Introduction
Peanut, Arachis hypogaea Linn is an important crop in Ghana, particularly in northern Ghana, which comprises the Northern, Upper East and Upper West Regions, and accounts for 92% of the total national production [Statistics, Research and Information Directorate (SRID), 2004]. The kernel has high protein and oil content and is important both as a human food and a source of cooking oil. Revenue from the sale of the kernels is an important source of income to farmers. Additionally, peanut hay is used extensively as fodder for livestock, particularly during the dry season after the crop has been harvested (Natural Research Institute, 1996; Marfo, 1997). The ability of peanut to fix atmospheric nitrogen in the soil also improves the fertility of the otherwise poor soils characteristic of northern Ghana (Kaleem, 1990).
Despite its importance, peanut farmers in northern Ghana continue to not fully recognize the negative impact of a number of production constraints among which are the problems of arthropod pests and diseases. In a preliminary survey of peanut production in the Northern Region, 45–65% of respondent farmers said arthropod pests negatively impact peanut production (Salifu, 1991). Peanut farmers cited subterranean damage and spoilage in storage as primary constraints to high and stable yields. Past studies have shown that more than 350 species of insects damage peanut in different parts of the world (Stalker and Campbell, 1983). The economic importance of the soil pests, termites (Isoptera: Termitidae), millipedes (Myriapoda: Odontopygidae) and white grubs (Coleoptera: Scarabaeidae) has been well documented in parts of Africa (Wightman et al., 1990; Umeh et al., 1999, 2001). These pests penetrate and feed on roots, developing pods, and kernels (Umeh et al., 1999; Wightman et al., 1990). This feeding behaviour also predisposes pods to attack by disease pathogens such as the aflatoxin-producing fungus, Aspergillus flavus (Link) (Lynch et al., 1990; Waliyar et al., 1994; Wightman et al., 1990). Yield losses of up to 70% have been attributed to termites alone in parts of Nigeria (Johnson et al., 1981; Umeh et al., 1999). In places where soil pests are a problem, insecticides are used for control (Wightman et al., 1990; Brandenburg and Herbert, 1991). There is scarce information on pest profiles and status on peanut in northern Ghana. In addition, there is no information on insecticide control and how it affects peanut yield.
The present studies were conducted to identify key soil arthropod pests of peanut, their distribution and density, and their effect on peanut quality in northern Ghana. The effects of insecticide treatments on control of these pests and on yield were also assessed.
Materials and Methods
Survey of soil arthropod pests
Surveys of soil arthropod pests were carried out on farmers' fields from August to October in the Northern, Upper East and Upper West Regions of Ghana during the cropping seasons of 2003–2005. The survey area lies between latitudes 8° 00′s and 11° 2′ N and longitudes 0° 30′ E and 2° 5′ W. This area has a monomodal rainfall pattern that ranges between 600 and 1253 mm per annum and a mean annual temperature between 26 C and 29 C (Kasei 1990). During the surveys farmers were either harvesting or preparing to harvest their peanut crops.
An average of seven major peanut growing villages in the Northern Region and five each in the Upper East and Upper West Regions were randomly selected each year and three to four farms per village were selected for sampling. Sampled farms were at least 2 kilometers apart. A total of 53 farms (one field per farm) each in 2003 and 2004, and 67 farms in 2005 were surveyed across the three regions. Five plants were randomly selected and dug out with about 60 cm3 of soil around the pod and root zone from a 5 × 5 m marked section in each farm. To account for any spatial variability, ten plant samples were taken on farms with less uniformity in plant architecture, or farms larger than two ha. The plants were lifted with the soil using a hand-hoe and the different arthropods in the soil and on the roots and pods were sorted in situ, counted and appropriately recorded. The numbers of plants with one or more soil arthropods present were also recorded. The percentage of pods with arthropod penetration holes or hull scarification was determined from the sampled plants.
Background information on farms
Prior to sampling each farm, a checklist was used to obtain information from farmers on peanut cultiror planted, time of planting, cropping sequence, and whether the field was solely peanut or interplanted with other crops. The soil type at the farm, weed management practices, farmer's knowledge of soil arthropod pests and control practices applied, if any, were also recorded.
Assessment of yield loss
Experiments were conducted at Nyankpala (9° 42′ N latitude, and 0° 92′ W longitude, and 184 m altitude) and Bagurugu (9° 53′ N latitude, and 0° 43′ W longitude, and 168 m altitude) from 2005–2006. The locations are major peanut growing areas in the Northern Region. Soil type at both locations was gravelly-sandy loam. Peanut cultivar ‘JL 24’ which is susceptible to soil arthropod pests was used. Treatments included chlorpyrifos (Dursban 4E, Dow AgroSciences, Indianapolis, IN, USA) or carbofuran (Furadan 3G, FMC Corp., Philadelphia, PA, USA) applied at the appropriate rates at planting by hand in-furrow or applied by hand along each row over the vines at pegging (50 days after planting) (Lynch et al., 1991). A non-treated control and one with insecticide treatments at both planting and pegging were included. Chlorpyrifos and carbofuran were applied at the rates of 2.2 kg ai ha−1 and 1.0 kg ai ha−1, respectively. At Nyankpala, applications at planting were made on 8 June in 2005 and 31 May in 2006; applications at pegging were made on 28 July and 20 July in 2005 and 2006, respectively. At Bagurugu, at-planting applications were made on 14 June in 2005 and 8 June in 2006 whereas at-pegging applications were made on 3 August in 2005 and 28 July in 2006. The treatments were arranged in a split-plot design and replicated four times. Insecticide treatment constituted the main plot effect, and application timing the sub-plot effect. Each sub-plot (10 m2) consisted of four 5 m long rows spaced 0.5 m apart. Plants within rows were spaced 0.2 m. There was 1 m spacing between plots in main plots and 2 m between main plots. The experiments at Nyankpala were planted on 8 June 2005 and 31 May 2006 whereas those at Bagurugu were planted on 14 June 2005 and 8 June 2006.
Five randomly selected plants and adhering soil were dug out as previously described within the outer two rows of each plot for soil arthropod pests at harvest. Arthropods in the soil and on the plant samples were sorted in situ, counted and recorded. Data on pod hull scarification and penetration, and yield in treated and untreated plots were also recorded. The two middle rows of each plot were harvested to determine yield.
Statistical analyses
All data were subjected to analysis of variance and where F-values were significant at P ≤ 0.05 means were separated using Fisher's protected least significant difference (LSD) test (SAS Institute, 1998). Data on arthropod count were transformed to log10 (X + 1) whereas data on percentage damage were subjected to square root or arcsine square root transformation to normalize variances before analyses (Gomez and Gomez, 1984). Chi-squared analyses were used to test the relationships between the cropping systems of the three regions. Pod yield losses were computed by subtracting yields of untreated plots from those of insecticide-treated plots and the difference calculated as a percentage. Correlation analyses were used to assess the relationships between pod yield and arthropod densities, percentage of pods scarified, and percentage of pods penetrated.
Results
Background information on farms: Across the regions surveyed, twenty seven percent of farmers (n = 173) planted peanut at the onset of the rains in April while 32% planted in May. The majority (41%) of the farmers, however, planted peanut when the rains stabilized in June (data not presented). Improved peanut cultirors with bunch or erect morphological characteristics such as Manipintar were cultivated by 96.4% of the farmers while the remaining 3.6% said they grew locally-derived cultirors such as ‘Bugla’ (data not presented). Generally, cereals such as corn (Zea mays L.) preceded peanut but peanut preceding peanut was also practiced. Intercropping peanuts with cereals (and occasionally with other legumes such as Bambara groundnuts, Voandzea subterranea) was common and more in the Upper East and Upper West Regions (65.7 and 45.5% of farmers, respectively) than in the Northern Region (31.1%) (χ2 = 9.4955, df = 2, P = 0.0087). The majority of the farmers in the Northern Region generally practiced peanut monoculture on large tracts of land. Peanut farms in this region ranged from 0.4–16 ha with a mean of 1.7 hectares as opposed to a range of 0.1–2.8 hectares with a mean of 0.6 ha in the Upper Regions (data not presented). The surface soil texture of peanut fields was generally sandy-silty loam or gravelly-sandy loam. Hoeing was the dominant weed control method and was practiced by 91% of farmers. Fifty-two percent of farmers weeded their fields only once at 3–5 weeks after planting (WAP), 36 percent weeded twice at 2–3 and 5–6 WAP while only 2% weeded three times (data not presented). About 65% of the farmers sampled across the regions had a general understanding of the soil arthropods attacking their peanut crop and the damage caused but none of them practiced any control (data not presented).
Survey of soil arthropod pests: The survey revealed that termites (Microtermes sp. and Odontotermes sp), millipedes (Peridontopyge sp.), white grubs (Schyzonicha africana Lap.) and wireworms (Elaterid larvae) were the main soil pests infesting peanut in the field (tables 1– 3). Termites and millipedes were the most abundant while wireworms were least in occurrence in all three years and regions of the study. Other insects with infrequent occurrence included mealy bugs (Homoptera:Pseudoccoccidae) and red ants (Hymenoptera:Formicidae) that were observed on peanut pods in three fields only in the Northern Region (data not presented).
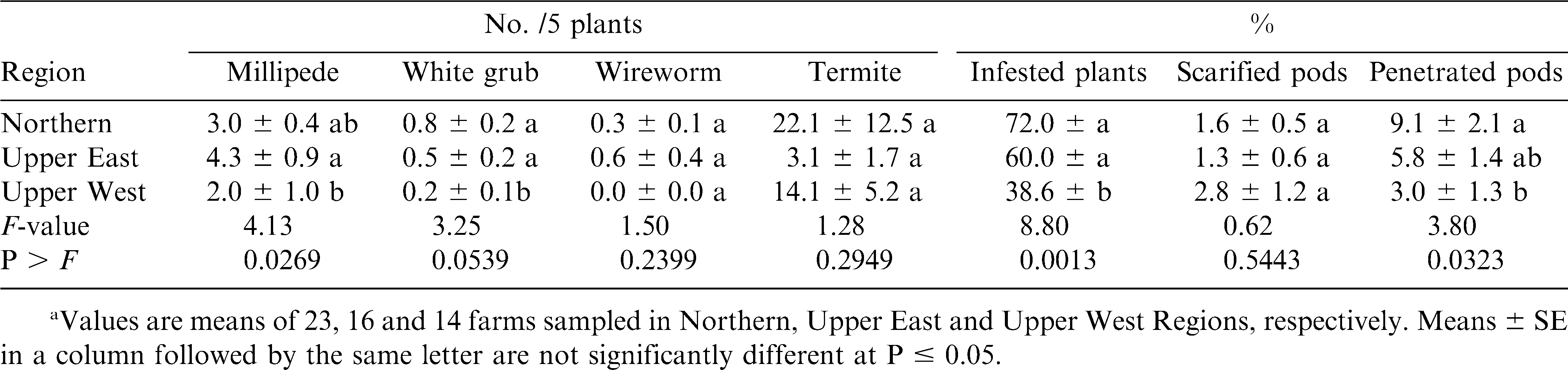
In 2003, significantly higher millipede densities were recorded in Upper East Region than in Upper West Region (table 1). However, millipede densities in Upper East Region did not differ from those of Northern Region. Densities of white grubs were higher in Northern and Upper East Regions than in Upper West Region. Wireworm and termite densities did not differ significantly among the regions. Significantly higher percentages of plants were infested in the Northern and Upper East Regions than in the Upper West Region. Pod hull scarification did not differ among the regions, but a significantly higher percentage of pods had arthropod penetration holes in Northern Region than in Upper West Region.
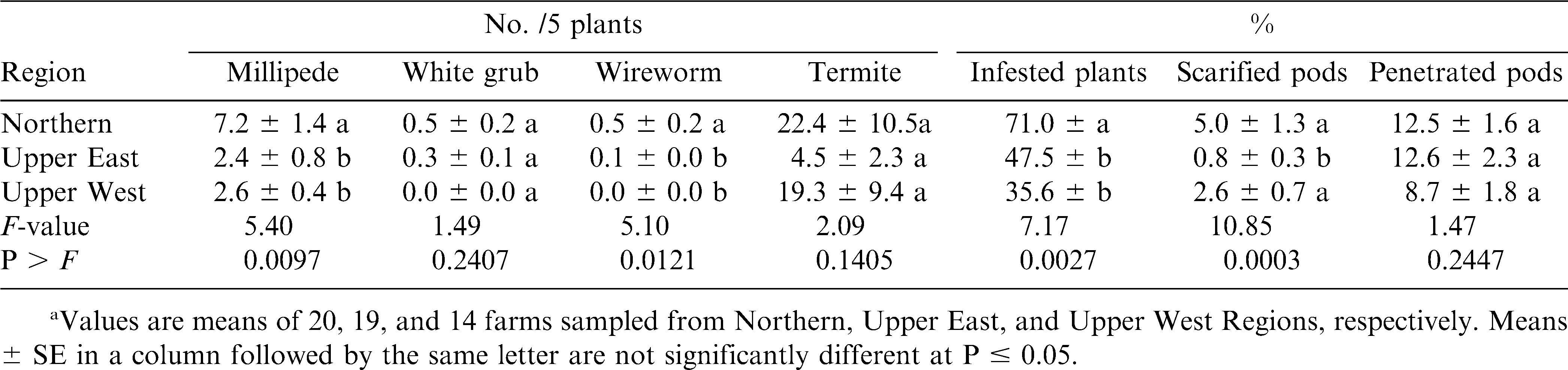
In 2004, higher millipede and wireworm densities were recorded in Northern Region compared with the other regions (table 2). The densities of the other pests did not differ significantly among the regions. A higher percentage of plants were infested in the Northern Region than in either Upper East or Upper West Regions. A significantly higher percentage of pods were scarified in Northern and Upper West Regions than in Upper East Region. However, percentage of pods penetrated did not differ among the regions.
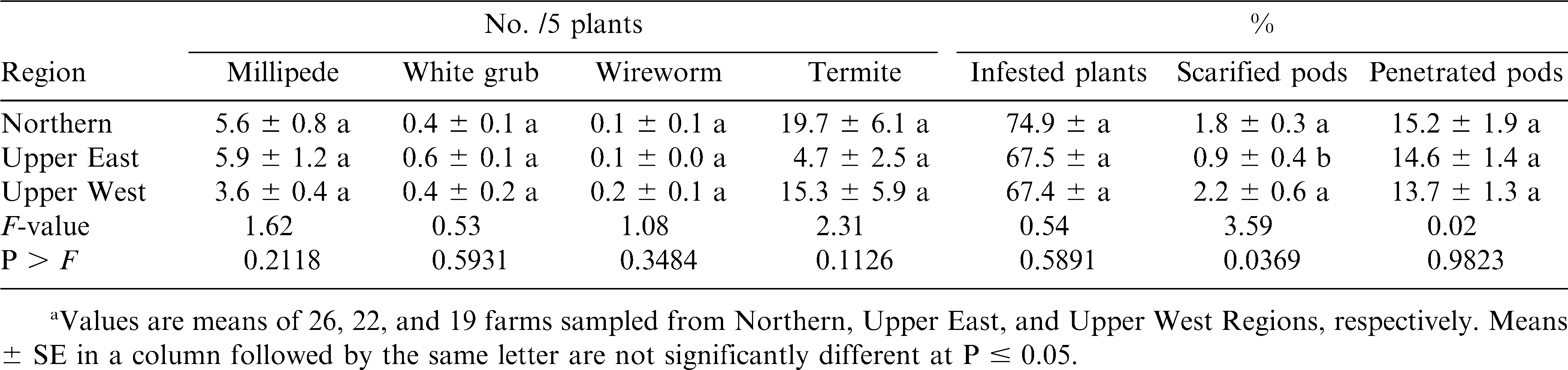
In 2005, soil arthropod pest densities, percentage of plants infested and percentage of pods penetrated were similar in all three regions (table 3). However, percentages of pods scarified were significantly higher in Northern and Upper West Regions than in Upper East Region.
Nyankpala
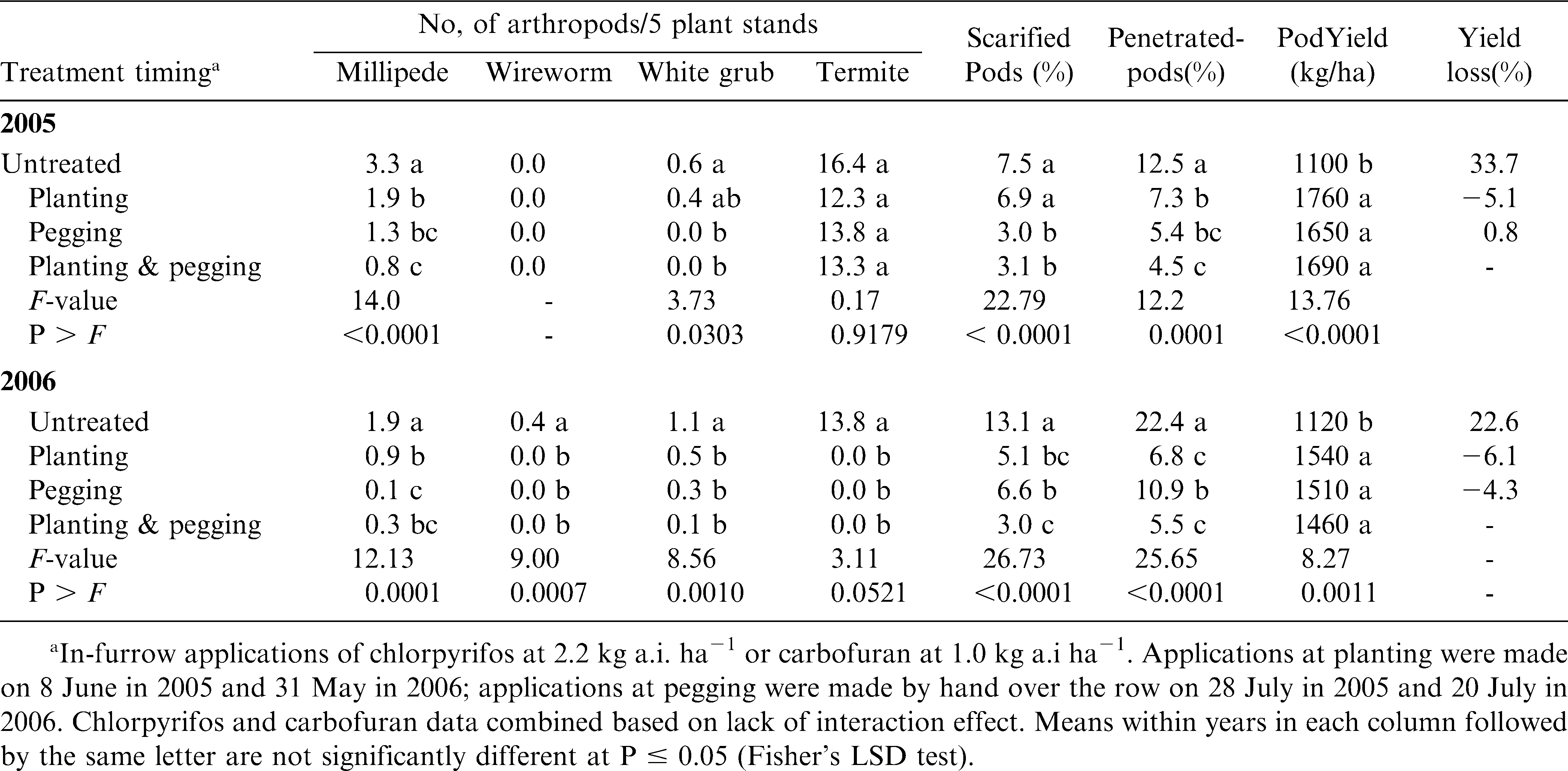
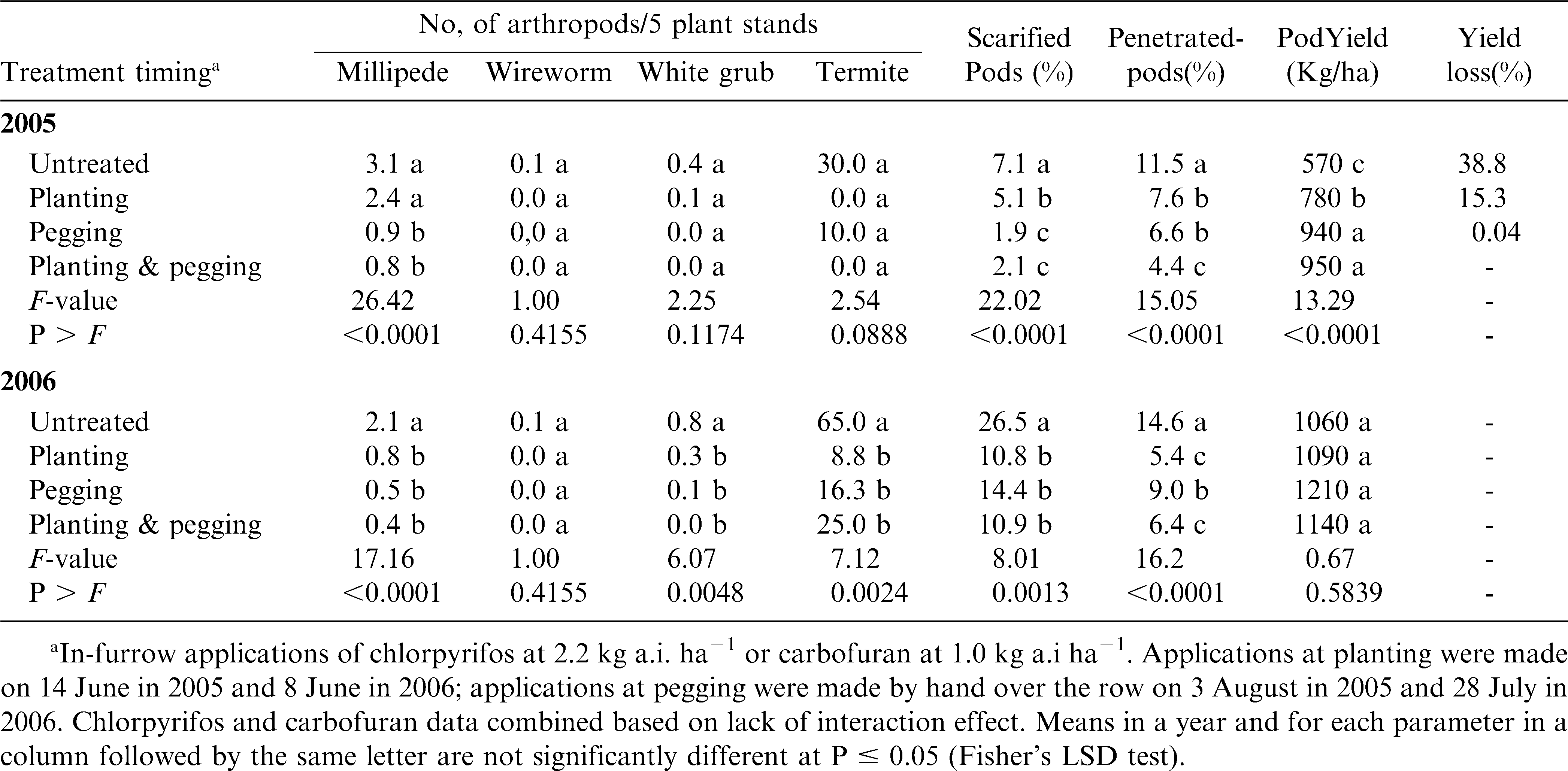
The insecticides, chlorpyrifos and carbofuran, did not differ significantly in their effects on soil pest densities, pod damage and yield in either year of the study. Also there were no significant interactions of insecticide and timing application with reference to the parameters tested, but treatment application timing did show significant effects (table 4). The effect of insecticide application at different peanut growth stages on densities of soil pests, peanut pod damage and yield were very similar for 2005 and 2006, averaged across insecticides (table 4). In both years, millipede densities were lower in treated plots compared with untreated control plots. Millipede densities were lowest and similar when plots were treated at pegging or at both planting and pegging. However, treatment at pegging in 2005 or at both planting and pegging in 2006 did not significantly lower millipede densities compared to treatment at planting. Wireworms were not recorded in 2005, but significantly higher densities were recorded in the untreated control than in treated plots in 2006. Except for plots treated at planting in 2005, insecticide treatment also significantly lowered white grub densities compared with untreated control in both 2005 and 2006. Termite densities were not significantly lowered by insecticide treatments compared with untreated control in 2005. However, in 2006 termite densities were significantly reduced in treated relative to untreated control plots. These results demonstrate that insecticides produce an overall reduction in all pest arthropods so there is an overall cumulative benefit even if individual species are present in subeconomic levels.
In 2005, percentage of pods scarified was significantly lower when plots were treated at pegging or at both planting and pegging when compared with untreated plots or treatment at planting. Pod scarification was significantly reduced in all treated plots compared with untreated plots in 2006 (table 4). For this year, percentage scarified pod damage was lowest when plots were treated at planting or at both planting and pegging. Percentage of pods that were penetrated by soil pests was significantly lower in treated plots compared with untreated controls in both 2005 and 2006. In 2005, the percentage of pods penetrated was lowest in plots that were treated at both planting and pegging. In 2006, the percentage of pods penetrated was lowest when plots were treated either at planting or at both planting and pegging.
Pod yield was significantly increased in treated compared with untreated plots in both 2005 and 2006 (table 4), and this increase was not influenced by timing of insecticide treatment. In 2005, percent yield losses in untreated plots were 35.6, 31.6 and 33.7 when compared to plots treated at planting, pegging, or at both planting and pegging, respectively. Yield losses in 2006 were 25.1, 25.7 and 22.6% relative to planting, pegging, and planting plus pegging treatment, respectively. In 2005, pod yield was negatively correlated to the percentage of penetrated pods (r = −0.51, P = 0.0031) and to some extent to millipede densities (r = −0.34, P = 0.0558). In 2006, pod yield per hectare was negatively correlated with total pest densities (r = −44, P = 0.0108), percentage scarified pods (r = −0.55, P = 0.0011) and percentage penetrated pods (r = −0.49, P = 0.0047). Among the pests in test plots, pod yield was most correlated with millipede (r = −0.44, P = 0.0118) and termite (r = −0.35, P = 0.0506) populations.
Bagurugu
As in Nyankpala, the effects of insecticide type as well as insecticide by application timing interactions were not significant for the parameters tested both for 2005 and 2006 (table 5). However, application timing showed significant effects. Averaged across insecticides, the effect of insecticide application at the different peanut growth stages on the parameters tested were similar for 2005 and 2006. In both years, except at planting in 2005, millipede densities were lower in treated than in untreated plots. Wireworm densities were low in both years, and there were no significant differences due to treatments at the different growth stages. White grub densities did not differ among the treatments in 2005. However, in 2006, white grub densities were significantly lower in treated than in untreated plots. Termite densities were not significantly lowered by insecticide treatment at the different growth stages compared with control in 2005. However, in 2006, termite densities were significantly lower in treated plots.
For both 2005 and 2006, percentage of pods scarified was significantly lower in treated than in untreated plots (table 5). Pod scarification was lowest in plots that were treated at pegging or at both planting and pegging in 2005. Also, percentage of pods penetrated was lower in treated than in untreated plots in both 2005 and 2006. Pod penetration damage in 2005 was lowest in plots that were treated at both planting and pegging and in 2006 it was lowest in plots treated at planting or at both planting and pegging.
Pod yield increased in treated plots over the untreated control in 2005, but not in 2006 (table 5). In 2005, yield was highest and similar in plots treated at pegging and at both planting and pegging. Yield losses in untreated plots were 26.6, 37.5 and 38.8% relative to plots treated at planting, pegging or at both planting and pegging, respectively. There were significant negative correlations between pod yield and millipede abundance (r = −0.70, P = <0.0001),white grub abundance (r = −0.38, P = 0.0343), percentage scarified pods (r = −0.56, P = 0.0009), and percentage penetrated pods (r = −0.45, P = 0.0094) in 2005. No significant correlations were detected among these parameters in 2006.
Discussion
Results from this study showed that termites, millipedes, white grubs and wireworms were the major soil arthropod pests of peanut in northern Ghana (Abudulai et al., 2007) as also reported for other parts of Africa (Wightman et al., 1990; Umeh et al., 1999; Owusu-Akyaw et al., 2005). Termites were the most abundant soil pests surveyed in both years, and accounted for >70% of all soil pests recorded from soil and plant samples. Millipedes were the next most abundant pests followed by white grubs. Wireworms were least in abundance, which also agrees with the findings by Umeh et al., (1999, 2001). The activities of these pests negatively affected the quality of harvested peanut as shown by the high percentages of infested plants, scarified and penetrated pods. The pests including white grubs, millipedes and wireworms may also have reduced plant stand and yield by severing the main stem at soil level (Johnson et al., 1981; Wightman et al., 1990; Lynch et al., 1991). In addition, their feeding activities increase the risk of aflatoxin contamination in peanut (Wightman et al., 1990). The high pest densities and pod damage by scarification and penetration in the Northern Region compared to the other regions was likely due to concentration of resources because of the greatest production in the Northern Region (SRID, 2004). The high incidence of peanut monoculture in this region may also enhance arthropod activities compared with the mixed cultural systems (Andow, 1991; Tonhasca and Byrne, 1994; Banks and Ekbom, 1999) such as those of the Upper Regions.
The reduction in the densities of all the four soil pests due to insecticide treatment resulted in the reduction of the numbers of pods damaged through scarification and penetration leading to increased pod yields. Generally, treatment at planting or pegging was sufficient for lowering pest densities and pod damage to increase yield. These results are in agreement with the findings of Brandenburg and Herbert (1991), Lynch et al. (1991), Anitha et al. (2005) and Owusu-Akyaw et al. (2005). Wightman et al. (1990), however, reported from field tests carried out in Malawi that insecticide treatments did not reduce densities of millipedes. Since millipedes are not insects their susceptibility to these products may be less than that of the other arthropods. There was no significant difference between carbofuran and chlorpyrifos on the parameters tested, indicating that the two insecticides had similar efficacy on peanut pests. Numerous studies also have shown that both chemicals are efficacious against soil pests of peanut (Wightman et al., 1990; Anitha et al., 2005; Owusu-Akyaw et al., 2005). Owusu-Akyaw et al., (2005), however, reported a higher efficacy for chlorpyrifos than carbofuran against soil pests of peanut in southern Ghana. Although carbofuran is no longer labeled for use by farmers because of environmental concerns (Pimentel et al., 1992), it was included in the present tests for yield loss assessment because of its known effectiveness against soil pests. The absence of any significant difference between carbofuran and chlorpyrifos confirmed the efficacy of chlorpyrifos which is currently used for pest control in peanut (Brandenburg and Herbert, 1991). In general, correlation analyses indicated that insecticide treatment had a positive effect on peanut pod yield by reducing pest densities and pod damage. Pod penetration was considered a good indicator of the degree of plant damage and thus yield loss in parts of Africa (Wightman et al., 1990). Surface scarification of pods probably weakened pods for easy penetration by arthropods (Abudulai et al., 2007). Peanut pod yield loss in untreated plots ranged between 23 and 36% in Nyankpala and between 26 and 39% in Bagurugu. Yields were generally lower at Bagurugu than at Nyankpala, which may be attributed to more consistent rainfall that may have influenced pest densities and crop performance (Padmalatha et al., 2006).
In conclusion, the results of these studies demonstrated that termites, millipedes, white grubs and wireworms are the major soil arthropods pests of peanut in northern Ghana. These arthropods impact negatively on peanut quality and yield. Insecticide protection either at planting or pegging lowered pest densities and resulted in increased pod yield, and therefore could be a useful management practice for peanut soil pests. At this time, however, insecticide control for peanut pests is currently not practiced by farmers in Ghana due largely to the high price of these products(Wightman et al., 1990). This demonstrates the need for further research on control options that are less expensive and can be readily obtained by farmers.
Acknowledgements
We thank Frederick Anaman and Misbaw Haruna for assistance in the field work. We are especially grateful to the farmers that cooperated with us during the surveys. Financial support provided by the United States Agency for International Development under the Peanut Collaborative Research and Support Program grant LAG-G-00-96-90013-00 is greatly appreciated.
Literature Cited
Abudulai M Dzomeku I.K Salifu A.B Nutsugah S.K Brandenburg R.L and Jordan D 2007 Influence of cultural practices on soil arthropods, leaf spot, pod damage, and yield of peanut in northern Ghana Peanut Sci. 34 : 72 – 78 .
Andow D 1991 Vegetational diversity and arthropod population response Ann. Rev. Entomol. 36 : 561 – 586 .
Anitha V Wightman J and Rogers D.J 2005 Management of white grubs (Coleoptera: Scarabaeidae) on groundnut in southern India Int'l J. Pest Manage. 51 : 313 – 320 .
Banks J.E and Ekbom B 1999 Modelling herbivore movement and colonization: pest management potential of intercropping and trap cropping Agric. Forest Entomol. 1 : 165 – 170 .
Brandenburg R.L and Herbert D.A 1991 Effect of timing on prophylactic treatments for southern corn rootworm (Coleoptera: Chrysomelidae) in peanut J. Econ. Entomol. 84 : 1894 – 1898 .
Johnson R.A Lamb R.W and Wood T.G 1981 Termite damage and crop loss studies in Nigeria – a survey of damage to groundnuts Trop. Pest Manage. 27 : 325 – 342 .
Kaleem F 1990 Assessment of nitrogen fixation by legumes and their relative contribution to a succeeding maize crop in the savannah zone of Ghana In: Proceedings of the second workshop on improving farming systems in the interior savanna zone of Ghana, 24–26th April, 1990 Rudat H Mercer-Quarshie H von Bargen J Marfo K.O Salifu A.B Nyamekye A.L Afuakwa J.J Dekuku R.C and Sallah P.Y.K (eds). , Nyankpala Agricultural Experiment Station , Nyankpala, Ghana pp. 123 – 127 .
Kasei C.N 1990 A synopsis on the climate of the North of Ghana In: Proceedings of the second workshop on improving farming systems in the interior savanna zone of Ghana, 24–26th April, 1990 Rudat H Mercer-Quarshie H von Bargen J Marfo K.O Salifu A.B Nyamekye A.L Afuakwa J.J Dekuku R.C and Sallah P.Y.K (eds.) , Nyankpala Agricultural Experiment Station , Nyankpala, Ghana pp. 2 – 9 .
Lynch R.E Ouedraogo A.P and Some S.A 1990 Effect of harvest date and termite-resistant varieties on termite and millipede damage to groundnut in Burkina Faso In: Summary proceedings of the first ICRISAT regional groundnut meeting for West Africa, 13–16 September 1988, ICRISAT Sahelian Centre, Niamey, Niger, pp. 87 – 90 .
Lynch R.E Dicko I.O Some S.A and Ouedraogo A.P 1991 Effect of harvest date on termite damage, yield and aflatoxin contamination in groundnut in Burkina Faso IAN 10 : 24 – 25 .
Marfo K.O 1997 The performance and association among some important peanut yield traits in northern Ghana In: Proceedings of the 3rd Workshop on improving farming systems in the interior Savanna zone of Ghana, 27–29th April, 1993 Mercer-Quarshie H Marfo K.O Langyintuo A.S and Owusu R.K (eds.) , Nyankpala Agricultural Experiment Station , Tamale, Ghana pp. 133 – 140 .
Natural Research Institute 1996 . Groundnuts. Pest control series Hobbs the printers Ltd , Totton, Hampshire .
Owusu-Akyaw M Afun J.V.K Mochiah M.B Adu-Mensah J Moses E Bolfrey-Arku G Osei K Osei-Yeboah S Dankyi A.A Adama I Brandenburg R.L and Jordan D.L 2005 Survey and management of soil arthropods in peanut (Arachis hypogaea L) in southern Ghana, West Africa Peanut Sci. 32 : 136 – 143 .
Padmalatha Y Reddy S.R and Reddy T.Y 2006 The relationship between weather parameters during developmental phase and fruit attributes and yield of peanut Peanut Sci. 33 : 118 – 124 .
Pimentel D Acquay H Biltonen H Rice P Silva M Nelson J Lipner V Giordano S Horowitz A and D'Amore M 1992 Environmental and economic costs of pesticide use Bioscience. 42 : 750 – 761 .
Salifu A.B 1991 . Nyankpala Agricultural Experimental Station, Crops Research Institute, Annual Report for 1990.
SAS Institute 1998 . SAS User guide statistics, 8th edn SAS Institute Inc. , Cary, NC .
SRID , 2004 . Production of major crops in Ghana, 2003 Statistics, Research and Information Directorate (SRID), Ministry of Food and Agriculture , Ghana .
Stalker H.T and Campbell W.V 1983 Resistance of wild species of peanut to an insect complex Peanut Sci. 10 : 30 – 33 .
Tonhasca A and Byrne D.A 1994 The effect of crop diversification on herbivorous insects: a meta-analysis approach Ecol. Entomol. 19 : 239 – 244 .
Umeh V.C Waliyar F Traore S and Egwurube E 1999 Soil pests of groundnut in West Africa – species diversity, damage and estimation of yield losses Insect Sci. Applic. 19 : 131 – 140 .
Umeh V.C Yuom O and Waliyar F 2001 Soil pests of peanut in sub-Saharan Africa – A Review Insect Sci. Applic. 21 : 23 – 32 .
Waliyar F Ba A Hamma H Bonkoungou S and Bosc J.P 1994 Sources of resistance to Aspergillus flavus and aflatoxin contamination in groundnut genotypes in West Africa Plant Dis. 78 : 704 – 708 .
Wightman J.A Dick K.M Ranga Raw G.V Shanower T.G and Gold C.G 1990 Pests of groundnut in the semi-arid tropics In: Insect pests of legumes Singh S.R (ed.) , Longman and Sons Ltd , New York pp. 243 – 322 .
Notes
- CSIR-Savanna Agricultural Research Institute, P. O. Box TL 52, Tamale, Ghana
- University for Development Studies, P. O. Box TL 1385, Tamale, Ghana
- North Carolina State University, Raleigh, N.C 27695, USA *Corresponding author's email: rick_brandenburg@ncsu.edu
Author Affiliations