Introduction
There is interest in diversifying peanut production in the southeastern U. S. to certified organic production to meet the consumer demand for organic peanut butter. Previously, the majority of the organic peanut production in the United States was in western Texas and eastern New Mexico (Guerena and Adam, 2008; USDA-ERS, 2013). Peanut production in this region is at elevations >1200 m and the growing season is short. As a result, organic peanut grown in that region are primarily Valencia peanut, which have a shorter growing season and lower yield potential than runner-type peanut. Runner peanut have superior yield, roasting qualities, and flavor making them the primary component of conventional peanut butter. Furthermore, runner peanut are commonly grown in the southeastern U. S. A persistent criticism of increased demand of organic agronomic commodities in the U. S. has been that much of resulting supply has been met by imports (Gianessi and Reigner, 2005), not by increased U. S. production. In the context of consumer demand for organic peanut butter, criticism could be deflected by increases in certified organic peanut production in the southeastern U. S. using runner-type peanut.
Difficulties in weed management in certified organic crops remains a nationwide priority for growers (Organic Farming Research Foundation, 2016), including peanut. Previous research on weed management in organic peanut has shown that herbicides derived from natural products (clove oil, acetic acid plus citric acid, and corn gluten) are generally ineffective (Johnson and Mullinix, 2008; Johnson et al., 2013). Propane flaming is a weed control option in certified organic systems, but in peanut trials propane flaming did not control annual grasses and the flaming implement was persistently difficult to use (Johnson and Mullinix, 2008).
Mechanical weed control has been consistently the most effective approach in organic peanut (Johnson et al., 2012a, 2012b; Wann 2011). Cultivation is a form of mechanical weed control that uses tillage tools to remove, cut, or disrupt weed growth in a manner that does not injure the crop. Sweep cultivation is a common form of mechanical weed control that cuts or buries seedling weeds using uniquely shaped blades (sweeps) that slice just under the soil surface in the area between the crop rows. However, weeds present in the crop row are not affected by the sweeps.
The tine weeder is a cultivation implement of European origin that operates on a different principle compared to sweeps (Colquhoun and Bellinder, 1997). The tine weeder uses multiple rows of closely spaced rods of spring steel (tines). The tine weeder uses vibratory action of the tines to displace weed seedlings (Ascard and Fogelberg, 2008; Melander and Rasmussen, 2001). In laboratory studies, a single cultivation using an implement conceptually similar to a tine weeder uprooted 51% of the emerged weed seedlings in a coarse-textured soil (Kurstjens et al., 2000). Tine weeders are very effective on annual grasses (Johnson et al., 2012a, 2012b) and seedling dicot weeds, with maximum performance when weed seed have germinated, but seedlings not emerged. Gunsolus (1990) referred to this stage of weed development as 'weeds in the white', with the white portion being the rapidly developing hypocotyl from a very young weed seedling. The tine weeder has shown great potential for use in organic peanut with the crop tolerating multiple early-season cultivations at weekly intervals (Johnson et al., 2012a; Wann et al., 2011).
The tolerance of peanut to aggressive cultivation with a tine weeder may be related to the evolutionary development of the genus. Arachis spp. evolved in South America, with A. hypogaea believed to have originated in Bolivia (Hammons, 1973). In this region, naturally occurring Arachis spp. are heavily trod and grazed by cattle. Under this selection pressure, Arachis spp. developed tolerance to that type of physical abuse and plants routinely survive. Despite the innate tolerance of Arachis spp. to abuse, subterranean fruiting of peanut can be disrupted by sweep cultivation that may unintentionally sever pegs and pods. Boyle (1952, 1956, 1961) and Mixon (1963) correlated the relationship between southern stem rot (Sclerotium rolfsii Sacc.) incidence with sweep cultivation that placed soil containing fungal sclerotia onto the lateral branches of peanut. Those findings were the basis for the long-standing peanut production recommendations to adjust sweep cultivators to minimize soil movement onto the peanut plant; colloquially termed 'non-dirting' cultivation. Since a tine weeder operates on a different principle than a sweep cultivator and does not displace soil to the same degree as sweeps, the effect of tine weeding on incidence of soil-borne diseases is unknown.
In earlier weed management trials in organic peanut, plots were cultivated as many as six times at weekly intervals with the tine weeder, sweeps, or the PTO-powered brush-hoe (Johnson et al., 2012a, 2012b; Wann 2011). No insecticides were applied for thrips (Thysanoptera: Thripidae), foliage-feeding Lepidopterans, or pod-feeding insects. Similarly, no conventional fungicides were applied for foliar or soil-borne disease control. While organic peanut in these trials were occasionally infested common insect pests, damage was never severe or judged to be yield limiting. Similar observations were made with peanut diseases; peanut were infected but epidemics were not severe.
Overall, there appears to be a distinct discrepancy between the long-standing peanut production philosophy successfully used by conventional peanut growers and the surprisingly effective systems used to manage organic peanut in the previous research trials. It was hypothesized that components in the organic peanut production system could be integrated into conventional peanut production to reduce inputs. Therefore, trials were initiated in 2012 to determine the interactive relationships among systems to manage weeds, insects, and diseases in organic peanut production.
Materials and Methods
Irrigated research trials were conducted at the University of Georgia Ponder Research Farm near Ty Ty, GA (31.510884°, -83.645913°) for three seasons from 2012 through 2014. The soil was a Tifton loamy sand (fine-loamy, kaolinitic, thermic Plinthic Kandiudults) with 88% sand, 6% silt, and 6% clay and 0.2% organic matter. The soil at this location is representative of soils in the southeastern U. S. peanut producing region and naturally infested with weeds that are common to the region.
The experimental design was a 3 x 2 x 3 factorial arrangement of treatments in a randomized complete block design with four replications. Treatments were all possible combinations of three levels of weed control, two levels of insect control, and three levels of fungal disease control. Weed control treatments were cultivation six times with a tine weeder (Aerostar Tined Weeder; Einböck GmbH & CoKG; Schatzdorf 7; 4751 Dorf an der Pram; Austria) at weekly intervals beginning four days after planting, weed-free with handweeding as-needed, and a weedy control. The tine weeder used in these trials tilled a swath 1.8 m wide. Tension of tines located immediately above the crop row was adjusted by the use of mechanical hangers, each having several hooks on which tines over the row were lifted to prevent crop damage. Gauge wheels were attached to the front of the tine weeder to add lateral stability of the implement.
Insect control was spinosad (Entrust® Naturalyte® Insect Control; Dow AgroSciences, LLC; 9330 Zionsville Road; Indianapolis, IN 46268-1189) (0.11 kg ai/ha) and a nontreated control. This formulation of spinosad is approved by the Organic Materials Review Institute (OMRI) for insect control in certified organic crop production, including peanut. Spinosad was applied twice; two weeks and four weeks after peanut emergence. Fungicide regimes were four foliar sprays of a combination of OMRI approved fungicides, a commonly used conventional fungicide for peanut disease control, and a nontreated control. The fungicide regime for certified organic crop production was cuprous oxide (2.2 kg/ha) (Nordox® 75DF; NORDOX Industrier AS; c/o Monterey Ag Resources; P. O. Box 35000; Fresno, CA 93745-5000) plus sulfur (9.0 kg/ha) (Microthiol® Disperss®; United Phosphorous Inc.; 630 Freedom Business Center, Suite 402; King of Prussia, PA 19406) (abbreviated "Cu+S"). The conventional fungicide chosen was azoxystrobin (0.45 kg ai/ ha) (Abound® Flowable; Syngenta Crop Protection, LLC; P. O. Box 18300; Greensboro, NC 27419). Both spray regimes were initiated in early-June each year and applied at three week intervals.
Plots were 1.8 m wide and 6.1 m in length. Land preparation included moldboard plowing in early May and seedbed conditioning using a power-tiller and field cultivator. Non-treated 'Georgia-04S' (Branch 2005) peanut were seeded mid-May each year in rows spaced 91 cm apart using vacuum planters (Monosem Inc.; 1001 Blake St.; Edwardsville, KS 66111) that placed seeds at a density of 20 seed/m, to a depth of 6.4 cm. The GA04-S peanut cultivar was used in these trials since it has excellent seedling vigor, rapid early season growth, and resistance to spotted wilt (tomato spotted wilt Tospovirus) (Branch 2005). This cultivar performed well in previous organic peanut research trials (Johnson et al., 2012a, 2012b).
Visual estimates of weed control compared to the nontreated control were assessed in mid-season using a scale of 0 to 100 where 0 = absolutely no weed control and 100 = complete weed control. Thrips feeding damage was monitored throughout the early season. Immediately prior to harvest, incidence of early leafspot (Cercospora arachidicola Hori), southern stem rot, and spotted wilt infection was determined. Incidence of early leafspot was evaluated using the Florida 1-to-10 intensity scale, where 1 = no leafspot disease or defoliation and 10 = plants completely defoliated or dead (Chiteka et al., 1988). Stem rot and spotted wilt incidence were measured by one or more plants affected by the diseases in a 30-cm section of row. Percent incidence of either disease were determined by number of 30-cm sections of disease loci divided by total number of 30 cm sections in the plot. Peanut yields were obtained by pre-harvest mowing to cut tops of tall weeds, digging/inverting, air-curing, and combining peanut from the entire plot using commercial two-row equipment. Yield samples were mechanically cleaned to remove foreign material, particularly weed biomass, with yields reported as cleaned farmer stock peanut.
Data were analyzed using a mixed-model analysis. Degrees of freedom were partitioned to test singularly and in combination the effects of weed control, insect control, and fungal disease control on the parameters measured. Means were separated using Fisher's LSD test (P<0.05).
Results and Discussion
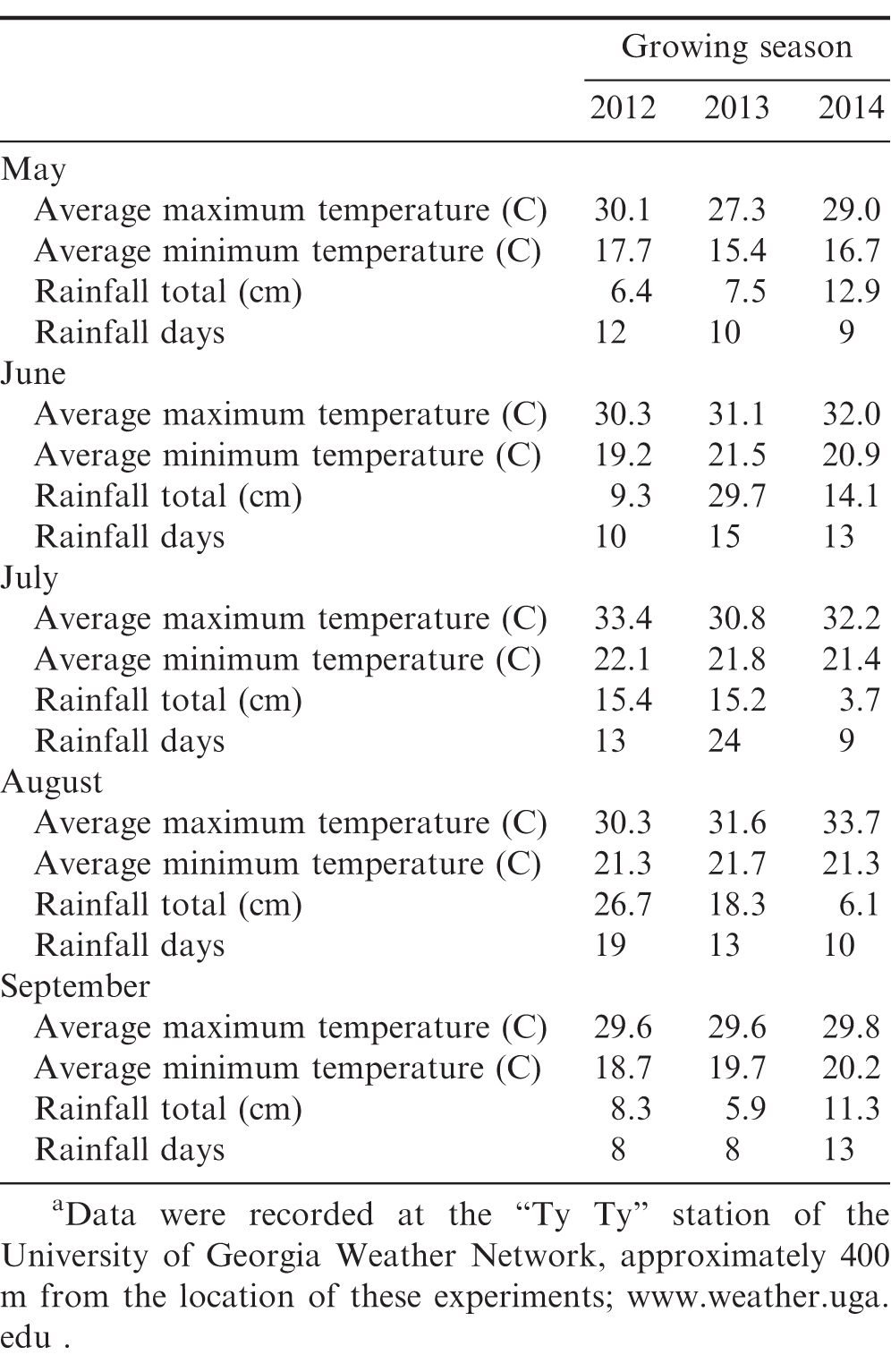
There were large differences in growing conditions among years that affected weed control performance with the tine weeder, with excessive rainfall during May and June (when initial cultivations were scheduled) in 2014 (Table 1). In addition, all field operations ceased for 17 days in October 2013 due to the Federal Government shut-down and which unfortunately coincided with peanut harvest and the delay reduced peanut yield potential. Therefore, all data were analyzed by year. Analysis of variance indicated no significant interactions among weed control, insect control, and fungal disease control for all parameters. Therefore, all data are presented as main effects.
Weed control
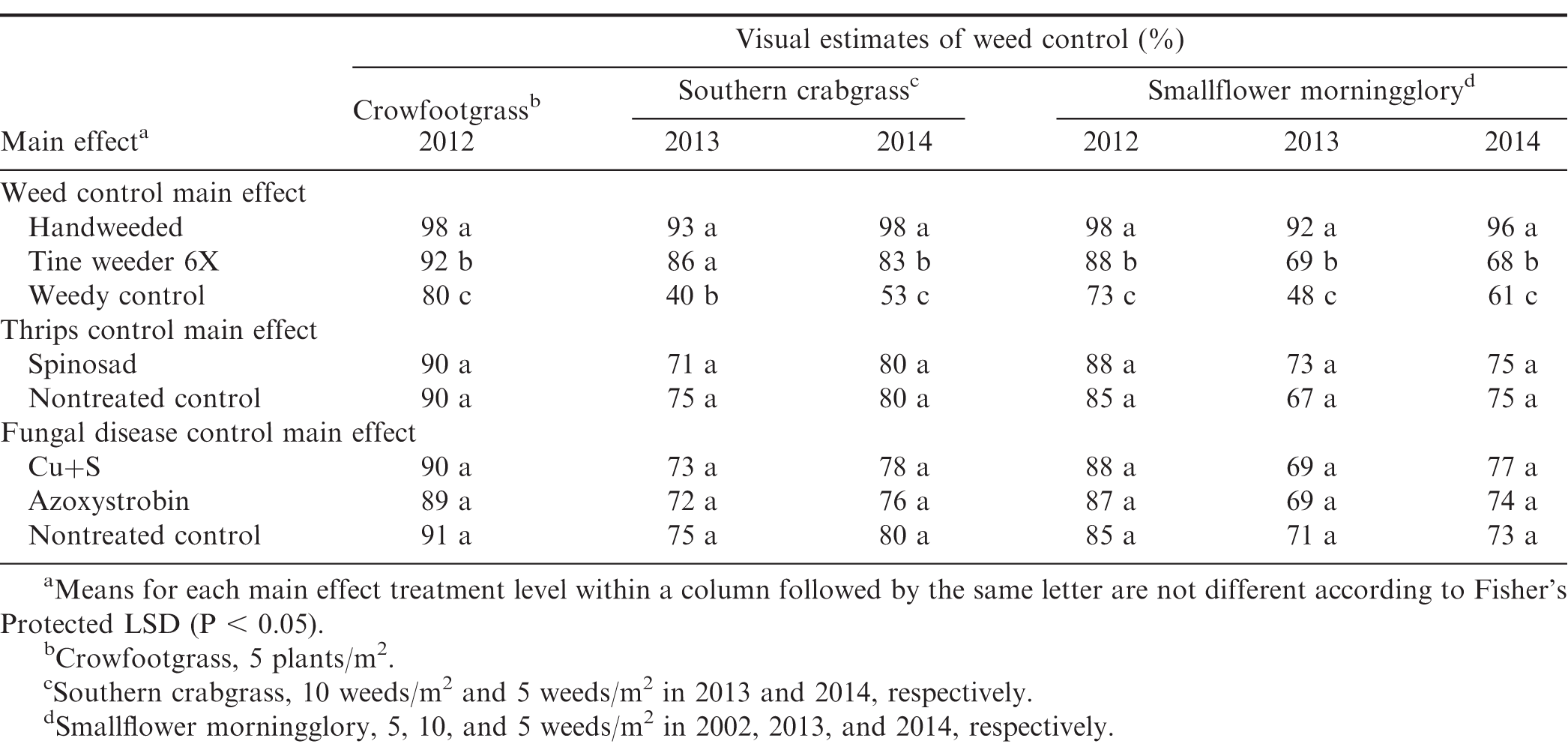
Crowfootgrass [Dactyloctenium aegyptium (L.) Willd.] (2012 density of 5 plants/m2) and southern crabgrass [Digitaria ciliaris (Retz.) Koeler] (2013 density of 10 plants/m2 and 2014 density of 5 plants/m2) were the annual grasses present during these trials. Crowfootgrass control in 2012 was improved with season-long handweeding compared to tine weeding 6X, with both handweeding and tine weeding 6X controlling crowfootgrass better than the weedy control (Table 2). Southern crabgrass control with handweeding and tine weeding 6X were similar in 2013, but handweeding was more effective than tine weeding 6X in 2014.
Smallflower morningglory [Jacquemontia tamnifolia (L.) Griseb.] was present every year of the trial at baseline densities of 5, 10, and 5 plants/m2 in 2012, 2013, and 2014, respectively. In each year of the study, smallflower morningglory control was greater when organic peanut plots were handweeded season-long compared to cultivation with a tine weeder 6X (Table 2). It is worth noting the variability in smallflower morningglory control with the tine weeder among the three years with visual control ranging from 68 to 88%. This was likely due to frequent rainfall (12.9 cm in nine rainfall events) in May 2014 (Table 1) that delayed the early cultivations and caused soils to be wet, which affected tine weeder performance. This highlights the heightened risk of dependence on cultivation for weed control in organic peanut production.
Neither insect control nor fungal disease control affected annual grasses or smallflower morningglory for the duration of the study (Table 2). This was predictable since there are no reports of these pesticides having weed control properties.
Thrips control
From early-season through mid-season, plots were monitored for indications of insect feeding, specifically thrips. There were no differences noted among treatments in peanut response to thrips feeding, and by mid-season evidence of thrips feeding was no long apparent (data not presented).
Peanut disease incidence
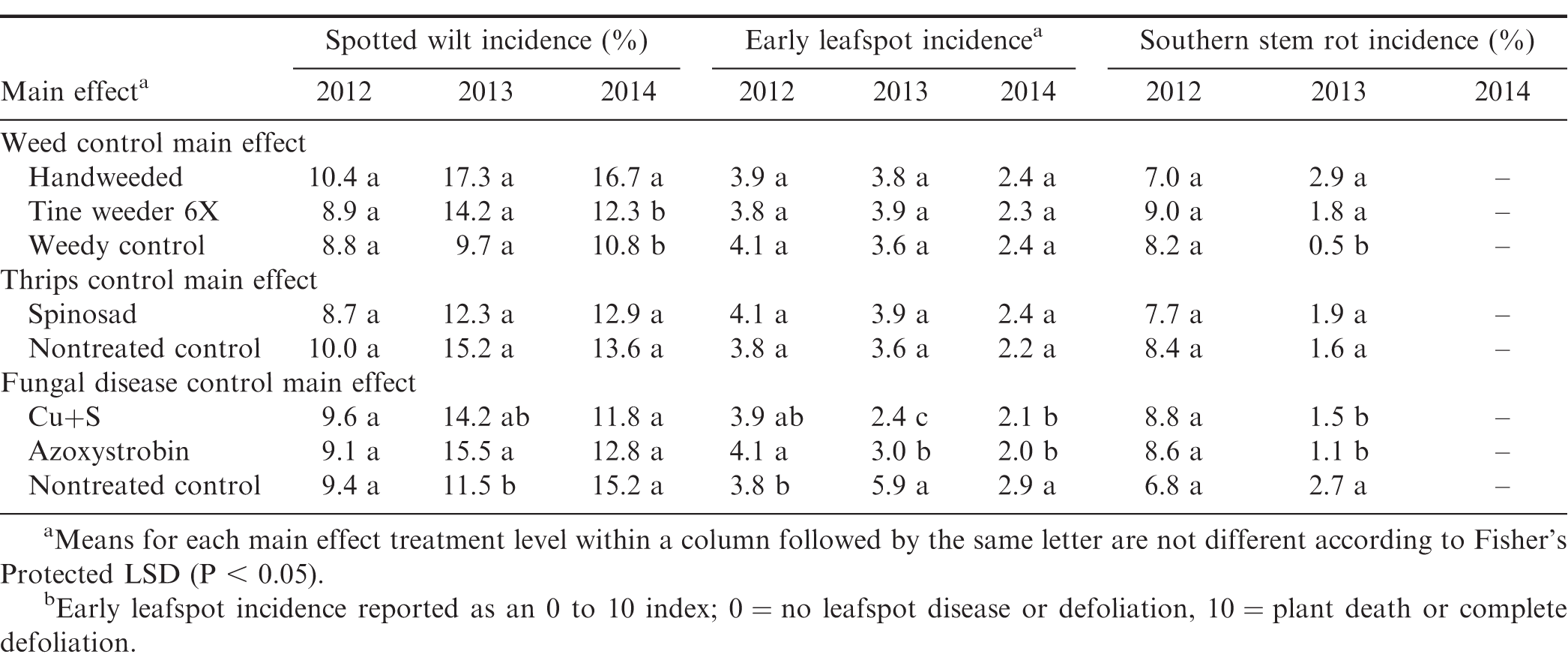
Incidence of spotted wilt was not affected by weed control two years out of three (Table 3). In 2014, spotted wilt incidence was greater in handweeded plots compared to plots cultivated with a tine weeder and the weedy control. We speculate that handweeded peanut were more attractive to thrips vectors compared to weedy peanut.
Thrips control with spinosad did not affect incidence of spotted wilt (Table 3). Spotted wilt is vectored by thrips species. However, previous research has shown that controlling thrips with insecticides is not a major factor affecting incidence of spotted wilt (Todd et al. 1996; Marasigan et al. 2016), and these results with spinosad are in agreement.
Fungal disease control did not affect incidence of spotted wilt two years out of three (Table 3). In 2013, spotted wilt incidence was greater when peanut were treated with azoxystrobin compared to the nontreated control. The reason for this occurrence is unknown.
Early leafspot incidence was not affected by weed control in any year of the study (Table 3). Similarly, insect control with spinosad had no effect on early leafspot incidence. In both cases, the lack of a treatment effect on early leafspot incidence was expected. Early leafspot response to fungal disease control varied among all three years (Table 3). In 2012, the greatest incidence of early leafspot was in plots treated with azoxystrobin compared to the nontreated control. The greatest early leafspot incidence in 2013 was in the nontreated control compared to either fungicide treatment. However, early leafspot incidence in 2013 was greater in plots treated with azoxystrobin compared to Cu+S. In 2014, early leafspot incidence was greatest in the nontreated control compared to either fungicide treatment. However, there was no difference in early leafspot incidence between applications of azoxystrobin or Cu+S. Cantonwine et al. (2008) reported fungicides containing copper sulfate and copper hydroxide provided effective control of early leafspot in peanut varieties with disease tolerance and those fungicides protected peanut yields. Our results using Cu+S are in agreement.
Southern stem rot was present in all years of the study, but not at levels high enough to report in 2014. Weed control did not affect incidence of stem rot in 2012 (Table 3). In 2013, the lowest levels of stem rot were in the weedy control, with no difference in stem rot incidence between the handweeded and tine-weeded plots. As expected, spinosad treatment had no effect on incidence of stem rot each year of the study. In 2012, incidence of stem rot was not affected by any of the fungicide treatments. In 2013, stem rot incidence was greatest in the nontreated control compared to either Cu+S or azoxystrobin, with no difference in stem rot incidence between Cu+S or azoxystrobin treated peanut.
Peanut yield
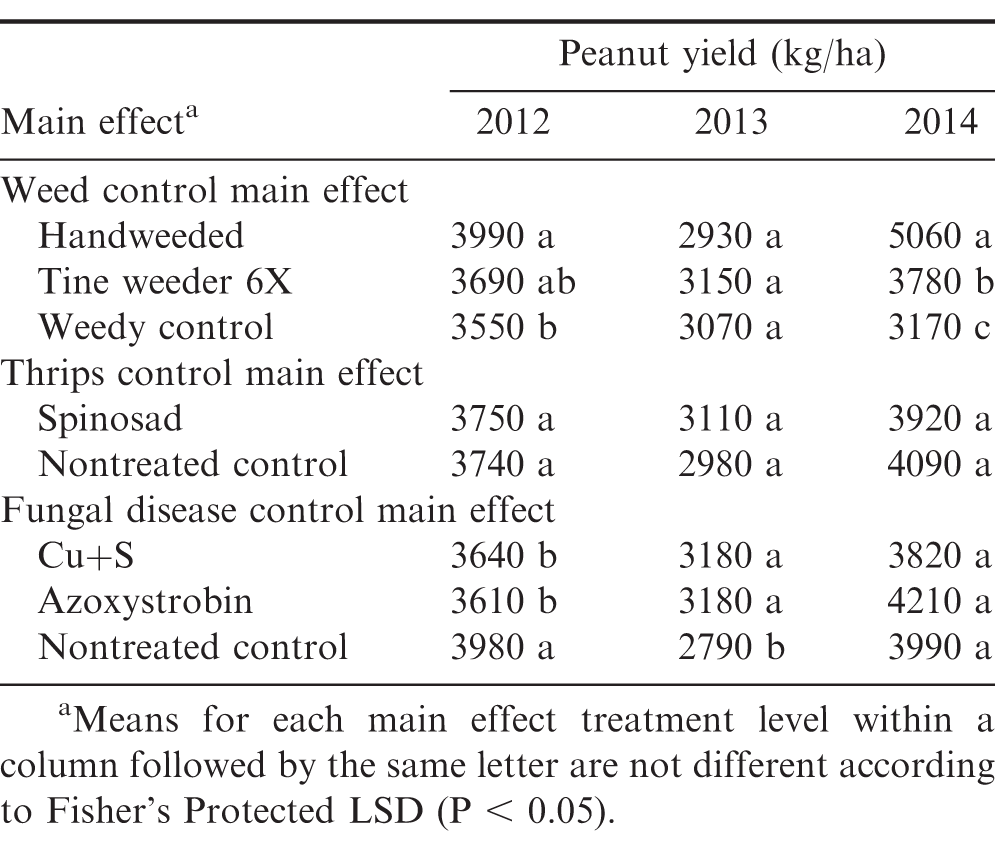
Peanut yield response to weed control varied among the three years of the study (Table 4). In 2012, peanut yields were lower in the nontreated (weedy) control compared to the handweeded (weed free), with no yield differences when organic peanut were handweeded or cultivated with a tine weeder 6X. In 2013, peanut yields did not differ among any of the weed control treatments. Peanut yields in 2014 were lowest in the nontreated control and greatest in the handweed plots. Plots cultivated with a tine weeder 6X in 2014 had peanut yields reduced by 25% compared to the handweeded plots. Insect control using spinosad had no effect on peanut yield in each year of the study. Peanut yield response to fungal disease control varied among the three years of the study. In 2012, peanut yields were higher in the nontreated control compared to fungal disease control using either Cu+S or azoxystrobin. In contrast, 2013 peanut yields were lowest in the nontreated control compared to either Cu+S or azoxystrobin. In 2014, there were no differences in peanut yield among the fungal disease control treatments.
The lack of interactions among weed control, insect control, and fungal disease control for any of the parameters measured presents an interesting perspective of organic peanut production. Many of the long-standing recommendations about avoiding soil movement onto the crown of the peanut plant are based on landmark studies that clearly showed a correlation and biological explanation between cultivation and incidence of stem rot of peanut (Boyle 1952, 1956, 1961 and Mixon 1963). In our studies, that correlation was not clearly evident. A probable factor in our results was the land-use history at the research sites which had corn rotated with peanut research on an annual basis and perhaps that rotation maintained low levels of disease inoculum in the soil, thus lessening stem rot incidence in our trials. Another possible factor was peanut cultivar. While the disease susceptibility of peanut cultivars used in the previous studies is unknown, our results suggest that the Georgia-04S cultivar has more stem rot tolerance than originally thought.
Of equal importance, the incidence of early leafspot in peanut treated with a fungicide regime suitable for certified organic peanut production (Cu+S) was generally similar to treatment with the conventional fungicide azoxystrobin. We acknowledge that there are many fungicide regimes commonly used in conventional peanut production and some may be more effective than azoxystrobin in controlling early leafspot. However, the performance of Cu+S in managing early leafspot in organic Georgia-04S peanut suggests similar results in conventional peanut production. Similarly, the lack of any significant effect from spinosad treatment on any parameter, including thrips-vectored spotted wilt, indicates that insecticides for thrips control are not universally needed for organic peanut production and that effect may have similar implications in conventional peanut production as well.
Finally, these results indicate that weed control in organic peanut production using intensive cultivation with a tine weeder has limitations, particularly in seasons where wet soils from frequent rainfall may impede implement performance. Overall, cultivation with a tine weeder was not as effective as season-long handweeding. However, organic peanut cultivated with a tine weeder 6X yielded similar to handweeded peanut two years of three. While weed control was greater when peanut were handweeded compared to plots cultivated with a tine weeder, the weeds escaping cultivation did not consistently reduce organic peanut yield. The weed control results also illustrate the heightened weed control risk that organic peanut growers face by relying on cultivation for weed control. In our trials, wet soils from frequent rainfall events in 2014 early in the cultivation period affected both the performance and scheduling of tine weeding. For organic peanut growers who have limited weed control options, these delays can cause numerous weed escapes and could be catastrophic when scaled-up to commercial scale production.
Summary and Conclusions
While these results directly affect pest management decisions in organic peanut production, there are possible implications for conventional peanut production. It can be extrapolated that under certain conditions insect and fungal disease control inputs in conventional peanut production can be reduced without significant yield effects and provide savings to conventional peanut growers. To adequately address the topic of reduced pest control inputs in conventional peanut production, further research using structured trials is needed across an array of growing conditions, along with demonstration on large-scale peanut plantings.
Acknowledgements
We acknowledge the contributions of Daniel R. Evarts, Terrell W. Jones, and Danielle B. Simmons whose technical skills made these studies possible.
Literature Cited
J. Ascard, and F. Fogelberg (2008). Mechanical in-row weed control in transplanted and direct-sown bulb onions. Biol. Agr. Hort 25: 235- 251.
L. W. Boyle, (1952). Factors to be integrated in the control of southern blight on peanut. Phytopathol 42: 282.
L. W. Boyle, (1956). Fundamental concepts in the development of control measures for southern blight and root rot on peanut. Plant Dis. Rep 40: 661- 665.
L. W. Boyle, (1961). The ecology of Sclerotium rolfsii with emphasis on the role of saprophytic media. Phytopathol 51: 117- 119.
W. D. Branch, (2005). Registration of 'Georgia 04S' peanut. Crop Sci 45: 1653- 1654.
Cantonwine, E. G., A. K. Culbreath, B. Shew, and M. A. Boudreau 2008 Efficacy of organically acceptable fungicides for management of early and late leaf spot diseases on partially resistant peanut cultivars Plant Health Progress https://www.plantmanagementnetwork.org/pub/php/research/2008/peanut/ (Accessed 24 July 2017).
Z. A., Chiteka, D. W. Gorbet, F. M. Shokes, T. A. Kucharek, and D. A. Knauft (1988). Components of resistance to late leaf spots in peanut. I. Levels of variability - implications for selection. Peanut Sci 15: 25- 30.
Colquhoun, J.and R. Bellinder 1997 New Cultivation Tools for Mechanical Weed Control in Vegetables. IPM Fact Sheet 102FSNCT Cornell Univ. Coop. Ext. Serv. Ithaca, NY.
Gianessi, L.and N. P. Reigner 2005 The outsourcing of organic crop production - research brief number 2 The Crop Life Foundation (http://s3.amazonaws.com/zanran_storage/www.croplifefoundation.org/ContentPages/43735339.pdf . (Accessed 26 July 2017).
Guerena, M.and K. Adam 2008 Peanuts: Organic Production National Sustainable Agriculture Information Service - National Center for Appropriate Technology (NCAT) https://attra.ncat.org/attra-pub/summaries/summary.php?pub=95 (Accessed 26 July 2017).
J. L. Gunsolus, (1990). Mechanical and cultural weed control in corn and soybeans. Am. J. Alter Agric 5: 114- 119.
Hammons, R. O. 1973 Early history and origin of the peanut Pp 17- 45 Peanuts - Culture and Uses Amer. Peanut Res. Educ. Assoc. Stillwater, OK.
W. C., Johnson, and B. G. Mullinix, (2008). Potential weed management systems for organic peanut production. Peanut Sci 35: 67- 72.
W. C., Johnson, M. A. Boudreau, and J. W. Davis (2013). Combinations of corn gluten meal, clove oil, and sweep cultivation are ineffective for weed control in organic peanut production. Weed Technol 27: 417- 421.
Johnson, W. C., III, M. A. Boudreau, and J. W. Davis 2012 a Implements and cultivation frequency to improve In-row weed control in organic peanut production Weed Technol 26: 334- 340.
Johnson, W. C., III, M. A. Boudreau, and J. W. Davis 2012 b Cultural practices to improve in-row weed control with cultivation in organic peanut production Weed Technol 26: 718- 723.
D. A., Kurstjens, U. D. Perdok, and D. Goense (2000). Selective uprooting by weed harrowing on sandy soils. Weed Res 40: 431- 447.
K., Marasigan, M. Toews, R. Kemerait, M. R. Abney, A. Culbreath, and R. Srinivasan (2016). Evaluation of alternatives to carbamate and organophosphate insecticides against thrips and tomato spotted wilt virus in peanut production. J. Econ. Entomol 109: 544- 557.
B. Melander, and G. Rasmussen (2001). Effects of cultural methods and physical weed control on intra-row weed numbers, manual weeding, and marketable yield in direct-sown leek and bulb onion. Weed Res 41: 491- 508.
Mixon, A. C. 1963 Effects of deep turning and non-dirting cultivation on bunch and runner peanuts. Auburn Univ. Agri., Exp. Stn. Bull. 344.
Organic Farming Research Foundation 2016 National Organic Research Agenda - Outcomes and Recommendations from the 2015 National Organic Farmer Survey and Listening Sessions http://ofrf.org/sites/ofrf.org/files/staff/NORA_2016_final9_28.pdf (last accessed 26 July 2017).
J. W., Todd, A. K. Culbreath, and S. L. Brown (1996). Dynamics of vector populations and progress of spotted wilt disease relative to insecticide use in peanuts. Acta Horti 431: 483- 490.
USDA - Economic Research Service 2013 Organic production for 2011 https://www.ers.usda.gov/data-products/organic-production/ . (last accessed 26 July 2017).
D. Q. Wann, R. S. Tubbs, W. C. Johnson, A. R. Smith, N. B. Smith, and A. K. Culbreath (2011). Cultivation frequency and duration effects on productivity and economics of peanut in organic management. Peanut Sci 38: 101- 110.
Notes
- Research Agronomist, USDA-ARS, Tifton Campus, P. O. Box 748, Tifton, GA 31793-0748; Professor, Dept. of Plant Pathology, Univ. of Georgia, Tifton Campus, Tifton, GA 31793; Res. Statistician, Univ. of Georgia, Tifton Campus, Tifton, GA 31793. [^] *Corresponding author's email: Carroll.Johnson@ars.usda.gov .
Author Affiliations