Introduction
Adequate control of tobacco thrips, Frankliniella fusca (Hinds), is necessary in North Carolina and Virginia to protect yield (Brandenburg, 2017; Drake et al., 2009; Herbert et al., 2007; Hurt et al., 2005; Marasigan et al., 2016) and minimize incidence of tomato spotted wilt (TSW) caused by tomato spotted wilt virus (family Tospoviridae, genus Orthotospovirus) of peanut (Culbreath et al., 2003, 2008; Culbreath and Srinivasan 2011). Although cultivar selection, plant population, and planting date can affect thrips populations and incidence of TSW, systemic insecticide applied in the seed furrow at planting is generally the most effective practice to suppress thrips and protect peanut yield in North Carolina (Brandenburg, 2017).
Aldicarb was considered the most frequently used active ingredient applied in the seed furrow at planting in North Carolina (Rhodes et al., 2008). However, availability of aldicarb was reduced following the 2009 growing season and farmers and their advisors began using other systemic insecticides to control thrips. Phorate applied in the seed furrow at planting is an effective alternative to aldicarb but can cause phytotoxicty under certain conditions (Herbert et al., 2007; Marasigan et al, 2016; Rhodes et al., 2008; Tubbs et al., 2013, 2015; Whalen et al., 2014). Acephate can also be applied in the seed furrow at planting, but in some instances slower emergence of peanut after application of acephate has been observed (Brandenburg, 2017). Imidacloprid received a registration for use in peanut in 2011 and has become popular with growers in North Carolina with 21% of growers surveyed in 2013 indicating that imidacloprid was applied in the seed furrow at planting (Morgan et al., 2014). Although greater incidence of TSW was reported following use of imidacloprid compared with non-treated peanut (Culbreath and Srinivasan, 2011), the most recently released Virginia market type cultivars including Bailey (Isleib et al., 2011) and Sullivan (Isleib et al., 2016) express resistance to TSW and have reduced concerns that imidacloprid might result in a higher incidence of TSW (Brandenburg, 2017; Isleib et al., 2011; Shew, 2017).
In many instances, thrips suppression by systemic insecticides applied in the seed furrow at planting can be adequate to protect yield (Brandenburg, 2017). However, Morgan et al. (2014) reported that 66% of farmers applied acephate after peanut emergence to control thrips regardless of in-furrow insecticide treatment. Herbert et al. (2007) reported that acephate further reduced injury above that of in-furrow insecticides and increased yield compared with non-treated peanut or peanut treated only with insecticides applied in the seed furrow at planting.
Bradyrhizobia is often applied in the seed furrow at planting as a liquid formulation to ensure biological nitrogen fixation (BNF), especially in fields without a previous history of peanut plantings (Elkan et al., 1995; Morgan et al., 2014; Rhodes et al., 2008). Efficacy of commercially-applied inoculant can be compromised by other products applied in the seed furrow in combination with the inoculant (Jordan et al., 2010). Peanut response to Bradyrhizobia was affected by pyraclostrobin but not prothioconazole (Jordan et al., 2010, 2017). Acephate applied as a liquid spray and phorate applied in a granular formulation did not affect peanut response to commercial inoculant (Jordan et al., 2017; Tubbs et al., 2015). Jordan et al. (2010) reported that imidacloprid did not affect peanut yield response to Bradyrhizobia inoculant but did affect peanut leaf color in a negative manner in fields without a recent history of peanut planting during 1 year out of 4 years. Thrips control by imidacloprid was not determined when applied alone or in combination with Bradyrhizobia in that research.
With the increase in use of imidacloprid in peanut to control thrips, determining the need to apply acephate to emerged peanut as compared to other systemic insecticides is important. Also, given the expense of seed and the expense in correcting nitrogen deficiency following failure of commercial Bradyrhizobia inoculant (Jordan et al., 2017), research is needed to determine if negative interactions occur when Bradyrhizobia inoculant is applied in the seed furrow with imidacloprid and to determine if thrips control is compromised by this combination. Therefore, research was conducted to 1) compare peanut injury from thrips feeding and pod yield when acephate, imidacloprid, and phorate were applied in the seed furrow at planting either with or with acephate applied to peanut foliage 3 weeks after emergence and to 2) determine compatibility of imidacloprid and Bradyrhizobia inoculant applied in the seed furrow in fields with and without a recent history of peanut plantings.
Materials and Methods
Comparison of In-furrow and Foliar Insecticide Treatments
Experiments were conducted in North Carolina from 2012 through 2014 at the Peanut Belt Research Station located near Lewiston-Woodville on a Norfolk sandy loam (fine loamy, siliceous, thermic, Aquic Paleudalts) with organic matter ranging from 0.5 to 1.2% and pH 5.9 to 6.1, during 2012 and 2014 at the Upper Coastal Plain Research Station on a Goldsboro loamy sand (fine-loamy, siliceous, thermic Aquic Paleudalts), and in 2013 and 2014 in farmer fields near Wilson on an Aycock fine sandy loam (fine-silty, siliceous, thermic, Typic Paleudults). The Virginia market type peanut cultivar 'Bailey' (Isleib et al., 2011) was planted at a seeding rate designed to achieve a final in-row population of 12 to 15 plants/m of row in all fields. Corn (Zea mays L.) or cotton (Gossypium hirsutum L.) preceded peanut. Peanut was planted between May 8 and May 25 into conventionally-prepared, raised seedbeds. Plot size was 2 rows spaced 91-cm by 9 m. Production and pest management practices other than those associated with thrips control were held constant across the entire test area and were based on Cooperative Extension Service recommendations for North Carolina (Jordan et al., 2017). Peanut had not been planted in fields near Wilson in at least the past 20 years. Fields at Lewiston-Woodville and Rocky Mount had peanut planted 3 years prior.
Treatments consisted of a factorial arrangement of 4 levels of insecticide applied in the seed furrow at planting (no insecticide, acephate, imidacloprid, and phorate) and 2 levels of acephate (none versus a single application 3 weeks after planting). Acephate (Orthene 97, Valent USA, Walnut Creek, CA), imidacloprid (Admire Pro, Bayer CropScience, Research Triangle Park), and phorate (Thimet 20 G; AMVAC Chemical Corporation, Los Angeles, CA) were applied in the seed furrow at 1.1, 0.21, and 0.56 kg ai/ha. Acephate and imidacloprid were applied in 18.9 L/ha aqueous solution immediately after seed drop but prior to slit closure. Acephate was applied 3 weeks after planting at 0.4 kg/ha using a CO2-pressurized backpack sprayer calibrated to deliver 140 L/ha at a pressure of 275 kPa.
Injury from thrips feeding was recorded 2 weeks after acephate was applied postemergence using an ordinal scale of 0 to 5, where 0 = no damage, 1 = noticeable feeding but no stunting, 2 = noticeable feeding and 25% stunting, 3 = feeding with blackened terminals and 50% stunting, 4 = severe feeding and 75% stunting, 5 = severe feeding and 90% stunting (Drake et al., 2009). Incidence of tomato spotted wilt was sporadic and did not exceed 5% for any experiment or treatment. The cultivar Bailey expresses resistance to TSW (Brandenburg, 2017; Isleib et al., 2011; Shew, 2017). Peanut pods were dug and vines inverted based on pod mesocarp color (Williams and Drexler, 1981). Final pod yield was adjusted to 8% moisture.
The experimental design was a randomized complete block with treatments replicated 4 times. Data for thrips injury and pod yield were subjected to ANOVA using the PROC GLIMMIX procedure in SAS 9.4 (SAS Institute Inc., Cary, NC) appropriate for the factorial treatment arrangement of 7 levels of location/year combination (referred to as experiment) by 4 levels of in-furrow insecticide treatment by 2 levels of acephate POST. In-furrow and foliar insecticide treatments were considered fixed effects with year and replication considered a random effect. Means of significant main effects and interactions were separated using Fisher's Protected LSD test at P ≤ 0.05.
Compatibility of Imidacloprid and Bradyrhizobia Inoculant
The experiment was conducted in North Carolina at the Border Belt Tobacco Research Station near Whiteville (2014), the Peanut Belt Research Station near Lewiston-Woodville (2012 and 2013), and the Upper Coastal Plain Research Station near Rocky Mount (2012-2014). The experiment was also conducted in commercial production fields near Elizabethtown (2012) and Wilson (2012-2014). Peanut had not been planted in fields near Wilson in at least 20 years while all other fields were planted in peanut within the previous 3 to 5 years. Soils at Lewiston-Woodville, Rocky Mount, and Wilson were the same as those described in the first experiment. Soil at Elizabethtown and Whiteville was a Norfolk sandy loam (fine loamy, siliceous, thermic, Aquic Paleudalts). Peanut was planted in conventionally-prepared seedbeds as described previously.
Treatments included a factorial arrangement of 2 levels of imidacloprid (0 and 0.21 kg/ha) and 2 levels of Bradyrhizobia inoculant (0 and 0.5 L/ha of the commercial inoculant Optimize Lift, Nitragin Corp., Brookfield, WI). The commercial inoculant at this rate delivers approximately 5.0 × 1012 viable Bradyrhizobia cells/ha. Treatments were applied in the seed furrow at planting as described previously.
Visible injury caused by thrips and pod yield were recorded as described previously. The experimental design was a randomized complete block with 4 replications. Data for injury caused by thrips and pod yield were subjected to ANOVA for a 10 (experiment) by 2 (imidacloprid treatment) by 2 (Bradyrhizobia treatment) factorial treatment arrangement. Experiment, imidacloprid treatment, and Bradyrhizobia treatment were considered fixed effects. Experiment was considered fixed in order to evaluate treatments over the locational peanut production histories. Means of significant main effects and interactions were separated using Fisher's Protected LSD at P ≤ 0.05.
Results and Discussion
Comparison of In-furrow and Foliar Insecticide Treatments
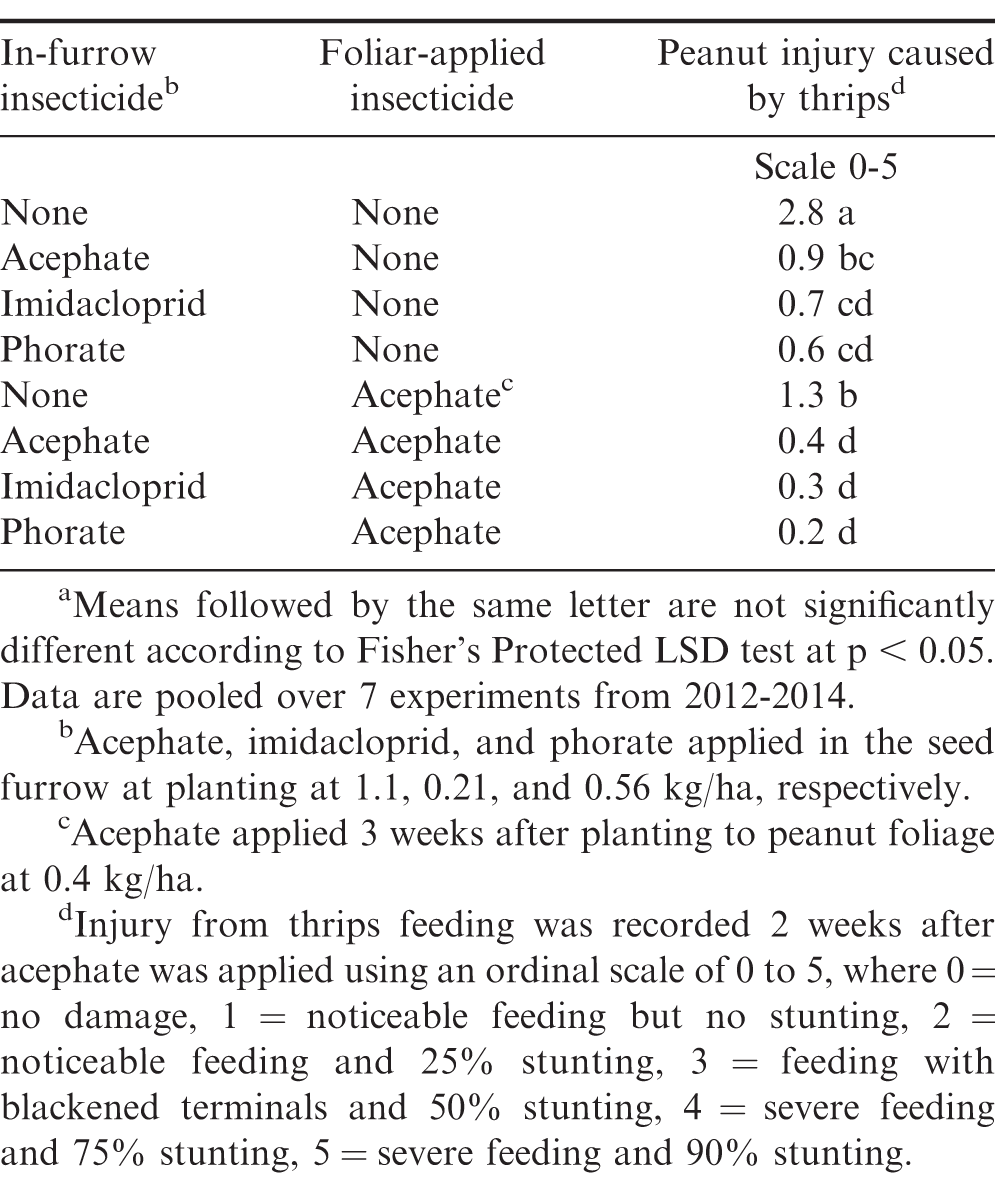
When pooled over the seven experiments, the interaction of in-furrow and post emergent insecticide treatment was significant (P = 0.0001; F = 12.8). Peanut receiving in-furrow and/or post emergent insecticide treatment reduced injury compared to the non-treated peanut (Table 1). Acephate applied in the seed furrow at planting or the POST treatment alone had increased injury compared to any combination of in-furrow insecticide followed by the POST treatment. When acephate was applied to emerged peanut there was no difference in injury when preceded by acephate, imidacloprid, or phorate applied in the seed furrow at planting. Previous research has shown that applying insecticides in-furrow and/or POST generally reduces thrips injury (Herbert et al., 2007; Hurt et al., 2005; Marasigan et al., 2016; Mahoney et al., 2018; Whalen et al., 2015). Herbert et al. (2007) reported acephate did not significantly improve thrips control when preceded by phorate or aldicarb in-furrow. However, when combined over in-furrow treatments, acephate POST applications following the in-furrow insecticide reduced thrips injury over time. In a one year study, Whalen et al. (2015) reported imidacloprid applied in the seed furrow at planting followed by acephate 2 weeks after planting decreased thrips injury compared to imidacloprid alone.
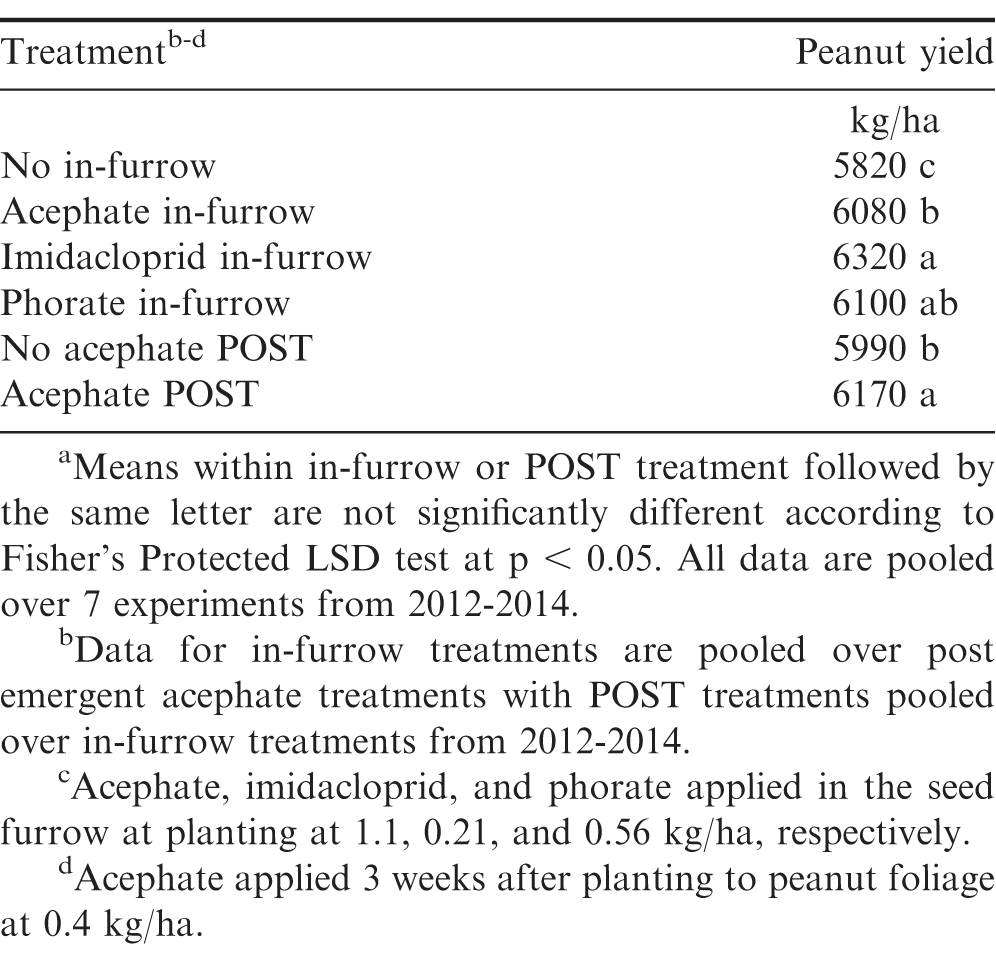
The main effects of in-furrow insecticide (P = 0.0020; F = 7.4) and POST acephate application (P = 0.0349; F = 7.4) were significant for peanut yield when pooled over all experiments. Increased peanut yield was observed when acephate, imidacloprid, or phorate were applied in the seed furrow at planting compared to no in-furrow treatment (260-500 kg/ha increase; Table 2). Imidacloprid further increased peanut yield when compared to acephate (240 kg/ha increase) while phorate provided similar yields to both acephate and imidacloprid. When pooled over in-furrow insecticides, an application of acephate to the peanut foliage 3 weeks after planting increased yields compared to no application (180 kg/ha increase).
Variation in yield response to thrips control by in-furrow insecticides has been reported previously in peanut (Brandenburg, 2017; Drake et al., 2009; Herbert et al., 2007; Hurt et al., 2005; Marasigan et al., 2016). Peanut yield response to acephate applied 3 weeks after peanut planting (approximately two weeks after emergence) was similar to a four year study by Mahoney et al. (2018), who reported increased peanut yield compared to non-treated peanut, but no yield increase when preceded by an in-furrow insecticide treatment.
Compatibility of Imidacloprid and Bradyrhizobia Inoculant
The main effects of experiment (P < 0.0001; F = 9.7) and imidacloprid treatment (P < 0.0001; F = 71.8) were significant, as was their interaction (P < 0.0001; F = 8.6). However, when the interaction was explored, only the magnitude of injury was different between experiments, but similar trends were observed across experiments with respect to the imidacloprid treatment. Given similar trends and the much larger F-value (7.4-8.3X higher) of imidacloprid in-furrow compared to the main effect of experiment and the interaction of these factors, only the main effect of imidacloprid treatment will be discussed.
When pooled over experiments and Bradyrhizobia inoculant treatments, injury ratings from thrips feeding was 0.5 (SE = 0.10) on an ordinal scale of 0 to 5 when imidacloprid was applied compared with only 1.7 (SE = 0.59) in absence of imidacloprid treatment (data not shown in tables). There was no interaction of imidacloprid with Bradyrhizobia inoculant treatment. These data suggest that co-application of Bradyrhizobia inoculant with imidacloprid will not adversely affect thrips control by imidacloprid.
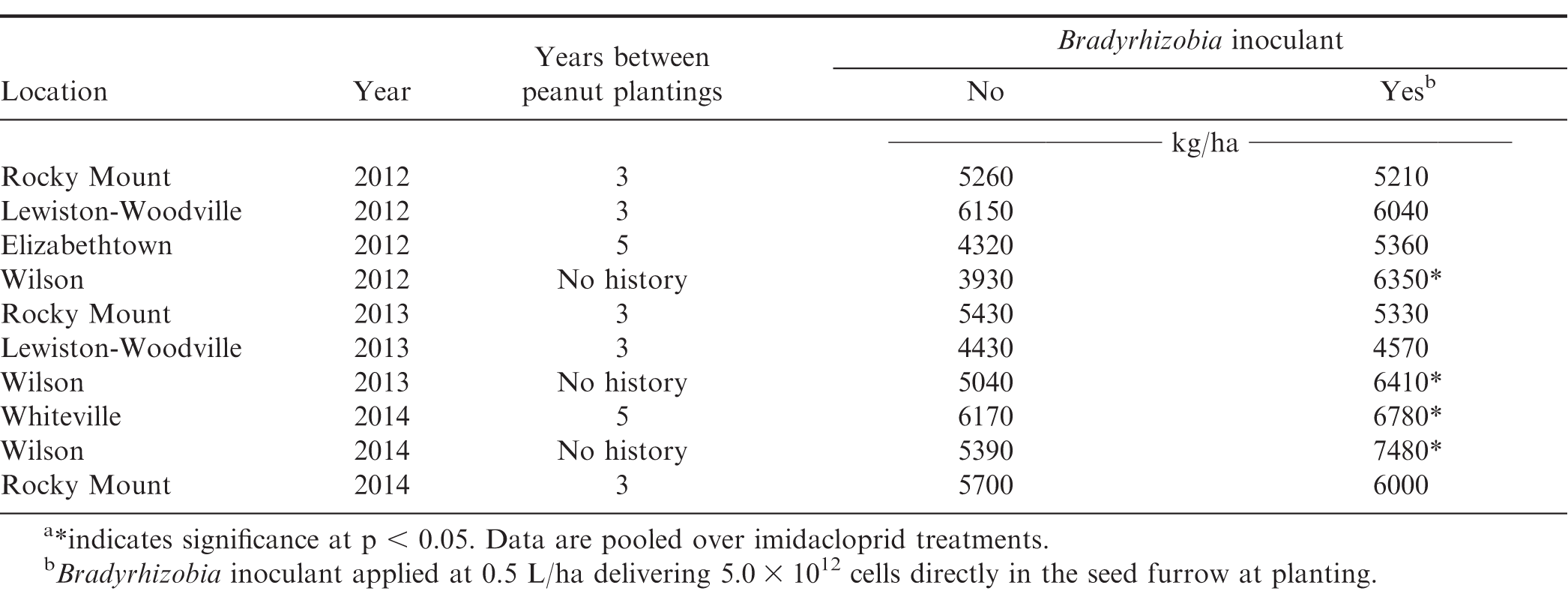
The interaction of experiment by Bradyrhizobia inoculant by imidacloprid treatment was significant (P = 0.0257; F = 2.3); therefore, the data was sorted by experiment. Bradyrhizobia inoculant, imidacloprid in-furrow, and their interaction significantly affected yields in three (Wilson 2012-2014), one (Rocky Mount 2014), and one (Whiteville 2014) experiment, respectively. With respect to the interaction in the Whiteville 2014 experiment, peanut yield for all treatment combinations were similar and increased compared to the non-treated peanut (1,190-1,400 kg/ha increase; data not shown in tables). In the Rocky Mount 2014 experiment, peanut receiving imidacloprid in-furrow had increased yields compared to those not receiving imidacloprid (1,270 kg/ha increase) regardless of the Bradyrhizobia inoculant addition (data not shown in tables).
Bradyrhizobia inoculant increased peanut yield in 4 of 10 experiments (Table 3). The increase in yield was most likely associated with previous history in these fields. A positive response to Bradyrhizobia inoculant was noted in one field without peanut in the previous 5 growing seasons or when there was no history of peanut production within the previous 20 years. In contrast, in those fields where peanut had been planted no more than 2 or 3 years prior to the experiment, yield was not affected. Previous research (Jordan et al., 2017, 2018) has shown that peanut often respond favorably to Bradyrhizobia inoculant when peanut is planted in new fields without a history of peanut production and that a modest response to Bradyrhizobia inoculant is observed in fields with recent plantings of peanut. Previous research (Jordan et al., 2017) also demonstrated that peanut yield response to acephate applied in the seed furrow at planting in a manner similar to the current study increased yield irrespective of Bradyrhizobia treatment. Additionally, response of peanut to Bradyrhizobia inoculant was independent of systemic insecticide applied in the seed furrow with inoculant. Tubbs et al. (2015) also reported no adverse effect of the systemic insecticide phorate on peanut yield response to Bradyrhizobia inoculant. These results are of practical value to growers attempting to suppress thrips and promote BNF simultaneously when planting peanut.
Results from these experiments indicate that yield response to systemic insecticides applied in the seed furrow at planting can vary while a more consistent response to acephate applied to peanut foliage was observed. When pooled over the seven experiments, imidacloprid applied in the seed furrow at planting provided increased peanut yield compared to acephate, but was similar to phorate. Consistent with other research (Jordan et al., 2010), the combination of imidacloprid and the commercial formulation of Bradyrhizobia inoculant used in these experiments are compatible.
Acknowledgements
The North Carolina Peanut Growers Association, the National Peanut Board, and the Feed the Future Peanut and Mycotoxin Innovation Lab provided partial funding for this research.
Literature Cited
Brandenburg, R.L 2017 Peanut insect and mite management Pages 81- 99 in 2017 Peanut Information. North Carolina Cooperative Extension Service Pub. AG-331 172 pages.
A.K Culbreath, and R Srinivasan (2011). Epidemiology of spotted wilt disease of peanut caused by tomato spotted wilt virus in the southeastern US. Virus. Res 159: 101- 109.
A. K., Culbreath, B. L Tillman, D. W Gorbet, C. C Holbrook, and C Nischwitz (2008). Response of new field-resistant peanut cultivars to twin row pattern or in-furrow applications of phorate for management of spotted wilt. Plant Dis 92: 1307- 1312.
A. K., Culbreath, J. W Todd, and S. L Brown (2003). Epidemiology and management of tomato spotted wilt in peanut. Annu. Rev. Phytopathol 41: 53- 75.
W. L., Drake, D. L Jordan, B. R Lassiter, P. D Johnson, R. L Brandenburg, and B. M Royals (2009). Peanut cultivar response to damage from tobacco thrips and paraquat. Agron. J 101: 1388- 1393.
Elkan, G. H 1995 Biological nitrogen fixation in peanuts pp 286- 300 in H. E Patteeand H. T Stalker, eds Advances in Peanut Science American Peanut Research and Education Society, Stillwater, OK.
D. A., Herbert, S Malone, S Aref, R. L Brandenburg, D. L Jordan, B. M Royals, and P. D Johnson (2007). Role of insecticides in reducing thrips injury to plants and incidence of tomato spotted wilt virus in Virginia market type peanut. J. Econ. Entomol 100: 1241- 1247.
C. A., Hurt, R. L Brandenburg, D. L Jordan, G. G Kennedy, and J. E Bailey (2005). Management of spotted wilt virus vector Frankliniella fusca (Thyanoptera: Thripidae) in Virginia market type peanut. J. Econ. Entomol 98: 1435- 1440.
T.G., Isleib, S.R Milla-Lewis, H.E Pattee, S.C Copeland, M.C Zuleta, B.B Shew, J.E Hollowell, T.H Sanders, L.O Dean, K.W Hendrix, M Balota, and J.W Chapin (2011). Registration of 'Bailey' peanut. J. Plant Reg 5: 27- 39.
Isleib, T. G., R. W Mozingo, J. B Graeber, J. E Hollowell, M. C Zuleta, P. W Rice, B. B Shew, H. E Pattee, S. R Milla-Lewis, and S Copeland 2016 Sullivan peanut. Technology No. 13236. Technology Transfer, North Carolina State University, Raleigh, NC.
Jordan, D.L., R Brandenburg, B Brown, G Bullen, G Roberson, and B Shew 2017 2017 Peanut Information North Carolina Cooperative Extension Service Pub. AG-331 172 pages.
D.L., Jordan, P.D Johnson, R.L Brandenburg, and J.L Faircloth (2010). Peanut (Arachis hypogaea L.) response to Bradyrhizobia inoculant applied in-furrow with agrichemicals. Peanut Sci 37: 32- 38.
Jordan, D.L., B.B Shew, and R.L Brandenburg 2017 Peanut response to acephate, Bradyrhizobia inoculant, and prothioconazole applied in the seed furrow at planting J. Crop, Forage, and Turfgrass Management 3: doi:10.2134/cftm2017.01.0007.
Mahoney, D.J., D.L Jordan, R.L Brandenburg, B.R Royals, M.D Inman, A.T Hare, and B.B. Shew 2018 Influence of planting date and insecticide on injury caused by tobacco thrips and peanut yield in North Carolina Peanut Sci (in press).
K., Marasigan, M Toews, R Kemerait, M.R Abney, A Culbreath, and R Srinivasan (2016). Evaluation of alternatives to carbamate and organophosphate insecticides against thrips and tomato spotted wilt virus in peanut production. J. Econ. Entomol 109: 544- 557.
J., Morgan, M Carroll, P Smith, R Rhodes, A Cochran, A Bradley, W Drake, C Ellison, A Whitehead, C Tyson, M Smith, T Britton, N Harrell, C Fountain, R Thagard, M Malloy, L Grimes, M Shaw, R Harrelson, D.L Jordan, P.D Johnson, R.L Brandenburg, B.B Shew, K Wells, M Parrish, G Slade, J Spencer, J Reiter, B Council, and W Marcus, M Balota, A Herbert, and H Mehl (2014). Survey of key production and pest management practices in peanut in North Carolina and Virginia during 2013. Proc. Am. Peanut Res. and Educ. Soc 46: 22- 23.
Rhodes, R., L Smith, M Williams, P Smith, F Winslow, A Cochran, B Simonds, A Whitehead, Jr., C Ellison, J Pearce, C Tyson, S Uzzell, R Harrelson, C Fountain, M Shaw, T Bridgers, D.L Jordan, R.L Brandenburg, and B.B Shew 2008 Summary of production and pest management practices by top growers in North Carolina Proc. Am. Peanut Res. Educ. Soc 40: 78- 79 (abstr.).
Shew, B.B 2017 Peanut insect and mite management Pages 100- 139 in 2017 Peanut Information. North Carolina Cooperative Extension Service Pub. AG-331 172 pages.
Tubbs, R., K Balkcom, M Toews, and R Srinivasan 2013 Effects of fertilization, tillage, and phorate on thrips and TSWV incidence in early planted peanuts Southern Cons. Agric. Syst. Conf. p. 34 (abst.).
R. S., Tubbs, R. C Kemerait, B Williams, and J. M Sarver (2015). Effect of Bradyrhizobia inoculant formulation with phorate in new peanut fields. Peanut Sci 42: 138- 144.
R., Whalen, D.A Herbert, S Malone (2014). Influence of seed treatment and granular insecticide on two peanut cultivars for thrips management. Arthropod Manag. Tests 39: F30.
R., Whalen, D.A Herbert, S Malone (2015). Evaluation of rates and combinations of liquid in-furrow and foliar insecticides for control of thrips in peanut. Arthropod Manag. Tests 40: F49.
E.J., Williams, and J.S Drexler (1981). A non-destructive method for determining peanut pod maturity, pericarp, mesocarp, color, morphology, and classification. Peanut Sci 8: 134- 141.