Introduction
Changes in farming practices and technology have led to the application of pesticides over a broader time period in a given d (Mohr et al., 2007). In recent years, average farm size in the U.S. has continued to increase while number of farms has decreased (Hoppe and Macdonald, 2015). Due to this increase in farm size, growers must cover more land area in a given time period in order to be timely with pesticide applications. The desire to minimize herbicide drift may also lead to applications early in the morning or late in the evening, when wind speed is generally lower. The addition of global positioning technology (GPS) to most modern application equipment has allowed for the accurate application of pesticides under all light conditions (Klassen et al., 1993; Mohr et al., 2007; Tillet, 1991). The increased occurrence of these practices is validating the well-researched fact that the efficacy of many herbicides is directly related to sunlight (Montgomery et al., 2017; Stewart et al., 2009; Stopps et al., 2013).
Reduced weed control due to variable application timing has previously been reported for bentazon, acifluorfen, fomesafen, glufosinate, and glyphosate (Doran and Andersen, 1976; Lee and Oliver, 1982; Martinson et al., 2002; Miller et al., 2003; Mohr et al., 2007). While there have been reported reductions in herbicide efficacy due to herbicide applications at varying times during the d, weed response is still largely species dependent. Species-specific time of d (TOD) effects for atrazine, bromoxynil, dicamba, glufosinate, glyphosate, and nicosulfuron were observed when applied to barnyardgrass (Echinochloa crus-galli L.), common lambsquarters (Chenopodium album L.), common ragweed (Ambrosia artemisiifolia L.), redroot pigweed (Amaranthus retroflexus L.), and velvetleaf (Abutilon theophrasti L.), (Stewart et al., 2009). Acifluorfen applied at night (21:00 hr) resulted in improved control of hemp sesbania (Sesbania herbacea Mill.), pitted morningglory (Ipomoea lacunose L.), and smooth pigweed (Amaranthus hybridus L.), while no TOD effect was observed for several other species (Lee and Oliver, 1982). Bentazon was less efficacious when applied before daybreak in the morning (6:00 hr) and after sunset in the evening (21:00 hr) (Doran and Andersen, 1976). However, other research reported no difference in bentazon efficacy when applied at various TOD to common ragweed, common lambsquarters, pigweed, and velvetleaf (Stopps et al., 2013).
Environmental and plant physiological factors can contribute to the varying degrees of control observed when herbicides are applied at different TOD. Environmental factors such as dew, temperature, and relative humidity (RH) can influence herbicide performance. Typically, dew can be present on leaf surfaces in early morning (6:00 hr) and late evening (19:00 to 24:00 hr). It is hypothesized that dew can intercept herbicide spray droplets potentially diluting the herbicide and increasing herbicide run-off from the leaf surface (Fausey and Renner, 2001; Kogan and Zuniga, 2001; Stewart et al., 2009). But, other research suggests that dew could potentially increase herbicide absorption and increasing efficacy (Caseley, 1989; Nalewaja et al., 1975). Increased air temperature and RH have also been shown to increase herbicide efficacy (Sharma and Singh, 2001). Temperatures below 25 C caused reduced control regardless of the TOD when the herbicide was applied (Friesen and Wall, 1991). Daytime air temperatures above 25 C increased control of pitted morningglory, common cocklebur, and velvetleaf when compared to temperatures below 25 C (Doran and Andersen, 1976; Lee and Oliver, 1982). Increases in RH have also been reported to increase herbicide efficacy over a range of herbicides and species, due to increased absorption and translocation of the herbicide (Johnson and Young, 2001; Willis, 1978; Willis and McWhorter, 1981).
Plant morphological and physiological factors at different TOD also influence the efficacy of the herbicide applied (Hess and Falk, 1990). Factors such as leaf orientation, exposed leaf surface area, thickness of epicuticular wax, and plant metabolic rate may all affect plant absorption and translocation of herbicides (Doran and Andersen, 1976; Hess and Falk, 1990; Mohr et al., 2007; Stewart et al., 2009; Waltz et al., 2004). Response to the factors listed above can also vary largely depending on weed species (Hess and Falk, 1990).
Additional information is needed to determine TOD effects on herbicides when multiple active ingredients are used in tank-mixtures. Most previous studies on TOD only evaluated treatments consisting of a single mode of action. Also, growers do not typically make one herbicide application per growing season. Multiple applications (i.e. programs) are made in order to have an effective season-long weed control program. Thus, research was conducted to determine the effects of TOD on the performance of peanut weed control programs.
Materials and Methods
Non-crop study
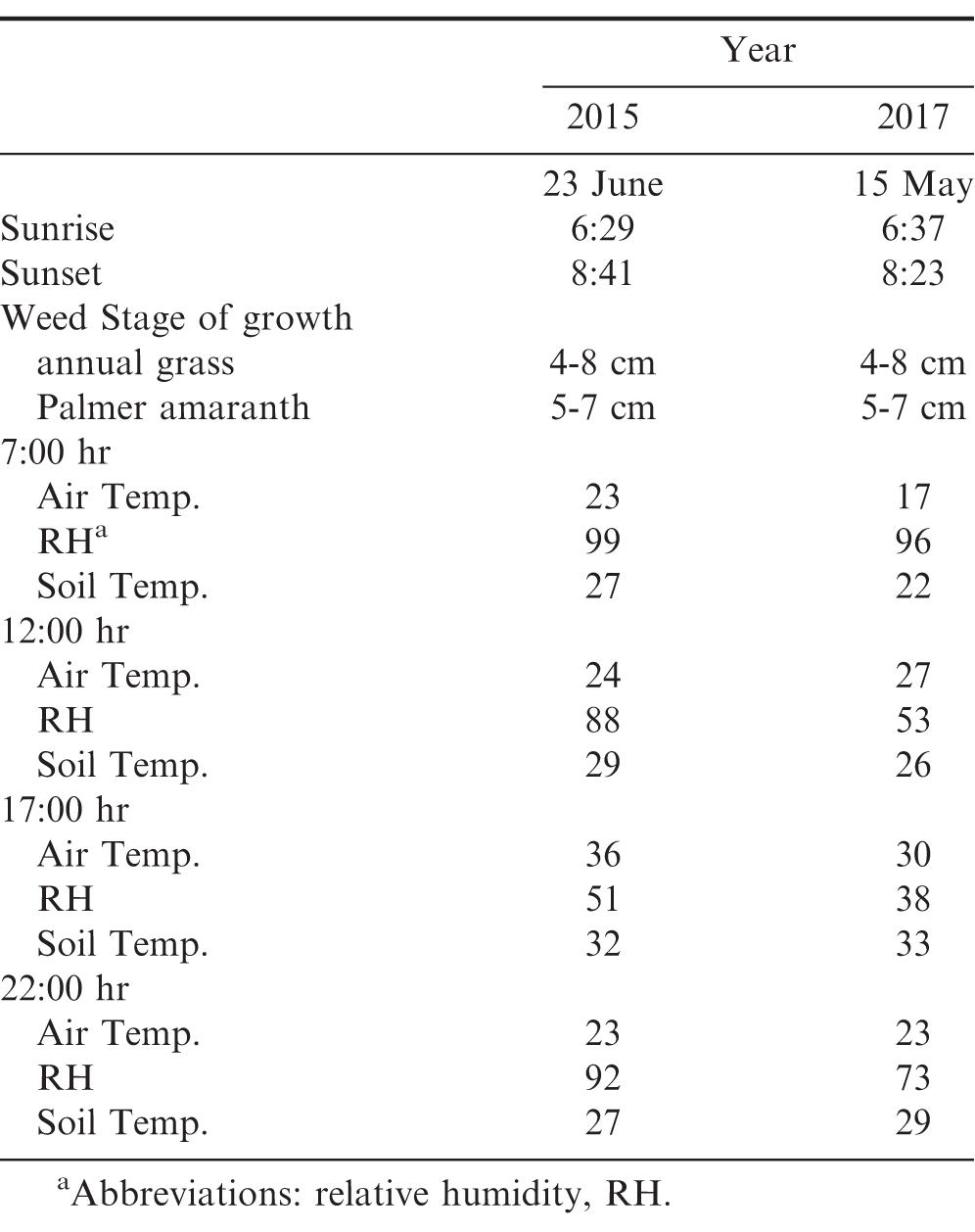
A non-crop study was conducted during 2015 and 2017 at the Ponder Research Farm located near Ty Ty, Georgia (31.507654 N, -83.658395 W) on a Tifton loamy sand (fine-loamy, Kaolinitic, thermic Plinthic Kandiudult) with 93% sand 3% silt, 4 % clay, 1% organic matter, and pH of 6.0. The trial was arranged in a randomized complete block design with a 3 (herbicide treatments) by 4 (different times during the d) factorial arrangement of treatments. The herbicide treatments included: paraquat (0.21 kg ai/ha) plus bentazon (0.37 kg ai/ha) plus acifluorfen (0.19 kg ai/ha) plus S-metolachlor (1.23 kg ai/ha); imazapic (0.07 kg ai/ha) plus S-metolachlor (1.23 kg ai/ha) plus 2,4-DB (0.25 kg ai/ha); and lactofen (0.22 kg ai/ha) plus S-metolachlor (1.23 kg ai/ha) plus 2,4-DB (0.25 kg ai/ha). A non-treated control (NTC) was included for comparison. Application timing were at 7:00, 12:00, 17:00, and 22:00 hr. Temperature, relative humidity (RH), and weed height for each application are presented in Table 1.
Plot size was 7.6 m by 0.9 m. Each treatment was replicated 3 or 4 times depending upon field size. Palmer amaranth and a non-uniform mixture of annual grasses including; Texas millet (Brachiaria texana, Buckley), crowfootgrass (Dactyloctenium aegyptium, L. Wild), goosegrass (Eleusine indica, L. Gaertn.), and crabgrass (Digitaria spp.) were present in the non-treated check plots at densities of 50 - 100 plants/m2 and 20 - 40 plants/m2, respectively. The treatments were applied using a CO2-pressurized backpack sprayer calibrated to deliver 141 L/ha at 262 kPa and 4.83 km/ha. An 11002DG flat fan nozzle was used for all applications (TeeJet, Springfield, IL 62701). Visual estimates of percent weed control were obtained at 7 and 14 d after treatment (DAT) using a scale of 0% = no control; 100% = complete control or plant death. Plant stunting, chlorosis, and necrosis were considered when making the visual estimates.
In-Crop study
An in-crop trial was also conducted at the Ponder Research Farm and the Attapulgus Research and Education Center (30.7636290N, -84.4799380W) on a Faceville loamy sand (Fine, kaolinitic, thermic Typic Kandiudults) with 84% sand, 10% clay, 6% silt, 1.6% organic matter, and pH of 6.0 during 2015, 2016, and only at the Ponder Research Farm in 2017 (4 site-years). Conventional tillage practices were used and 'Georgia-06G' (Branch, 2007) peanut was planted at both locations. A vacuum planter (Monosem Precision Planters, 1001 Blake St., Edwardsville, KS 66111) was calibrated to deliver 18 peanut seed/m at a depth of 5 cm. Peanut was planted in 2 twin rows (90 cm by 22 cm spacing) at Ponder and 2 single rows (90 cm spacing) in Attapulgus. Plot size was 7.6 m by 0.9 m.
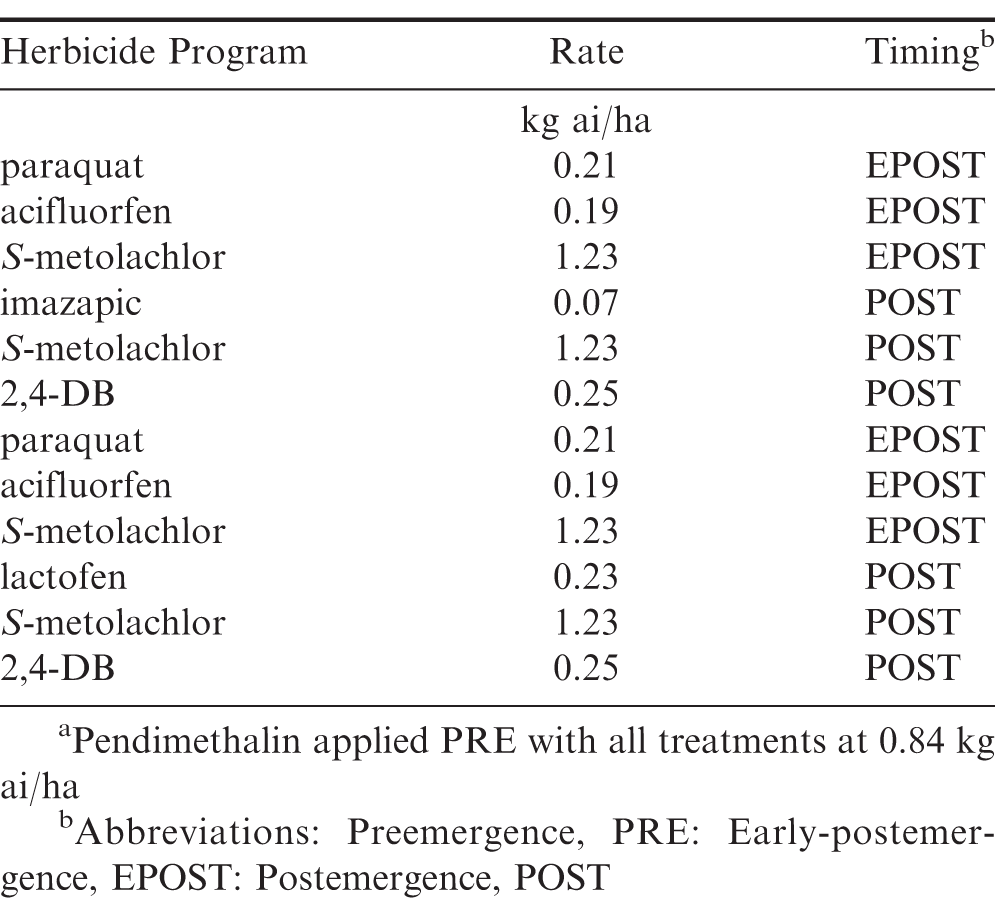
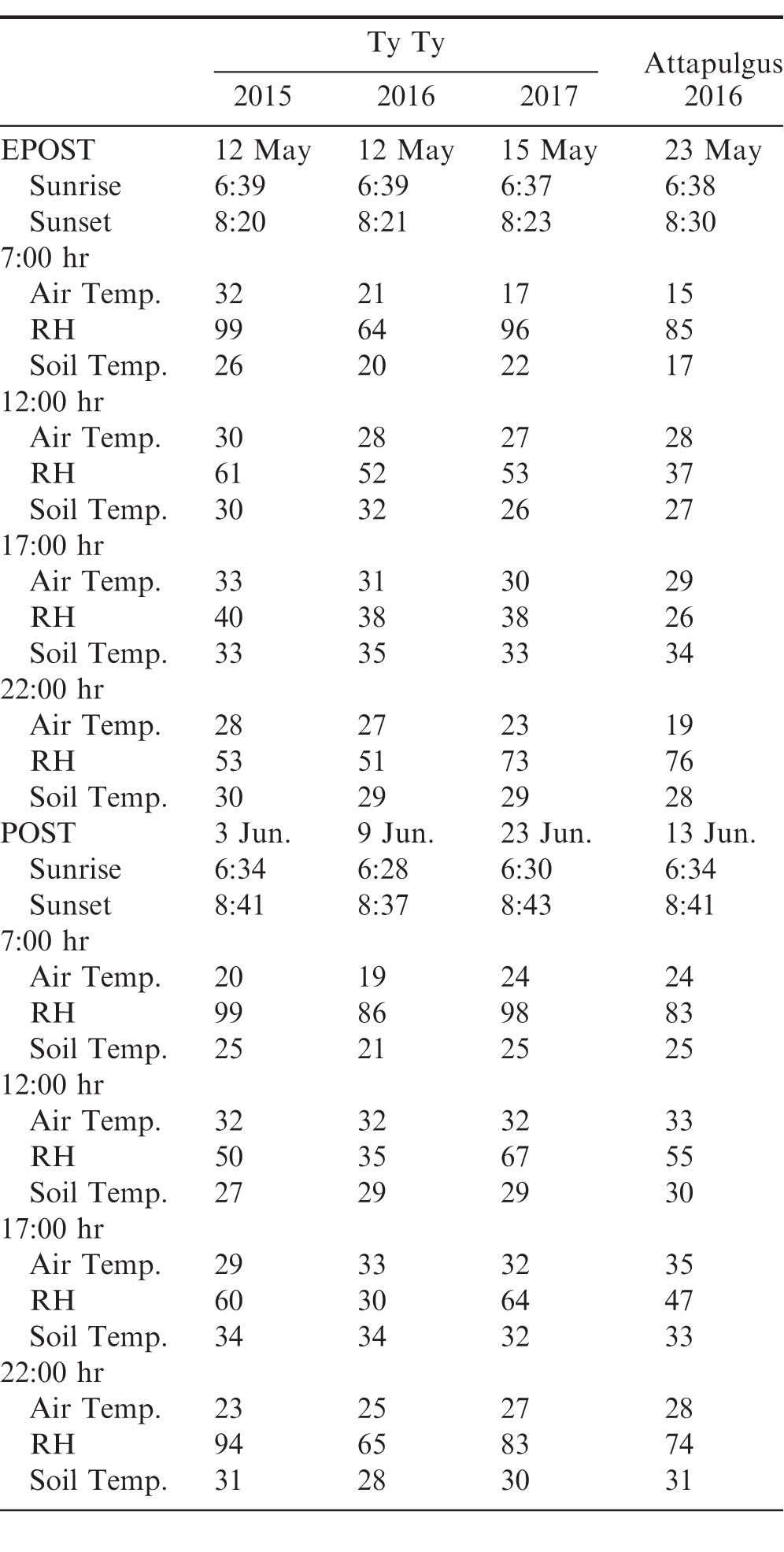
The trial was arranged in a randomized complete block design with a 2 (herbicide programs) by 4 (different TOD) factorial design with 4 replications. The herbicide programs used are presented in Table 2. Each herbicide program was applied at each TOD throughout the entire season (7:00, 12:00, 17:00, and 22:00 hr). Temperature, RH, and weed size at each treatment are presented in Table 3. Herbicides were applied using a CO2-pressurized backpack sprayer calibrated to deliver 141 L/ ha at 262 kPa and 4.83 km/h. Visual estimates of peanut crop injury were obtained 7 to 14 after the EPOST and 7 to 14 d after POST treatments. Visual estimates of crop injury consisted of a combination of leaf burn and stunting (0%= no crop injury; 100%= no crop present). Visual estimates of weed control were recorded at 7 and 14 d after the EPOST treatment and 7 to 14 and 50 d after the POST treatment. Weed species were rated including Palmer amaranth, sicklepod, and a non-uniform mixture of annual grasses including, Texas millet, crowfootgrass, goosegrass, and crabgrass. Peanuts were inverted, allowed to air dry, and harvested 4 d later using commercial equipment. Peanut yields were adjusted to 10% moisture.
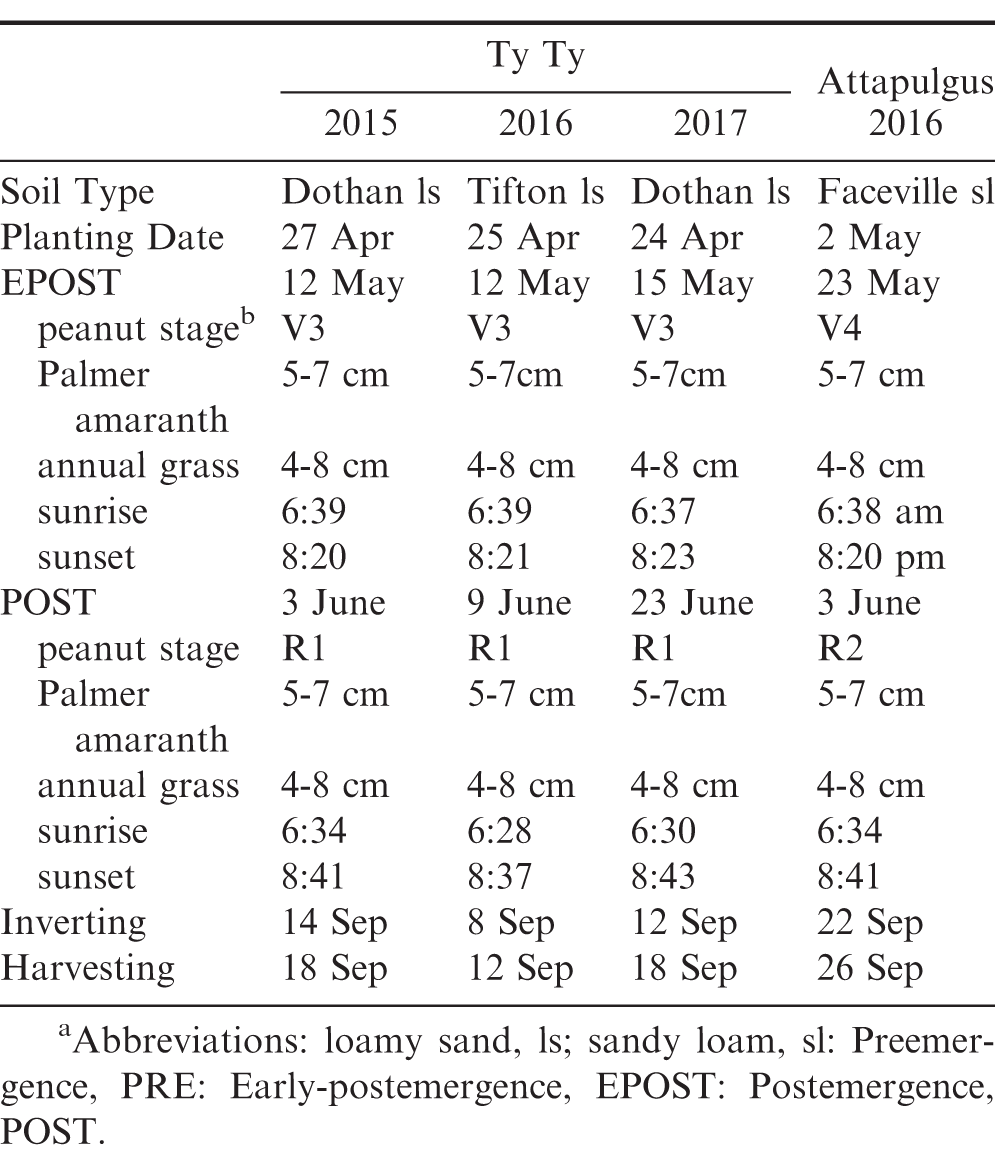
University of Georgia Extension peanut production recommendations were used and supplemental irrigation was applied to maximize peanut growth and development (Anonymous 2017). Soil characteristics, planting date, peanut stages of growth at application, weed heights, and harvest dates are presented in Table 4 (Boote 1982).
Data for all parameters in both the non-crop and in-crop studies were analyzed as factorial plot designs and subjected to ANOVA using the PROC MIXED procedure in SAS (SAS Institute 107 Inc., Cary, NC 27511). TOD and herbicide treatment/program were considered fixed effects and locations and replications (nested within year) were considered random effects. Least square means of significant main effects were separated using Tukey's HSD test (alpha=0.10).
Results and Discussion
Non-crop study (Data not shown)
For the non-crop study there was a significant interaction between TOD and herbicide treatment for the 7 DAT rating for Palmer amaranth control. Palmer amaranth control was lower when imazapic plus S-metolachlor plus 2,4-DB were applied at 22:00 hr, when compared to applications made during daylight hours. Palmer amaranth control was unacceptable (< 70%) with imazapic because the population at this location is known to be ALS-resistant (Wise et al. 2009). At 14 DAT, there was no interaction between treatment and TOD. Palmer amaranth control was reduced with the combination of imazapic plus S-metolachlor plus 2,4-DB when compared with other treatments and no TOD effects were observed. Previous research has shown that reduced control of barnyardgrass can occur when using the ALS-herbicide nicosulfuron at different TOD (Stewart et al., 2009). There is no published evidence that barnyardgrass exhibits diurnal leaf movements, therefore reduced herbicide interception is not the cause of the reduction in control (Mohr et al., 2007).
There was no significant interaction between TOD and treatment for the 7 and 14 DAT rating timing for annual grass control. At 7 DAT, annual grass control was reduced with the imazapic plus S-metolachlor plus 2,4-DB and lactofen plus s-metolachlor plus 2,4-DB treatments. At 14 DAT, only the lactofen plus S-metolachlor plus 2,4-DB treatment provided unacceptable control of annual grasses (< 35%). Lactofen is a broadleaf herbicide and has limited efficacy on grass weed species (Grichar, 1991; Minton et al., 1989). TOD had no effect on annual grass control. This is contrary to previous research where a TOD effect was observed for barnyardgrass control with nicosulfuron (Stewart et al., 2009).
Crop Injury
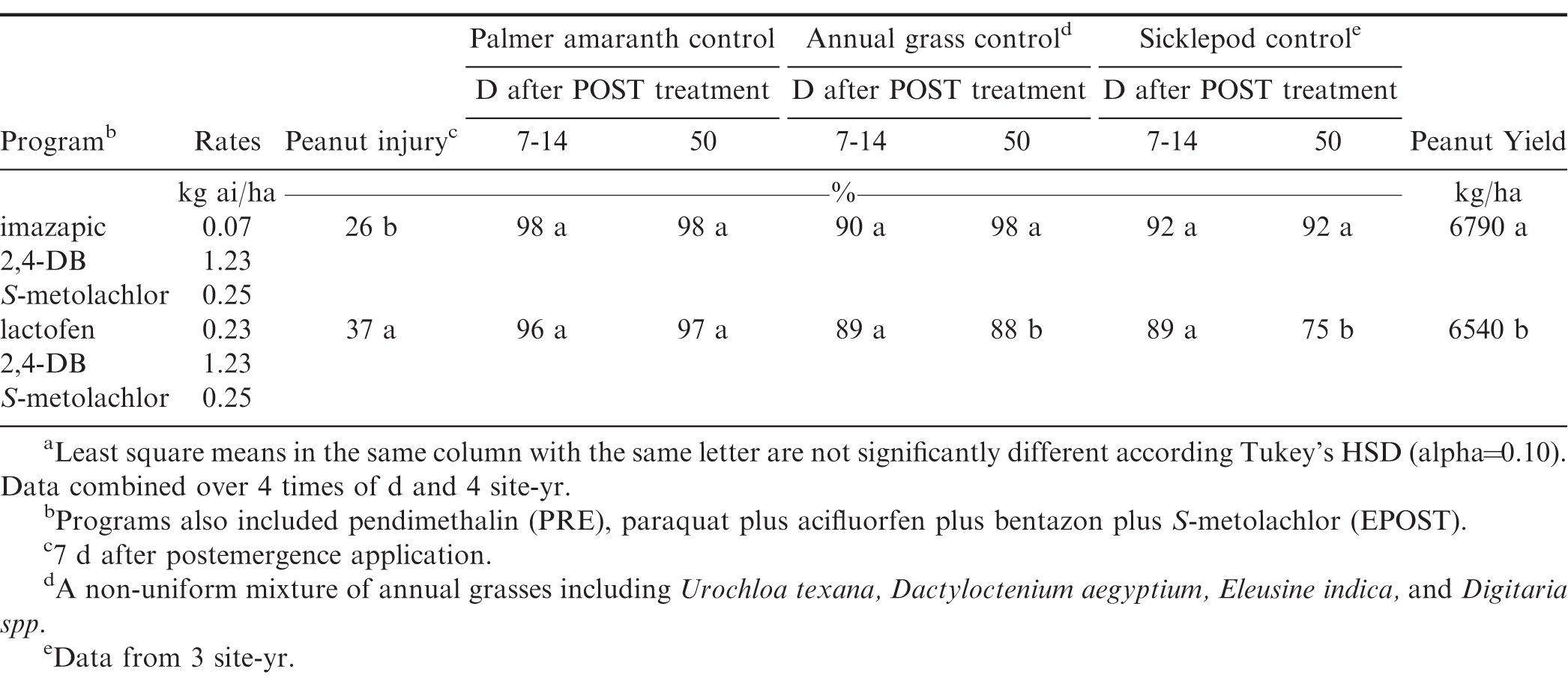
Peanut crop injury was evaluated 1 wk after the EPOST and POST applications. Significant differences in injury were observed at both times. Generally, herbicide programs were less injurious when applied at 7:00 hr and 22:00 hr (Tables 5 and 7). When averaged over TOD, the lactofen program was more injurious than the imazapic program (Table 6). Peanut injury from lactofen has been observed in other research ranging from 20 to 48%, with no observed yield losses (Boyer et al., 2011; Ferrell et al., 2013). However, yield losses of 5% were observed from applications of lactofen applied approximately 60 d after planting (Dotray et al., 2012).
Palmer amaranth
Palmer amaranth was completely controlled by a combination of the PRE and EPOST herbicide applications both 1 and 2 wk after the EPOST application was made (data not shown). For Palmer amaranth control after the POST herbicide applications, there was no significant interaction for any rating. There were also no significant differences between programs or TOD (Tables 6 and 7). Reduced control of Palmer amaranth when applying 2,4-D, imazethapyr, dicamba, glufosinate, and bentazon late in the evening or at night, has been observed in other research (Doran and Andersen, 1976; Johnston et al., 2018; Montgomery et al., 2017; Stopps et al., 2013). However, these studies differ from ours in that a single herbicide was used, not a tank-mixture with multiple active ingredients. TOD does not appear to influence Palmer amaranth control when a complete peanut herbicide program is used consisting of multiple active ingredients at a single application timing.
Annual grass
After the EPOST applications, annual grass control at 7 to 14 DAT was significantly lower at 7:00 hr (Table 5). Paraquat has been reported to be more efficacious when applied at night or later in the evening for weed control due to minor intercellular translocation occurring (Brian, 1967; Montgomery et al., 2017; Putnam and Ries, 1968). It has been reported that annual grasses are not as sensitive as broadleaf weeds to TOD effects with herbicides (Stewart et al., 2009). One possible explanation for the reduction in control observed at 7:00 hr is that dew was present on the weeds and on the crop. Dew presence has been reported to both increase or decrease herbicide efficacy depending on the herbicide and weed species involved (Caseley, 1989; Fausey and Renner, 2001; Nalewaja et al., 1975; Wanamarta and Penner, 1989). There was no significant TOD by program interaction for grass control after the POST applications, thus data is averaged over TOD and program. At 1 wk after the POST application there was no difference in herbicide program for annual grass control (Table 6). However, at the end of season rating (50 DAT) there was a significant difference in control of annual grasses. Annual grass control with the lactofen program was significantly lower than the imazapic program. Although primarily used for nutsedge (Cyperus spp.) and broadleaf weed control in peanut, imazapic provides various levels of annual grass control depending upon the species and stage of growth (Monks et al., 1996; Wilcut et al., 1999; Jordan et al., 2009). Lactofen has also been reported to have limited activity on grasses (Grichar, 1991; Minton et al., 1989).
At 7 to 14 d after the POST application, reduced control of annual grass was observed at 7:00 and 22:00 hr (Table 7). However, at the end of season control rating, there was no significant difference in TOD for annual grass control. As previously mentioned grass species are less sensitive to TOD effects when compared to broadleaf weed species (Stewart et al., 2009). Additionally, the dense canopy and spreading growth habit of the peanut crop likely resulted in the peanut plants out-competing the few grasses that were present at the earlier application timing (Leon et al., 2016).
Sicklepod
There was a significant TOD effect for control of sicklepod at 7 to 14 d after the EPOST application with less control, observed at 22:00 hr (Table 5). The diurnal leaf movement of sicklepod has been reported to reduce herbicide interception and control (Norsworthy et al. 1999). There was no interaction between herbicide program and TOD for the visual control ratings of sicklepod after the POST application was made, data presented are pooled over herbicide treatment and TOD. Differences for sicklepod control were observed for both program and TOD at 7 to14 and 50 d following the POST application. The application made at 22:00 hr resulted in less sicklepod control when compared to all other application timings. The imazapic program was more effective than the lactofen program for the control of sicklepod (Table 6). Sicklepod control with imazapic has been well documented (Grey et al., 2003; Grey and Wehjte, 2005).
Peanut Yield
There was no interaction between TOD and herbicide program for peanut yield; data are pooled over TOD, herbicide program, and site-years. Herbicide program had a significant effect on peanut yield. The imazapic program resulted in higher yields than the lactofen program. The reduction in yield observed between the two programs is potentially due to less annual grass and sicklepod control that was observed between the two treatments. Control of annual grass species for 8 to 10 wks after peanut emergence has been shown to be critical in maintaining a high yielding peanut crop (Everman et al., 2008; Grichar, 1991; Johnson and Mullinix, 2006). Peanut injury from lactofen has been observed in other research ranging from 20 to 48%, with no observed yield losses (Boyer et al. 2011; Ferrell et al. 2013). But other research has reported a yield loss of 5% from lactofen 60 DAP (Dotray et al. 2012). TOD of did not affect peanut yield. While reductions in sicklepod control were observed from applications made at 22:00 hr those reductions did not result in yield loss.
Summary and Conclusions
TOD influenced peanut injury and weed control, but did not affect peanut yield. Peanut growers who choose to spray early in the morning or late in the evening should be aware of the possibility of reduced control of certain weed species, especially sicklepod, that exhibit diurnal leaf movements. The diurnal leaf movements of sicklepod and several other weed species can greatly reduce herbicide interception (Norsworthy et al. 1999). The use of a complete herbicide program, i.e. multiple active ingredients in a tank-mixture and multiple applications, has been shown to reduce TOD effects on herbicide efficacy (Sellers et al. 2003).
Acknowledgements
The authors wish to thank Dwayne Dales, Charlie Hilton, and Tim Richards for technical assistance.
Literature Cited
Anonymous. Peanut Production Update. Cooperative Extensions Service Series CSS-17-0118. ed.. W.S Monfort (2017). Athens, GA: : University of Georgia. Pages. 67.
K.J Boote, (1982). Growth stages of peanut (Arachis hypogaea L.). Peanut Sci 9: 35- 40.
Boyer, J.A., Ferrell, J MacDonald, G. Tillman, B. and Rowland, D 2011 Effect of acifluorfen and lactofen application timing on peanut injury and yield Online. Crop Manag. doi:10.1094/CM-2011-0519-01-RS.
W.D Branch, (2007). Registration of 'Georgia-06G' peanut. J Plant Regist 1: 120.
R.C Brian, (1967). Darkness and the activity of diquat and paraquat on tomato, broad bean and sugar beet. Annals of Applied Bio 60: 77- 85.
J.C Caseley, (1989). Variation in foliar pesticide performance attributable to humidity, dew and rain effects. Aspects Appl. Biol. CAB Abstr 21: 215- 225.
D.L Doran, and R.N Andersen (1976). Effectiveness of bentazon applied at various times of the d. Weed Sci 24: 567- 570.
P.A., Dotray, W.J Grichar, T.A Baughman, E.P Prostko, T. L Grey, and L. V Gilbert (2012). Peanut (Arachis hypogaea L.) response to lactofen at various postemergence timings. Peanut Sci 39: 9- 14.
W.J., Everman, S.B Clewis, W.E Thomas, I.C Burke, and J.W Wilcut (2008). Critical period of weed interference in peanut. Weed Technol 22: 63- 67.
J.C Fausey, and K.A Renner (2001). Environmental effects on CGA-248757 and flumiclorac efficacy/soybean tolerance. Weed Sci 49: 668- 674.
J.A., Ferrell, R.G Leon, B Sellers, D Rowland, and B Brecke (2013). Influence of lactofen and 2,4-DB combinations on peanut injury and yield. Peanut Sci 40: 62- 65.
G.H Friesen, and D.A Wall (1991). Effect of application factors on efficacy of fluazifop-P-butyl in flax. Weed Technol 5: 504- 508.
T.L., Grey, D.C Bridges, and E.P Prostko (2003). Residual weed control with imazapic, diclosulam, and flumioxazin in Southeastern peanut (Arachis hypogaea). Peanut Sci 30: 23- 28.
T.L Grey, and G.R Wehtje (2005). Residual herbicide weed control systems in peanut. Weed Technol 19: 560- 567.
W.J Grichar, (1991). Sethoxydim and broadleaf herbicide interaction effects on annual grass control in peanuts (Arachis hypogaea). Weed Technol 5.2: 321- 324.
F.D Hess, and R.H Falk (1990). Herbicide deposition on leaf surfaces. Weed Sci 38: 280- 288.
R., Hoppe, and J MacDonald (2015). America's Diverse Family Farms, 2015 Edition, EIB-146, U.S. Department of Agriculture, Economic Research Service. .
B.C Johnson, and B.G Young (2002). Influence of temperature and relative humidity on the foliar activity of mesotrione. Weed Sci 50: 157- 161.
W.C Johnsonand B.G Mullinix (2006). Texas panicum (Panicum texanum) interference in peanut (Arachis hypogaea) and implications for treatment decisions. Peanut Sci 32: 68- 72.
C.R., Johnston, P.M Eure, T.L Grey, A.S Culpepper, and W.K Vencill (2018). Time of application influences translocation of auxinic herbicides in Palmer amaranth (Amaranthus palmeri). Weed Sci 66: 4- 14.
D. L., Jordan, S.H Lancaster, J.E Lanier, B.R Lassiter, and P.D Johnson (2009). Weed management in peanut with herbicide combinations containing imazapic and other pesticides. Weed Technol 23: 6- 10.
N.D., Klassen, R.J Wilson, J.N Wilson (1993). Agricultural vehicle guidance sensor, ASAE Paper No. 931008. St. Joseph, Mich.: ASAE..
M Kogan, and M Zúñiga (2001). Dew and spray volume effect on glyphosate efficacy. Weed Technol 15: 590- 593.
S.D Lee, and L.R Oliver (1982). Efficacy of acifluorfen on broadleaf weeds. Times and methods for application. Weed Sci 30: 520- 526.
R.G., Leon, M.J Mulvaney, and B.L Tillman (2016). Peanut cultivars differing in growth habit and canopy architecture respond similarly to weed interference. Peanut Sci 43: 133- 140.
K.B., Martinson, R.B Sothern, W.L Koukkari, B.R Durgan, and J.L Gunsolus (2002). Circadian response of annual weeds to glyphosate and glufosinate. Chronobiol. Int 19: 405- 422.
R., Miller, K.B Martinson, R.B Sothern, B.R Durgan, and J.L Gunsolus (2003). Circadian response of annual weeds in a natural setting to high and low application doses of four herbicides with different modes of action. Chronobiol. Int 20: 299- 324.
W.M., Minton, M.E Kurtz, and D.R Shaw (1989). Barnyardgrass (Echinochloa crus-galli) control with grass and broadleaf weed herbicide combinations. Weed Sci 37: 223- 227.
K., Mohr, B.A Sellers, and R.J Smeda (2007). Application time of d influences glyphosate efficacy. Weed Technol 21: 7- 13.
C.D., Monks, J.W Wilcut, J.S Richburg, J.H Hatton, and M.G Patterson (1996). Effect of AC 263,333, imazethapyr, and nicosulfuron on weed control and imidazolinone-tolerant corn (Zea mays) yield. Weed Technol 10: 822- 827.
G.B., Montgomery, J.A Treadway, J.L Reeves, and L.E Steckel (2017). Effect of time of d of application of 2,4-D, dicamba, glufosinate, paraquat, and saflufenacil on horseweed (Conyza canadensis) Control. Weed Technol 31.4: 550- 556.
J.D., Nalewaja, J Pudelko, and K.A Adamczewski (1975). Influence of climate and additives on bentazon. Weed Sci 23: 504- 507
J.K., Norsworthy, L.R Oliver, and L.C Purcell (1999). Diurnal leaf movement effects on spray interception and glyphosate efficacy. Weed Technol 13: 466- 470.
A.R., Putnam, S.K Ries, (1968). Factors influencing the phytotoxicity and movement of paraquat in quackgrass. Weed Sci 16: 80- 83.
B.A., Sellers, R.J Smeda, and W.G Johnson (2003). Diurnal fluctuations and leaf angle reduce glufosinate efficacy. Weed Technol 17: 302- 306.
S.D Sharma, and M Singh (2001). Environmental factors affecting absorption and bio-efficacy of glyphosate in Florida beggarweed (Desmodium tortuosum). Crop Prot 20: 511- 516.
C.L., Stewart, R.E Nurse, and P.H Sikkema (2009). Time of d impacts POST weed control in corn. Weed Technol 23: 346- 355.
G.J., Stopps, R.E Nurse, P.H Sikkema (2013). The effect of time of d on the activity of postemergence soybean herbicides. Weed Technol 27: 690- 695.
R.D Tillett, (1991). Image analysis for agricultural processes: a review of potential opportunities. J. Agric. Engin. Res 50: 247- 258.
A.L., Waltz, A.R Martin, F.W Roeth, and J.L Lindquist (2004). Glyphosate efficacy on velvetleaf varies with application time of d. Weed Technol 18: 931- 939.
G Wanamarta, and D Penner (1989). Foliar penetration of herbicides. Rev. Weed Sci 4, 215- 231.
J.W., Wilcut, J.S Richburgand F.R Walls (1999). Response of johnsongrass (Sorghum halepense) and imidazolinone-resistant corn (Zea mays) to AC 263,222. Weed Technol 13: 484- 488.
G.D Wills, (1978). Factors affecting toxicity and translocation of glyphosate in cotton. Weed Sci 26: 509- 513.
G.D Wills, and C.G McWhorter (1981). Effect of environment on the translocation and toxicity of acifluorfen to showy crotalaria (Crotalaria spectabilis). Weed Sci 29: 397- 401.
A.M., Wise, T.L Grey, E.P Prostko, W.K Vencill, and T.M Webster (2009). Establishing the geographic distribution level of acetolactate synthase resistance of Palmer amaranth (Amaranthus palmeri) accessions in Georgia. Weed Technol 23: 214- 220.
Notes
- First and second authors: Former Graduate Research Assistant and Professor, Department of Crop & Soil Sciences, The University of Georgia, Tifton, GA 31794. [^] *Corresponding author's E-mail: wenc@amvac-chemical.com
Author Affiliations