Introduction
Lignins are complex phenolic macromolecules produced by plants that are composed of three phenylpropane alcohols: p-coumaryl, coniferyl, and sinapyl (Vance et al., 1980; Hatfield and Fukushima, 2005). In addition to providing structural support to tissues, lignin protects plants from herbivores and pathogens (Taiz and Zeiger, 1991). Most microorganisms, including many fungi, are unable to break down lignin, and lignin is known to be a critical component in multiple pathosystems for plant defense responses (Vance et al., 1980; Banniza et al., 2005; Bhuiyan et al., 2009; Xu et al., 2011; Eynck et al., 2012). Inhibition of lignin formation in resistant wheat genotypes resulted in increased susceptibility to Puccinia gramins f. sp. tritici (Moerschbacher et al., 1990). Lines of camelina resistant to Sclerotinia sclerotiorum, and cotton resistant to Verticillium dahliae, produced more lignin precursors upon infection than susceptible lines (Xu et al., 2011; Eynck et al., 2012). In peas, preformed stem lignin content was negatively associated with disease severity of Mycosphaerella pinodes (Banniza et al., 2005). However, the association between lignin content on diseases in other crops was less favorable. Lignin levels in alfalfa lines did not appear to be related to alfalfa rust resistance (Webb et al., 1996), and higher lignin content in soybean was positively correlated with disease susceptibility to Sclerotinia sclerotiorum (Peltier et al., 2009).
Sclerotinia blight, caused by S. minor Jagger, is a major disease in the Virginia-Carolina and Southwest peanut production regions of the U.S. Progress in breeding disease-resistant peanuts may be accelerated if physical or biochemical traits associated with disease resistance were available, particularly if such screening methods were more efficient than conducting field trials. Few studies have examined the relationship between lignin and disease resistance in peanut. Godoy et al. (1985) found higher levels of lignin in pods in breeding lines that were more resistant to pod rot than in susceptible lines. Similarly, Liang and colleagues reported a negative correlation between lignin content in peanut seed and rate of infection by Aspergillus flavus (2006). While evaluating detached stem assays for resistance to Sclerotinia blight, Brenneman et al. (1988) observed that older stem tissue was less susceptible than younger tissue to Sclerotinia blight, and hypothesized Sclerotinia minor might be deterred by the greater lignin and/or decreased sugar content in older stems.
Few studies have quantified lignin in peanut stems and most reports are for perennial forage species such as Arachis glabrata, A. pintoi, and A. repens (Terrill et al., 1996; Gomes et al., 2011; Ferreira et al., 2012). In crops produced for forage or biofuels, cultivars with lower levels of lignin are generally more desirable than those with high levels (Hisano et al., 2009; Jung, 1989). Foster et al. (2012) evaluated the digestibility of peanut stover from three cultivars (C-99-R, Georgia 01-R, York) and also measured lignin content at growth stages 2 and 8 (Boote, 1982). As expected, more lignin was present in the dried, above-ground tissue at growth stage R8 (95-104 g/kg) than at R2 (42-71 g/kg). However, significant differences in lignin content among the three cultivars were not observed at either growth stage (Foster et al., 2012). Forage quality is currently not a priority for U.S. peanut breeding programs, but low-lignin haulm may be a valuable trait in developing countries.
The objectives of this study were to quantify lignin in multiple genotypes of peanut, and to test if preformed lignin content could be used as a predictor of susceptibility or resistance to Sclerotinia blight in genotypes of runner, spanish, and virginia peanuts.
Materials and Methods
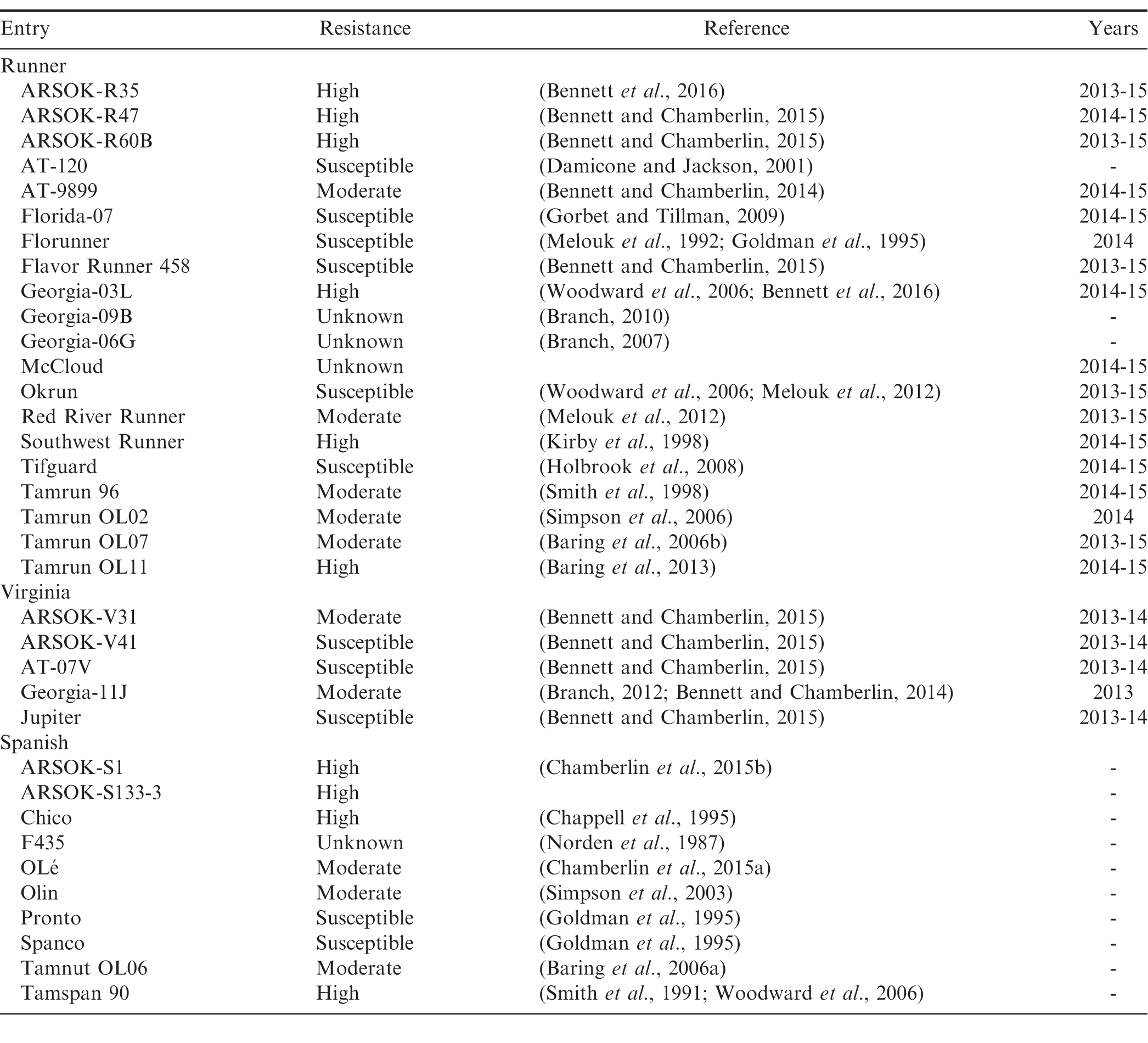
A total of 35 peanut cultivars and advanced breeding lines, including twenty runner (Arachis hypogaea subsp. hypogaea var. hypogaea), ten spanish (A. hypogaea subsp. fastigiata var. vulgaris), and five Virginia (A. hypogaea subsp. hypogaea var. hypogaea) market types were evaluated for preformed lignin content (Table 1). The entries included genotypes known to vary in susceptibility to Sclerotinia minor. To avoid confounding effects from natural infections and because lignin content in greenhouse-grown corn was shown to be comparable to lignin from field-grown plants (Hatfield et al., 2008), peanuts were grown in the greenhouse. Four seeds of each genotype were planted in a 30.5-cm-diam bulb pan filled with a soil-less potting mix (Metro-Mix 350, Sun Gro Horticulture, Agawam, MA). Seedlings were culled to one plant per pot at the 2 to 3 true-leaf stage. Plants were grown in a greenhouse set at 22-32 C with 14 hrs of supplemental lighting from 600-watt LED lights (Grow Pro, LED Lighting for Less). Plants were fertilized with 100 ml of 0.2%; ammonium nitrate biweekly, beginning at five weeks after planting. Three plants of each genotype were used per experiment, and genotypes were randomized within blocks by market type on greenhouse benches. The first experiment was planted on 18 Mar. 2014; the second experiment was planted on 15 Apr. 2014.
Plants were uprooted and stem samples were collected at 127 to 129 days after planting. Five-cm-long stem pieces were cut from the second pair of lateral branches from the bottom. The stem sections were cut so that the third node on the lateral branch (counting from the main stem) was in the center. The two stem pieces were dried for three to seven days in an envelope at 55C, and then stored in a closed plastic container containing desiccant at room temperature.
Lignin Extraction
The two stem pieces were ground together using a Wiley Mini-Mill (Thomas Scientific, Swedesboro, NJ) with a 0.425-mm screen mesh. The Mini-Mill was cleaned thoroughly between samples by vacuuming out remaining material and wiping down surfaces with 70%; ethanol. The ground tissue, approximately 500 mg, was weighed into a 30-mL Oak Ridge centrifuge tube (Thermo Fisher Scientific, Waltham, MA). Cell wall components were isolated using a series of 14 extractions with 10 mL each of the following solutions: 50 mM NaCl buffer (1x), 80%; ethanol (4x), acetone (2x), 2:1 chloroform:methanol (5x), and acetone (2x). The extractions removed compounds such as sugars, organic acids, fatty acids, flavenols, and pigments, which interfere with spectrophotometric analyses (Peltier et al., 2009). For the few samples that were significantly greater than 500 mg, an additional 2 mL of each solution was used for every additional 100 mg. After each solution was added, tubes were capped, placed horizontally, and shaken on an orbital shaker for 15 min. The tubes were then centrifuged for 10 min at 8360 x g, and the supernatant was removed with an aspirator. After the last acetone extraction, samples were allowed to air-dry in a fume hood, and were dried further at 55C overnight, at which point the samples remained at a constant weight.
The acetyl bromide method was used to estimate lignin content (Hatfield et al., 1999; Fukushima and Hatfield, 2004). A subsample (approximately 25 mg weighed accurately ±0.1mg) of dried sample was transferred to a 10-mL glass culture tube with screw-top Teflon-lined cap. Acetyl bromide reagent (2.5 mL) was added to the sample and the tube was incubated in a 50 C water bath for 2 hr. After cooling, the sample was mixed and a 1-mL subsample was transferred to a microcentrifuge tube. Non-dissolved proteins and carbohydrates were pelleted by centrifugation at 10000 x g, and 500 μL of the supernatant was mixed with 9.5 L of glacial acetic acid (7.15 mL), 2 M NaOH (2 mL), and 0.5 M hydroxyl amine (0.35 mL) before measuring UV absorption spectrum (250 to 350 nm) in a scanning spectrophotometer. Concentrations of lignin were based upon the maximum absorbance reading at 280 nm, and controls for background absorbance were included. Two subsamples were analyzed for each genotype, and the mean of both subsamples was used in the data analyses. Alfalfa stem standards were included with each batch of samples in order to standardize acetyl bromide lignin responses. The extinction coefficient for alfalfa was used to determine the relative lignin content in each sample. Total lignin was measured as proportion of the total cell wall (mg/mg CW).
Field Data
Resistance to Sclerotinia minor in the field was evaluated in unrelated experiments in 2013 to 2015 for a subset of the runner entries, and in 2013 and 2014 for the virginia entries, (Bennett and Chamberlin, 2014, 2015, 2016). Entries were screened at the Caddo Research Station in Fort Cobb, OK, in naturally infested fields that are planted every other year in peanuts. Only data from runner and virginia entries evaluated for two or three years were used. Seed of the entries were planted in two 4.6-m-long rows with 0.9-m beds at a density of 14.4 seeds per meter. A randomized complete block design was used, with a minimum of three blocks/replications. Field plots were irrigated using a center pivot and managed for weeds. Foliar diseases and southern blight were managed over the three years with applications of the following fungicides: chlorothalonil, chlorothalonil + difenoconazole, cyproconazole, flutriafol, propiconazole, tebucanozole, and/or trifloxystrobin. Plots were not sprayed to control Sclerotinia blight. Disease ratings for Sclerotinia blight were taken approximately one to two weeks before harvest, by counting the number of 15.2-cm-sections within each plot that had symptoms caused by S. minor.
Statistical Analyses
Mean stem lignin content among genotypes were compared within market types by one-way ANOVA using PROC GLIMMIX of SAS (SAS, ver. 9.3, SAS Institute, Cary, NC), with block and experiment as random factors. Mean incidence of Sclerotinia blight was analyzed similarly, but with year and block as random factors. The Type I error rate for pairwise comparisons of breeding lines and cultivars was controlled at 𝜶 = 0.05 using the ADJUST=TUKEY option. The relationship between mean stem lignin content and mean incidence of disease in the field was estimated using Pearson's correlation coefficient SAS with PROC CORR.
Results and Discussion
When the stems were harvested at 127-129 days after planting, the Spanish entries were approximately at the R8 to R9 growth stages, and the runner and most virginia entries were at the R8 stage (Boote, 1982). The virginia entry Georgia-11J was an exception by being at the R7 stage at harvest.
Lignin content
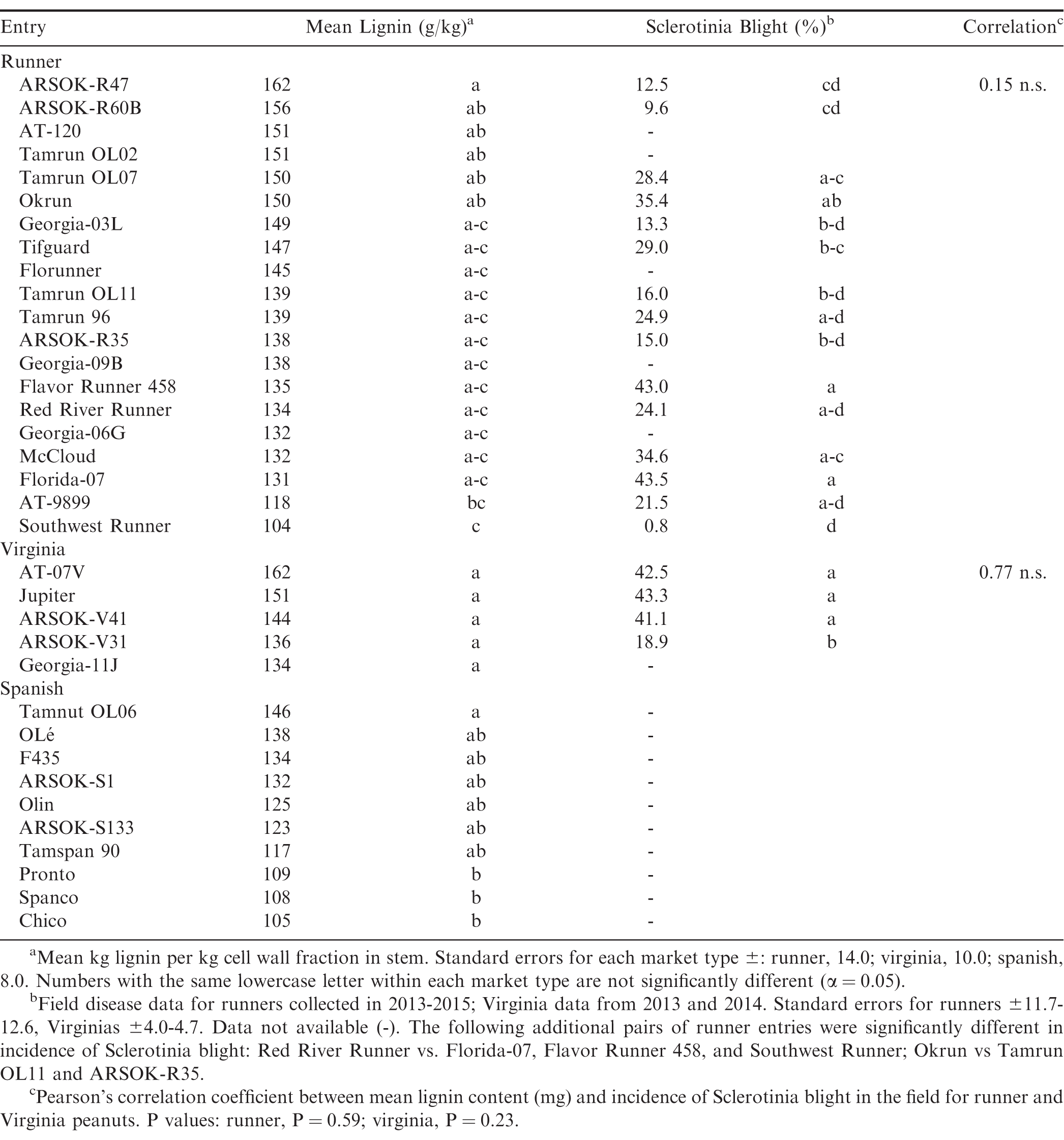
Significant differences in lignin content were found within the runner market types (F = 2.65; df = 19, 94; P 0.01; Table 2). The runner entry with the numerically highest lignin content was the advanced breeding line ARSOK-R47 (162 g/kg). ARSOK-R47 did not differ significantly from the other entries in lignin content except AT-9899 (P = 0.03) and Southwest Runner (P 0.01). Mean lignin content in the six Virginia entries varied from 162 g/kg (AT-08V) to 134 g/kg (Georgia-11J), but the differences were not statistically significant (F = 1.34; df = 4, 24; P = 0.28; Table 2). The Spanish entries differed in lignin content (F = 3.39; df = 9, 48; P 0.01; Table 2), and Tamnut OL06 had the highest stem lignin (146 g/kg). Tamnut OL06 was statistically similar to all other entries in lignin content except Pronto (P = 0.03), Spanco (P = 0.03), and Chico (P = 0.01).
Field resistance to Sclerotinia blight and association with lignin content
Environmental conditions for Sclerotinia blight were moderately conducive in 2013, favorable in 2014, and not favorable in 2015. The mean incidence of Sclerotinia blight for the highly susceptible cultivar Flavor Runner 458 was 42.1%; in 2013, 72%; in 2014, and 13.5%; in 2015. Significant differences among entries were found for both runner (F = 9.09; df = 14, 152.1; P 0.01) and virginia (F = 6.67; df = 3, 46; P 0.01) entries (Table 2). The most susceptible entries were Florida-07 (43.5%; diseased), Flavor Runner 458 (43.0%;), and Okrun (35%;). ARSOK-R47, ARSOK-R60B, and Southwest Runner were most resistant to Sclerotinia blight with disease incidences of 12.5%;, 9.6%;, and 0.8%;, respectively. Among the virginia entries, ARSOK-V31 was significantly more resistant than AT-07, Jupiter and ARSOK-V41 (P 0.01). No significant correlations between lignin content and field resistance to Sclerotinia blight were found within the runner (r = 0.15; F = 0.30; P = 0.59) or virginia entries (r = 0.84; F = 7.34; P = 0.07; Table 2). Therefore, lignin content does not appear to be reliable predictor of Sclerotinia blight resistance.
The number of genotypes sampled for virginia and spanish peanuts were small and may not be a representative sample of either market type. A larger number (n = 20) of runner entries with a range of susceptibility to Sclerotinia blight were evaluated. While some of the more resistant entries, such as ARSOK lines R47 and R60B, had high levels of lignin, others did not. Southwest Runner, the cultivar most resistant to S. minor in the field trials, had the least stem lignin of all peanut entries. Thus, the negative correlation between lignin content and disease resistance found in other pathosystems (e.g. Banniza et al., 2005; Xu et al., 2011) does not seem to apply to peanuts and Sclerotinia blight. Others have also failed to find strong evidence supporting the role of preformed lignin in plant defense. Peltier et al. (2009) found soybean lines with greater stem lignin were actually more susceptible to S. sclerotiorum. Pea lines with high lignin content had slower lesion development but did not survive beyond two weeks, unlike the most resistant lines (Porter et al., 2009). However, this study only examined preformed lignin in peanut, and the quantity of lignin formed on-site in response to infection may be better correlated with disease resistance. In addition, further study is needed to determine if peanut lignin content is as consistent as it is in corn (Hatfield et al., 2008) when grown in different environments such as the field and greenhouse.
It was expected that more lignin would be present in spanish peanut stems to support the upright spanish plant architecture than in the prostrate runner and virginia lines. However, as a group, the spanish entries had lower mean lignin (x̄ = 124 g/kg; SD = 13.9) than virginias (x̄ = 140 g/kg; SD = 13.4) or runners (x̄ = 145 g/kg; SD = 11.5). Lignin content in plants generally increases with age (Buxton and Russell, 1988), but the Spanish market types were slightly more mature at harvest than the runners and virginias. Lignin is not the only component within the cell wall that contributes to plant structural integrity, and the density of specific cell types, such as fiber, vascular, and pith cells, can have a major impact upon total lignin within a whole stem, regardless of growth stage. In addition, cultivated peanut is known to have low genetic variability (Cuc et al., 2008), and peanut breeders have crossed different market types to introgress desirable traits into currently available cultivars. For example, many high-oleic runner and virginia cultivars available today derive their high-oleic trait from F435, a spanish peanut (Norden et al. 1987). While none of the high-oleic runner and virginia cultivars grown today have the upright canopy characteristic of the spanish market type, plant breeding may have affected lignin content among market types, if differences originally existed.
There are several methods for quantifying lignin (Hatfield and Fukushima, 2005), but most published studies of Arachis spp. use the acid detergent method (Van Soest, 1967). Foster and colleagues (2012), using the acid detergent method on three runner cultivars, found 95-104 g/kg lignin in a sample containing all aboveground tissue collected at the R8 stage. This study, using the acetyl bromide method on twenty runner entries at the same growth stage, obtained 104-162 g/kg lignin from stem sections of the second lowermost branch. Despite the popularity of the acid detergent protocol, several studies demonstrated that this method consistently underestimates lignin relative to other approaches such as the acetyl bromide method used here (Iiyama and Wallis, 1990; Hatfield et al., 1994; Jung et al., 1997; Fukushima and Hatfield, 2004). Results produced by different methods within and among crops are therefore difficult to compare, even before considering effects of the plant part analyzed or plant maturity. Fukushima and Hatfield (2004) compared several methods for quantifying lignin in several forage species including lower stems of alfalfa (in full bloom) and corn stalk (past anthesis). Mean estimates of alfalfa lignin were 92.5 g/kg with the acid detergent assay and 134.7 g/kg using acetyl bromide method. A greater difference between the two methods was found for the corn stalk: 24.8 g/kg for the acid detergent versus 91.9 g/kg for the acetyl bromide. From soybean stems collected at the R5 (beginning seed) stage, Peltier et al. (2009) obtained an average of 211 g of lignin/kg using the acetyl bromide method. Estimates of stem lignin content for the peanut genotypes in this study varied from 104 to 167 g/kg collected at the R8-R9 stage. Stem lignin levels in peanut thus appear to be lower than in soybean, and may be comparable to alfalfa. The new reduced-lignin cultivars of alfalfa, Hi-Gest (Alforex Seeds, Woodland, CA) and HarvXtra (Forage Genetics International, Nampa, ID), reportedly have 7 to 15%; less lignin than conventional alfalfa (company websites). In this study, the spanish cultivar Chico had 15%; less lignin (105 g/kg) compared to the mean for all spanish peanuts (124 g/kg), and cultivar Southwest Runner had 28%; less lignin (104 g/kg) than the mean for the runner genotypes (145g/kg). Chico (Chappell et al., 1995) and Southwest Runner are also resistant to Sclerotinia blight. Both cultivars may be good options for low-input producers in developing countries, and organic producers in this country, seeking to add crop value by feeding peanut hay to livestock.
Acknowledgements
The authors thank Angela Harting and Miranda Zibolski-Meyer for technical assistance. This research was supported by USDA-ARS CRIS Project No. 3072-21220-007-00D. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Literature Cited
S., Banniza, P Hashemi, T.D Warkentin, A Vandenberg, and A.R Davis (2005). The relationships among lodging, stem anatomy, degree of lignification, and resistance to mycosphaerella blight in field pea (Pisum sativum). Can. J. Bot 83: 954- 967.
Baring, M.R., Y Lopez, C.E Simpson, J.M Cason, J Ayers, and M.D Burow 2006 a Registration of 'Tamnut OL06' peanut Crop Sci 46: 2720- 2721.
Baring, M.R., C.E Simpson, M.D Burow, M.C Black, J.M Cason, J Ayers, Y Lopez, and H.A Melouk 2006 b Registration of 'Tamrun OL07' peanut Crop Sci 46: 2721- 2722.
M.R., Baring, C.E Simpson, M.D Burow, J.M Cason, and J.L Ayers (2013). Registration of 'Tamrun OL11' peanut. J. Plant Regist 7: 154- 158.
Bennett, R.S., and K.D Chamberlin 2014 Disease evaluations and agronomic traits of advanced peanut breeding lines in 2013 Oklahoma Agric. Exp. Sta., P-1042.
Bennett, R.S., and K.D Chamberlin 2015 Disease evaluations and agronomic traits of advanced peanut breeding lines in 2014 Oklahoma Agric. Exp. Sta., P-1044.
Bennett, R.S., and K.D Chamberlin 2016 Disease evaluations and agronomic traits of advanced peanut breeding lines in 2015 Oklahoma Agric. Exp. Sta., P-1047.
R.S., Bennett, M.E Payton, and K.D Chamberlin (2016). Three inoculation methods for evaluating Sclerotinia blight resistance in peanut. Peanut Sci 43: 52- 58.
N.H., Bhuiyan, G Selvaraj, Y Wei, and J King (2009). Role of lignification in plant defense. Plant Signal. Behav 4: 158- 159.
K.J Boote, (1982). Growth stages of peanut (Arachis hypogaea L.). Peanut Sci 9: 35- 40.
W.D Branch, (2007). Registration of 'Georgia-06G' peanut. J. Plant Regist 1: 120.
W.D Branch, (2010). Registration of 'Georgia-09B' peanut. J. Plant Regist 4: 175- 178.
W.D Branch, (2012). Registration of 'Georgia-11J' peanut. J. Plant Regist 6: 281- 283.
T.B., Brenneman, P.M Phipps, and R.J Stipes (1988). A rapid method for evaluating genotype resistance, fungicide activity, and isolate pathogenicity of Sclerotinia minor in peanut. Peanut Sci 15: 104- 107.
D.R., Buxton, and J.R Russell (1988). Lignin constituents and cell-wall digestibility of grass and legume stems. Crop Sci 28: 553- 558.
Chamberlin, K.D., R.S Bennett, J.P Damicone, C.B Godsey, H.A Melouk, and K Keim 2015 a Registration of 'OLé' peanut J. Plant Regist 9: 154- 158.
Chamberlin, K.D., J.P Damicone, M.R Baring, M.D Burow, C.B Godsey, R.S Bennett, H.A Melouk, and C.E Simpson 2015 b Registration of high-oleic peanut germplasm line ARSOK-S1 (TX996784) with enhanced resistance to Sclerotinia blight and pod rot J. Plant Regist 9: 103- 107.
G.F., Chappell, B.B Shew, J.M Ferguson, and M.K Beute (1995). Mechanisms of resistance to Sclerotinia minor in selected peanut genotypes. Crop Sci 35: 692- 696.
L.M., Cuc, E.S Mace, J.H Crouch, V.D Quang, T.D Long, and R.K Varshney (2008). Isolation and characterization of novel microsatellite markers and their application for diversity assessment in cultivated groundnut (Arachis hypogaea). BMC Plant Biol 8: 55.
Damicone, J.P., and K.E Jackson 2001 Field studies for the control of peanut diseases Oklahoma Agric. Exp. Sta., P-983.
C., Eynck, G Séguin-Swartz, W.E Clarke, and I.A Parkin (2012). Monolignol biosynthesis is associated with resistance to Sclerotinia sclerotiorum in Camelina sativa. Mol. Plant Pathol 13: 887- 899.
A.L., Ferreira, R.M Maurcio, F.D Fernandes, M.A Carvalho, A.K.B Ramos, and R.G Junior (2012). Ranking contrasting genotypes of forage peanut based on nutritive value and fermentation kinetics. Anim. Feed Sci. Technol 175: 16- 23.
J.L., Foster, G.C Lamb, B.L Tillman, J.J Marois, D.L Wright, and M.K Maddox (2012). In sacco degradation kinetics of fresh and field-cured peanut (Arachis hypogaea L.) forage harvested at different maturities. Anim. Feed Sci. Technol 171: 52- 59.
R.S., Fukushima, and R.D Hatfield (2004). Comparison of the acetyl bromide spectrophotometric method with other analytical lignin methods for determining lignin concentration in forage samples. J. Agric. Food Chem 52: 3713- 3720.
R., Godoy, O.D Smith, R.A Taber, and R.E Pettit (1985). Anatomical traits associated with pod rot resistance in peanut. Peanut Sci 12: 77- 82.
J.J., Goldman, O.D Smith, C.E Simpson, and H.A Melouk (1995). Progress in breeding Sclerotinia blight-resistant runner-type peanut. Peanut Sci 22: 109- 113.
D.I., Gomes, E Detmann, S. de C Valadares Filho, R.S Fukushima, M.A de Souza, T.N.P Valente, M.F Paulino, and A.C de Queiroz (2011). Evaluation of lignin contents in tropical forages using different analytical methods and their correlations with degradation of insoluble fiber. Anim. Feed Sci. Technol 168: 206- 222.
D.W., Gorbet, and B.L Tillman (2009). Registration of 'Florida-07' peanut. J. Plant Regist 3: 14- 18.
R.D., Hatfield, H.-J.G Jung, J Ralph, D.R Buxton, and P.J Weimer (1994). A comparison of the insoluble residues produced by the Klason lignin and acid detergent lignin procedures. J. Sci. Food Agric 65: 51- 58.
R., Hatfield, and R.S Fukushima (2005). Can lignin be accurately measured?. Crop Sci 45: 832- 839.
R.D., Hatfield, J Grabber, J Ralph, and K Brei (1999). Using the acetyl bromide assay to determine lignin concentrations in herbaceous plants: some cautionary notes. J. Agric. Food Chem 47: 628- 632.
R.D., Hatfield, J.M Marita, and K Frost (2008). Characterization of p-coumarate accumulation, p-coumaroyl transferase, and cell wall changes during the development of corn stems. J. Sci. Food Agric 88: 2529- 2537.
H., Hisano, R Nandakumar, and Z.-Y Wang (2009). Genetic modification of lignin biosynthesis for improved biofuel production. In Vitro Cell. Dev. Biol.-Plant 45: 306- 313.
C.C., Holbrook, P Timper, A.K Culbreath, and C.K Kvien (2008). Registration of 'Tifguard'peanut. J. Plant Regist 2: 92- 94.
K., Iiyama, and A.F.A Wallis (1990). Determination of lignin in herbaceous plants by an improved acetyl bromide procedure. J. Sci. Food Agric 51: 145- 161.
H.G Jung, (1989). Forage lignins and their effects on fiber digestibility. Agron. J 81: 33- 38.
H.G., Jung, D.R Mertens, and A.J Payne (1997). Correlation of acid detergent lignin and Klason lignin with digestibility of forage dry matter and neutral detergent fiber. J. Dairy Sci 80: 1622- 1628.
J.S., Kirby, H.A Melouk, T.E Stevens, D.J Banks, J.R Sholar, J.P Damicone, and K.E Jackson (1998). Registration of 'Southwest Runner' peanut. Crop Sci 38: 545- 546.
X.A., Liang, M Luo, and B.Z Guo (2006). Resistance mechanisms to Aspergillus flavus infection and aflatoxin contamination in peanut (Arachis hypogaea). Plant Pathol. J 5: 115- 124.
H.A., Melouk, C.N Akem, and C Bowen (1992). A detached shoot technique to evaluate the reaction of peanut genotypes to Sclerotinia minor. Peanut Sci 19: 58- 62.
H.A., Melouk, K Chamberlin, C.B Godsey, J Damicone, M.D Burow, M.R Baring, C.E Simpson, K.E Dashiell, and M Payton (2012). Registration of 'Red River Runner' Peanut. J. Plant Regist 7: 22- 25.
B.M., Moerschbacher, U Noll, L Gorrichon, and H.-J Reisener (1990). Specific inhibition of lignification breaks hypersensitive resistance of wheat to stem rust. Plant Physiol 93: 465- 470.
A.J., Norden, D.W Gorbet, D.A Knauft, and C.T Young (1987). Variability in oil quality among peanut genotypes in the Florida breeding program. Peanut Sci 14: 7- 11.
A.J., Peltier, R.D Hatfield, and C.R Grau (2009). Soybean stem lignin concentration relates to resistance to Sclerotinia sclerotiorum. Plant Dis 93: 149- 154.
L.D., Porter, G Hoheisel, and V.A Coffman (2009). Resistance of peas to Sclerotinia sclerotiorum in the Pisum core collection. Plant Pathol 58: 52- 60.
C.E., Simpson, M.R Baring, A.M Schubert, M.C Black, H.A Melouk, and Y Lopez (2006). Registration of 'Tamrun OL 02' peanut. Crop Sci 46: 1813.
C.E., Simpson, M.R Baring, A.M Schubert, H.A Melouk, Y Lopez, and J.S Kirby (2003). Registration of 'OLin' Peanut. Crop Sci 43: 1880- 1881.
O.D., Smith, C.E Simpson, M.C Black, and B.A Besler (1998). Registration of 'Tamrun 96' peanut. Crop Sci 38: 1403.
O.D., Smith, C.E Simpson, W.J Grichar, and H.A Melouk (1991). Registration of 'Tamspan 90' peanut. Crop Sci 31: 1711.
L., Taiz, and E Zeiger (1991). Plant Physiology. Redwood City, CA: Benjamin/Cummings Publishing Company, Inc. ., .
T.H., Terrill, S Gelaye, S Mahotiere, E.A Amoah, S Miller, R.N Gates, and W.R Windham (1996). Rhizoma peanut and alfalfa productivity and nutrient composition in central Georgia. Agron. J 88: 485- 488.
C.P., Vance, T.K Kirk, and R.T Sherwood (1980). Lignification as a mechanism of disease resistance. Annu. Rev. Phytopathol 18: 259- 288.
P.J Van Soest, (1967). Development of a comprehensive system of feed analyses and its application to forages. J. Anim. Sci 26: 119- 128.
Woodward, J.E., T.B Brenneman, R.C Kemerait Jr, A.K Culbreath, and J.R Clark 2006 Sclerotinia blight in Georgia and evidence for resistance to Sclerotinia sclerotiorum in runner peanuts Plant Health Prog. Available at http://www.plantmanagementnetwork.org/pub/php/research/2006/runner/.
L., Xu, L Zhu, L Tu, L Liu, D Yuan, L Jin, L Long, and X Zhang (2011). Lignin metabolism has a central role in the resistance of cotton to the wilt fungus Verticillium dahliae as revealed by RNA-Seq-dependent transcriptional analysis and histochemistry. J. Exp. Bot 62: 5607- 5621.
Notes
- First and fourth authors: Research Plant Pathologist and Research Biologist, USDA-ARS, Wheat, Peanuts and Other Field Crops Research Unit, Stillwater, OK 74075. Second author: Research Plant Physiologist, USDA-ARS, U.S. Dairy Forage Research Center, Madison, WI 53706. Third author: Professor, Department of Statistics, Oklahoma State University, Stillwater, OK 74078. [^] *Corresponding author's E-mail: rebecca.bennett@ars.usda.gov.
Author Affiliations