Introduction
Cylindrocladium black rot (CBR) is a serious disease of peanut (Arachis hypogaea L.) caused by the soilborne fungal pathogen Cylindrocladium parasiticum Crous, Wingfield, and Alfenas. The disease was first described in Georgia (Bell and Sobers, 1966), and has since spread throughout the southern US. CBR has caused the most losses in areas of North Carolina and Virginia (Garren et al., 1972). The first known incidence of CBR in Texas occurred in a field of virginia peanuts grown in Terry County during the summer of 2004 (Wheeler and Black, 2005). Above ground symptoms on affected plants include chlorosis followed by wilting and eventually death. The disease can affect all below-ground structures including hypocotyls, pods, roots, and pegs. Losses of 50% have been reported in the southeast (Phipps and Beute, 1997).
Management strategies that reduce C. parasiticum soil inoculum densities include fumigation with metam sodium (Cline and Beute, 1986), rotations with non-hosts (Black and Beute, 1984; Phipps and Beute, 1979), use of fungicides (Kucharek, 2005), and partially resistant cultivars (Lemay, 1999). As with many soil-borne fungi, an integrated approach is necessary to effectively manage C. parasiticum.
Programs that breed for CBR resistance typically use infested fields to screen germplasm for resistance. This approach allows for disease screening in an environment where a multitude of resistance mechanisms are expressed and simultaneous evaluation of germplasm for agronomic traits. Limitations of this approach include non-uniform distribution of fungal inoculum in field plots, years where the environment limits disease development, and confounding interactions with other pathogens such as Tomato spotted wilt virus that cause similar symptoms. As a result of these problems, some breeding programs are utilizing greenhouse screening to augment field trials. However, there are examples of poor correlation between greenhouse and field screening results (Dong et al., 2008; Lemay, 1999; Pataky et al., 1983). Hollowell et al. (2008) found that their greenhouse technique correlated well with field studies (r = 0.83) when the field studies were repeated over 4 years, but not with single year field studies. This suggests that field studies were less consistent than greenhouse screening.
Methodology can affect greenhouse screening for resistance to C. parasiticum. Some factors that affect greenhouse assays include isolate virulence (Rowe and Beute, 1975) and density and size of microscloertia (Dong et al., 2008). Additional factors that may influence the effectiveness of greenhouse assays are container size, duration of the test, and method of evaluating symptoms.
Our objective was to develop a protocol for screening peanuts in a growth chamber using a C. parasiticum isolate obtained from West Texas. Our strategy was to inoculate two peanut genotypes known to differ in resistance to C. parasiticum in field and controlled environments. We examined the effect of two container sizes, two inoculum density levels, three disease assessment times, and three methods of rating disease.
Materials and Methods
C. parasiticum isolate TW1 (originally obtained from Terry Co., TX, August 2004), was re-isolated from lesions on greenhouse-grown peanut plants before these experiments were performed and between trials. Symptomatic tissue was surface sterilized with a 10% NaOCl solution for 2 minutes and placed on petri dishes containing a C. parasiticum selective medium (Phipps et al., 1976) for 5 days. Pure cultures were transferred to potato dextrose agar (PDA) plates and grown for 8 weeks at 25 C. PDA plates were then comminuted with tap water in a Waring blender for 2.5 minutes at low speed and 1.5 minutes at high speed. The extraction was poured through a 500 µm opening sieve onto a 74 µm opening sieve with copious tap water. The microsclerotia (ms) retained were suspended in 500 ml of tap water and sampled for quantification. The suspensions were adjusted to inoculate autoclaved soil at rates of 15 and 25 ms/g soil. These two densities were chosen based on results from preliminary experiments where multiple densities were tested.
Autoclaved sandy loam soil (72% sand, 10% silt, 18% clay, 0.3% organic matter) from Gaines Co, TX was mixed with Bradyrhizobium spp. inoculant (1g/50 g soil; Soil Implant® +, EMD Crop BioScience, Milwaukee, WI) to promote nodulation. C. parasiticum ms suspensions were added to soil in polyethylene bags (4.8 ml suspension/100 g soil) and mixed for 2 minutes. Approximately 70 g of infested soil were placed in small cone containers (2.2 × 16 cm, 66 cm3, RL-C4 Pine Cell, Cone-tainers™, Stuewe & Sons, Inc., Corvallis, OR) and 205 g of infested soil were placed in large cone containers (3.8 × 21 cm, 164 cm3, SC-10 Super Cell, Cone-tainers™). Filter paper (9 cm diameter) was placed in the bottom of each container to retain soil.
Seeds of susceptible cultivar ‘NC 7’ (Brune, 1996; Lemay, 1999) and resistant genotype ‘NC 3033’ (Beute et al., 1976) were pre-germinated in vermiculite for 3 days at 27 C. Germinated seedlings were selected for uniform radicle size (2 to 3 cm) before planting in containers. The upper 25% of cotyledons was left exposed and the top of the seed testa was split using a razor blade. Containers were placed in a growth chamber set at a constant 23 C, which produced 12 hrs of light from fluorescent lamps followed by 12 hrs of darkness in a 24 h cycle. Soil was kept moist using hand watering, usually on a daily basis.
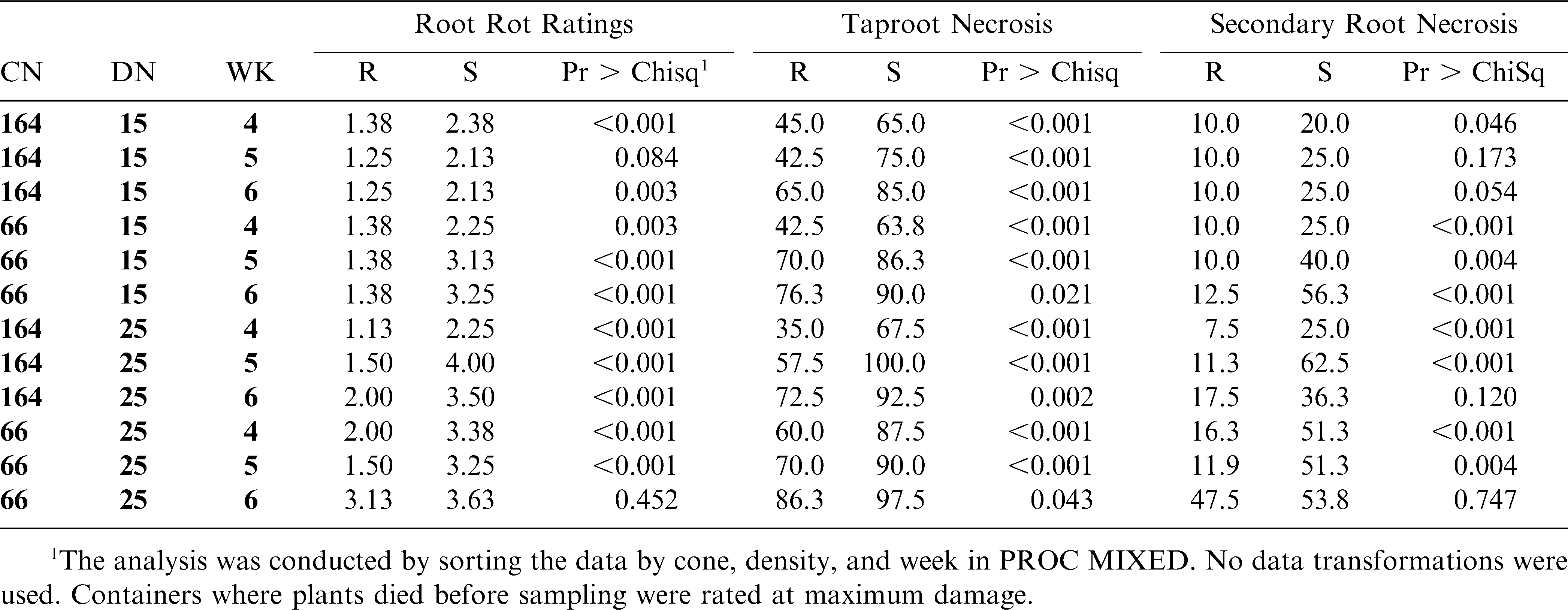
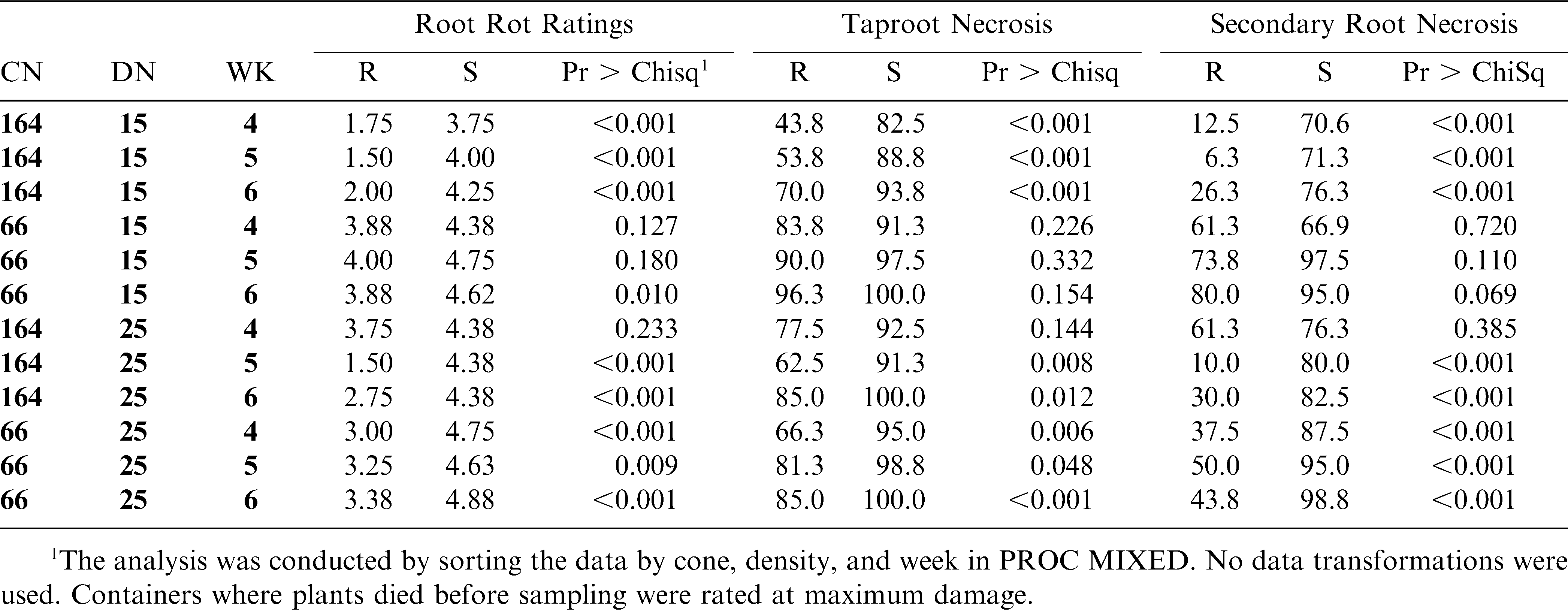
This experiment was set up as a randomized complete block design (RCBD) with 24 replications. Eight replications were randomly selected to be terminated at 4, 5, or 6 weeks after planting. The experiment was repeated. Within each replication was a factorial arrangement of inoculum density (0, 15, 25 ms/g soil), container size (66, 164 cm3), and genotype (NC 7, NC 3033). Root rot ratings, percent taproot necrosis, and percent secondary root necrosis were analyzed using Proc MIXED (PC SAS, SAS Institute, Ver. 9.1, Cary, NC). Root rot ratings were assigned using a 0–5 scale (0 = no lesions, 1 = few lesions on secondary roots and or taproot, 3 = many lesions on secondary roots and many lesions on taproot with several secondary roots missing, 5 = completely decayed roots with most of secondary root and part of taproot missing, with 2 and 4 = intermediate levels) (Black and Beute, 1984; Rowe and Beute, 1975). Containers where plants died before sampling were rated at maximum damage. Incidence of dead plants was low (less than 5%) across each trial. Percent taproot and secondary root necrosis ranged from 0 to 100% and was scored in 10% increments. Containers where the entire taproot or secondary root system were missing due to disease were rated at 100%; otherwise necrosis measurements were based on roots present. Root rot ratings and taproot and secondary root ratings were completed on each plant at the same time.
The analysis was conducted by sorting data for each disease assessment method by trial, container, inoculum density, and week of sampling and comparing the resistant and susceptible genotype response for each treatment combination with the chi-square option at a significance level of P = 0.05. Degrees of freedom were calculated using the Satterthwaite method. Trials, replicates within trials, and the interaction between genotype and trials were considered random effects, while genotypes were considered fixed effects.
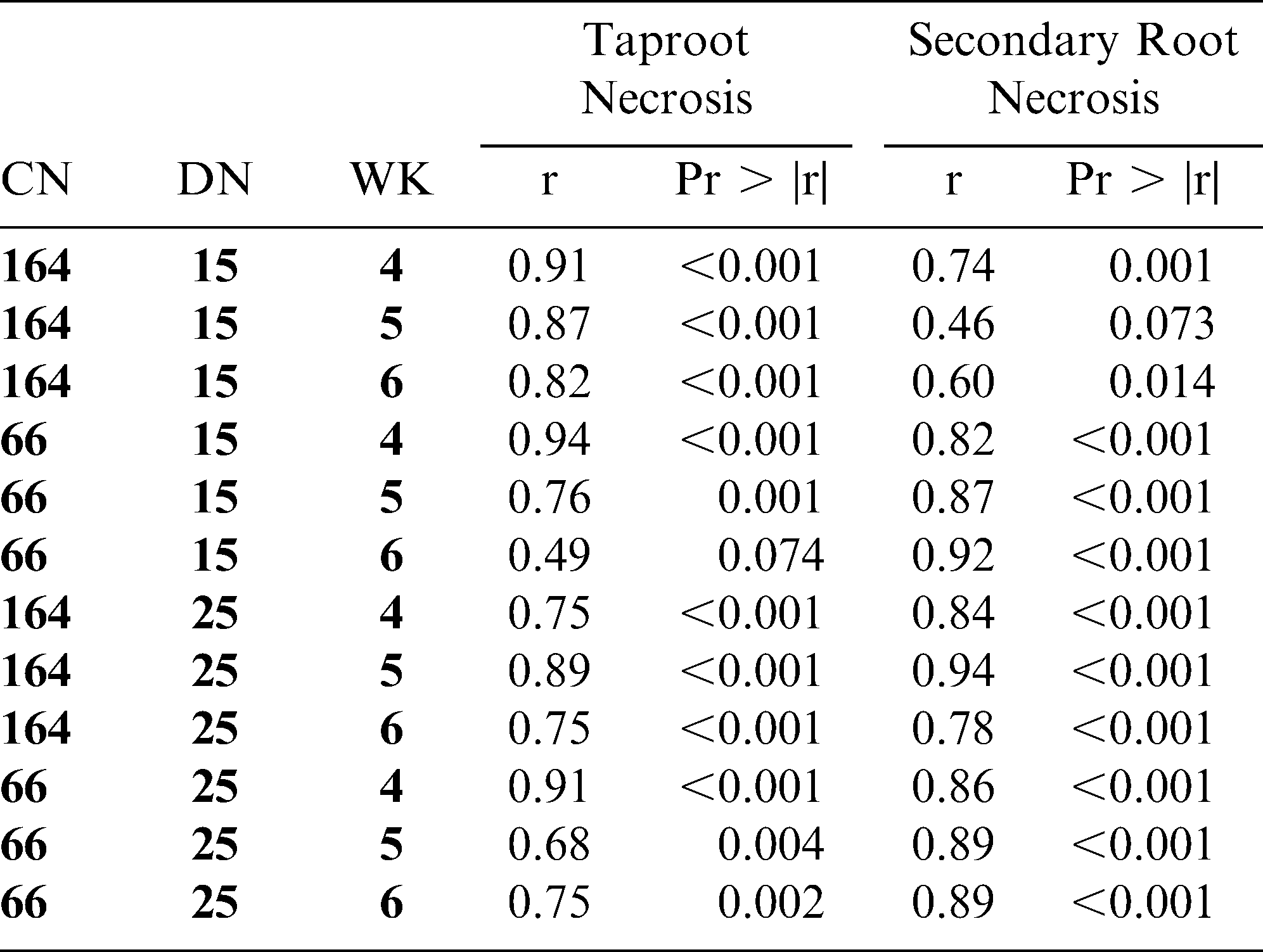
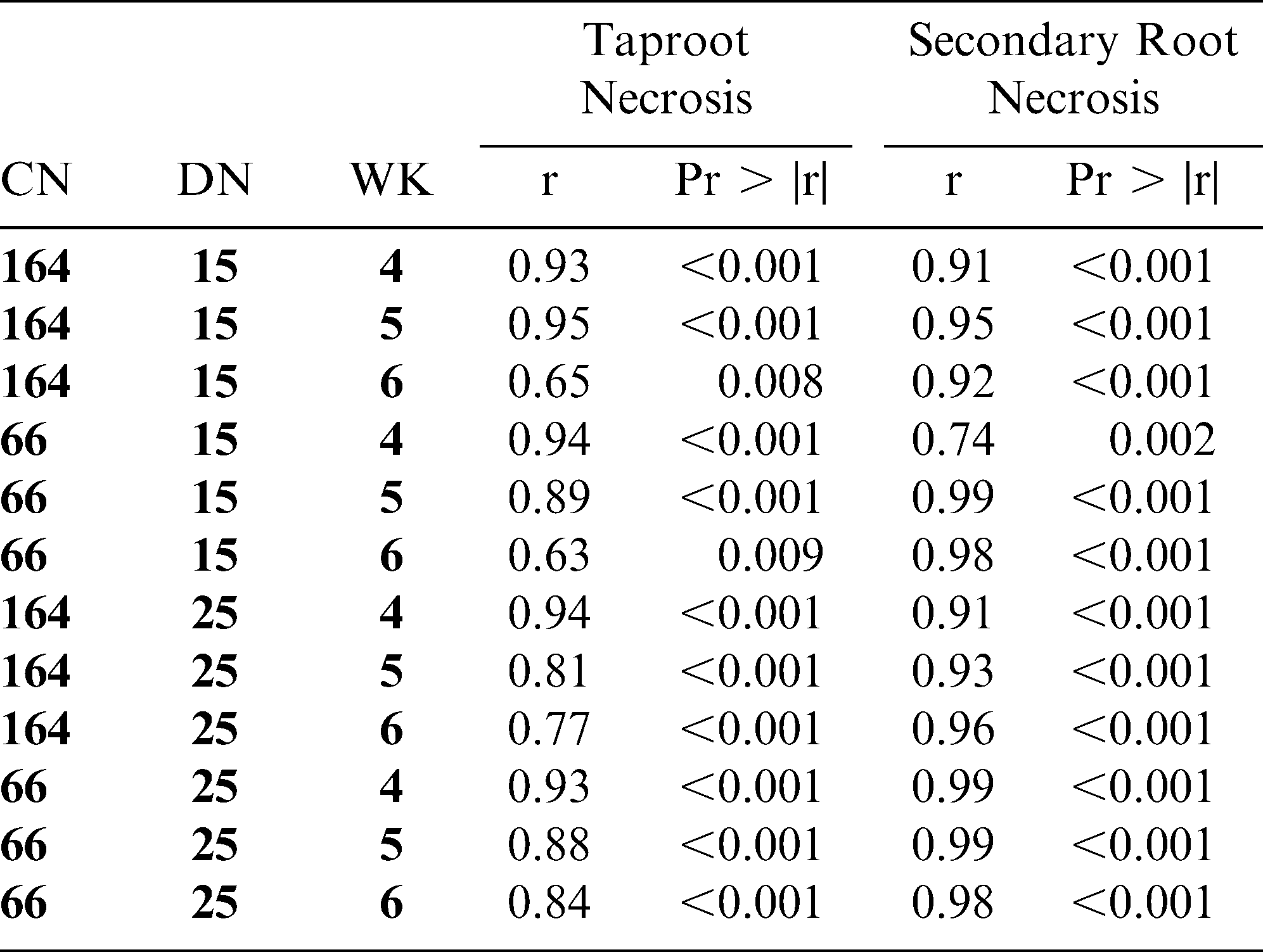
Analyses of non-parametric data in factorial experiments are problematic, although modified chi-square and analysis of variance tests have been developed to examine main effects and interactions (Shah and Madden, 2004). Our approach disregarded the factorial structure and analyzed data with a one-way design. This allowed for direct comparisons of disease severity ratings for NC 3033 and NC 7 via a chi-square test for each treatment combination. Spearman's nonparametric rank correlation in Proc CORR was used to test the potential relations between root rot ratings and taproot and secondary root necrosis for each treatment combination.
Results
Examination of combined analyses revealed that the contribution to overall variance was excessively high for the random effects of trial and trial by genotype interaction in multiple treatment combinations for each disease assessment; therefore data were analyzed by trial.
In trial I, root rot ratings for resistant and susceptible genotypes were different (P < 0.05) for all treatment combinations except in small containers at the 25 ms/g soil density at week 6 (P = 0.5) and in large containers at the 15 ms/g density at sampling week 5 (P = 0.08) (Table 1). Root rot ratings had identical means in large containers at the 15 ms/g density at both sampling weeks 5 and 6, but in week 5, cultivar differences were not significant (P = 0.084) and in week 6 they were significant (P = 0.003). Under the same conditions, means of secondary root necrosis in week 5 were the same as week 6, but the two cultivars were not different (P = 0.173) at week 5. However, at week 6, they were much closer to being significant (P = 0.054). While the means were the same for both root rot rating and secondary root necrosis at both times, the variability across the different replications decreased over time. The taproot necrosis ratings in the same combination indicated highly significant differences between cultivars (P < 0.001) for both times (Table 1).
In trial II, genotypes differed in all treatment combinations except in small containers at the 15 ms/g density at weeks 4 (P = 0.1) and 5 (P = 0.2) and in large containers at the 25 ms/g soil density at week 4 (P = 0.2) (Table 2). No single factor (container size, density, or duration) always differentiated the two genotypes. However, both container sizes and inoculum densities allowed differentiation with the root rot scale in all cases in one or more of the durations.
Significant genotype differences were detected with taproot necrosis ratings for each treatment combination in trial I. Four treatment combinations were similar for taproot necrosis between genotypes in trial II, including all three durations in small containers at the 15 ms/g soil density. As with root rot ratings, no single factor (container size, density, or week of evaluation) always differentiated the two genotypes.
For secondary root necrosis in trial I, genotypes were similar in three combinations with the large container size and one combination with small container size. In trial II, three combinations with the small container size and one combination with a large container resulted in non-significant genotype differences. One third of the total combinations between the two trials resulted in non-significant genotype differences with this rating method. All non-significant root rot rating and taproot necrosis treatment combinations were also non-significant for secondary root ratings in both trials. No one combination was always non-significant in trial I and trial II for any disease assessment method.
Root rot ratings were highly correlated with taproot necrosis ratings at every treatment combination (Tables 3 and 4) except in small containers at the 15 ms/g soil density at week 6 in trial I (r = 0.49) (P = 0.074) Root rot ratings were also highly correlated with secondary root necrosis ratings (Tables 3 and 4) except in large containers at the 15 ms/g soil density at week 5 in trial I (r = 0.46) (P = 0.073).
Discussion
In most greenhouse experiments, disease severity has been assessed using a 0–5 scale (Black and Beute, 1984; Rowe and Beute, 1975). This rating scale requires evaluation of secondary roots and the taproot, an assessment of missing roots, and fails to clearly define each level. Ordinal disease rating scales, including the 0–5 CBR scale, produce data that are not based on degree of infection on a continuous scale and therefore are not normally distributed. This type of data should not be subjected to parametric statistical methods such as analysis of variance (Madden et al., 2007; Snedecor and Cochran, 1989). Researchers have long argued over whether analysis of variance is robust to violations of assumptions such as normal data distribution. Previous studies utilizing root rot ratings to measure CBR disease severity have simply disregarded potential violations of assumptions and analyzed data using analysis of variance rather than employing non-parametric tests (Dong et al., 2008; Hollowell et al., 2008).
Our research demonstrates that taproot necrosis measurements are a valid alternative to root rot ratings. Taproot necrosis ratings are highly correlated to root rot ratings, can be evaluated with an image analysis system that is consistent, and do not present as many difficulties with data analysis. Finite-class disease scales such as taproot necrosis are potentially appropriate for analysis with statistical procedures such as analysis of variance and t-tests (Snedecor and Cochran, 1989). Taproot necrosis ratings separated NC 3033 and NC 7 in 20 out of 24 total treatment combinations and were slightly more reliable for separating the two cultivars than the traditional root rot ratings across both trials. For taproot necrosis, three of the four non-significant treatment combinations included the small container size and two occurred after 4 weeks, therefore sampling larger containers after 5 weeks provided the most consistent results.
For root rot ratings, the large container size was highly reliable in separating the two genotypes at the 25 ms/g soil density at week 6 in both trials. Using the small container size, there were no differences detected at all three rating times. With secondary root necrosis ratings, results were inconsistent across container sizes, weeks of sampling, and inoculum densities between trials. However, results were very similar for secondary root and taproot necrosis ratings in trial II. Both taproots and primary branch roots on NC 3033 are capable of forming additional periderm to seal off lesions produced by CBR (Harris and Beute, 1981; Harris and Beute, 1982), while NC 7 does not form periderm layers capable of impeding the pathogen (Brune, 1996). However, secondary root necrosis ratings alone do not appear to be robust enough to consistently differentiate between resistant and susceptible peanut genotypes.
We identified a protocol to reliably distinguish between resistant and susceptible genotypes. Experimental conditions in the growth chamber were highly consistent and therefore the rate of plant growth within and between tests was similar. However, there was still variability in severity from trial to trial. The variability between trials is likely due to the different sizes of sclerotia that are counted or not counted due to a continuum of microsclerotia sized (Dong, 2008) and/or changes in isolate aggressiveness between trials. Pataky et al. (1983) suggested that pronounced differences in the response of Florigiant to increasing inoculum densities between trials were a result of changes due to long term storage of C. parasiticum cultures. In this study, cultures were not stored for long periods (2 months), but the pathogen was re-isolated from peanuts grown in a growth chamber before the experiment and between trials, which could have increased pathogen aggressiveness between the trials.
Partial resistance in NC 3033 was overwhelmed by disease in small containers at the 15 ms/g soil density in trial II. Previous research has shown NC 3033's resistance to be inoculum density dependent (Harris and Beute, 1982; Pataky, 1983) and related to secondary root re-growth (Brune, 1996; Harris and Beute, 1982). Different results between the two trials are likely due to density differences and the potential for density to interact with root growth (which is affected by container size and time of evaluation). Container size may interact with root growth and inoculum density, and may be critical for optimal evaluation of resistance.
Acknowledgements
We appreciate the support of the Texas Peanut Producers Board and Texas AgriLife Research. NC 3033 seed was provided by Dr. T.G. Isleib at North Carolina State University, Raleigh.
Literature Cited
Bell D.K and Sobers E.K 1966 A peg, pod, and root necrosis of peanuts caused by a species of Calonectria Phytopathology 56 : 1361 – 1364 .
Beute M.K Wynne J.C and Emery D.A 1976 Registration of NC 3033 peanut germplasm Crop Sci. 16 : 887 .
Black M.C and Beute M.K 1984 Effects of rotations with susceptible and resistant peanuts, soybeans, and corn on inoculum efficiency of Cylindrocladium crotalaria on peanuts Plant Dis. 68 : 401 – 405 .
Brune P.D 1996 Differences in peanut (Arachis hypogaea L.) root growth dynamics which may provide field resistance to Cylindrocladium black rot PhD. Disseration, North Carolina State University , Raleigh .
Cline W.O and Beute M.K 1986 Effect of metam sodium, peanut genotype, and inoculum density on Cylindrocladium block rot Peanut Sci. 13 : 41 – 45 .
Dong W.B Brenneman T.B Holbrook C.C and Culbreath A.K 2008 Evaluation of resistance to Cylindrocladium parasiticum in runner-type peanut in the greenhouse and field Peanut Sci. 35 : 139 – 148 .
Garren K.H Beute M.K and Porter D.M 1972 The Cylindrocladium black rot of peanut in Virginia and North Carolina Proc. Am. Peanut Res. Ed. Assoc. 4 : 67 – 71 .
Harris N.E and Beute M.K 1981 Histological responses of peanut germplasm resistant and susceptible to Cylindrocladium crotalariae in relationship to inoculum density Phytopathology 72 : 1250 – 1256 .
Harris N.E and Beute M.K 1982 Cylindrocladium crotalariae-induced periderm formation in taproot and fibrous roots of Arachis hypogaea Peanut Sci. 10 : 66 – 69 .
Hollowell J.E Isleib T.G Tallury J.P Copeland S.C and Shew B.B 2008 Screening of Virginia-type peanut breeding lines for resistance to Cylindrocladium black rot and Sclerotinia blight in the greenhouse Peanut Sci. 35 : 18 – 24 .
Kucharek T.A 2005 Disease control program for peanuts Extension Plant Pathology Report No. 12, 24th revision, University of Florida , Gainesville http://plantpath.ifas.ufl.edu/takextpub/ExtPubs/ppp1205.pdf .
Lemay A.V 1999 The management of Sclerotinia minor and Cylindrocladium parasiticum on peanut (Arachis hypogaea L.) with host resistance and chemical control M.S. Thesis, North Carolina State University , Raleigh .
Madden L.V Hughes G and van den Bosch F 2007 The study of plant disease epidemics APS Press , St. Paul, MN .
Pataky J.K Black M.C Beute M.K and Wynne J.C 1983 Comparative analysis of Cylindrocladium black rot resistance in peanut: Greenhouse, microplot, and field testing procedures Phytopathology 73 : 1615 – 1620 .
Phipps P.M and Beute M.K 1979 Population dynamics of Cylindrocladium crotalariae microsclerotia in naturally infested soil Phytopathology 69 : 240 – 243 .
Phipps P.M Beute M.K and Barker K.R 1976 An elutriation method for quantitative isolation of Cylindrocladium crotalariae microsclerotia from peanut field soil Phytopathology 66 : 1255 – 1259 .
Phipps P.M and Beute M.K 1997 Cylindrocladium black rot pp. 12 – 15 In: Compendium of peanut diseases. 2nd ed , Kokalis-Burelle N Porter D.M Rodriguez-Kabana R Smith D.H and Subrahmanyan P (eds.) , APS Press , St. Paul, MN .
Rowe R.C and Beute M.K 1975 Variability in virulence of Cylindrocladium crotalariae isolates on peanut Phytopathology 65 : 422 – 425 .
Shah D.A and Madden L.V 2004 Nonparametric analysis of ordinal data in designed factorial experiments Phytopathology 94 : 33 – 43 .
Snedecor G.W and Cochran W.G 1989 Statistical methods. 8th ed Iowa State University Press , Ames .
Wheeler T.A and Black M.C 2005 First report of Cylindrocladium parasiticum on peanut in Texas Plant Dis. 89 : 1245 .
Notes
- Dept. of Soil and Crop Sciences, Texas A&M University, College Station, TX 77843
- Texas AgriLife Research, 1102 East FM 1294, Lubbock, TX 79403
- Texas AgriLife Extension Service, P. O. Box 1849, Uvalde, TX *Corresponding author: J.N. Wilson (E-mail: jwilson@ag.tamu.edu)
Author Affiliations