Introduction
Peanut is an important commodity in North Carolina planted on over 48,000 ha in 2017, which had an estimated value that exceeded $113,000,000 (USDA, 2018). Peanut producers must manage a wide range of biotic and abiotic stresses that affect peanut in order to maximize yield and economic returns (Jordan et al., 2018). Tobacco thrips, Frankliniella fusca (Hinds), is a common insect pest in North Carolina and can injure peanut in a manner that limits vegetative growth, delays pod maturation, and reduces yields through direct feeding on seedlings early in the growing season (Drake et al., 2009; Herbert et al., 2007; Marasigan et al., 2016).
Research in North Carolina has shown altering planting date can influence thrips injury and peanut yield with the consistent trend of decreased injury and increased yield when peanut is planted in mid- to late-May compared to earlier plantings (Carley et al., 2008; Drake et al., 2014; Hurt et al., 2005; Mahoney et al., 2018). Peanut can be planted anytime in the month of May in North Carolina and realize optimum yield, but growers are cautioned that weather during the season and at harvest may limit yields from late May plantings (Drake et al., 2014). Regardless of planting date, systemic insecticides are recommended at planting to minimize injury from thrips and protect yield (Herbert et al., 2007; Hurt et al., 2005; Marasigan et al., 2016; Tubbs et al., 2013). Phorate is often applied in the seed furrow at planting in North Carolina to suppress thrips and minimize injury, with an acephate application within the first month after planting to further protect peanut from thrips injury (Brandenburg, 2018).
In addition to causing direct injury, thrips (Frankliniella spp.) vector Tomato spotted wilt orthotospovirus (family Tospoviridae, genus Orthotospovirus) which results in restricted plant growth, reduced pod quality, and lower yields when tomato spotted wilt (TSW) incidence is high (Culbreath et al., 2003; Culbreath and Srinivasan, 2011; Lassiter et al., 2016). Certain cultural practices and systemic insecticides applied in a timely manner can reduce the impact of TSW (Brandenburg, 2018; Culbreath et al., 2003).
Planting date and phorate in-furrow each have been investigated for their effects on thrips injury and peanut yield, but their interactions have not been well characterized in North Carolina. In a two-year study, Hurt et al. (2005) reported reduced thrips injury in late May planted peanut compared to early May and noted a further reduction in injury, following the same trend, when phorate was applied in the seed furrow at planting. However, the influence of the interaction on peanut yield was not assessed. Likewise, in 3 of 4 years, Mahoney et al. (2018) noted less injury from thrips on peanut planted in late May compared to early- and mid-May plantings when phorate was applied in-furrow. Similar frequencies were observed when comparing mid- to early-May planted peanut. While their interaction was not significant, phorate in-furrow increased peanut yield with mid-May planted peanut providing greater yields 3 of 4 years compared to early- and late-May. To date, only Mahoney et al. (2018) has explored the interaction of phorate in-furrow application and planting date on thrips injury and peanut yield suggesting further research is needed to elucidate these effects.
Planting in cool soils can slow emergence and vigor of peanut, especially if seeds are not protected against seedling pathogens and the disease they cause (Shew, 2018). The vast majority of seed planted in commercial peanut fields in North Carolina are treated before sale with products containing a mixture of fungicides that inhibit seed- and soil-borne pathogens (Shew, 2018). However, peanut grown under organic certification cannot be treated with synthetic fungicides and insecticides that are used in conventionally-grown peanut (Cantonwine et al., 2011; Ruark and Shew, 2010). While delaying planting until soil temperatures increase facilitating the likelihood of establishing an adequate stand (Prasad et al., 2006), delayed planting in North Carolina can put peanut at risk of not reaching full maturity before temperatures cool late in the season (Carley et al., 2008). Determining the impact of planting date on peanut stand establishment would be informative for both conventional and organic production. Although organic peanut production is virtually non-existent in North Carolina, there appears to be demand for organically-grown peanut. Determining the most effective approach to obtaining an adequate stand of peanut in absence of synthetic fungicide applied to seed would increase the likelihood of success with organic peanut production in North Carolina.
The influence of plant population has largely focused on TSW, a disease vectored by thrips. Greater TSW incidence was noted when peanut was established at lower populations compared to higher populations (Black et al., 1994; Branch et al., 2003; Culbreath et al., 2013; Gorbet and Shokes, 1994; Lassiter et al., 2016; Tillman et al., 2006; Tubbs et al., 2011; Wehtje et al., 1994). Similar trends have been reported for thrips injury with less injury observed as plant populations increased (Hurt et al., 2004, 2005, 2006). In these previous studies, all plantings received aldicarb or phorate in the seed furrow at planting; thus the effect of plant population was not considered independent of insecticide effects (Hurt et al., 2004, 2005, 2006). It has been hypothesized that increased peanut populations creates canopy closure earlier and does not allow thrips to locate the host plant as easily (Culbreath et al., 2003; Hurt et al., 2006). Additionally, these authors suggested that the adverse effects were diluted, or compensated for, by higher plant populations. Better characterization of plant population effects on thrips injury is needed as well as the interaction across planting dates. These data would help growers determine how to address thrips management when plant populations are low in both conventional and organic production systems. Therefore, the objective of this research was to determine the impact of planting date on stand establishment and peanut injury trends over time caused by thrips when peanut seed was treated or not treated with a synthetic fungicide and the systemic insecticide phorate was applied in the seed furrow at planting in North Carolina.
Materials and Methods
Experiments were conducted in North Carolina from 2013 through 2016 in different fields at the Peanut Belt Research Station located near Lewiston-Woodville (36.07 N, -77.11W) on a Norfolk sandy loam (fine loamy, siliceous, thermic, Aquic Paleudults) with organic matter ranging from 0.5 to 1.2% and pH 5.9 to 6.1. The Virginia market type peanut cultivar 'Bailey' (Isleib et al., 2011) was planted at an in-row seeding rate of 18 seed/m in conventionally-prepared raised seedbeds. Two years of corn (Zea mays L.) and one year of cotton (Gossypium hirsutum L.) preceded peanut in these fields.
Treatments consisted of a factorial arrangement of 3 levels of planting date (May 2-4, May 16-18, and May 28), 2 levels of commercial seed treatment prior to planting (with or without), and 2 levels of systemic insecticide applied in the furrow at planting (with or without). The commercial seed treatment consisted of azoxystrobin plus fludioxonil plus mefenoxam (Dynasty PD, Syngenta, Greensboro, NC) at 0.08, 0.05, and 0.01 g/kg seed, respectively. The systemic insecticide phorate (Thimet 20 G; AMVAC Chemical Corporation, Los Angeles, CA) was applied at 1.12 kg ai/ha. Plot size was 2 rows spaced 91 cm by 9 m. Production and pest management practices other than those associated with seed treatment and thrips control were held constant across the entire test area and were based on Cooperative Extension Service recommendations for North Carolina (Jordan et al., 2018).
Peanut stand count and injury from thrips feeding were recorded 3 and 4 weeks after planting, respectively. The number of emerged peanut plants from a 3-m section in each of the 2 plot rows was determined. A variety of fungi and aspergillus crown rot (caused by Aspergillus niger) most likely contributed to differences in peanut stand when comparing fungicide seed treatment (Shew, 2018) but the exact causal agent relative to stand was not documented in this research. Injury from thrips feeding was recorded using an ordinal scale of 0 to 5, where 0 = no damage, 1 = noticeable feeding but no stunting, 2 = noticeable feeding and 25% stunting, 3 = feeding with blackened terminals and 50% stunting, 4 = severe feeding and 75% stunting, 5 = severe feeding and 90% stunting (Drake et al., 2009; Mahoney et al., 2018). Peanut pods were dug and vines inverted based on pod mesocarp color (Williams and Drexler, 1981). Final pod yield was adjusted to 8% moisture.
The experimental design was a split-plot with planting date serving as whole plot units and combinations of seed treatment and phorate serving as sub-plot units. Treatments were replicated 8 times. Data for stand, thrips injury and pod yield were subjected to ANOVA using the PROC MIXED procedure in SAS 9.4 (SAS Institute Inc., Cary, NC) appropriate for the factorial treatment arrangement. Planting date, fungicide seed treatment, phorate treatment, and year were considered fixed effects in order to examine trends over time with replication considered a random effect. Means of significant main and interaction effects were separated using Fisher's Protected LSD test at P ≤ 0.05. Relationships among variables were determined using Pearson Correlation coefficients with the PROC CORR in SAS (SAS Institute Inc., Cary, NC).
Results and Discussion
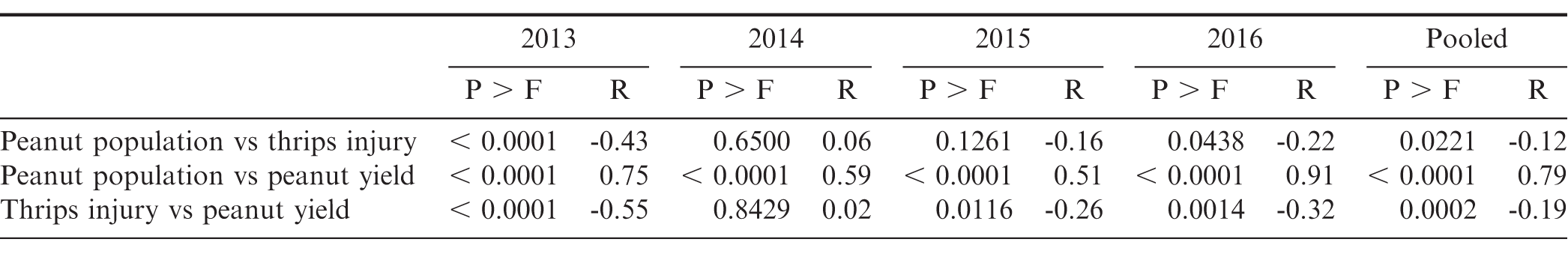
A significant negative relationship between plant population and thrips injury was observed in 2013 and 2016, in addition to data pooled over years, suggesting that thrips injury increased as plant population decreased (Table 1). Plant population was highly correlated with peanut yield within years and in data pooled across years. The relationship between thrips injury and peanut yield was also significant in 2013, 2015, and 2016, but modest overall, suggesting protecting peanut from thrips injury has a positive impact on yield.
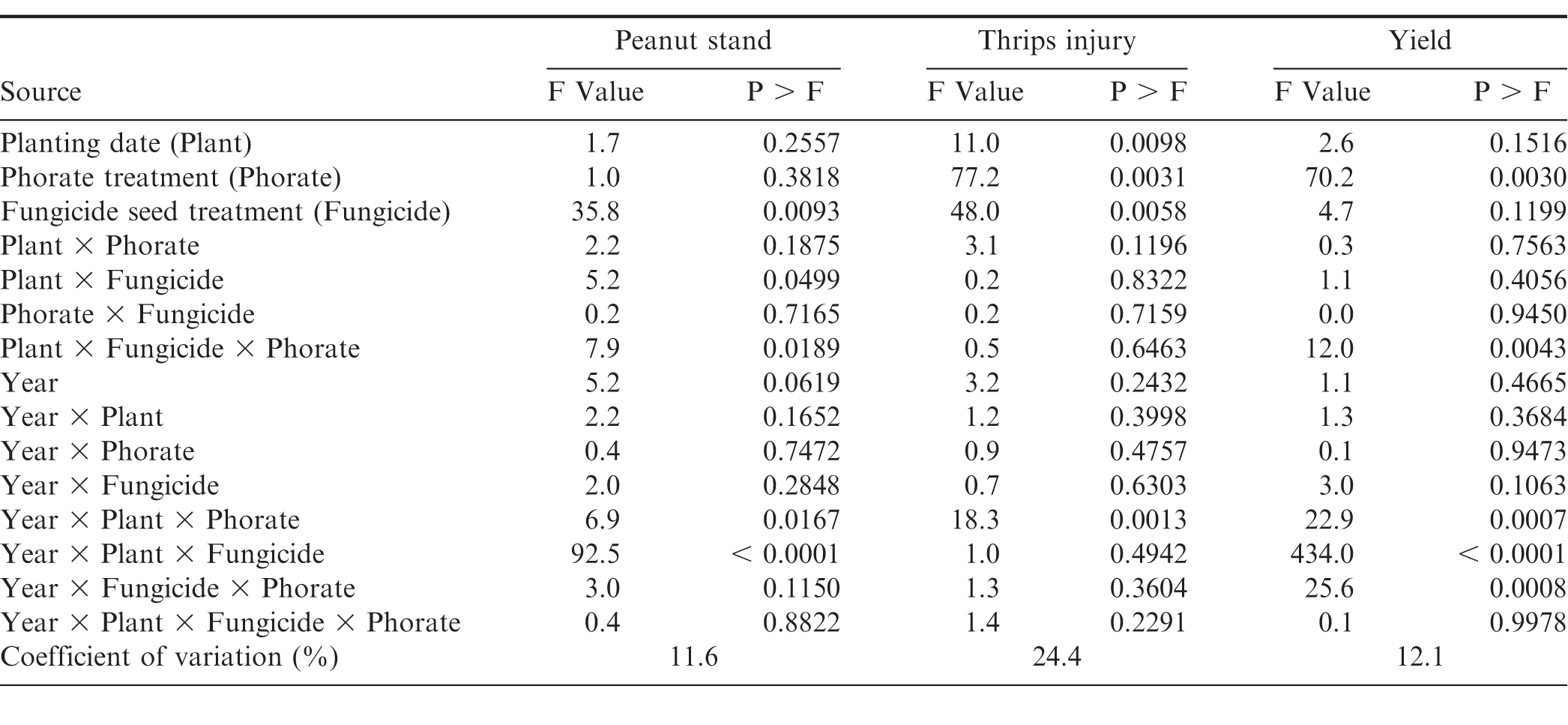
The interaction of experimental year × planting date × phorate was significant for peanut stand, but no clear trend was identified (Table 2). Phorate application did not affect stands in 2013 and 2014 at any planting date (data not shown). Decreased plant stands were observed when phorate was applied compared to those not receiving phorate in 2015 early May (65 and 73 plants/6 m of row, respectively) and 2016 mid-May plantings 33 and 37 plants/6 m of row, respectively; data not shown). However, in 2016 late May plantings, peanut receiving phorate in-furrow had increased stands when compared across all planting date and phorate treatments (76 and ≤ 69 plants/6 m of row, respectively; data not shown). Overall, changes in plant stands (No./6 m) did not exceed 8 plants, suggesting the impact is likely minimal.
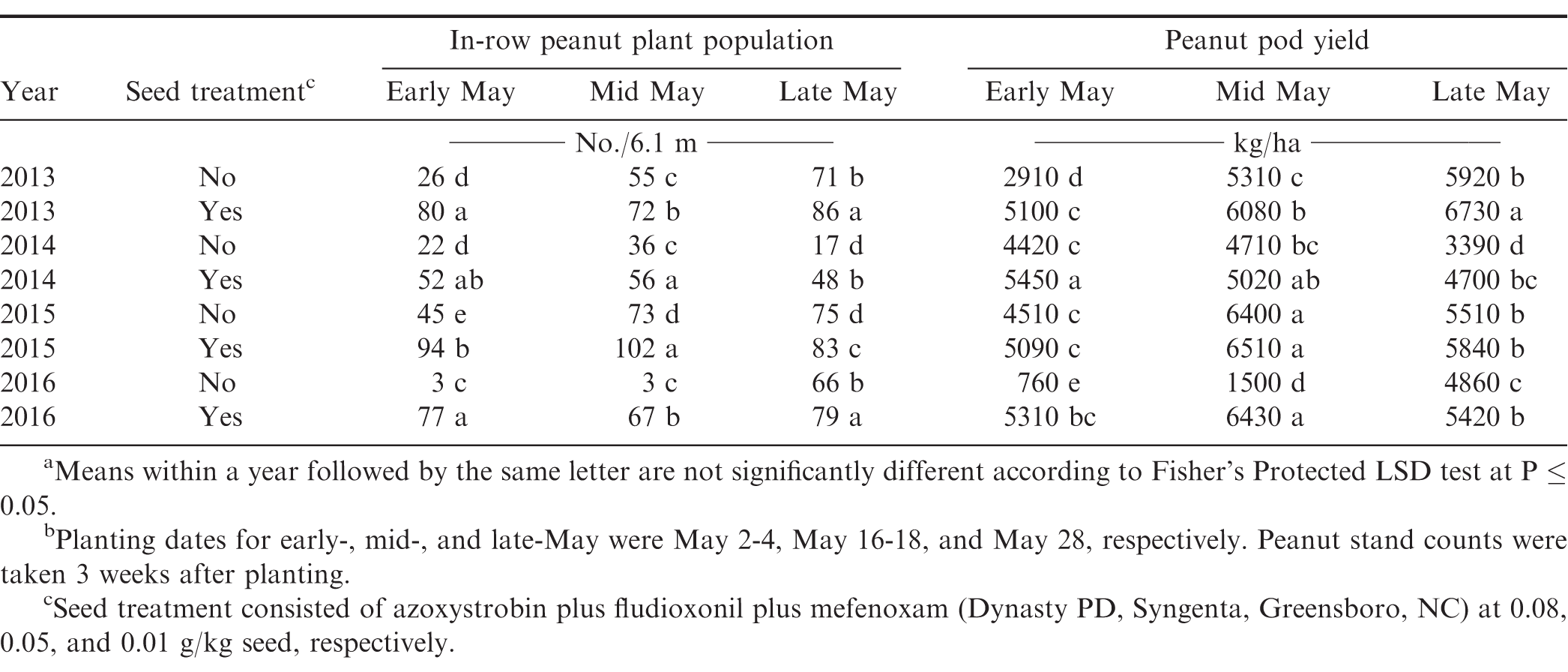
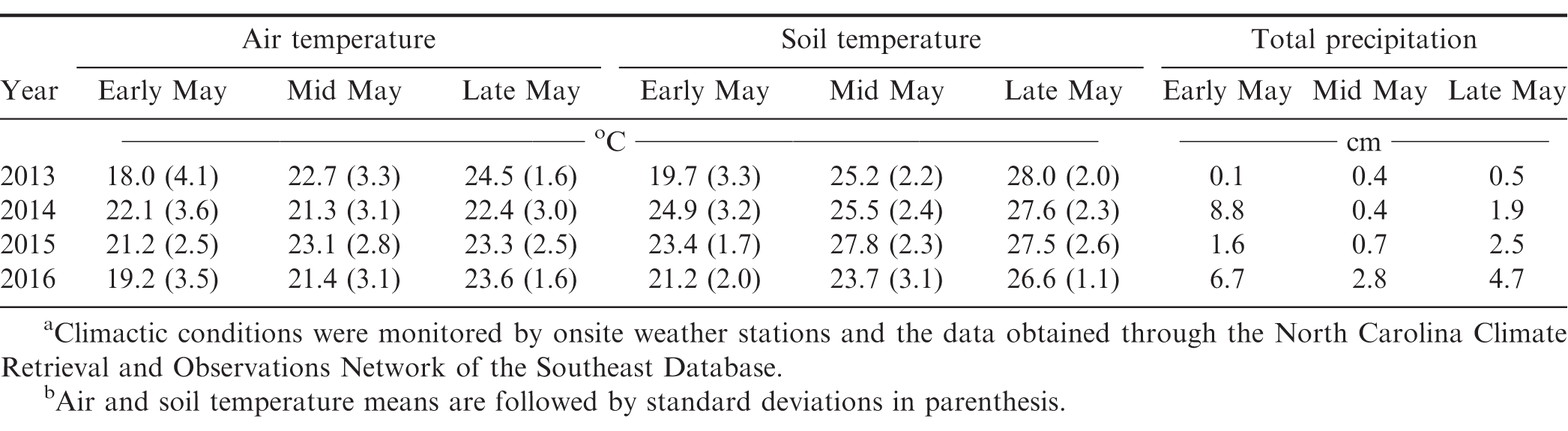
Peanut population was affected by interactions of experimental year × planting date × seed treatment (Table 2). At each planting date, treated seed consistently increased stands compared to non-treated seed, yet the magnitude of these effects generally became less impactful as planting date moved later into May with the exception of 2014 (Table 3). This deviation does not seem to be associated with climactic conditions (Table 4) and cannot be fully explained. The aforementioned trend can be seen by comparing peanut stand increases using treated vs non-treated seed in early- (2.1 to 25.7-fold increase), mid- (1.3 to 22.3-fold increase), and late-May (1.1 to 2.8-fold increase; Table 3). The appreciable differences in the 2016 early- and mid-May plantings may be due to generally cooler and wetter conditions present 14 days after planting compared to other years (Prasad et al., 2006; Table 4). In a 12-year study by Smith et al. (2000), the authors reported consistent and significant increases in peanut stand when treated seed was planted compared to non-treated seed. Stands increased by a 1.6-fold average, which is in good agreement with the presented increases (excluding early- and mid-May 2016). When looking within treated peanut seed, plant stand differences were less than 1.2-fold across planting dates suggesting more flexibility in planting dates are available when it is utilized. Within non-treated peanut seed, mid- and late-May plantings generally increased peanut stands compared to early May. These trends largely translated into yield increases as well and will be discussed further below.
Thrips injury was influenced by the main effect of seed treatment and the interaction of year × planting date × phorate application (Table 2). When pooled over years, observed thrips injury was significantly less on peanut that received a seed treatment compared to non-treated peanut seed (1.4 and 1.7 on a scale of 5, respectively; P < 0.0001). This is reflected by the significant correlation between peanut stand and thrips injury that occurred when pooled over years (Table 1). Correlations were moderate to weak, but trends suggest that thrips injury increases as stands decrease. Stands with higher plant populations may have had fewer thrips per plant, thus incurring less injury. This trend was previously reported by Hurt et al. (2004, 2005, 2006). Hurt et al. (2004) reported reduced thrips injury (8-23%) at in-row densities of 79 and 104 plants/6.1 m compared to 43 plants/6.1 m at 4 of 5 sites over two years. Using similar plant populations, Hurt et al. (2005) reported that as populations increased, thrips damage decreased. A trend of decreased thrips injury with increased in-row plant populations (79 and 104 plants/6.1 m) was also noted (Hurt et al., 2006). The authors suggested earlier canopy cover associated with increased plant populations may affect migrating thrips. Culbreath et al. (2003) suggested canopy closure may affect the thrips ability to recognize the host plant. Additionally the authors discussed the role of population, suggesting higher populations result in fewer thrips per plant or the percent of infected plants decreases within the population, thus reducing observed injury.
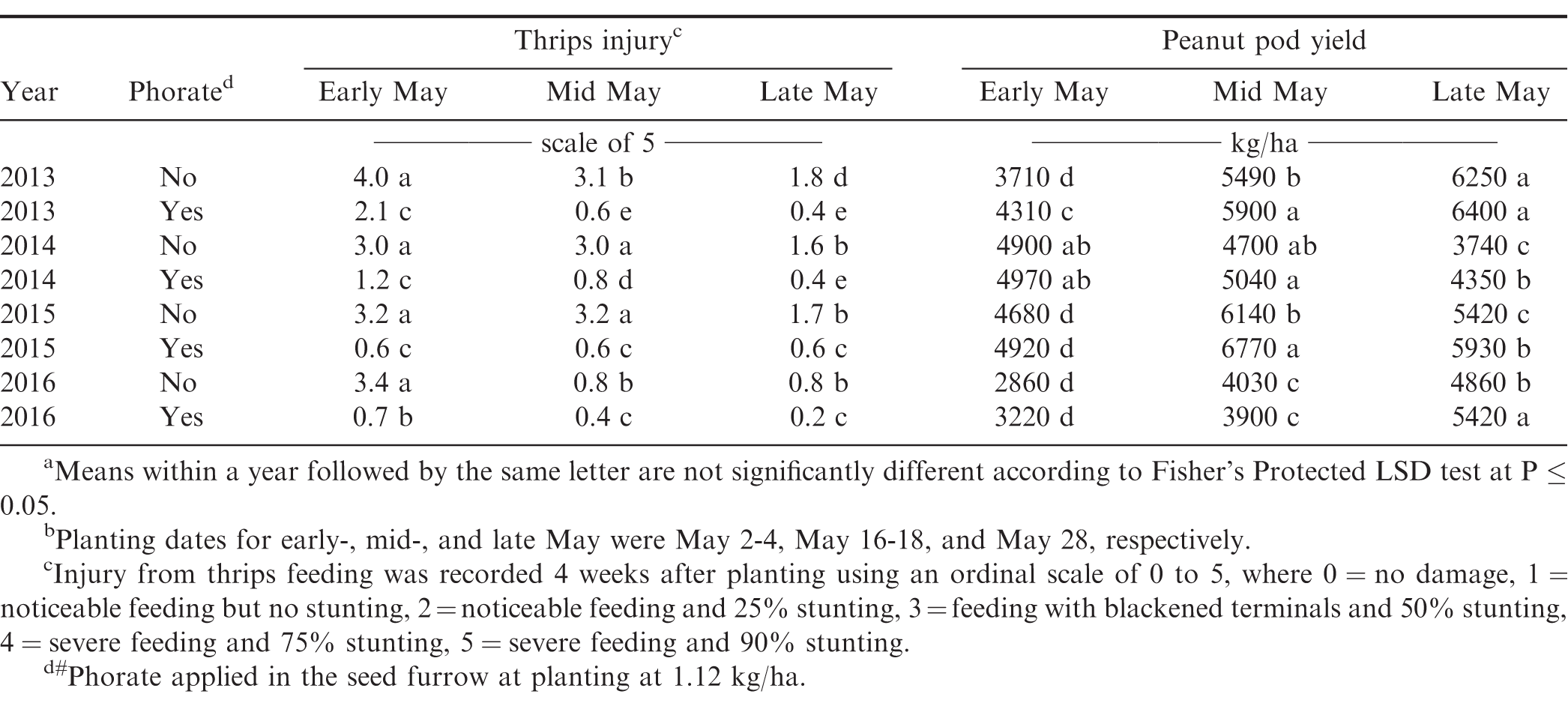
Thrips injury varied with planting date, phorate application, and year. In each experimental year, whether comparing within or across planting dates, peanut receiving phorate in-furrow had reduced injury compared to those not receiving phorate (excluding early May planted peanut receiving phorate in 2013 and 2016; Table 5). Planting date significantly affected observed thrips injury whether or not phorate was applied. When phorate was not applied, mid-May planted peanut had less injury than early planted in 2013 and 2016 while being similar during 2014 and 2015. The late May planting incurred less injury than early May in all years and in all but 2016 when compared to mid-May. When phorate was applied in the seed furrow at planting, thrips injury was greatest on early May planted peanut during 2013, 2014, and 2016. Thrips injury on mid- and late-May planted peanut was similar in 2013 and 2016, with late May having the least injury in 2014. Injury was similar on all planting dates during the 2015 season. Previous research has illustrated that peanut planted in North Carolina during early May often incurs greater thrips injury than peanut planted in mid- or late-May (Brandenburg, 2018; Drake et al., 2009; Hurt et al., 2005). Peak thrips dispersal time in North Carolina typically occurs from early- to mid-May (Morsello et al., 2008), which may explain these trends.
Peanut yield was affected by the interaction of year with planting date × phorate application, planting date × seed treatment, and phorate application × seed treatment (Table 2). Peanut yield for the early May planting increased by 600 kg/ha when phorate was applied in 2013 (Table 5). Phorate increased yields by 410 and 630 kg/ha for mid-May planted peanut in 2013 and 2015, respectively. Increases were also recorded for the late May planting in 2014, 2015, and 2016 (600, 510, and 560 kg/ha, respectively) when phorate was applied. Applying phorate in-furrow has previously been shown to increase yield compared to no systemic insecticide treatment in numerous studies (Culbreath et al., 2008; Herbert et al., 2007; Mahoney et al., 2018; Marasigan et al., 2016). Within each phorate application, planting date significantly affected yields. Mid- and late-May planted peanut had increased yields compared to early May in 3 of 4 years with average increases of 1,570 and 1,620 kg/ha for peanut receiving phorate in-furrow or not, respectively. In 2013, late May planted peanut yielded greater than mid-May when not receiving phorate in-furrow; however, yields were similar when phorate was applied. In 2014 and 2015, regardless of phorate treatment, peanut yield was greater for mid-May planting compared to late May planting with the opposite being the case in 2016. When comparing yield from mid- to late-May plantings for peanut not receiving phorate the average increase and decrease was 840 and 800 kg/ha, respectively. For peanut receiving phorate in-furrow, these were 760 and 1,010 kg/ha, respectively. This may be attributed in part to peanut stand as the increase yield trends measured generally had increased peanut stands (Table 3).
Planting date has previously been shown to affect peanut yields in North Carolina. Carley et al. (2008) reported mid-May planted peanut produced the greatest yields in 2003 and 2004, but yields were similar to early- and late-May planted peanut in 2005. Yield from early- and late-May plantings was similar across years. Drake et al. (2014) also reported no significant difference in yield among early and late planting dates in 2010 and 2011, but yield was greatest in early May plantings compared with late May in 2008 and 2009. In the presented research, late May planted peanut generally out yielded early May. Differing responses reported by Carley et al. (2008) may be attributed to cultivar as they utilized ('VA 98R'; Mozingo et al., 2000) one no longer planted in NC (Jordan et al., 2018). Drake et al. (2014) focused on multiple cultivar responses to tillage and planting date with respect to disease management instead of thrips management. More recently, Mahoney et al. (In press) observed peanut yield from mid- and late-May plantings were increased in 3 and 2 years, respectively, when compared yield with early May plantings.
Interacting effects of planting date and seed treatment on yield varied among years. Planting treated seed increased yields 3 of 4 years for early- and late-May planted peanut by an average of 2,590 and 890 kg/ha, respectively compared to non-treated seed (Table 3). Seed treatment increased peanut yield for mid-May plantings in 2 of 4 years by an average of 2,850 kg/ha. Middleton and Mayer (1985) previously reported on the influence of seed treatment on peanut yield as reflected by crop value. The authors reported a 29-43% increase in crop value when treated seed was planted compared to non-treated. Smith et al. (2000) reported a 13-73% increase (37% average increase) in yield when peanut seed was treated compared to non-treated seed over a 12-year period. More recently, Jadon et al. (2015) reported 32-45.6% yield increases when using fungicide treated seed compared to seed not treated with fungicide for Spanish market type peanut.
Within seed treatments, planting date affected yields. When non-treated peanut was planted, yield increased from early- to mid-May plantings and from mid- to late-May plantings in 2013 (2,400 and 600 kg/ha increase, respectively) and 2016 (740 and 3,360 kg/ha increase, respectively; Table 3). In 2014, early- and mid-May planted peanut yields were similar and increased more than 1,000 kg/ha) compared to those planted in late May. Similar to 2013 and 2016, mid- and late-May planted peanut in 2015 yielded more than early May (1,890 and 1000 kg/ha increase, respectively); however, mid-May yield was greater than late May in this year. Within treated peanut seed, yields increased from early- to mid-May and mid- to late-May in 2013 (980 and 650 kg/ha increase, respectively). In 2014, early May planted peanut had increased yields compared to late May (760 kg/ha increase), but was similar to mid-May. In 2015 and 2016, mid-May planted peanut yielded greater than did early- or late-May planted peanut (670 to 1,420 kg/ha increase) with late May out yielding (1000 kg/ha) early May in 2015. When comparing across seed treatment and planting date, these data suggests using treated seed extends the planting window while maintaining comparable yields. For example, when treated seed was used, early May plantings had increased yields compared to mid-May planted non-treated peanut in 2014 and 2016 while yielding similar in 2013.
The increase in yields when comparing treated and non-treated seed is likely caused by increased plant stands that were obtained by planting treated seed (Table 3). This supported by correlations between peanut stand and yields which showed significant, moderate to very strong relationships in 2013, 2014, 2015, and 2016 suggesting as peanut stand increases, so do yields (Table 1).
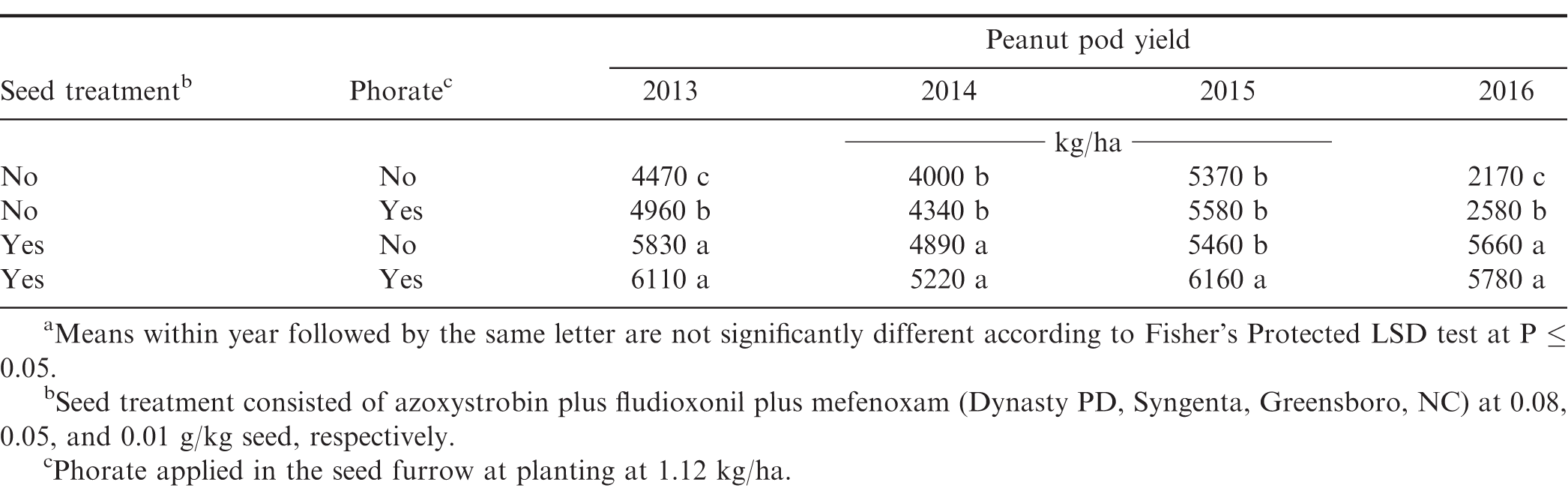
Peanut yield was affected by the interaction of year × phorate application × seed treatment. General trends show increased yields from non-treated < phorate only < seed treatment only < phorate plus fungicide seed treatment (Table 6). Excluding one instance in 2015, utilizing treated peanut seed significantly increased yields regardless of phorate application. On the other hand, phorate application significantly increased yields in one and two instances when treated and non-treated seed were planted, respectively. These data illustrate achieving adequate stands is imperative for reducing overall thrips pressure and maximizing peanut yield.
The presented research provides further evidence of important pest management strategies for peanut production, including planting date strategies, using treated seed, increasing plant stands, and applying phorate in the seed furrow at planting. With respect to conventional production, the data suggest that planting fungicide-treated seed and applying insecticide, in this case phorate, is recommended across planting dates. These data also illustrates the expanded planting window gained by using treated peanut seed as resource, or other constraints, allows planting during early May. Further, treated seed increased peanut stands, which aided in reducing thrips injury and increased yields.
In regards to organic producers, these data suggest mid- to late-May plantings may experience more favorable climatic conditions that improve plant vigor and growth, thus promoting emergence and improved plant stands. Later planting dates and increased plant stands also aided in thrips management as less injury was seen in both cases and improved peanut yields. Seeding rate may also be increased to maximize the aforementioned benefits. The above recommendations will aid in improving pest management and peanut yields for North Carolina producers. These data can also be used to assist growers in determining the impact of poor stands on yield and the economic value of replanting.
Acknowledgements
The North Carolina Peanut Growers Association, the National Peanut Board, and the Feed the Future Peanut and Mycotoxin Innovation Lab provided partial funding for this research.
Literature Cited
Black, M.C., H Tewolde, C.J Fernandez, and A.M Schubert 1994 Effects of seeding rate irrigation and cultivar on spotted wilt, rust, and southern blight diseases of peanut Proc. Am. Peanut Res. Ed. Soc 26: 50 (Abstr.)
W.D., Branch, J.A Baldwin, and A.K Culbreath (2003). Genotype x seeding rate interaction among TSWV-resistant runner-type peanut cultivars. Peanut Sci 30: 108- 111.
Brandenburg, R.L 2018 Peanut insect and mite management Pages 80- 99 in 2018 Peanut Information. North Carolina Cooperative Extension Service Pub. AG-331 170 pages.
E.G., Cantonwine, C.C Holbrook, A.K Culbreath, R.S Tubbs, and M.A Boudreau (2011). Genetic and seed treatment effects in organic peanut. Peanut Sci 38: 115- 121.
D.S., Carley, D.L Jordan, C.L Dharmasri, T.B Sutton, R.L Brandenburg, and M.G Burton (2008). Peanut (Arachis hypogaea L.) response to planting date and potential of canopy reflectance as an indicator of pod maturation. Agron. J 100: 376- 380.
A.K Culbreath, and R Srinivasan (2011). Epidemiology of spotted wilt disease of peanut caused by tomato spotted wilt virus in the southeastern US. Virus. Res 159: 10- 109.
A.K., Culbreath, B.L Tillman, D.W Gorbet, C.C Holbrook, and C Nischwitz (2008). Response of new field-resistant peanut cultivars to twin row pattern or in-furrow applications of phorate for management of spotted wilt. Plant Dis 92: 1307- 1312.
A.K., Culbreath, J.W Todd, and S.L Brown (2003). Epidemiology and management of tomato spotted wilt in peanut. Annu. Rev. Phytopathol 41: 53- 75.
A.K., Culbreath, R.S Tubbs, B.L Tillman, J.P Beasley W.D Branch, C.C Holbrook, A.R Smith, and N.B Smith (2013). Effects of seeding rate and cultivar on tomato spotted wilt of peanut. Crop Prot 53: 118- 124.
W.L., Drake, D.L Jordan, P.D Johnson, B.B Shew, R.L Brandenburg, and T Corbett (2014). Peanut response to planting date, tillage, and cultivar in North Carolina. Agron. J 106: 486- 490.
W.L., Drake, D.L Jordan, B.R Lassiter, P.D Johnson, R.L Brandenburg, and B.M Royals (2009). Peanut cultivar response to damage from tobacco thrips and paraquat. Agron. J 101: 1388- 1393.
Gorbet, D.W and F.M Shokes 1994 Plant spacing and tomato spotted wilt virus Proc. Am. Peanut Res. Ed. Soc 26: 50 (Abstr.)
D.A., Herbert, S Malone, S Aref, R.L Brandenburg, D.L Jordan, B.M Royals, and P.D Johnson (2007). Role of insecticides in reducing thrips injury to plants and incidence of tomato spotted wilt virus in Virginia market type peanut. J. Econ. Entomol 100: 1241- 1247.
C.A., Hurt, R.L Brandenburg, D.L Jordan, G.G Kennedy, and J.E Bailey (2004). Effect of cultivar and plant population on spotted wilt in Virginia market-type peanut. Peanut Sci 31: 101- 107.
C.A., Hurt, R.L Brandenburg, D.L Jordan, G.G Kennedy, and J.E Bailey (2005). Management of spotted wilt virus vector Frankliniella fusca (Thyanoptera: Thripidae) in Virginia market type peanut. J. Econ. Entomol 98: 1435- 1440.
C.A., Hurt, R.L Brandenburg, D.L Jordan, B.M Royals, and P.D Johnson (2006). Interactions of tillage with management practices designed to minimize tomato spotted wilt of peanut. Peanut Sci 33: 83- 89.
T.G., Isleib, S.R Milla-Lewis, H.E Pattee, S.C Copeland, M.C Zuleta, B.B Shew, J.E Hollowell, T.H Sanders, L.O Dean, K.W Hendrix, M Balota, and J.W Chapin (2011). Registration of 'Bailey' peanut. J. Plant Reg 5: 27- 39.
K.S., Jadon, P.P Thirumalaisamy, V Kumar, V.G Koradia, and R.D Padavi (2015). Management of soil borne diseases of groundnut through seed dressing fungicides. Crop Prot 78: 198- 203.
Jordan, D.L., R Brandenburg, B Brown, G Bullen, G Roberson, and B Shew 2018 2018 Peanut Information North Carolina Cooperative Extension Service Pub. AG-331 170 pages.
B.R., Lassiter, D.L Jordan, G.G Wilkerson, B.B Shew, and R.L Brandenburg (2016). Influence of planting pattern on pest management in Virginia market type peanut (Arachis hypogaea L.). Peanut Sci 43: 59- 66.
Mahoney, D.J., D.L Jordan, R.L Brandenburg, B.R Royals, M.D Inman, A.T Hare, and B.B Shew 2018 Influence of Planting Date and Insecticide on Injury Caused by Tobacco Thrips and Peanut Yield in North Carolina Peanut Sci 45: (in press)
K., Marasigan, M Toews, R Kemerait, M.R Abney, A Culbreath, and R Srinivasan (2016). Evaluation of alternatives to carbamate and organophosphate insecticides against thrips and tomato spotted wilt virus in peanut production. J. Econ. Entomol 109: 544- 557.
K.J Middleton, and R.J Mayer (1985). Liquid formulations of seed-treatment fungicides suitable for use on peanuts. Crop Prot 4: 494- 500.
S.C., Morsello, R.L Groves, B.A Nault, and G.G Kennedy (2008). Temperature and precipitation affect seasonal patterns of dispersing tobacco thrips, Frankliniella fusca, and onion thrips, Thrips tabaci (Thysanoptera: Thripidae) caught on sticky traps. Environ. Entomol 37: 79- 86.
R.W., Mozingo T.A Coffelt, and T.G Isleib (2000). Registration of 'VA 98R' peanut. Crop Sci 40: 1202- 1203.
P.V.V., Prasad, K.J Boote, J.M.G Thomas, L.H Allenand D.W Gorbet (2006). Influence of soil temperature on seedling emergence and early growth of peanut cultivars in field conditions. J. Agron. Crop Sci 192: 168- 177.
S.J Ruark, and B.B Shew (2010). Evaluation of microbial, botanical, and organic treatments for control of peanut seedling diseases. Plant Dis 94: 445- 454.
Shew, B.B 2018 Peanut disease management Pages 100- 137 in 2018 Peanut Information. North Carolina Cooperative Extension Service Pub. AG-331 170 pages.
D.T., Smith, M.C Black, W.J Grichar, and A.J Jaks (2000). Economic assessment and fungicide use on peanut seed in the Southwestern United States. Peanut Sci 27: 39- 44.
Tillman, B.L., D.W Gorbet, A.K Culbreath, and J.W Todd 2006 Response of peanut cultivars to seeding density and row patterns Crop Manag doi:10.1094/CM2006-0711-01-RS.
Tubbs, R., K Balkcom, M Toews, and R Srinivasan 2013 Effects of fertilization, tillage, and phorate on thrips and TSWV incidence in early planted peanuts Southern Cons. Agric. Syst. Conf. p. 34 (abst.).
R.S., Tubbs, J.P Beasley A.K Culbreath, R.C Kemerait, N.B Smith, and A.R Smith (2011). Row pattern and seeding rate effects on agronomic, disease, and economic factors in large-seeded runner peanut. Peanut Sci 38: 93- 100.
[USDA] United States Department of Agriculture 2018 2017 state agriculture overview https://www.nass.usda.gov/Quick_Stats/Ag_Overview/stateOverview.php?state=north%20carolina. Accessed July 24, 2018.
G., Wehtje, R Weeks, M West, L Wells, and P Pace (1994). Influence of planter type and seeding rate on yield and disease incidence in peanut. Peanut Sci 21: 16- 19.
E.J., Williams, and J.S Drexler (1981). A non-destructive method for determining peanut pod maturity, pericarp, mesocarp, color, morphology, and classification. Peanut Sci 8: 134- 141.