Introduction
Defoliation of peanut (Arachis hypogaea L.) vegetation by insects is a concern for peanut growers across the southeastern United States. Little is known about peanut economic injury levels for defoliating caterpillars in Mississippi. While no research on defoliation thresholds in Mississippi peanuts exists, there is also a general lack of understanding across the Southeast on damage thresholds, especially on newer cultivars.
The peanut plant canopy is susceptible to a range of insects and diseases. Defoliation by pests can impede photosynthetic potential by reducing leaf area, and in turn, light interception and photosynthesis (Boote et al. 1980, Bourgeois and Boote 1992). The reduction in photosynthates can reduce vegetative and reproductive growth (Boote et al. 1980, Bourgeois and Boote 1992).
Pest damage from defoliating insects in peanut varies from incidental feeding to near plant consumption, with the level of defoliation determining yield loss (Stalker and Campbell 1983). Corn earworm (CEW) [Helicoverpa zea (Boddie)], fall armyworm (FAW) [Spodoptera frugiperda (J. E. Smith)], granulate cutworm (GCW) [Feltia subterranean (F.)], velvetbean caterpillar (VBC) [Anticarsia gemmatalis (Hübner)], and other Lepidoptera species are pests that negatively impact the plant canopy via physical defoliation (Deitz et al. 1992, Jones et al. 1982, Lynch 1996, Minton et al. 1991, Stalker and Campbell 1983). While all of these insects can affect the plant canopy, their feeding behaviors vary among species and crop growth stages, meaning not every insect listed invades the peanut plant at the same time or damages the plant the same way. Some insects prefer young terminal vegetation, while other pests may favor older vegetation based on nutritional requirements (Deitz et al. 1992, Stalker and Campbell 1983).
Previous research addressed the feeding behaviors of defoliating caterpillars in peanut (Deitz et al. 1992, Endan et al. 2006, Garner and Lynch 1981, Todd et al. 1991). According to Deitz et al. (1992) appropriate thresholds for FAW, GCW, and CEW are 13 larvae per row meter in South Carolina. However, sampling difficulty for these pest species was noted because of feeding site preference between the larvae and the time of day that sampling occurred. Moreover, the impact of larval defoliation was underestimated because feeding in the axillary bud region, especially by GCW, retarded development of new leaves and reproductive branches. While previous research helps to explain feeding patterns, more work is needed in a field-scale situation to quantify yield consequences of defoliation.
Defoliation from disease pathogens has received more attention than defoliation by insects in peanut. Early and late leaf spot [caused by Cercospora arachidicola S. Hori and Phaeoisariopsis personatum (Berk. & M.A. Curtis), respectively] are the two most common defoliating fungal pathogens affecting peanut fields across the southeastern peanut belt (Adomu et al. 2005, Boote et al. 1980, Bourgeois et al. 1991, Bourgeois and Boote 1992). Defoliation resulting from severe incidence of leaf spot can reduce yield up to 50% if preventative and curative measures are not taken (Bourgeois et al. 1991). Even when precautions are taken and a high-risk fungicide plan incorporated, yield losses up to 10% can occur (Pixley et al. 1990). Previous research on defoliation from the leaf spot diseases in peanut may help refine defoliating-insect management decisions.
Soybean experiences indirect feeding much like that of peanut. Owen et al. (2013) noted that feeding on the foliage, stems, and/or roots of plants can lead to yield reductions by stressing the plant. Owen et al. (2013) used hand removal of foliage at different growth stages in soybean to simulate feeding by defoliating caterpillar pests and determine the impact on yield. Based on that research, they were able to establish accurate defoliation thresholds at different soybean growth stages regardless of insect species. Similarly, the erratic feeding patterns across species that affect peanut and the difficulty of accurately estimating caterpillar densities make it difficult to use insect counts as a trigger for control measures. As a result of this and a lack of recent work on insect defoliation effects on peanut, the objective of this research was to determine canopy defoliation thresholds at multiple growth stages in peanut. Ultimately, this work will be important for developing recommendations that will allow extension personnel, producers, and consultants to make informed management decisions when dealing with peanut canopy defoliation.
Materials and Methods
Field research was conducted on a Leeper silty clay loam (fine, smectitic, nonacid, thermic Vertic Epiaquepts) (USDA-NRCS, 2016) at the Mississippi State University R.R. Foil Research Center in Starkville, Mississippi and on a Bosket very fine sandy loam (fine-loamy, mixed, active, thermic Mollic Hapludalfs) (USDA-NRCS, 2016) at the Mississippi State University Delta Research and Extension Center (MSU DREC) near Stoneville, Mississippi in 2015 and 2016. Both locations were furrow irrigated.
Land preparation at the Starkville location included a ripper-hipper single bed formation, with a do-all over the top prior to planting, and a roller packer to firm the seed bed. Single beds were 0.97-m wide. Soil preparation at MSU DREC was similar in that 1.02-m wide beds were ripped and hipped and then rolled to firm the seed bed. Fertilizer requirements and applications, which include those for calcium and boron, were based on MSU Extension recommendations (Oldham 2017). Immediately after planting in 2015, a pre-emergent herbicide tank-mix of pendimethalin (930 g a.i./ha), diclosulam (27 g/ha), and flumioxazin (107 g/ha) was applied. Pre-emergent herbicides in 2016 consisted of a tank-mix of S-metolachlor (650 g/ha) and flumioxazin (107 g/ha). Fungicide programs were based on guidelines obtained from the medium risk model of the Peanut Disease Risk Index (Kemerait et al. 2015). Chlorantraniliprole at 75 g/ha was applied once across all plots at both locations in 2016, due to fall armyworm pressure that could have potentially confounded results if left untreated.
Peanut cultivar Georgia-06G (Branch, 2007) was planted in Starkville, MS using a two-row Monosem precision air planter (Monosem, Inc., Edwardsville, KS). Seed were planted at a depth of 5.1 cm at a rate of 20 seed/m of row in two row plots that were 1.94-m wide and 4.57-m long. At Stoneville, a John Deere MaxEmerge2 four-row vacuum planter (John Deere, Moline, Illinois) was used to seed the same cultivar at a similar seeding depth and rate as those at the Starkville site. Two row plots at Stoneville measured 2.04-m wide and 6.10-m long. Seed at both locations had a fungicide seed treatment (azoxystrobin, fludioxonil, and mefenoxam, Syngenta Crop Protection, Greensboro, NC). Planting dates for each site year are reported in Table 1.
For both locations the experimental design was a six (defoliation level) by two (defoliation timing) factorial arranged in a randomized complete block, with four replications per site-year. The levels of defoliation were 0, 20, 40, 60, 80, and 100% of the peanut foliage. Defoliation was achieved by hand removal of completely expanded leaves, while ensuring that flowers on the plant, pods in the ground, and leaf buds were undisturbed. The defoliation events occurred at either 40 or 80 d after emergence (DAE). These timings correspond closely with the beginning of pegging and peak pod fill, respectively.
Plots in Starkville were evaluated for above-ground plant and pod biomass immediately following each defoliation and at two and four wk after defoliation. Above-ground biomass and pod samples were taken from 0.3-m of row at each sample timing and were placed in forced air dryers for 48 hrs at 46 C before biomass readings were recorded. Canopy height and width measurements were taken from 10 random locations in each plot and averaged at each site-year at two and four wk after each defoliation event. Plots were also evaluated for pod yield and grade. Harvest timing was determined at each site-year by the hull-scrape maturity profile method (Williams and Drexler 1981). Plots were inverted using a two-row KMC digger-shaker-inverter (Kelley Manufacturing, Tifton, GA) and harvested using a two-row KMC peanut combine. Inversion and harvest dates are reported in Table 1. Yield was adjusted to 10.5% moisture and graded.
To determine the impact of defoliation on peanut grade, biomass, and canopy development; data were analysed with analysis of variance (PROC GLM, SAS 9.4, SAS Institute, Cary, NC). Peanut grade, biomass, and canopy development were dependent variables and defoliation level was the independent variable. When effects were found to be significant, least significant differences (LSD, 𝜶 = 0.05) were calculated to separate means. For the purpose of determining the impact of defoliation on peanut yields, data were analysed with regression analysis (PROC GLM, SAS 9.4, SAS Institute, Cary, NC). Defoliation level was the independent variable and peanut yield was the dependent variable in the model. No significant interaction occurred between defoliation levels and site-years, so analyses are reported with all data combined across locations and years. Analysis of covariance was used to compare the slopes of the regression equations for levels of defoliation at each time of defoliation.
Data from the regression equations were used to estimate an economic injury level (EIL) for regression equations that had a significant relationship between level of defoliation and peanut yield. The equation
(Pedigo et al. 1986) was used to calculate the economic injury level (EIL), C is the cost of control, V is the value of the crop in $/metric tonne, b is the yield loss per 1% defoliation value derived from the slope of the regression equation, and K is the control tactic or application (%). This is not a specific guide to any one control measure or tactic, and K was assumed to have an 85% control level.
Results and Discussion
Yield and grade
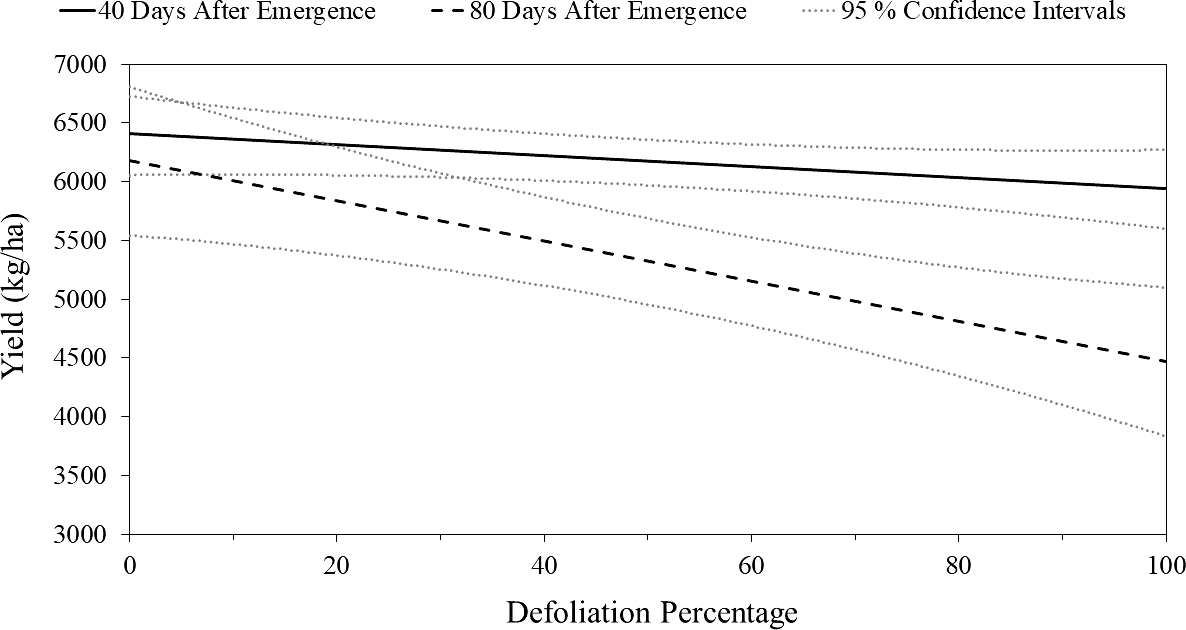
.The relationship between canopy defoliation at 40 DAE and pod yield of peanut was not significant (P = 0.16, R-squared = 0.57), suggesting that defoliation occurring at this timing does not impact peanut yields (Figure 1). In contrast, the relationship between canopy defoliation at 80 DAE and pod yield of peanut was significant (P < 0.01, r2 = 0.84), suggesting that defoliation at 80 DAE impacts peanut yields (Figure 1). There was a significant interaction between timing of defoliation and level of defoliation (P = 0.01) indicating that there were differences between the slopes of the regression equations at 40 and 80 DAE. At 80 DAE, the regression equation indicated a yield decrease of 18.6 kg/ha for every 1% increase in canopy defoliation. The greater impact on yield from defoliation at 80 DAE relative to 40 DAE can likely be explained by the fact that plants are at the height of reproductive growth at 80 DAE. Conversely, at 40 DAE, peanut plants are in late vegetative or early reproductive stages, giving them time to compensate from injury.
Yield regressions for the 40 and 80 d after emergence (DAE) defoliation timing across all site-years. The solid line (—) is the linear trend line for predicted values with the upper and lower dotted lines (•••••) giving the 95% confidence interval for defoliation at each given level for 40 DAE. The 40 DAE trend line equation Y= -3.08x + 6285 represents a pod yield reduction of 3.08 kg/ha per 1% of canopy defoliation. The long dashed line (— —) is the linear trend line for predicted values with the upper and lower short dashed lines (- - -) giving the 95% confidence interval for defoliation at each given level for 80 DAE. The 80 DAE trend line equation of Y= -18.6x 6285 represents a pod yield reduction of 18.6 kg/ha per 1% of canopy defoliation.
Defoliation did not affect market grade at any defoliation timing or level (P = 0.99). Market grades based on total sound mature kernels (TSMK) were 71.7 to 73.8 across defoliation treatments and the control. Because grade can be correlated with maturity (Court et al., 1984; Knauft et al., 1986; Mozingo et al., 1991) it can be assumed that defoliation did not affect optimum harvest timing.
Plant growth characteristics
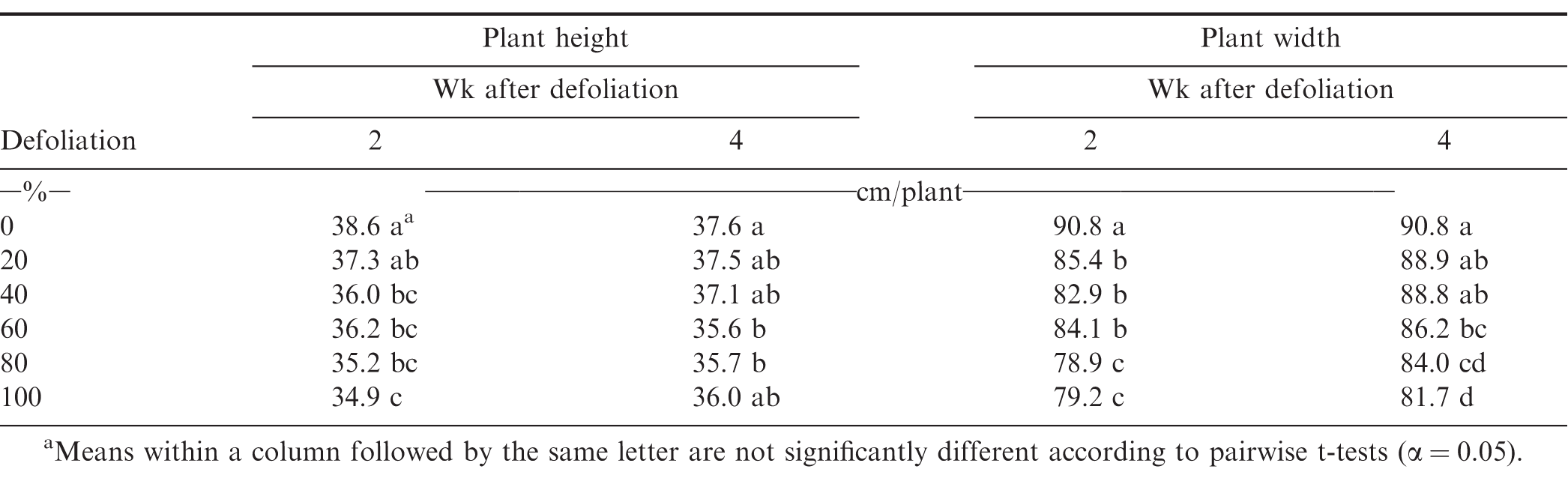
.Canopy height was reduced at all levels of defoliation compared to the non-defoliated treatment at 40 DAE. Plants receiving 80 and 100% defoliation were significantly different from those defoliated 20 and 40% two wk after defoliation occurred. However, there are no differences amongst defoliated treatments four wk after the defoliation timing. This suggests that peanut plants are able to respond similarly to severe defoliation events relative to more minor defoliation. When the defoliation event takes place early in the season, although none of the heights in defoliated plots were equal to those found in non-defoliated plots (Table 2). Canopy widths responded in a similar fashion at this timing. Plant widths were reduced when measured two wk following defoliation across all treatments when compared to the non-defoliated control. Four wk after the 40 DAE defoliation event, defoliated plant canopy widths were still significantly reduced when compared to the non-defoliated treatment, with the completely defoliated treatment seeing the largest reduction.
At 80 DAE, defoliation of 40% and greater reduced plant height when measured two wk after defoliation (Table 3). At four wk post-defoliation, plant heights in those plots receiving 40% defoliation were equal to the untreated, but defoliation of 60% or 80% still showed reductions in height. Plant widths measured two wk post-defoliation were reduced at all levels of defoliation, with defoliation of 80 and 100% being impacted more severely than defoliations of 20 to 60%. At the four wk post-defoliation, all treatments receiving defoliation of 60% or more had canopies narrower than those defoliated at 20 and 40%, and the non-defoliated control. Canopies that received defoliation of 20 and 40% were not significantly narrower than the non-defoliated canopy four wk after defoliation at 80 DAE. This data shows that peanut canopies are able to respond well to lower levels of defoliation imposed at peak pod filling growth stages.
Complete canopy closure is beneficial to production for a number of reasons. Hauser and Buchanan (1981) noted that earlier canopy closure increased weed suppression, which in turn resulted in fewer herbicide applications and increased yield. Butzler et al., (1998) reported that soil temperature was consistently 1 C warmer beneath plots that were pruned when compared to non-pruned peanut plots, with bare soil temperatures sometimes reaching 8 to 9 C warmer than non-pruned plots. These micro-climate differences were attributed to both increased sunlight penetration and air movement which increased soil temperature and moisture loss. Research conducted by Dreyer et al. (1981) reported that pod weights were lower when soil temperatures reached 37 C when compared to optimal soil temperatures of 30 and 34 C, meaning that defoliation from insects could potentially also affect yield.
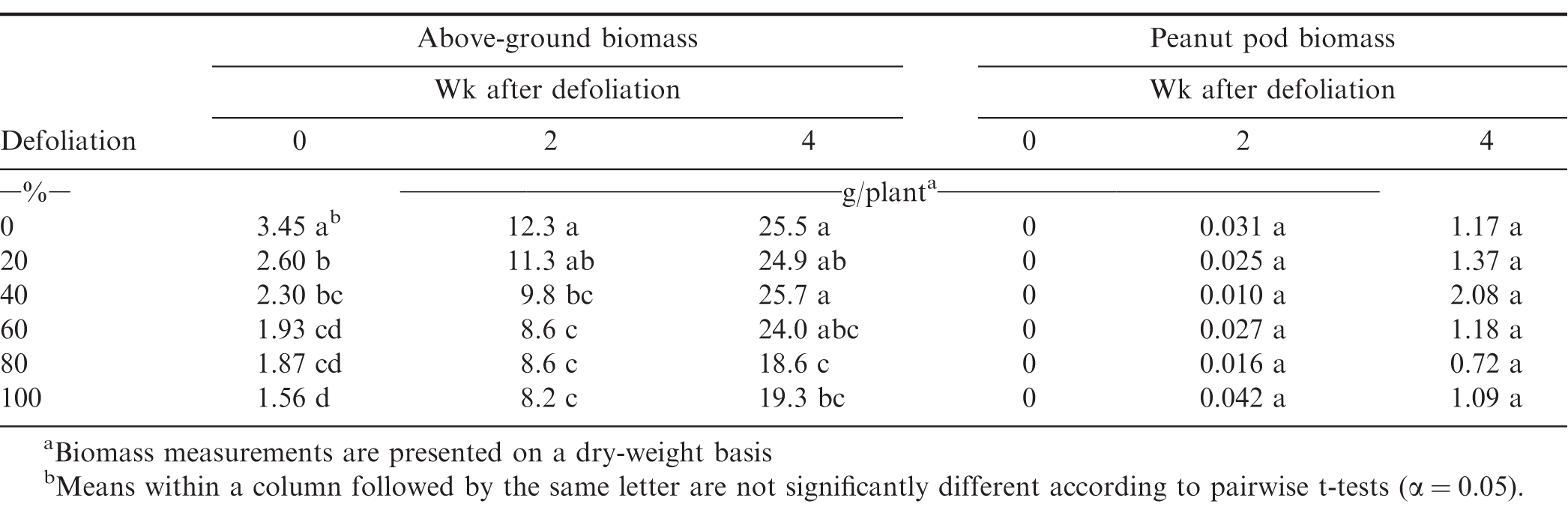
Above-ground plant and pod biomass samples provided further information on canopy and pod development following defoliation at both the 40 and 80 DAE timings. Data in Table 4 for plant and pod response to defoliation 40 DAE at three intervals - 0, 2 and 4 wk after defoliation. Immediately following defoliation, plants from defoliated treatments weighed significantly less than the non-defoliated control on a mass per plant basis. Treatments that received 40% defoliation and greater had significantly less plant biomass than the non-defoliated control two wk following defoliation. By four wk following the defoliation event, plants receiving the 20, 40, and 60% defoliation treatments were equal in size to the non-defoliated control, while plots receiving the 80 and 100% defoliation treatments had not fully recovered. There were no differences in pod weight per plant at any time following the 40 DAE defoliation, perhaps because pod set had yet to begin at the time of the defoliation event.
Immediately following the 80 DAE defoliation, plant weights from all defoliated plots were significantly less than the non-defoliated control (Table 5). The 60, 80 and 100% treatments had less biomass than the 20 and 40% treatments. Two wk after defoliation, plant weights for 60% defoliation and higher treatments were still less than those from the non-defoliated control, but were equal to 20 and 40% treatments. Four wk following defoliation, plants from plots that received either 80% or 100% defoliation were still significantly reduced in weight when compared to the control plots. Two wk following defoliation, the non-defoliated control plots had greater pod weights per plant than those receiving defoliation of 60% or greater (Table 5). Similar to the two week pod weights, the non-defoliated treatment had heavier pod weights per plant compared to 60, 80 and 100% defoliation treatments four wk after defoliation. At this timing, pods of plants receiving 80 and 100% treatments weighed less than those receiving 20 and 40% defoliation.
Economic Injury Levels
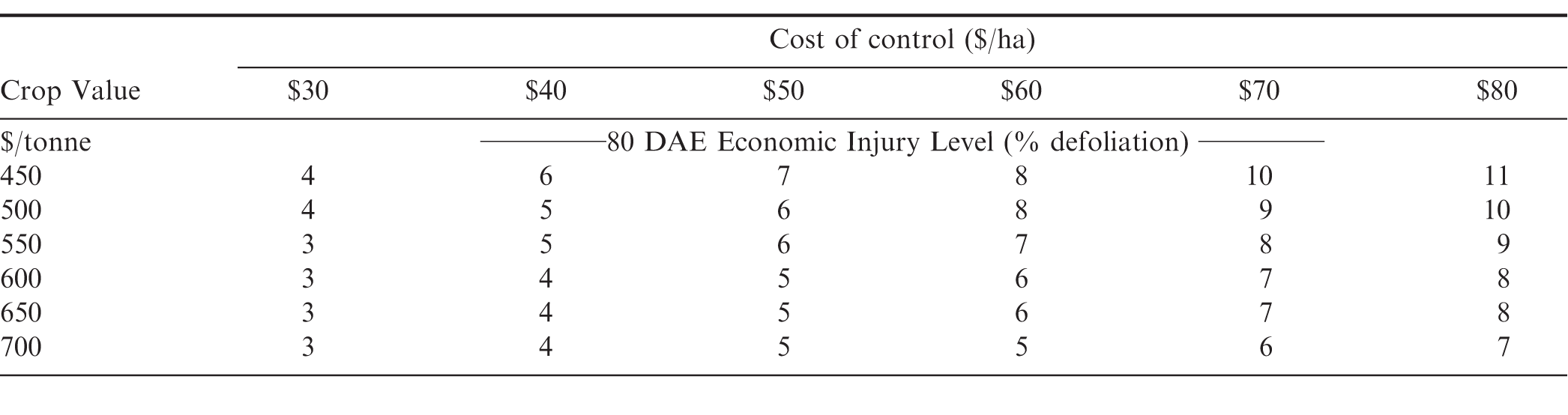
.Because defoliation had an effect on peanut pod yield during 80 DAE, EIL's for peanuts based on canopy defoliation at this time were established. Based on the expected yield losses from the regression equation at 80 DAE, EIL's were 2 to 10% depending on crop value and control costs (Table 6). These values fall well below the actual defoliation levels imposed on peanut plants in this experiment. These values are based on the assumption of a linear relationship for yield loss between 0 to 20%. To determine if that relationship is linear, more research is needed with multiple levels of defoliation between 0 and 20% defoliation during the pod filling stages.
Summary and Conclusions
Yield and economic analyses show that peanut is able to compensate for various levels of defoliation early in the growing season. While not totally eliminating all cause for concern early in this season, this finding should allow producers to remain judicious with insecticide applications at this time period. From a practical perspective, however, growers should manage defoliating insects prior to high levels (>60%) being reached, in order to reduce insect numbers as the crop enters reproductive growth. Conversely, yield and economic loss estimates following defoliation around peak pod fill (80 DAE) show the importance in minimizing defoliation during reproductive growth. Generally, insect control measures have not often been employed at defoliation levels below 10%. These data suggest that managing defoliating caterpillars at lower levels than previously thought may be warranted.
A limitation of this work is that determining defoliation percentages in peanut fields is often difficult for consultants, growers, and Extension personnel as each person's opinion is subjective. In addition, data from this study represent defoliation levels that occur only once at one particular time, and do not represent the likelihood of previous or future damage that occurs over time. Lastly, figures obtained from the EIL analysis represent peanuts yielding over 6,000 kg/ha. Yield projections must be considered when using this analysis, as peanuts with a higher potential value may be more sensitive to defoliation, while a crop with a lower potential may be able to withstand more damage before an economic loss is reached.
Future research is needed to help validate these EIL's. Economic thresholds need to be developed using defoliation from insects as well as looking at other times in the crop growth cycle that are outside the scope of this research. Understanding crop growth stages, crop price, control costs, and yield potential is imperative for those making management decisions in peanut. These data along with these careful considerations will allow for a more efficient integrated pest management strategy to be implemented in Mississippi, as well as the rest of the peanut producing belt.
Acknowledgements
Many thanks to the field crews of Drs. Darrin Dodds, Trent Irby, and Erick Larson. Appreciation is extended to Tanner Ainsworth, Jake Dupree, Josh Rusche, Nolan Stapleton and Logan Yancey for their dedication to this project as well. We are also grateful to the Mississippi Peanut Promotion Board and Mississippi Agriculture and Forestry Experiment Station for financial support.
Literature Cited
M., Adomu, P. V. V., Prasad, K. J., Boote, and J. Detongnon. (2005). . Disease assessment methods and their use in simulating growth and yield of peanut crops affected by leafspot disease. . Annals of Applied Biology . 146: 469- 479.
K. J., Boote, J. W., Jones, G. H., Smerage, C. S., Barfield, and R. D. Berger. (1980). . Photosynthesis of Peanut Canopies as Affected by Leafspot and Artificial Defoliation. . Agronomy Journal . 72: 247- 252.
G., Bourgeois, and K. J. Boote. (1992). . Leaflet and canopy photosynthesis of peanut affected by late leaf spot. . Agronomy Journal . 84: 359- 366.
G., Bourgeois, K. J., Boote, and R. H. Berger. (1991). . Growth, development, yield and seed quality of Florunner peanut affected by late leaf spot. . Peanut Sci . 18: 137- 143.
W. D. Branch, (2007). . Registration of 'Georgia-06G' peanut. . Journal of Plant Registrations . 1: 20.
T. M., Butzler, J., Bailey, and M. K. Beute. (1998). . Integrated management of sclerotinia blight in peanut: Utilizing canopy morphology, mechanical pruning, and fungicide timing. . Plant Dis . 82: 1312- 1318.
W.A., Court, R.C., Roy, and J.G. Hendel. (1984). . Effect of harvest date on agronomic and chemical characteristics of Ontario peanut. . Can. J. Plant. Sci . 64: 521- 528.
S. S., Deitz, J. W., Chapin, and P. H. Adler. (1992). . Feeding-site preference of fall armyworm, corn earworm, and granulate cutworm (Lepidoptera: Noctuidae) on Florunner peanut. . Peanut Sci . 19: 63- 68.
J., Dreyer, W. G., Duncan, and D. E. McCloud. (1981). . Fruit temperature, growth rates, and yield of peanuts. . Crop Sc . 21: 686- 688
J. B., Endan, J. H., Young, and M. A. Awal. (2006). . Effect of hand defoliation on peanut growth. . Asian Journal of Plant Science . 5: 281- 286.
J. W., Garner, and R. E. Lynch. (1981). . Fall armyworm leaf consumption and development on Florunner peanuts. . J. Econ. Entomol . 74: 191- 193.
E. W., Hauser, and G. A. Buchanan. (1981). . Influence of row spacing, seeding rates and herbicide systems on the competiveness and yield of peanuts. . Peanut Sci . 8: 74- 81.
J. W., Jones, C. S., Barfield, K. J., Boote, G. H., Smerage, and J. Mangold. (1982). . Photosynthetic recovery of peanuts to defoliation at various growth stages. . Crop Sci . 22: 741- 746.
Kemerait, R., Culbreath, A., Prostko, E., Brenneman, T., Smith, N., Tubbs, S., Rajagolpalbabu, S., Abney, M., Monfort, S. 2015. Peanut Rx: Minimizing diseases of peanuts in the Southeastern United States. p. 50- 57. In: E.P. Prostko. (ed.) 2015 Peanut Update. Spec. Pub. CSS-10-0125. Univ. of Georgia Coop. Ext. Serv., Athens, GA.
D.A., Knauft, A.J., Norden, and D.W. Gorbet. (1986). . The effect of three digging dates on oil quality, yield, and grade of five peanut genotypes grown without leafspot control. . Peanut Sci . 13: 86- 89.
R. E. Lynch, (1996). . Peanut fungicides: effect on survival and development of the corn earworm, fall armyworm, and velvetbean caterpillar. . Peanut Sci . 23: 116- 123.
N. A., Minton, A. S., Csinos, R. E., Lynch, and T. B. Brenneman. (1991). . Effects of two cropping and two tillage systems and pesticides on peanut pest management. . Peanut Sci . 18: 41- 46.
R.W., Mozingo, T.A., Coffelt, and F.S. Wright. (1991). . The influence of planting and digging dates on yield, value, and grade of four Virginia-type peanut cultivars. . Peanut Sci . 18: 55- 62.
Oldham, L. 2017. Micronutrients in Crop Production. Mississippi State University Extension Service. MAFES publication No. IS1038.
L. N., Owen, A. L., Catchot, F. R., Musser, J., Gore, D. C., Cook, R., Jackson, and C. Allen. (2013). . Impact of defoliation on yield of group IV soybeans in Mississippi. . Crop Protection . 54: 206- 212.
K. P., Pedigo, S. H., Hutchins, and L. G. Higley. (1986). . Economic injury levels in theory and practice. . Annu. Rev. Entomol . 31: 341- 368.
K. V., Pixley, K. J., Boote, F. M., Shokes, and D. W. Gorbet. (1990). . Growth and partitioning characteristics of four peanut genotypes differing in resistance to late leafspot. . Crop Sci . 30: 796- 804b.
H. T. Stalker, and W. V. Campbell. (1983). . Resistance of wild species of peanut to an insect complex. . Peanut Sci . 10: 30- 33.
J. W., Todd, R. M., Beach, and W. D. Branch. (1991). . Resistance in eight peanut genotypes to foliar feeding of fall armyworm, velvetbean caterpillar, and corn earworm. . Peanut Sci . 18: 38- 40.
USDA-NRCS. 2016. Official soil series descriptions. [Online]. Available at http://soilseries.sc.egov.usda.gov/osdname.asp (verified 17 February 2016). USDA Nat. Resour. Conserv. Serv., Washington D.C.
Williams, E. J. and J. S. Drexler. 1981. A non-destructive method for determining peanut pod maturity. Peanut Sci. 8: 134- 141
Notes
- Dept. of Plant and Soil Sciences, Mississippi State University, Mississippi State, MS. [^]
- Dept. of Biochemistry, Entomology and Plant Pathology, Mississippi State University, Stoneville, MS. [^]
- Dept. of Biochemistry, Entomology and Plant Pathology, Mississippi State University, Mississippi State, MS. [^]
- Dept. of Plant and Soil Sciences, Mississippi State University, Stoneville, MS. [^] *Corresponding author's E-mail: JGore@drec.msstate.edu
Author Affiliations