Introduction
Establishing a non-yield-limiting, uniform plant stand is an important factor in growing a successful peanut ( Arachis hypogaea L.) crop. The University of Georgia's recommendation for peanut seeding rate in single-row peanuts is 19.7 seeds/m of row in an effort to obtain a final stand of 13.1 plants/m (Beasley et al., 1997). While 13.1 plants/m is the standard by which recommendations have been made, research has shown that in some cases, yield potential can be maintained at reduced plant stands (Augusto et al., 2010; Bell et al., 1987; Tewolde et al., 2002).
While data is limited on true plant stand effects on peanut yields, numerous other studies have described the relationship between seeding rates and pod yields. Sorenson et al. (2004) reported an 8.5% pod yield increase when seeding rates were increased from 10 to 20 seeds/m. Sconyers et al. (2007) showed increased yield at 22.6 seeds/m versus 12.5 seeds/m, although yields at the high rate were not greater than those at 17.4 seeds/m. When testing seeding densities from 34 to 123 kg/ha, Wehtje et al. (1994) reported maximum yield at 101 kg seed/ha. With Spanish peanuts in Oklahoma, yield increased linearly as seeding rate increased from 7 to 22 seeds/m (Chin Choy et al., 1982). That research went on to show that yield per plant decreased as population increased, although the total yield increased at a greater rate than the yield per plant decreased. Sternitzke et al. (2000) described similar results, in which pod mass per plant increased at lower plant stands; however, total pod yield was higher at higher plant stands. When testing intra-row spacings from 5 to 40 cm, Kvien and Bergmark (1987) reported increased yields at decreased spacing.
Research has shown that farmer stock grade (% total sound mature kernels, TSMK) can also be affected by plant stand. Mozingo and Coffelt (1984) reported a higher farmer stock grade in a virginia-type peanut in a higher versus a lower plant population. These results were similar to Sorenson et al. (2004), which showed a 0.7 point increase in TSMK when seeding rate was increased from 10 to 20 seeds/m. Sconyers et al. (2007) found similar results, with higher TSMK at a seeding rate of 17.4 seeds/m versus a rate of 12.5 seeds/m in one study and higher TSMK at 22.6 seeds/m than at 12.5 seeds/m in another. Numerous other studies have reported increased grade at increased plant population (Cox and Reed, 1965; Wynne et al., 1974). However, plant stand and seeding rate effects on market grade are not always consistent. Chin Choy et al. (1982) found no differences for TSMK values between seeding rates of 7, 15, and 22 viable seeds/m while Knauft et al. (1981) showed a significant difference in grade in only one of six cultivars when varying plant population. Hurt et al. (2004) reported mixed results with a 7 plants/m stand having a higher TSMK than a 17 plants/m stand in two of five experiments, the greater stand having a higher TSMK in two of five experiments, and no difference in TSMK in the fifth experiment.
It has been widely reported that increasing plant stands decreases incidence of tomato spotted wilt Tospovirus (TSWV). While the mechanism behind this phenomenon is not completely understood, there are multiple theories as to why it occurs. One potential reason for the decrease could be less exposed ground at higher plant stands. Two species of thrips; western flower thrips [Frankliniella occidentalis (Pergande)], and tobacco thrips [Frankliniella fusca (Hinds)] which vector the disease in peanut, are thought to be more attracted to this exposed ground than to ground covered by a crop canopy (Reddy and Wightman, 1988). Another line of thought is that the reduction may be a function of fewer plants being fed on by thrips as a percentage of the total number of plants in the field (Brown et al., 2005). Wehtje et al. (1994) reported a decrease in TSWV incidence when increasing seeding rates from a low of 34 to a high of 123 kg/ha. Similarly, Gorbet and Shokes (1994) reported increased TSWV as within-row plant spacing increased. Field surveys in Georgia in 1992 revealed a reduction in the percentage of peanut plants infected with TSWV when plant density increased from <6.6 to 6.6-13.12 to >13.12 plants/m (Culbreath et al., 1999). While data trends toward reduced TSWV at increased population, this does not always hold true, as Sconyers et al. (2007), found no differences in TSWV when rating the disease across three seeding rates.
Stem rot, caused by the fungus Sclerotium rolfsii, has also been shown to be affected by seeding rates and plant populations. Sconyers et al. (2005) reported increases in stem rot incidence when plant spacing decreased from 30 cm to 5 cm in 5-cm increments. Wehtje et al. (1994) found that stem rot increased in a linear fashion when seeding rate increased from 34 kg/ha to 124 kg/ha. Similarly, Sconyers et al. (2007) reported increased stem rot in plots seeded at 22.6 seeds/m when compared to those seeded at 12.5 and 17.4 seeds/m, respectively. Augusto et al. (2010) reported consistent increases in stem rot at increased plant populations in areas with significant levels of the pathogen.
The multitude of potential causes for a poor plant stand and a lack of previous results in peanut necessitated research designed to better understand the agronomic and pathological ramifications of replant decisions. There were three main objectives of this study. The first was to determine the effect of plant stand on pod yield, market grade, TSWV, and stem rot in peanut seeded in single rows. Building on the first objective; the second objective was to determine at what plant stands a peanut crop gains an advantage from replanting. The last objective was to determine the method of replanting that was most advantageous when replanting is warranted.
Materials and Methods
Irrigated field trials were conducted on a Tifton loamy sand (fine-loamy, kaolinitic, thermic Plinthic Kandiudults) at the University of Georgia (UGA) Ponder Farm in 2012 and at the NESPAL Farm in 2013; both near Tifton, GA. Tests were also conducted under irrigation on a Greenville sandy loam (fine, kaolinitic, thermic Rhodic Kandiudults) at the UGA Southwest Research and Education Center near Plains, GA in 2011, 2012, and 2013.
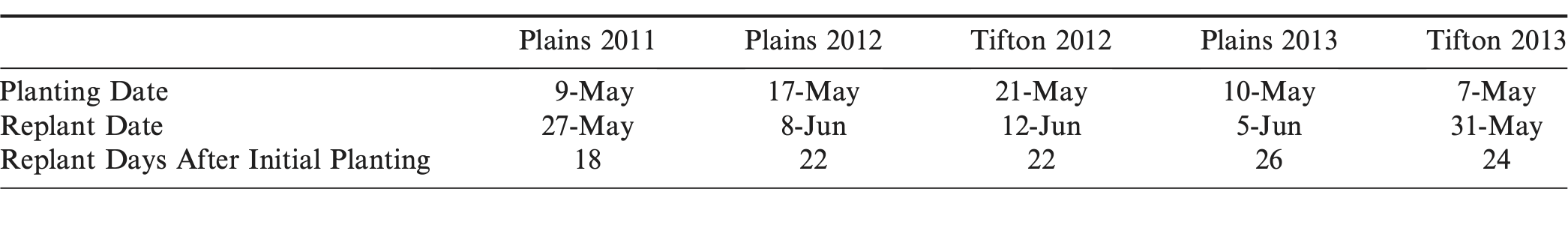
Land preparation at the Tifton sites included disc-harrowing, deep-turning with a moldboard plow to a depth of 30 to 35 cm, and rotary-tilling to form peanut beds 1.8 m wide. Preparation at the UGA Southwest Research and Education Center was similar with the exception of 2012, in which moldboard plowing did not occur. All fertilizer requirements and applications, including those for Ca and B, were based on UGA Extension recommendations (Harris, 1997). Peanut cultivar Georgia-06G (Branch, 2007) was planted in single rows at a depth of 5 cm and a row spacing of 0.91 m in rows 12.2 m in length. Phorate insecticide was applied in-furrow at a rate of 1.12 kg a.i./ha. Planting dates and replant dates for each site-year (location by year) are listed in Table 1.
Trials were designed as a randomized complete block design with four replications, in a six by three factorial arrangement with six peanut plant stands (3.3, 4.9, 6.6, 8.2, 9.8, and 11.5 plants/m) and three replant options of either 1) leave the initial peanut plant stand and do not replant; 2) retain the original plant stand, move the planter units 8.9 cm to the side and supplement with additional seed at a reduced seeding rate approximately 7-14 days after full stand emergence (Table 2); or 3) terminate the original peanut plant stand with glufosinate ((RS)-2-Amino-4-(hydroxy(methyl)phosphonoyl)butanoic acid) herbicide using a backpack sprayer at a rate of 655 g a.i./ha and replant at the full 19.7 seeds/m seeding rate.
A non-replanted control plot of the UGA recommended 13.1 plants/m was also included. All plots were initially planted at 19.7 seeds/m and upon full emergence were thinned by hand to the desired plant stands. To hand thin, all plants within the row were counted and then plants were removed until the desired number of plants per row was achieved. While plant-to-plant spacing was not exact, it was generally consistent.
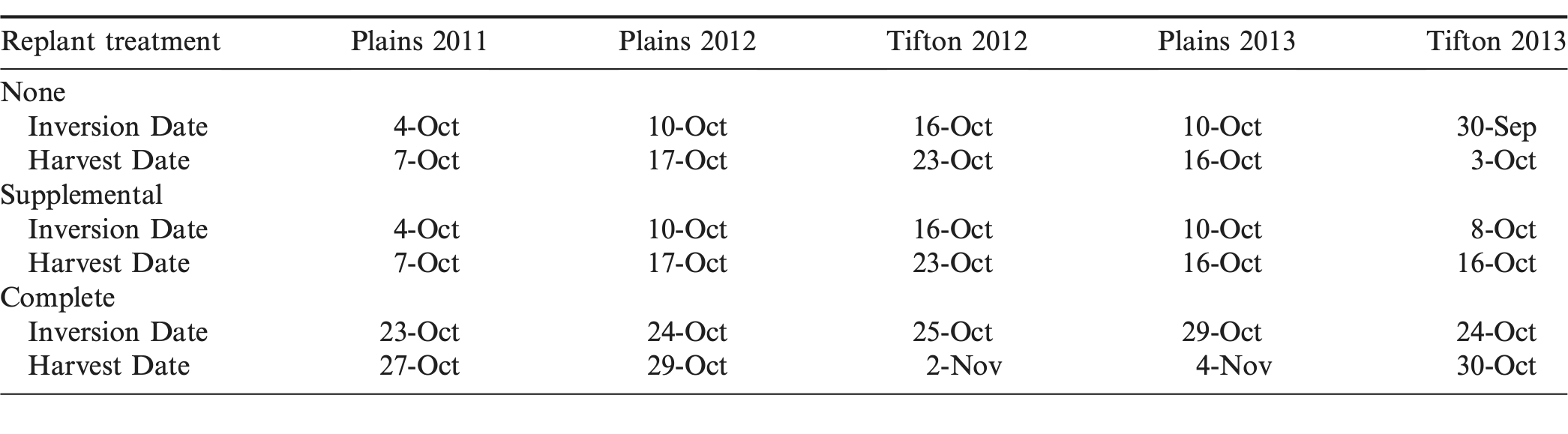
Fungicide applications were made based on guidelines provided by the high risk model of the Peanut Disease Risk Index (Kemerait et al., 2011). As such, fungicide programs were started at the onset of flowering, which was typically 35-40 days following the initial planting. Treatments were evaluated for pod yield, grade, incidence of TSWV (Plains 2011 and 2012; Tifton 2012 and 2013) and incidence of stem rot (Plains 2012; Tifton 2012 and 2013). Tomato spotted wilt virus levels were too low to warrant rating in Plains 2013 and stem rot levels were too low to warrant rating in Plains in 2011 and 2013. Ratings for TSWV were conducted on 23 September 2011, and 28 September 2012 in Plains; and 28 September 2012 and 30 September 2013 in Tifton. Peanut maturity was determined at each site-year using the hull-scrape maturity profile method (Williams and Drexler, 1981). Inversion and harvest dates are listed in Table 3. Ratings for stem rot were conducted on the date of plant inversion for each treatment. There were two inversion and two harvest dates for each site-year, except in Tifton 2013 which had three inversion and harvest dates. All peanuts receiving the no-replant and supplemental seed treatments were inverted and harvested earlier than those destroyed and replanted at the full seeding rate. In Tifton 2013, the no-replant treatment was inverted and harvested first, followed by the supplemental and then the complete replant treatment. Peanuts were inverted using a two-row KMC digger-shaker-inverter (Kelley Manufacturing, Tifton, GA) and harvested using a two-row Lilliston peanut combine. Yields were adjusted to 7% moisture. Peanuts were graded by the USDA Federal-State Inspection Service in Tifton, GA (Davidson et al., 1982).
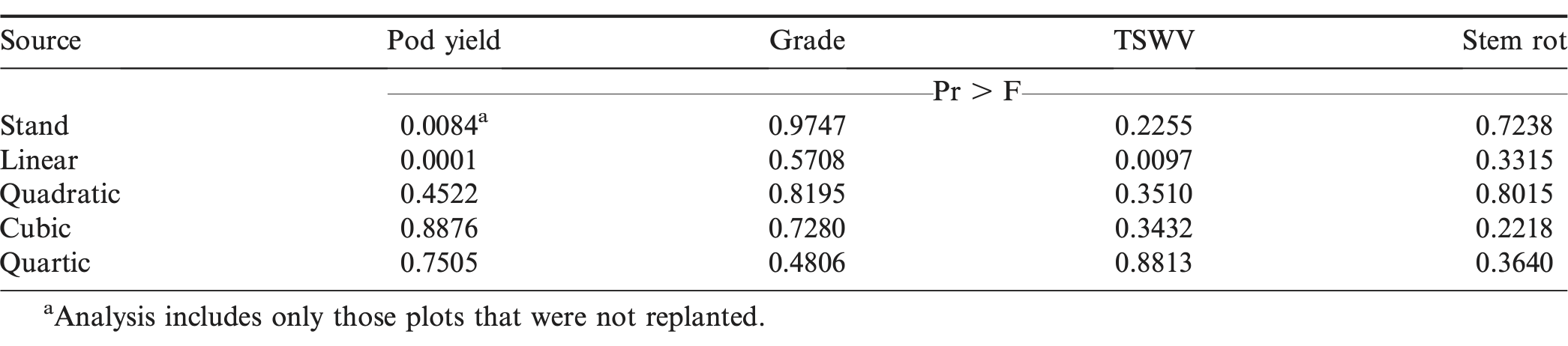
For the purpose of determining plant stand effects on pod yield, grade, TSWV and stem rot; non-replanted plots were analyzed separately with PROC MIXED in SAS 9.3 (SAS Institute, Cary, NC). Plant stand was treated as a fixed effect, while site-year (representing location by year effect) were treated as random effects. There were no interactions between site-years and plant stand for any of the factors measured, so further analyses were completed with data combined across locations and years. Because plant stand followed a logical structure, further analyses were completed using orthogonal polynomial contrasts in PROC MIXED in SAS 9.3.
For the purpose of determining replant treatment and replant treatment by plant stand effects, all data were analyzed together using PROC MIXED in SAS 9.3. Replant data were analyzed by analysis of variance and differences among least square means were determined using multiple pairwise t-tests (P≤0.05). Replant treatment and plant stand were treated as fixed effects, while site-years, replications, and interactions with these factors were treated as random effects. Because replant treatment by plant stand interactions were present for yield, replant treatment effects are reported for each plant stand separately. There were no interactions between site-years, plant stands, and replant treatments. As a result, plant stand by replant treatment data is combined and reported across locations and years.
Results
Plant stand
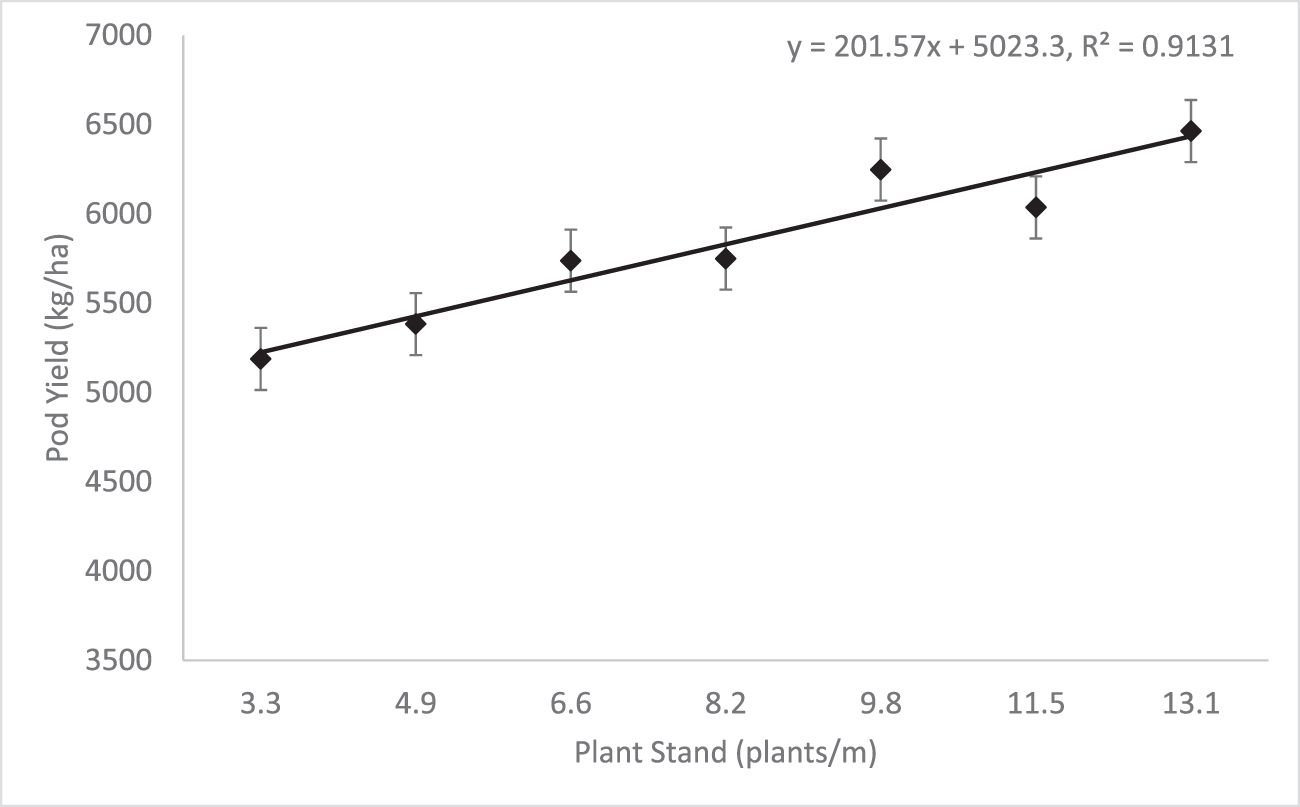
Plant stand significantly affected pod yield (Table 4). Only non-replanted plots were used in this analysis, so as not to confound plant stand results with planting dates or replanting treatment. When looking further at the data, there was a significant positive linear trend for yield (P<0.0001, R2=0.9131), indicating that as plant stand increased, pod yield increased proportionately (Figure 1). This positive linear trend in pod yield is similar to results reported by Chin Choy et al. (1982), who found a linear yield trend in seeding rates of 7, 15, and 22 seeds/m. Linear trendline analysis of the data indicated that for every unit increase in plant stand, a resultant 202 kg/ha increase in pod yield would be expected.
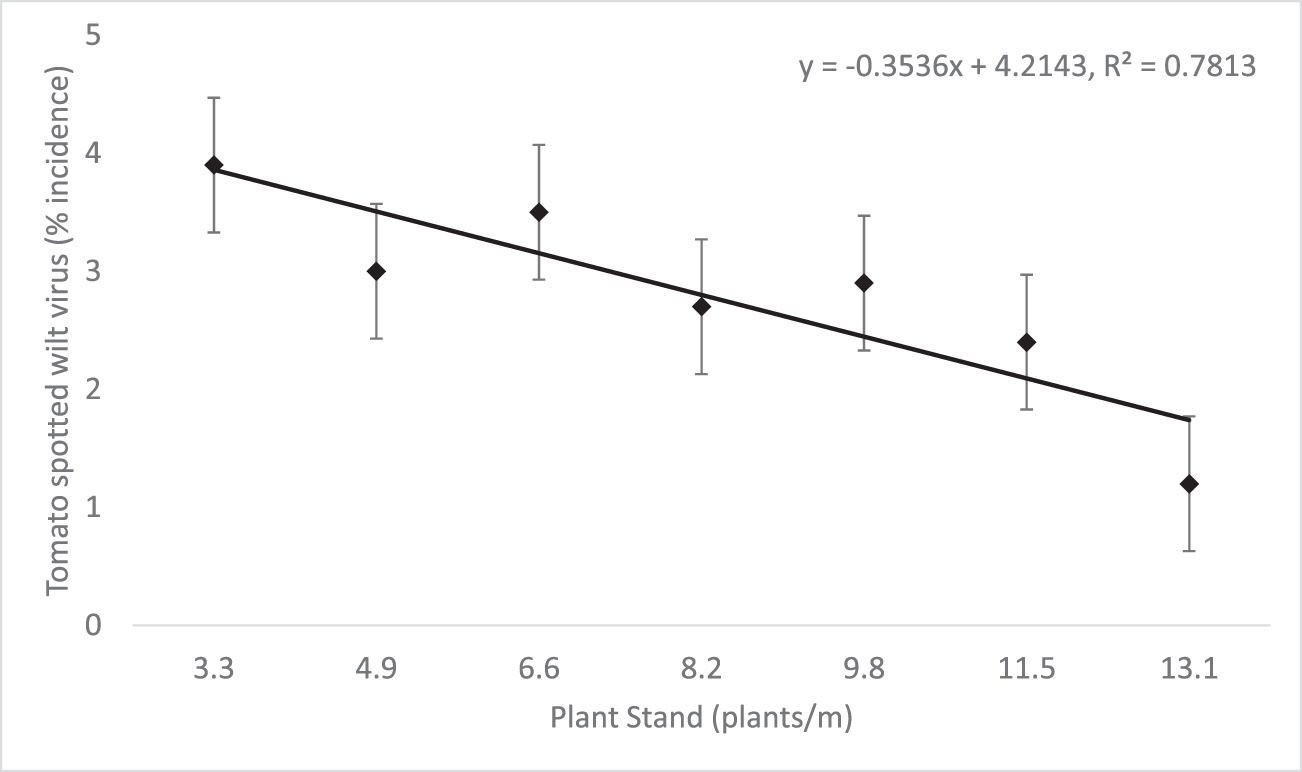
Overall, TSWV incidence was low. While the main effect of plant stand was not significant for TSWV incidence (Table 4), there was a significant negative linear trend indicating that for every unit increase in plant stand, TSWV incidence decreased by 0.4 percentage points (Figure 2). This reduction in TSWV as plant stand increased is similar to findings in multiple other studies (Gorbet and Shokes, 1994; Wehtje et al., 1994; Culbreath et al., 1999; Hurt et al. 2004).
Neither grade nor stem rot incidence were affected by plant stand. The majority of literature reports that increased plant stand leads to increased stem rot incidence (Wehtje et al. 1994; Sconyers et al., 2005; Sconyers et al., 2007). The lack of differences observed in our trials could be due to the exhaustive methods used to control initiation and spread of the disease. In the reference studies where disease incidence increased as plant stand increased, either a less-intensive fungicide program was used or plots were inoculated with the pathogen in order to ensure heavy pressure and uniformity of disease across the field. In this study, the high-risk model of the Peanut Disease Risk Index (Kemerait et al., 2011) was used at all site-years resulting in a robust fungicide program for preventative maintenance of incidence and spread of disease, and overall stem rot pressure was low. Augusto et al. (2010) reported that a greater number of plants/m of row are allowable without increasing stem rot incidence in areas of low pressure. While they described low incidence as 3% and below, average incidence of 4% in our study was only slightly above that level.
Replant Treatment
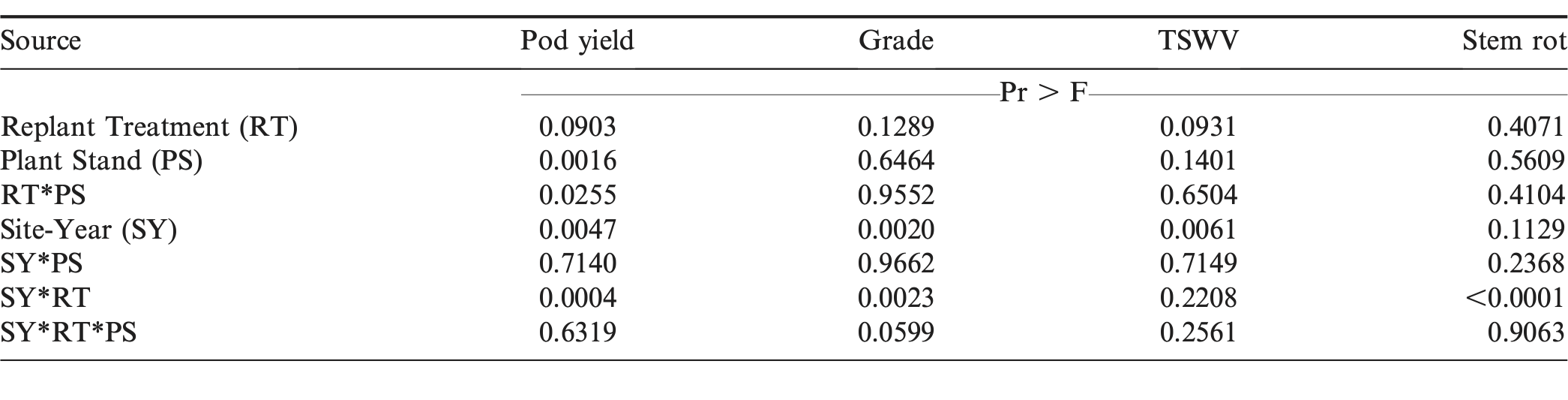
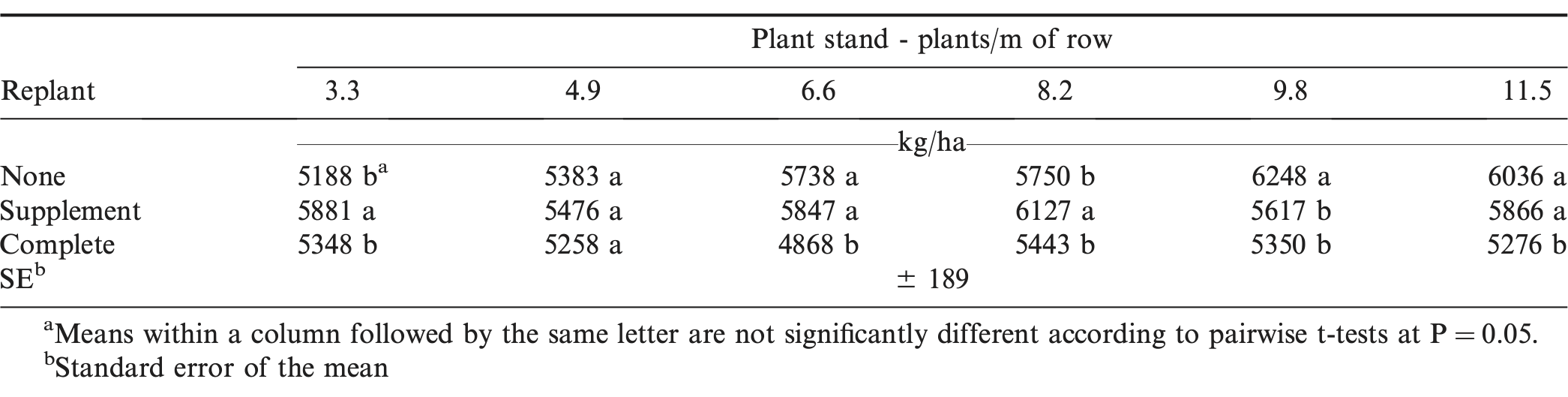
The interaction of plant stand by replant treatment for yield indicated that the optimum replant treatment was dependent on initial plant stand (Table 5). The supplemental replant treatment significantly increased yield over not replanting at two initial plant stands; 3.3 plants/m and 8.2 plants/m (Table 6). At an initial stand of 3.3 plants/m, yield increased by 13.4% by supplementing with additional seed, while at an initial stand of 8.2 plants/m, supplementing with additional seed increased yield by 6.6%. Pod yields were similar between non-replanted and supplemental treatments at initial stands of 4.9, 6.6, and 11.5 plants/m and yields were reduced by 10.1% when supplementing an initial stand of 9.8 plants/m. Completely replanting did not improve yield over the non-replanted treatment at any initial plant stand. At 6.6, 9.8, and 11.5 plants/m, yield was reduced by a range of 12.6 to 15.2% when completely replanting versus not replanting, while at 3.3, 4.9, and 8.2 plants/m, no yield differences were observed. When comparing supplemental and complete replanting, a yield advantage ranging from 9.1 to 16.7% was observed for the supplemental treatment at four of six initial plant stands (3.3, 6.6, 8.2, and 11.5 plants/m), while yields were equal at 4.9 and 9.8 plants/m. When taking all of these results into account, the supplemental replant treatment was the superior option when compared to completely replanting. Grade, TSWV, and stem rot were not affected by replant treatment or the interaction of plant stand by replant treatment.
Discussion
The results from these trials illustrate the importance of establishing the recommended plant stand on the initial planting date. The strong, positive linear trend observed between plant stand and pod yield shows that pod yield potential is increased as plant stand per meter of row increases. A minimum of 9.8 plants/m were needed in order to statistically equal the yield obtained at the 13.1 plants/m standard (Fig. 1). While overall pressure of the disease was low, increased plant stand also helped to reduce TSWV incidence in a linear fashion (Fig. 2), which is consistent with previously reported results. While yield and TSWV trended in opposite directions in relation to plant stand, effects of the disease on yield was likely minimal due to low overall disease pressure, so differences are almost exclusively attributed to the changes in stand.
Supplementing the initial stand increased yield at initial stands of 3.3 and 8.2 plants/m by 13.4 and 6.5%, respectively, and did not increase yield at any other stand. While supplemental seed addition benefitted both of these initial stands, the smaller increase observed at 8.2 plants/m is likely due to that stand being closer to optimum than the 3.3 plants/m initial stand. Destroying the initial stand and completely replanting was never a viable option when compared to either the non-replanted or supplemental replant treatments. While the completely replanted plots were not limited by season length and were harvested separate from the other treatments according to maturity determination via the hull scrape method, the later planting date was considered the primary cause of lost yield when compared to the other replant options. Initial planting dates ranged from 7 May to 21 May, with an average date of 13 May; while replant date ranged from 27 May to 12 June, with an average date of 4 June. The yield reduction at the later planting date was consistent with multiple other studies that showed decreased yield for peanut planted in late-May and June versus peanut planted in mid-May (Beasley, 2013; McKeown et al., 2001; Tillman et al., 2007). This is not always the case, however, as extreme weather conditions or pest pressure may affect an earlier planting more than a later planting in a given season (Moss et al., 2012). An initial concern when implementing the trials was the question of when to harvest those plots that receive the supplemental replant treatments and, as a result, had plants from two different planting dates maturing at different times. Along with yield, grade was a production factor of notable concern in this replant scenario considering varying peanuts at varying maturities would be present within the field. This concern was not warranted according to the results, as grade was not affected by replant treatment and showed not to be a limitation when deciding on what replant method to employ. However, additional research is needed to address the topic of the optimal timing of digging peanut when a supplemental replant occurs because of the different maturity progression of the plants.
When considering the entirety of the results, a primary recommendation to peanut growers would be to do everything possible to ensure an adequate initial plant stand. This is supported by both the linear trend for pod yield across plant stands and the finding that peanut only benefitted from replanting at two initial plant stands. Replanting should not be considered at plants stands greater than or equal to 9.8 plants/m, because replanting never resulted in a yield benefit at that stand or above and would cost a grower time and the expense of an additional trip across the field (seed, labor, fuel, wear on equipment, etc.). Because pod yield increases were observed at 8.2 plants/m, it would be advisable for a grower to replant at that level if the replant treatment can be applied in a reasonable time window after the initial planting date. Based on previous research, this recommendation holds especially true in areas of higher TSWV pressure or when planting cultivars with less resistance to TSWV than the cultivar used in this study. When the decision is made to replant, the best option is to supplement the initial stand with a reduced seeding rate rather than destroying the initial stand and completely replanting. Because grade and stem rot were unaffected by plant stand and replant method, TSWV risk, pod yield, and ultimately profitability should be the deciding factors when making decisions about replanting a peanut field.
Literature Cited
J., Augusto, T.B Brenneman, J.A Baldwin, and N.B Smith (2010). Maximizing economic returns and minimizing stem rot incidence with optimum plant stands of peanut in Nicaragua. Peanut Sci 37 : 137 – 143 .
Beasley, J.P., Jr., J.A Baldwin, S.L Brown, S.M Brown, B Padgett, M.J Bader, and D Shurley 1997 Georgia peanut production guide, Univ. of Georgia Coop Ext. Serv. Guide. Agron. 95-001, Athens, GA.
Beasley, J.P., Jr 2013 Planting dates, p 70 – 73 In: J.P Beasley (ed.) 2013 Peanut Update, Spec. Pub. CSS-13-0110. Univ. of Georgia Coop. Ext., Athens, GA.
M.J, Bell, R.C Muchow, and G.L Wilson (1987). The effect of plant population on peanuts (Arachis hypogaea) in a monsoonal tropical environment. Field Crops Res 17 ((2)) : 91 – 107 .
W.D Branch, (2007). Registration of ‘Georgia-06G' peanut. J. Plant Reg 1 : 120 .
S.L., Brown, A.K Culbreath, J.W Todd, D.W Gorbet, J.A Baldwin, and J.P Beasley (2005). Development of a method of risk assessment to facilitate integrated management of spotted wilt in peanut. Plant Dis 89 : 348 – 356 .
E.W., Chin Choy, J.F Stone, R.S Matlock, and G.N McCauley (1982). Plant population and irrigation effects on Spanish peanuts. Peanut Sci 9 : 73 – 76 .
F.R., Cox, and R.H Reed (1965). Interaction of plant population factors and level of production on the yield and grade of peanuts. Agron. J 57 : 455 – 457 .
A.K., Culbreath, J.W Todd, S.L Brown, J.A Baldwin, and H Pappu (1999). A genetic and cultural “package” for management of tomato spotted wilt virus in peanut. Biolog. Cult. Tests 14 : 1 – 8 .
Davidson, J.L., T.B Whitaker, and J.W Dickens 1982 Grading, cleaning, storage, shelling, and marketing of peanuts in the United States pp 571 – 623 In Peanut Science and Technology , H.E Pattee and C.T Young (eds.) , Amer. Peanut Res. and Educ. Soc., Inc. Yoakum, TX.
Gorbet, D.W., and F.M Shokes 1994 Plant spacing and tomato spotted wilt virus Proc. Am. Peanut Res. Educ. Soc 26 : 50 (abstract) .
Harris, G 1997 Fertilization. Pages 29-31 in: Peanut Production Field Guide, Bulletin 1146. Georgia Coop. Ext. Serv., Univ. of Georgia, Athens, GA .
C.A., Hurt, R.L Brandenburg, D.L Jordan, G.G Kennedy, and J.E Bailey (2004). Effect of cultivar and plant population on spotted wilt in Virginia market-type peanut. Peanut Sci 31 ((2)) : 101 – 107 .
Kemerait, R., A Culbreath, J Beasley, E Prostko, T Brenneman, N Smith, S Tubbs, R Olatinwo, R Srinivasan, M Boudreau, B Tillman, D Rowland, N Dufault, A Hagan, and W Faircloth 2011 Minimizing diseases of peanut in the southeastern United States, The 2011 version of the peanut disease risk index p 100 – 116 In: J.P Beasley (ed.) 2011 Peanut Update, Spec. Pub. CSS-11-0110. Univ. of Georgia Coop. Ext., Athens, GA.
D.A., Knauft, A.J Norden, and N.F Beninati (1981). Effects of intrarow spacing on yield and market quality of peanut genotypes. Peanut Sci 8 : 110 – 112 .
C.S., Kvien, and C.L Bergmark (1987). Growth and development of the Florunner peanut cultivar as influenced by population, planting date, and water availability. Peanut Sci 14 : 11 – 16 .
S.P, McKeown, J.W Todd, A.K Culbreath, D.W Gorbet, and J.R Weeks (2001). Planting date effects on tomato spotted wilt resistant and susceptible peanut cultivars. Phytopath 91 : S60 .
Moss, J.W., R.S Tubbs, T.L Grey, N.B Smith, J.W Johnson, and J.W Davis 2012 Agronomic and economic comparisons of double-crop and relay-intercropping systems of peanut with wheat Online. Crop Man. doi: 10.1094/CM-2012-0925-03-RS .
R.W., Mozingo, and T.A Coffelt (1984). Row pattern and seeding rate effects on value of Virginia type peanuts. Agron. J 76 : 460 – 462 .
Reddy, D.V.R., and J.A Wightman 1988 Tomato spotted wilt virus: Thrips transmission and control . pp 203 – 220 , In: Advances in Disease Vector Research , Vol. 5, Springler-Verlag, New York, NY. 100231.
L.E., Sconyers, T.B Brenneman, K.L Stevenson and B.G Mullinix (2005). Effects of plant spacing, inoculation date, and peanut cultivar on epidemics of peanut stem rot and tomato spotted wilt. Plant Dis 89 : 969 – 974 .
L.E., Sconyers, T.B Brenneman, K.L Stevenson, and B.G Mullinix (2007). Effects of row pattern, seeding rate, and inoculation date on fungicide efficacy and development of peanut stem rot. Plant Dis 91 : 273 – 278 .
R.B., Sorenson, L.E Sconyers, M.C Lamb, and D.A Sternitzke (2004). Row orientation and seeding rate on yield, grade, and stem rot incidence of peanut with subsurface drip irrigation. Peanut Sci 31 : 54 – 58 .
D.A., Sternitzke, M.C Lamb, J.I Davidson, R.T Barron, and C.T Bennet (2000). Impact of Plant Spacing and Population on Yield for Single-Row Nonirrigated Peanuts ( Arachis hypogaea L.). Peanut Sci 27 : 52 – 56 .
H., Tewolde, M.C Black, C.J Fernandez, and A. M Schubert (2002). Pod yield response of two runner peanut cultivars to seeding rate and irrigation. Peanut Sci 29 ((1)) : 1 – 8 .
B.L., Tillman, D.W Gorbet, and P.C Andersen (2007). Influence of peanut planting date on yield and spotted wilt of runner market type peanut. Peanut Sci 34 : 79 – 84 .
G., Wehtje, R Weeks, M West, L Wells, and P Pace (1994). Influence of planter type and seeding rate on yield and disease incidence in peanut. Peanut Sci 21 : 16 – 19 .
E. J., Williams, and J.S Drexler (1981). A non-destructive method for determining peanut pod maturity. Peanut Sci 8 : 134 – 141 .
J.C., Wynne, W.R Baker and P.W Rice (1974). Effects of spacing and a growth regulator, Kylar, on size and yield of fruit of Virginia-type peanut cultivars. Agron. J 66 : 192 – 194 .
Notes
- Dept. of Plant and Soil Sciences, Mississippi State University, Mississippi State, MS 39762 [^]
- Dept. of Crop and Soil Sciences, University of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793. [^]
- Dept. of Crop, Soil, and Environmental Sciences, Auburn University, Auburn, AL 36849 [^]
- Dept. of Plant Pathology, University of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793 [^]
- Agronomy Dept., University of Florida, Gainesville, FL 32611 [^]
- Department of Agricultural and Environmental Sciences, Clemson University, Sandhill Research and Education Center, Columbia, SC 29229 [^] *Corresponding author's E-mail: jason.sarver@msstate.edu
Author Affiliations