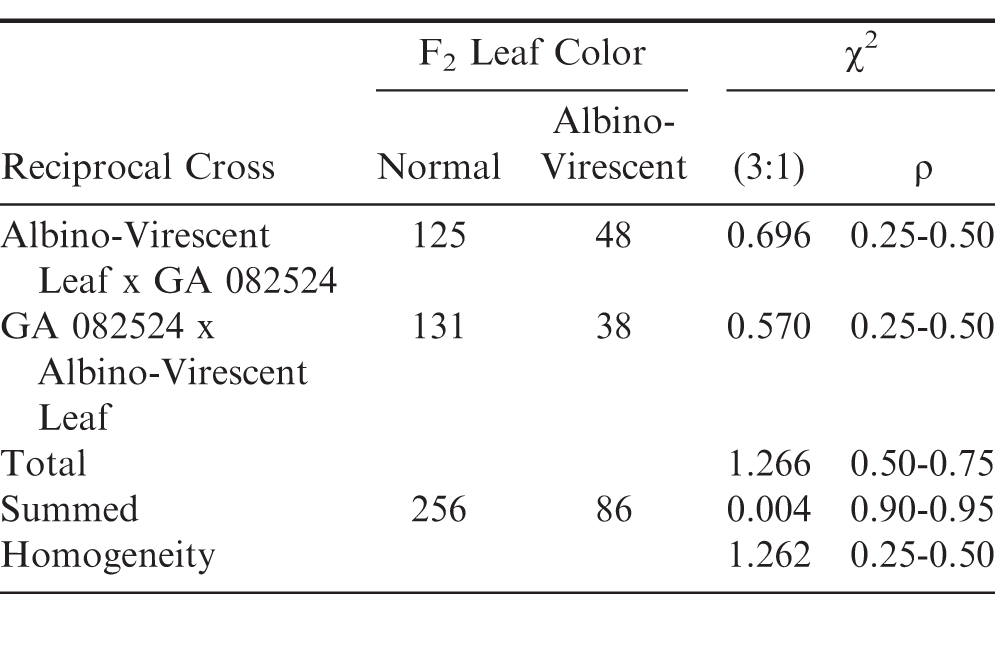
Segregation in the F2 population from the reciprocal crosses showed an acceptable fit for a 3 normal green, to 1 Albino-Virescent leaf ratio (Table 1). Total, summed, and homogeneity chi-square values were all found acceptable for the 3:1 expected ratio. These results agree with an earlier report for an unrelated virescent, v, type which originated from a radiation-induced mutation study (Tai et al., 1977). The gene symbol, avl, is proposed for the Albino-Virescent leaf color trait. An allelism test would have been of interest between the Albino-Virescent mutant and the virescent mutant identified by Tai et al., (1977) if seed were available of the latter.
Albino-Virescent leaf mutant seedlings have an almost albino appearance after emergence except for some light greenish color along the midrib of the leaflets (Fig. 1). As the plants begin to age, older leaves begin to darken and turn green, and only new young leaves are apparently deficient in chlorophyll (Fig. 2). During the latter half of the growing season, the older plants appear almost totally green (Fig. 3). Plant maturity thus changes leaflet appearance of the Albino-Virescent leaf mutant.
The Albino-Virescent mutant could be utilized as a delineating border plot to separate different tests due to the striking appearance of the leaf. It also could be used to measure natural crossing because of its single recessive inheritance. Future studies will be conducted to determine chlorophyll content and changes with age on individual plants of the Albino-Virescent leaf mutant. Such information should be of physiological interest to many peanut researchers.
Literature Cited
C. R Benedict, and D. L Ketring (1972). Nuclear gene affecting greening in virescent peanut leaves. Plant Physiol 49: 972- 976.
Branch, W.D 2001 Inheritance of a lutescent-leaf color trait in peanut J. Hered. 92: 436- 437.
W. D Branch, (2002). Registration of 'Georgia-01R' peanut. Crop Sci 42: 1750- 1751.
W. D Branch, (2003). Registration of 'Georgia-02C' peanut. Crop Sci 43: 1883- 1884.
W. D Branch, and C. K Kvien (1992). Cytoplasmically inherited albinism in peanut seedlings. J. Hered 83: 455- 457.
W. D., Branch, T. B Brenneman, and G Hookstra (2014). Field test results versus marker assisted selection for root-knot nematode resistance in peanut. Peanut Sci 41: 85- 89.
R. O Hammons, (1973). Genetics of Arachis hypogaea. In:. Peanuts-Culture and Uses. Am. Peanut Res. and Educ. Assoc., Stillwater, OK,: pp. 135- 173.
Murthy, T. G. K., and P. S Reddy 1993 Genetics of groundnut In: Cytogenetics and Genetics of Groundnuts, Intercept Ltd, Andover, UK.
S. H., Patil, and K. C Bora (1963). Radiation induced mutations in groundnut. I. Chlorophyll mutations. Indian J. Genet. Plant Breeding 23: 47- 49.
C. E Simpson, and J. L Starr (2001). Registration of 'COAN' peanut. Crop Sci 41: 918.
Strickberger, M. W 1968 Chap. 21 Chromosome variation in number Pp. 449-478 In Genetics. The Macmillan Co, New York, NY.
P.Y.P., Tai, R. O Hammons, and R. S Matlock (1977). Genetic relationships among three chlorophyll-deficient mutants in peanut, Arachis hypogaea L. Theor. Appl. Genet 50: 35- 40.
Tripp, L. D 1968 Germplasm evaluation and inheritance studies in peanuts, Arachis hypogaea L Ph.D. diss Oklahoma State University, Stillwater, OK.
Notes
- Professor and 2Post-Doctoral Research Assistant, Dept. of Crop and Soil Science, University of Georgia, Coastal Plain Experiment Station, 2360 Rainwater Road, Tifton, GA 31793-5766. [^] *Corresponding author email: wdbranch@uga.edu
Author Affiliations