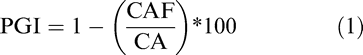Introduction
One of the most important diseases in peanut is stem rot also known as southern blight, caused by Sclerotium rolfsii Sacc. (teleomorph Athelia rolfsii (Curzi) C.C. Tu and Kimbr.), which was first reported by Rolfs in 1892 on tomato (Aycock 1966). This pathogen causes reductions in pod quality and numbers resulting in yield losses ranging from 10 to 80 (Melouk and Backman 1995). Production value losses from this disease in 2011 were estimated to be $50 million including both yield reductions and additional management costs such as fungicides (Woodward 2011). While fungicides are an effective strategy for managing stem rot in peanut (Kemerait et al. 2015), they can cost producers approximately $60 to $150 per acre each year. However, many producers consider this cost to be acceptable compared to overall risk associated from not utilizing them. An improved understanding about S. rolfsii populations in peanut could provide the industry with more information about how to manage this devastating pathogen while reducing production expenses such as fungicides.
S. rolfsii has a high level of phenotypic and genetic diversity (Xie et al. 2014; Remesal et al. 2012; Bagwan 2010; Punja and Sun 2001; Okabe and Matsumoto 2000). Regional samples of this fungus have been observed to produce multiple mycelia compatibility groupings with isolates of a particular MCG typically clustering together genetically (Punja and Sun 2001; Okabe and Matsumoto 2000). MCGs can also provide insights about S. rolfsii's relative aggressiveness and virulence (Xie et al. 2014; Remesal et al. 2012) However, considerable variation is present among the MCGs of this pathogen that does not allow these trends to remain consistent. Assessing the diversity of this pathogen in southeastern peanut production areas is a critical step in developing comprehensive resistance screening programs as well as understanding variation in the biological traits of this peanut pathogen.
Knowledge about pathogen diversity is an important component for developing useful integrated management strategies, but so is monitoring the pathogen phenotype fungicide sensitivity (Brent, 1998). There are multiple fungicidal products (e.g. tebuconazole, prothioconazole, flutolanil) available for S. rolfsii control, however, extensive sampling for pathogen sensitivity to these products has been limited in recent years (Franke et al. 1998a; Franke et al. 1998b; Le et al. 2012). Reduced sensitivity to the fungicide pentachloronitrobenzene (PCNB) was observed in Texas during the 1985 peanut growing season (Nalim et al. 1995). However, it was noted that this reduced sensitivity was limited to one isolate. This isolate belonged to a unique mycelial compatibility group which was not found in isolates collected between 1990 and 1994. Thus, it is possible that this trait is limited to S. rolfsii individuals that were no longer observed in the field. There is also an indication that in areas where fungicides are frequently used to manage S. rolfsii that the pathogen's sensitivity to these fungicides decreases (Franke et al., 1998a; Le et al. 2012). Thus, continued monitoring through intensive sampling (many single infections on several occasions) (Brent, 1998) of S. rolfsii population responses to different fungicide chemistries is needed to be sure that this pathogen remains relatively sensitive to these products.
Based on the S. rolfsii diversity reported by Xie et al. (2014), it was hypothesized that a high level of phenotypic diversity exists within S. rolfsii individuals from Florida's major peanut producing areas. We also hypothesized that reduced sensitivity to fungicides would be present in isolates collected from peanut fields. The objectives of our study were to phenotypically characterize S. rolfsii isolates collected from Florida peanut fields based on morphology and mycelial compatibility, and assess the difference in sensitivity of these S. rolfsii isolates for two commercial fungicides (flutolanil and tebuconazole) and survey the sensitivity of four novel commercial fungicides (azoxystrobin, penthiopyrad, prothioconazole, and solatenol) using intensive sampling techniques.
Materials and methods
Isolate collection
During the 2013 growing season, 15 S. rolfsii isolates were collected from diseased peanut plants in the Florida counties of Gadsden (6 isolates), Jackson (1 isolate), Hamilton (1 isolate), Levy (1 isolate), Marion (4 isolates) and Alachua (2 isolates). Twelve of the isolates were collected from production fields growing the runner type variety Georgia 06-G', and 3 were from the perennial peanut forage variety Floragraze'. Production fields were irrigated, had a history of peanut plantings with 2 to 3 years between crops, and consistently used 5 to 7 spray fungicide programs. Fields from Gadsden and Jackson counties typically used more flutolanil, estimated at 2 sprays per year, than production fields in the other counties sampled in this study. Tebuconazole and azoxystrobin were commonly used in all the production fields ranging between 2 to 4 sprays a year. Penthiopyrad was more commonly used in Levy, Marion, Alachua and Hamilton Counties with sprays ranging between 1 and 3 each year. Fungicide sprays had not been used in the perennial peanut site at the time the isolates had been collected with the previous crop being Bahia-grass. Plant samples were kept in cold storage at 6 2C for 3 to 5 days after which the fungus was isolated from 1 cm long diseased stem segment containing mycelium or a lesion. These segments were surface sterilized for 5 minutes using a 10 bleach solution and rinsed with sterile distilled water (Domsch et al. 1980; Okabe and Matsumoto 2000). Quarter strength potato dextrose agar (QPDA) was prepared by mixing 5 g of potato dextrose agar (PDA, Difco) in 1 L of distilled water. The sterilized stem segments were transferred to QPDA and incubated at 26 2C for 3 days. After incubation, a 6 mm diameter hyphal disk was harvested from the actively growing edge of the colony and transferred to another QPDA plate. This process was repeated 3 times to ensure that isolations were clean of contamination from other fungal species and bacteria. Single sclerotia from 10-day-old cultures were transferred to new QPDA plates for long- term storage at room temperature (22 5C) (Bagwan, 2011). To maintain the original characteristics (e.g. pathogenicity) of the isolates (Ryan et al. 2012), isolates were inoculated onto peanut plants and then re-isolated as previously described every six months (Shokes et al. 1996). To obtain mycelial plugs for each experiment isolates from long-term storage were revived by inoculating one sclerotium from each isolate onto QPDA plates and incubating for 4 days at 26 2C in complete darkness.
Morphological characterization
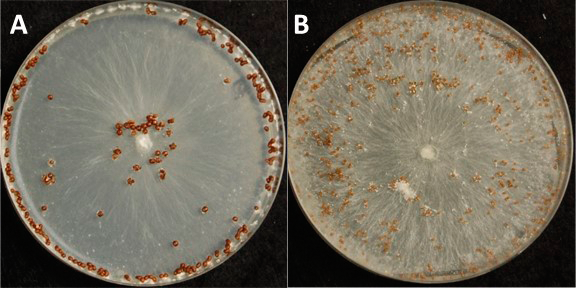
Morphological characteristics of each isolate were assessed using a media growth assay. Actively growing mycelial plugs of 6 mm diameter were harvested from the edge of the growing colony, transferred to the center of a PDA petri plate and incubated for 2 days at 26 2C in complete darkness. Digital images of the plates with a scale were captured for intrinsic growth analysis. The total area of the colony growth was measured using the digital image measurement software KLONK (KLONK Image Measurement, Denmark, Copenhagen). The area of the inoculum plug was subtracted from the total colony area to find colony growth after inoculation. Petri dishes were placed back in the growth chamber (26 2C; complete darkness) for an additional 14 days to assess sclerotial characteristics. Again, digital images of the plates with a scale were captured for analysis (Fig. 1). The number of sclerotia and average size of sclerotia were determined by using the ImageJ software's (National Institute of Health, United States) particle counting and analysis protocol based on the ImageJ tutorials (https://imagej.nih.gov/ij/docs/examples/index.html, accessed June of 2016).
Mycelial compatibility groups (MCGs)
Isolates were tested for mycelial compatibility by placing 2 agar plugs of the same isolate and 1 agar plug of a different isolate onto PDA (20 g PDA, Difco in 1 L of distilled water) plates equidistant (5 - 6 cm) from each other (Kohn et al. 1990; Kohn et al. 1991). The plugs of the same isolate served as the control for determining compatible mycelial reactions. After inoculation, the plates incubated at 26 2C in complete darkness. Plates were rated 2 days after inoculation once the control colonies had grown together for a compatible reaction. Compatibility between plugs was visually assessed to determine if they form a merged confluent colony as seen for a compatible reaction or a dark inhibition line as seen for an incompatible reaction (Leslie, 1993; Xie et al. 2014). Each replication of the experiment consisted of 3 sub-samples and the whole experiment was repeated 3 times.
One representative isolate from each MCG identified within the peanut isolates was also compared to a selection of the established MCGs reported by Xie et al. (2014) which included S. rolfsii isolates from cotton, peanut, tomato and other vegetable crops. Only 15 MCGs were tested because 8 type isolates (MCG 4, 8, 14, 15, 16, 17, 18, and 20) were no longer viable.
Mycelial growth inhibition assay
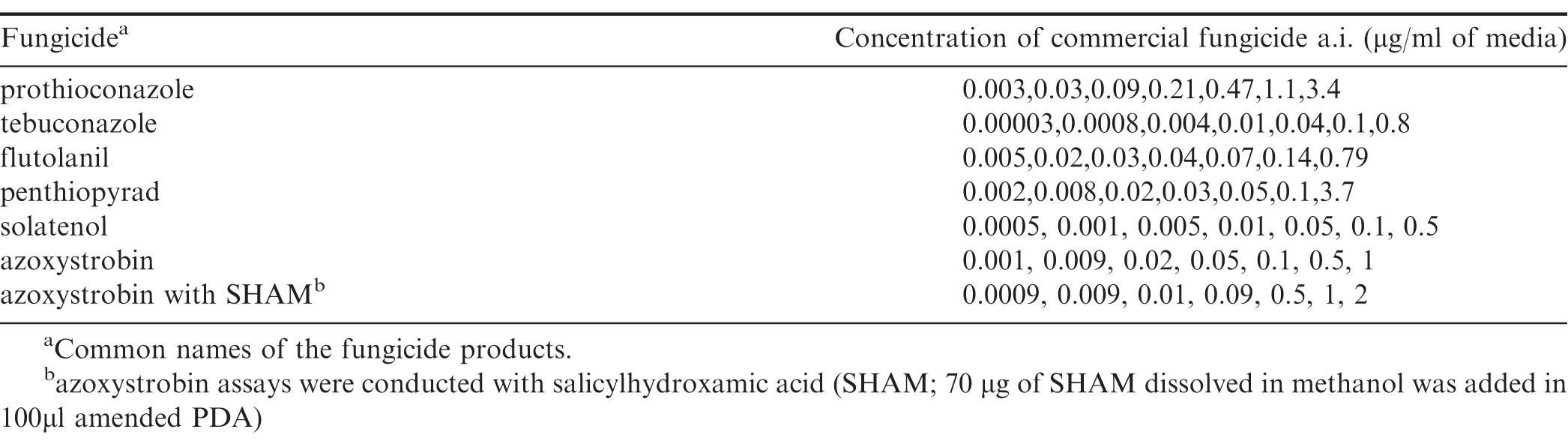
A mycelial growth assay was used to test for isolates sensitivities to the commercial fungicides Abound (azoxystrobin 22.9, Syngenta U.S., Wilmington, DE), Proline (prothioconazole 41, Bayer U.S., Research Triangle Park, NC), TebuStar 3.6 L (tebuconazole 38.7, Agristar US, Ankeny, IA), Convoy (flutolanil 40, Nichino America, Inc. U.S., Wilmington, DE ), FontelisTM (penthiopyrad 20.4, DuPont U.S., Wilmington, DE), and Solatenol (benzovindiflupyr, Syngenta U.S., Wilmington, DE). The commercial fungicide products were diluted in distilled water to amend QPDA with the concentrations of 5, 1, 0.5, 0.1, 0.05, 0.01, 0.005, 0.001, 0.0005, and 0.0001 g of the fungicide's active ingredient per ml of medium. Fungicide solutions were added to sterile QPDA that had been cooled to 50C using a water bath. Solutions were regularly ( every 1 to 2 min) swirled for homogenous mixing. For the azoxystrobin sensitivity test, a stock solution of salicylhydroxamic acid (SHAM) (Sigma Chemical Co., St. Louis, MO) with methanol was prepared by adding 100 mg of SHAM to 1 ml of methanol in a microcentrifuge tube and warming in a water bath at 50C for 1-2 minutes to completely dissolve SHAM in methanol. 70 l of this SHAM stock solution (7 mg) was added to every 100 ml of autoclaved liquid QPDA media that was cooled in a water bath to 60C (Pasche et al. 2004). Azoxystrobin sensitivity assay without SHAM was also conducted to evaluate the effect of SHAM on azoxystrobin sensitivity since it has been indicated that other fungal pathogens can survive media amended with the fungicide by using an alternative respiration pathway (Wood and Hollomon, 2003).
Mycelial inhibition was calculated by transferring an actively growing mycelial plug (6 mm diameter) of S. rolfsii to the center of a petri-dish (100 mm15 mm, Fisherbrand, USA) containing approximately 22 ml of fungicide amended PDA. Inoculated plates were incubated in complete darkness at 26 2C for 48 hours. After incubation, digital images of the plates with a scale were captured and stored for analysis. The total area of the colony growth was measured using the digital image measurement software KLONK (KLONK Image Measurement, Denmark, Copenhagen). The area of inoculated plug was subtracted from the total colony area to find the total colony growth after inoculation.
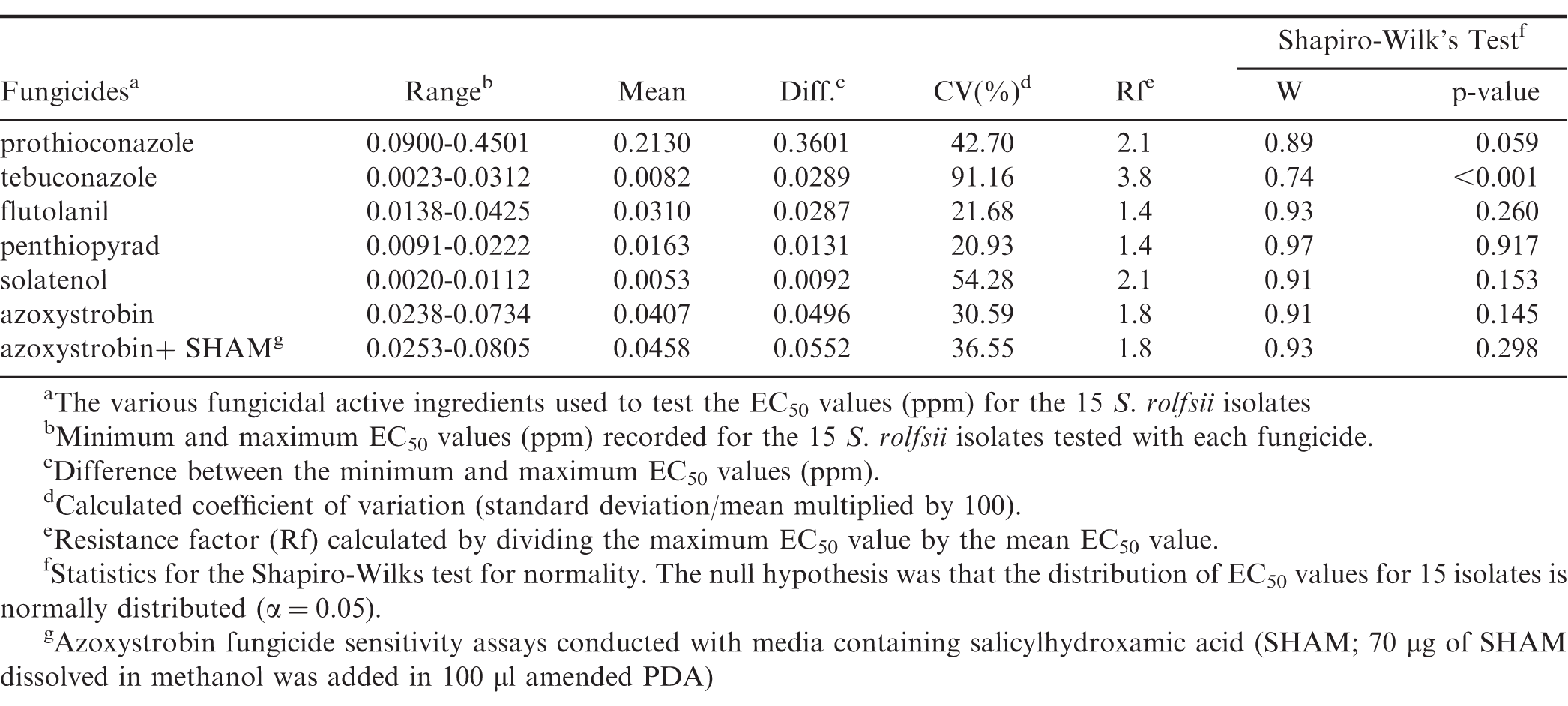
Isolates were exposed to these 10 concentrations for two experimental repeats and analyzed using nonlinear regression as described below. The resulting regression model for each isolate was used to estimate the concentrations causing percentage inhibition of 5, 20, 35, 50, 65, 80, and 95 which were used in the final three repeats to estimate EC50 values. The concentrations of fungicide used for the final three repeats are listed in Table 1.
Concentration of the commercial fungicide's active ingredient (a.i.) used for the final three runs of fungicide sensitivity assay. These concentrations were the estimated values that caused 5, 20, 35, 50, 65, 80, 95 percent growth inhibition of the isolates based on 2 previous runs using standard concentrations.
The growth inhibition at each fungicide concentration was calculated using the following equation:
In which, PGI is the percentage of colony growth inhibited by the fungicide compared to un-amended checks, CAF is the recorded colony area in plates amended with fungicide and CA is the colony area in the unamended control plates (Franke et al. 1998a). A total of 41 PGI values were calculated for each fungicide product using all five replications of the inhibition assay. The sensitivity of each isolate was determined by fitting a sigmoid nonlinear regression model with three parameters using SigmaPlot 12.5 (Systat Software Inc. San Jose, CA) (Franke et al. 1998a). The nonlinear equation was:
in which f is the percent growth inhibition at fungicide concentration x, a is the maximum percent growth inhibition with the fungicide, xo is the inflection point or EC50 value, and b is the shape parameter determining steepness of the curve (Damgaard and Nielsen 1999). This equation was fit to each of the repetitions and analyzed for goodness of fit to calculate the mean EC50 value for each isolate.
Data analysis
All phenotypic data analyses were completed using SAS (version 9.4; SAS Institute Inc, Cary, NC, USA). Phenotypic data were analyzed as a completely randomized design using the PROC GLM command to conduct an analysis of variance (ANOVA) with S. rolfsii isolate as the fixed effect for each of the dependent variables of radial hyphal growth (mm), sclerotia size (mm) and sclerotia number. Pairwise comparisons of isolate means for each dependent variable were analyzed using Fisher's protected least significant difference test ( 0.05). Both the Simpson's and Shannon-Weiner's diversity indices were calculated using the formulas from Zar, 1999 and Magurran, 2004 to quantify the diversity of S. rolfsii related to MCGs observed in this study and for the isolate data from different hosts published in Xie et al. 2014. The distributions of the EC50 values for all the isolates in relation to the various fungicide products were compared to a normal distribution using the Shapiro-Wilks Test (shapiro.test()) in RStudio (package statversion 0.99.451, RStudio, Inc.). and Pearson correlation coefficients were calculated using cor() in RStudio to examine the linear relationship between the EC50 results of the six different fungicide products across the S. rolfsii isolates (Erickson and Wilcox, 1997). The Rf was calculated by dividing the maximum EC50 by the mean EC50 value for the respective fungicides.
Results
Phenotypic Traits
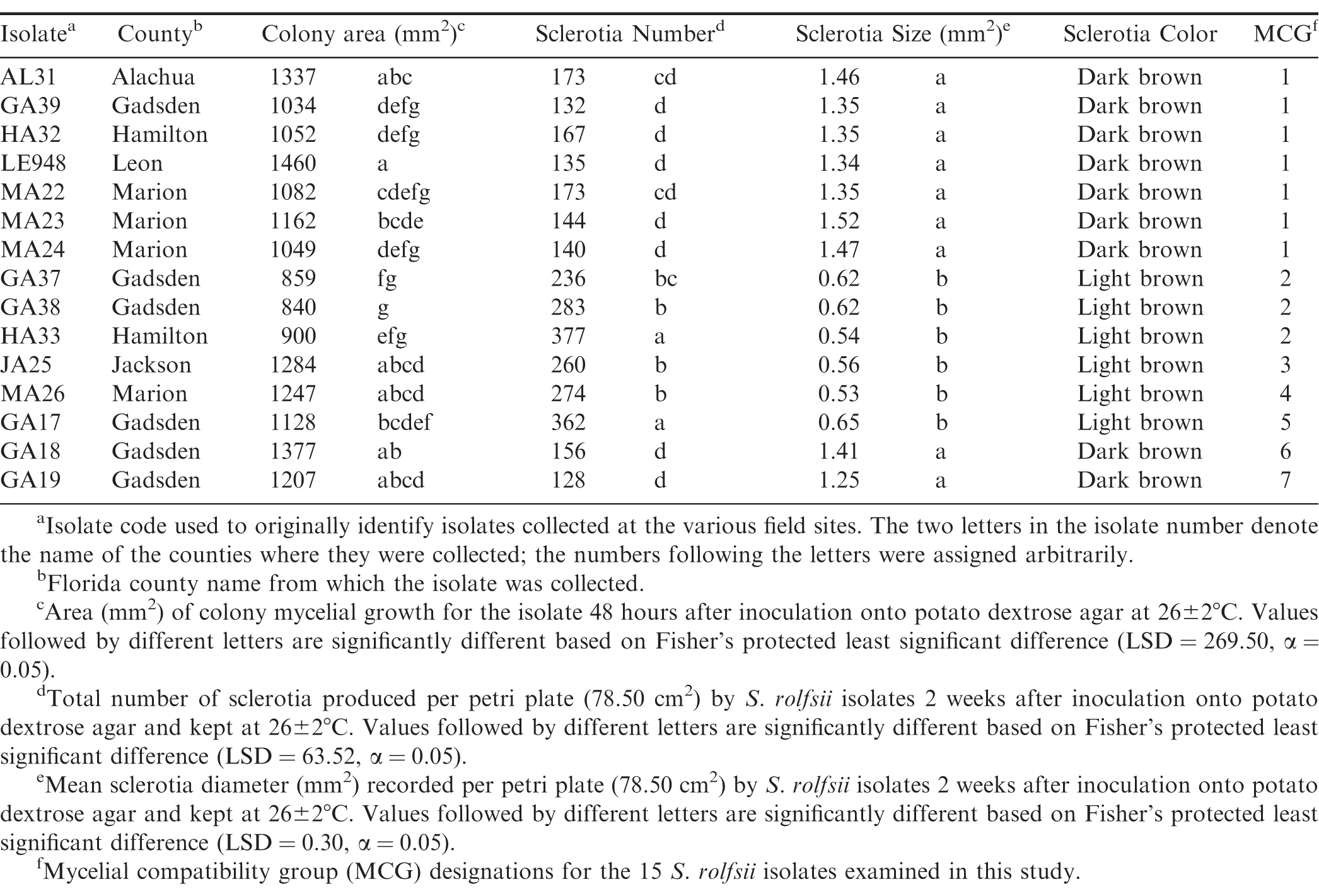
Isolates differed in their average colony growth on PDA two days after inoculation and sclerotial traits, including color, size and total number produced 14 days after inoculation (Table 2). In general, isolates were divided into two groups, those that produced large (> 1 mm2), less numerous (< 200), dark brown sclerotia, and small (< 1 mm2), more numerous (> 200), light brown sclerotia (Table 2, Fig. 1). These morphological characters varied within and across the Florida counties and fields from which the isolates were collected.
A total of seven MCGs were identified among the 15 isolates of S. rolfsii examined in this study (Table 2). These groups were arbitrarily assigned as MCG 1 to MCG 7. A total of seven isolates collected belonged to MCG 1, three isolates were categorized in MCG 2, and MCGs 3, 4, 5, 6 and 7 each contained one isolate. The three isolates collected from one perennial peanut site in Gadsden County (GA17, GA18, and GA19) belonged to three separate MCGs and varied significantly in their sclerotia size, number and color (Table 2). The representative isolate from MCG 1 in this study was compatible with MCG 11, reported by Xie et al. 2014, but MCG 2 through 7 were not compatible with any of the other MCGs from Xie et al (2014) that were tested.
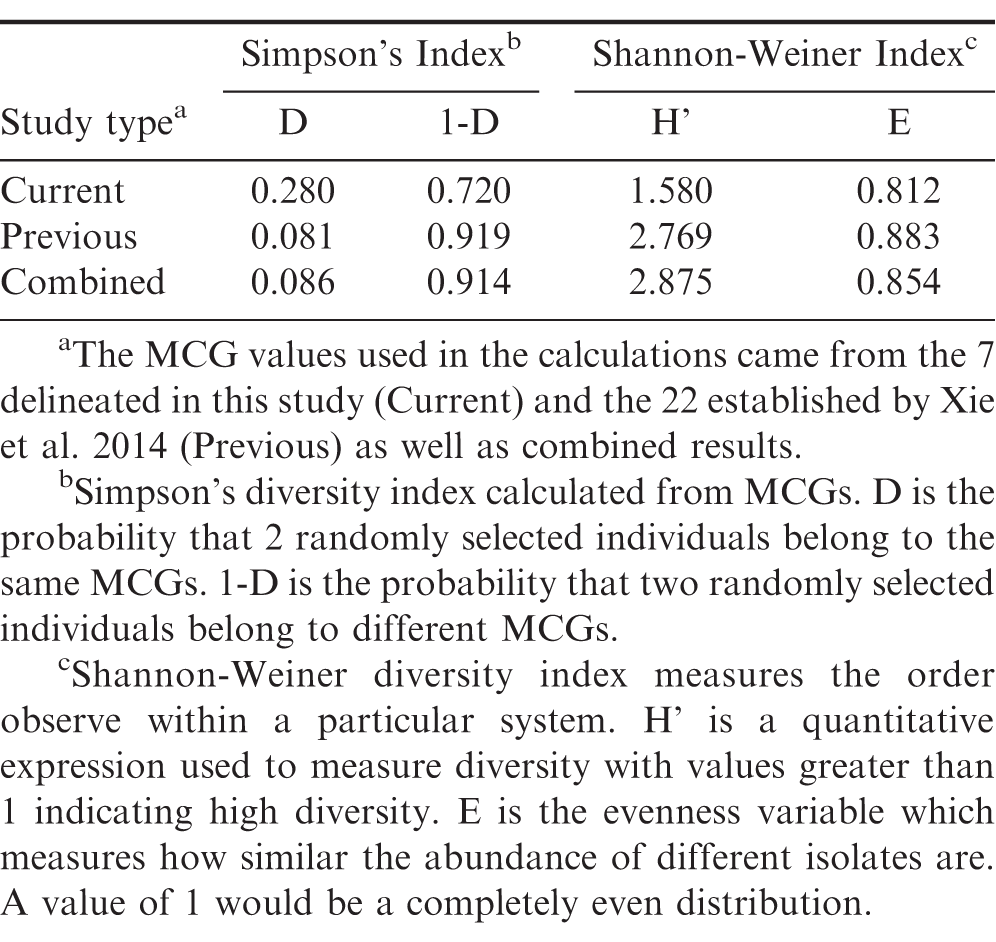
Simpson's diversity index indicated the probability of randomly selecting two isolates of the same MCG in this study is approximately 0.28 and 0.09 when studies were combined with published data by Xie et al. 2014 (Table 3). The Shannon-Weiner diversity index values were greater than one for all studies reported indicating a high level of diversity. Evenness variables were greater than 0.80 indicating that the abundance in each MCG is similar or homogenous.
Fungicide Sensitivity Monitoring and Survey
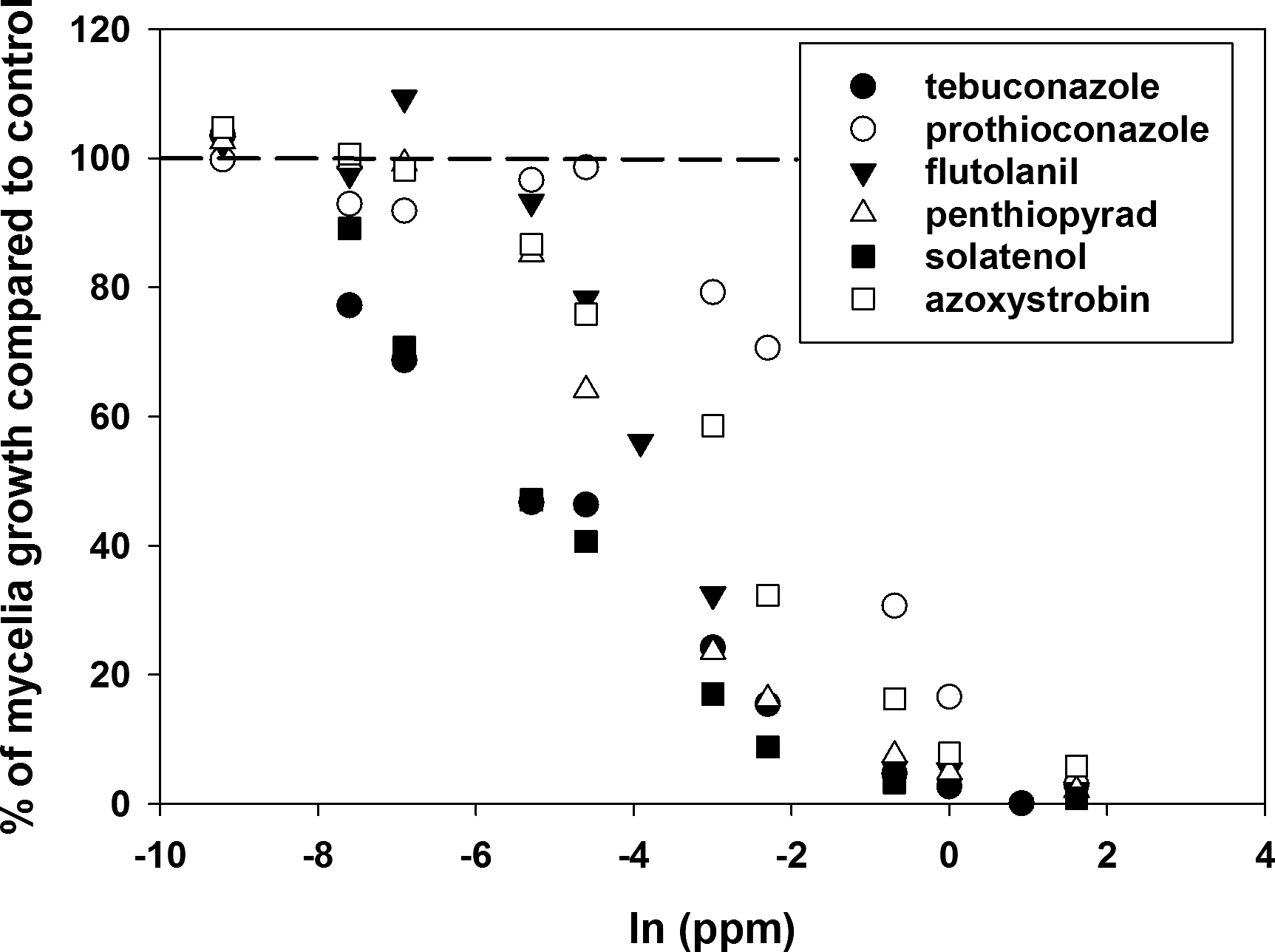
Ranges of EC50, mean EC50 and overall inhibition response of the S. rolfsii isolates were different for all fungicide products tested (Table 4, Fig. 2). Prothioconazole had the highest mean and range EC50 values and solatenol had lowest mean and range EC50 values (Table 4). The ranges calculated for tebuconazole and flutolanil in this study were 4.8 and 4.7 times smaller than those reported previously by Franke et al. 1998a. The coefficient of variation (CV) based on the EC50 values was more than 30 for all the products except flutolanil and penthiopyrad. Tebuconazole had the highest CV, which was about 1.68 times higher than the next highest fungicide product, solatenol (Table 4). Tebuconazole had the highest resistance factor which was 1.81 times greater than the next highest products, prothioconazole and solatenol.
The average response of the S. rolfsii isolates towards different concentrations of the various fungicide products tested. Mycelia growth of the isolates on the media amended with the various concentrations of fungicide were compared to a control, which consisted of media not amended with the fungicide compound. Non-amended plates were considered to have 100 of the possible growth for the isolates. A trend was apparent that the isolates produced more mycelia growth than the un-amended media (hormesis) for select fungicides at values of ln (6 ppm) or less.
The distribution of EC50 values for the 15 isolates was not significantly different (p > 0.05) from a normal distribution for all the isolates tested except tebuconazole (Table 4). The Shapiro-Wilk's test indicated the EC50 distribution for tebuconazole isolates was skewed (p < 0.01) with a long tail towards the maximum (0.031 ppm) value observed. The distribution for prothioconazole was not significantly different from normal (p 0.06), however, the largest EC50 range difference (0.360 ppm) was observed for this product. This range difference was 12.46 times greater than other triazole, tebuconazole (0.029 ppm).
A significant correlation was observed between the EC50 values of flutolanil and penthiopyrad (r 0.69, p-value 0.004). No significant correlations (p-value > 0.100) were observed for the rest of the fungicide products tested; however, a positive correlation coefficient was noted between flutolanil and solatenol (r 0.41, p-value 0.134).
Discussion
Phenotypic traits, such as morphological characteristics and MCGs, have been used to assess the diversity of S. rolfsii on different hosts and in many regions throughout the world (Xie et al. 2014; Remesal et al. 2012; Sarma et al. 2002; Punja and Sun 2001; Cilliers et al. 2000; Harlton et al. 1995; Nalim et al. 1995). Previous studies have shown that S. rolfsii isolates can vary both genetically and morphologically within the same MCG (Punja et al. 2001; Cilliers et al. 2000), and that the diversity of MCGs can be high within a field or across a region (Xie et al. 2014; Remesal et al. 2012). In this study, our results support these general conclusions about S. rolfsii MCGs. For example, seven separate MCGs were recognized among the six Florida counties in this study of which three distinct MCGs (5, 6, & 7) were all from a single field site location. This high level of diversity was also supported by both the Shannon-Weiner and Simpson's indices. The Shannon-Weiner index implies that the MCGs will have similar abundances across the Florida peanut regions and the southern region when combining this data with results from Xie et al (2014). Simpon's index indicates that the likelihood of randomly selecting two individuals from the same MCG is low for Florida peanut fields by county. It is important to note that all diversity indices are affected by sample size, which was low in this study (Gotelli and Ellison, 2013; Barrantes and Sandoval, 2009; Soetaert and Help, 1990). However, both the Shannon and Simpson indices are widely used for discerning population diversity and their agreement supports the conclusion that high level of diversity was present. This high level of diversity is likely a result of the relatively high MCG category number, which can mask the effects of large proportions being present within one category as observed in this study (Gotelli and Ellison, 2013). This means that the large number of MCG categories with low isolate numbers (< 4) is what the indices are indicating for the high diversity in the population. Thus, these results support the conclusion that S. rolfsii has a diverse population, but there does appear to be some clustering of isolates into one MCG.
Despite the high level of diversity indicated by MCGs, other morphological characteristics, such as number, size and color of sclerotia, remained more consistent with each phenotypic grouping. Sclerotia that were dark brown tended to have larger and less numerous sclerotia produced on media when compared to light brown sclerotia. Also, all the sclerotia in the largest MCG, group 1, were dark brown compared with light brown sclerotia for the second largest MCG, group 2. These results are different from Xie et al. (2014) and Le et al. (2012) in that they reported peanut isolates of S. rolfsii generally producing larger sclerotia compared to those from other crops such as tomato. These studies also did not notice a relationship between sclerotia size and number, which was apparent in this study. Xie et al. (2014) observed that most of their peanut isolates had tan to brown sclerotia with very few having dark brown color compared to a majority of the isolates in this study having dark brown color. Even though these studies had significantly higher sample sizes, they were still not able to capture all the variability associated with morphological traits of S. rolfsii, especially on peanut. In fact, it was discovered that possibly six new MCGs were identified in this study when compared to those previously established by Xie et al. (2014). Thus, high levels of diversity in S. rolfsii make it difficult to adequately describe a population using MCGs as well as other phenotypic traits even with an 80 isolate sample size. These results indicate that some population inferences can be observed from both large and small sample sizes, especially when monitoring is the goal of the study. However, it will be important in future studies to further classify S. rolfsii populations and possibly the genus using genetic data.
Fungicide sensitivity is another important phenotypic trait that can be used to better understand fungal populations exposed to selection pressures (Franke et al. 1998a; Franke et al. 1998b; Brent and Hollomon 2007; Milgroom 2015). However, S. rolfsii is considered a low risk pathogen for the development of fungicide insensitivity, and thus there should be minimal variation in the response of this pathogen to various fungicidal products. Our monitoring results support this hypothesis with low levels of variation observed between the isolates in regards to their fungicide sensitivity when exposed to commercial grades of the different active ingredients. Only tebuconazole had a coefficient of variation greater than 90 and produced an EC50 distribution that was not normally distributed. These results indicate that monitoring the effectiveness of tebuconazole products in the field will be important in the years to come, as a trend towards possible isolate insensitivity was noticed in this small sample size. As for the other products tested, no case of field resistance has currently been reported in Florida indicating if resistance is present it is in low proportions. Based on the sample size of 15, Russel (2008) indicated that we would have a 97 chance of identifying at least one resistant isolates in the population if the true frequency of resistance was 0.20 or greater. Thus, based on these results it appears that a majority, greater that 80, of the S. rolfsii population is sensitive to fungicides surveyed in this study, but that it will be important to continue field monitoring for reductions in efficacy of any of these products when managing this pathogen.
The efficacy of various fungicidal compounds on the inhibition of S. rolfsii intrinsic growth in-vitro has been previously examined in multiple studies (Bhulyan et al. 2012; Akgul et al. 2011; Franke et al. 1998a; Shim et al. 1998; Waterfield and Sisler 1989; Csinos 1987). In these studies, only Franke et al. (1998a) and Csinos (1987) examined similar compounds which were tebuconazole and flutolanil. Csinos (1987) indicated that the EC50 value for flutolanil was < 0.01 ppm, which was more than 2 times lower than the mean EC50 value reported in the extensive study by Franke et al. (1998a) and in this intensive, monitoring study. One possible reason for this difference could be that Csinos (1987) only used 1 isolate to test for flutolanil sensitivity, and the range from Franke et al. (1998a) and this study both had isolates with EC50 values near or below 0.01 ppm. Thus, a better comparison for shifts in sensitivity would be by comparing these monitoring results EC50 values with those of Franke et al. (1998a). This comparison shows no indication of changes in sensitivity of S. rolfsii to flutolanil as the values reported in this small sample size study tended to match the lower values reported Franke et al. (1998a). While continued monitoring will be important for this fungicide, it is apparent from these results that current fungicide usage has not selected for large populations with reduced-sensitivity in Florida.
Similar conclusions can be made about tebuconazole, in that the mean EC50 values reported in this monitoring study were generally lower than those observed by Franke et al. (1998a). Our observation can be further supported by the comparison of triazole product results from this study with those previously reported for propiconazole (Waterfield and Sisler 1989; Bhulyan et al. 2012). In these studies, the EC50 identified for propiconazole was around 0.25 ppm. This EC50 value was similar to the one identified for prothioconazole, and provides relative support that reduced sensitivity to triazole fungicides is still not prevalent in S. rolfsii populations in Florida. Overall, even though our results indicated a non-normal EC50 distribution was observed for tebuconazole, there was no indication from these comparisons that major changes in fungicide efficacy have occurred in this class of fungicides.
Correlation analysis is commonly used to identify the possibility of cross-resistance between fungicide compounds. Positive correlation values were among between the SDHI compounds of flutolanil, penthiopyrad and solatenol, in which the greatest correlations were seen between flutolanil and the other two compounds. This is concerning since many new products being released for peanut disease control often contain a SDHI product. This means that there could be an increased selection pressure put on S. rolfsii populations in the coming years that could lead to the establishment of resistant populations in peanut fields. Thus, it will be important to continue monitoring product efficacy in the field and test isolates that exhibit insensitivity to SHDI products by using the estimated EC50 values indicated here and the baseline value established Franke et al. (1998a) for flutolanil.
A unique outcome observed while conducting the fungicide sensitivity assay was that sub-lethal concentrations of flutolanil, as well as four other fungicide products, produce apparent increases in mycelial growth compared to the non-amended checks. This process, called hormesis, has been documented with other fungicidal compounds, primarily pertaining to oomycete control (Flores and Garzon, 2013). While current field doses of these products should provide effective control of S. rolfsii, there are concerns about the effects this observation will have on management. The dilution of fungicide products by plant canopies as well as producers reducing fungicide rates to save costs, could create a situation in which products can promote the development of the pathogen producing what appears to be an insensitive response. Further research is needed to better understand the impact hormesis will have on S. rolfsii growth and infection, and how it might influence disease development in the field. This trait could lead to false identification of field resistance related to the various fungicidal compounds.
Summary and Conclusion
Despite the high levels of phenotypic variation noticed in MCGs of S. rolfsii, from this small sample size study there was no indication of changes in sensitivity of S. rolfsii to flutolanil or tebuconazole. Phenotypic traits provide some insights about the population structure of this pathogen in Florida's peanut producing regions; however, more detailed information could be gained through the use of genetic data analysis and extensive sampling. These results imply that current fungicide reommendations should continue to provide effective control of this disease in peanut. Further research is needed to determine the true variations in the pathogen's diversity as well as the population's responses to the novel fungicidal compounds tested here, but our results provide essential reference points for researchers working on this pathogen in Florida and possibly throughout the southeast.
Acknowledgements
This research was supported by grants from the Florida Peanut Producers Association and the National Peanut Board as well as general support from the University of Florida Department of Plant Pathology. We want to acknowledge Kris Beckham for her assistance with instruction of basic laboratory techniques and maintenance of the S. rolfsii collection for Mr. Khatri. We are greatful for Mathew Beckham's assistantance with colony growth analysis. We would like to thank Dr. Gary Vallad for providing S. rolfsii isolates for comparison from his collection. Finally, we would like to acknowledge Ms. Patricia Soria for her thorough review of this manuscript.
Literature Cited
D.S., Akgul, H., Ozgonen, and A Erkilic, (2011). The effects of seed treatments with fungicides on stem rot caused by Sclerotium rolfsii Sacc., in peanut. Pak. J. Bot 43: 2991 2996.
R Aycock, (1966). Stem rot and other diseases caused by Sclerotium rolfsii. N.C. Agr. Expt. St. Tech. Bul. No. 174.,
N.B Bagwan, (2010). Morphological variation in Sclerotium rolfsii Sacc. isolates causing stem rot in groundnut (Arachis hypogaea L.). Inter. J. of Plant Prot 4: 68 73.
G., Barrantes, and L Sandoval, (2009). Conceptual and statistical problems associated with the use of diversity indices in ecology. Rev. Biol. Trop 47: 451 460.
M.A.H.B., Bhulyan, M.T., Rahman, and K.A Bhulyan, (2012). In vitro screening of fungicides and antagonists against Sclerotium rolfsii. African J. of Biotech 11: 14822 14827.
Brent, K. J 1998 Monitoring for Fungicide Resistance In: C. J Delp (Ed) Fungicide Resistance in North America (pp. 9- 11). APS Press St. Paul, Minnesota.
Brent, K.J., and Hollomon, D.W 2007 Fungicide resistance in crop pathogens: How can it be managed? 2nd ED. Brussels, Belgium Croplife International.
A.J., Cilliers, L., Herselman, and Z.A. Pretorius (2000). Genetic variability with and among mycelial compatibility groups of Sclerotium rolfsii in South Africa. Phytopathology 90: 1026 1030.
A.S Csinos, (1987). Control of southern stem rot and Rhizoctonia limb rot of peanut with flutolanil. Peanut Sci 14: 55 58.
C., Damgaard, and B.J Nielsen, (1999). The effect of fungal density on fungicide dose-response curves in barley powdery mildew (Erysiphe graminis f.sp. hordei). Plant Pathol . 48: 402 407.
K.H., Domsch, W Gamsand T.H Anderson, (1980). Compendium of Soil Fungi, Page 859. London: Acad. Press. , .
E. O., Erickson, and W. F Wilcox, (1997). Distributions of sensitivities to three sterol demethylation inhibitor fungicides among populations of Uncinula necator sensitive and resistant to triadimefon. Phytopathology 87: 784 791.
J.F., Flores, and C.D Garzon, (2013). Detection and assessment of chemical hormesis on the radial growth in vitro of oomycetes and fungal plant pathogens. Int. Dose Resp. Soc . 11: 361- 373.
Franke, M. D., Brenneman, T. B., Stevenson, K. L and Padgett, G. B 1998 a Sensitivity of isolates of Sclerotium rolfsii from peanut in Georgia to selected fungicides Plant Dis 82: 578 583.
Franke, M. D., Brenneman, T. B., and Stevenson, K. L 1998 b Stem rot of peanut: relationship between in vitro fungicide sensitivity and field efficacy of fungicides , Peanut Sci. 25: 76- 80.
N. J Gotelli, and A. M Ellison, (2013). A Primer of Ecological Statistics, 2nd edition. Sunderland, MA: : Sinauer Associates, Inc. .
C.E., Harlton, C.A., Levesque, and Z.K Punja, (1995). Genetic diversity in Sclerotium (Athelia) rolfsii and related species. Phytopathology 85: 1269 1281.
R., Kemerait, A., Culbreath, J., Beasley, E., Prostko, T., Brenneman, N., Smith, S., Tubbs, R., Srinivasan, M., Abney, B., Tillman, D., Rowland, N., Dufault, A., Hagan, and S Monfort, (2015). Minimizing disease of peanut in the southeastern United States. Peanut Rx. Auburn University, Clemson University: University of Georgia, University of Florida. , .
L.M., Kohn, I., Carbone J.B Anderson, (1990). Mycelial interactions in Sclerotinia sclerotiorum. Exp. Mycol 14 ((3)): 255 267.
L.M., Kohn E., Stasoviski I., Carbone J., Royer J.B Anderson (1991). Mycelial incompatibility and molecular markers identify genetic variability in field populations of Sclerotinia sclerotiorum. Phytopathology 81 : 480 485.
C. N., Le, R., Mendes, M., Kruijt, and J.M Raaijmakers, (2012). Genetic and phenotypic diversity of Sclerotium rolfsii in groundnut fields in central Vietnam. Plant Dis 96: 389 397.
J. F Leslie, (1993). Fungal vegetative compatibility. Annu. Rev. of Phytopathol 31: 127 150
Magurran, A.E 2004 Measuring biological diversity Blackwell Science LTD Oxford, UK pg: 215.
Melouk, H.A., and Backman, P.A 1995 Management of soil borne fungal pathogen Pages 75 85 In: H.A Melouk, and F.M Shokes, (eds) Peanut Health Management APS Press, St. Paul, MN.
M.G Milgroom, (2015). Population Biology of Plant Pathogens: Genetics, Ecology and Evolution. St. Paul, MN: American Phytopathological Society Press. , .
F.A., Nalim, J. L., Starr, K.E., Woodard, S., Segner, and N. P Keller, (1995). Mycelial compatibility groups in Texas peanut field populations of Sclerotium rolfsii. Phytopathology 85: 1507 1512.
I., Okabe, and N Matsumoto, (2000). Population structure of Sclerotium rolfsii in peanut fields. Mycoscience 41: 145 148.
Pasche, J.S., Wharam, C.M., and Gudmestad, N.C 2004 Shift in sensitivity of Alternaria solani in response to QoI fungicides, Plant Dis.88:181-187.
Z. K Punja, (1985). The biology, ecology, and control of Sclerotium rolfsii. Annu. Rev. Phytopathol 23: 97 127
Z. K., Punja, and L.J Sun, (2001). Genetic diversity among mycelial compatibility groups of Sclerotium rolfsii, Sclerotium delphinii. Mycology 105: 537 546.
E., Remesal, R., Ramirez, R., Diaz, and J Cortes, (2012). Mycelial compatibility groups and pathogenic diversity in Sclerotium rolfsii populations from sugar beet crops in Mediterranean-type climate regions. Plant Pathol 61: 739 753.
P.E Russel, (2008). Sensitivity baselines in fungicide resistance research and management. FRAC Monograph no. 3. United Kingdom: AIMPRINT. .
Ryan, M., Ritchie, B. J., and Smith, D 2012 Maintenance and storage of fungal plant pathogens In: Lane, C., Beales, P., and Hughes, K (Eds), Fungal plant pathogens (pp. 223- 250). Cambridge, MA: CAB International.
B.K., Sarma, U.P Singh, and K.P Singh, (2002). Variability in Indian isolates of Sclerotium rolfsii. Mycologia , 94: 1051 1058.
M.Y., Shim, J.L., Starr, N.P., Keller, K.E Woodard, and T.A Lee, (1998). Distribution of isolates of Sclerotium rolfsii tolerant to pentachloronitrobenzene in Texas peanut fields. Plant Dis 82: 103 106.
F.M, Shokes, K., Rozalski, D.W., Gorbet, T.B., Brenneman, and D.A Berger, (1996). Techniques for inoculation of peanut with Sclerotium rolfsii in the greenhouse and field. Peanut Sci 23: 124 128.
K., Soetaert, and C Heip, (1990). Sample-size dependence of diversity indices and the determination of sufficient sample size in a high-diversity deep-sea environment. Mar. Ecol. Prog. Ser 59: 305 307.
W.F Waterfield, and H.D Sisler, (1989). Effect of propiconazole on growth and sterol biosynthesis by Sclerotium rolfsii. Neth. J. P1. Path 95: 187 195.
P.M Wood, and D.W Hollomon, (2003). Review: A critical evaluation of the role of alternative oxidase in the performance of strobilurin and related fungicides acting at the Qo site of Complex III. Pest Manag Sci 59: 499 511.
J.L Williams-Woodward, (2013). 2011 Georgia Plant Disease Loss Estimates. Univ. Georgia Coop. Ext. Annual Publication 102-4,
C., Xie, C.-H., Huang, and G. E Vallad, (2014). Mycelial compatibility and pathogenic diversity among Sclerotium rolfsii isolates in the southern United States. Plant Dis 98: 1685 1694.
Z., Xu, M. L., Gleason, D. S., Mueller, P. D., Esker, C. A., Bradley, J. W., Buck, D. M., Benson, P. M., Dixon, and J. E. B. A Monteiro, (2008). Overwintering of Sclerotium rolfsii and S. rolfsii var. delphinii in different latitudes of the United States. Plant Dis 92: 719 724.
Zar, J.H 1999 Biostatical analysis, 4th ed Prentice Hall, Inc Upper Saddle River, NJ pg 40- 44.
Notes
- First, second, third and fourth author: Graduate student, Graduate student, Post-doctoral Scholar and Assistant Professor. Department of Plant Pathology, University of Florida, Gainesville, Florida, 32611-0680. [^] Corresponding author. Telephone: 352 273 4623; Email address: nsdufault@ufl.edu
Author Affiliations