Introduction
In the Virginia-Carolina (VC) area, peanut is an important cash crop. According to the United Nations Conference on Desertification (UNEP, 1992), this region is characterized as a moist sub-humid to humid region. This classification is based on the climatic aridity index: P/ETP, where P is annual precipitation and ETP is annual potential evapotranspiration calculated by the method of Penman (Doorenbos and Pruitt, 1977). Under historically moist conditions, over 90% of the current peanut farming is rainfed in the VC area. However, increased frequency and intensity of dry spells were recently observed due to uneven precipitation distribution during the summer months with negative effects on peanut yield and quality in the VC region. To increase peanut production under rainfed cropping systems in dry years, peanut cultivars with more effective use of water need to be developed for this region.
In peanut (Devi et al., 2010) and soybean [Glycine max (L.) Merr.] (Fletcher et al., 2007; Sinclair et al., 2008; Sadok and Sinclair, 2009) it has been shown that transpiration rate linearly increased with increasing VPD at low VPD values, but it was stable at VPD over 2.5 kPa. Devi et al., (2010) suggested that stomata closure at midday in response to high midday VPD will limit water loss by peanut crops. Evidence by Wright et al. (1994) that peanut can maintain high photosynthetic activity while minimizing water loss, determined Devi et al. (2010) to propose midday stomata closure in response to increase VPD as a mechanism of more effective use of water. These authors' study was performed in a greenhouse and under well watered conditions, and included peanut genotypes previously identified with different transpiration efficiency.
Virginia-type is the predominant peanut market type grown in the VC region. To our knowledge, there is no information on the response of stomatal conductance (gs) to midday VPD of the virginia-type genotypes currently grown in the VC region. Given the erratic nature of rainfall patterns, efforts are needed to enhance the effective water use of the virginia-type peanut in this region. Change of transpiration with increasing VPD it would seem to be an effective mechanism of soil moisture conservation and better water use during dry spells and later in the season during seed filling. This study was conducted to determine the relationship of gs to midday VPD in virginia-type cultivars and advanced breeding lines under field conditions.
Materials and Methods
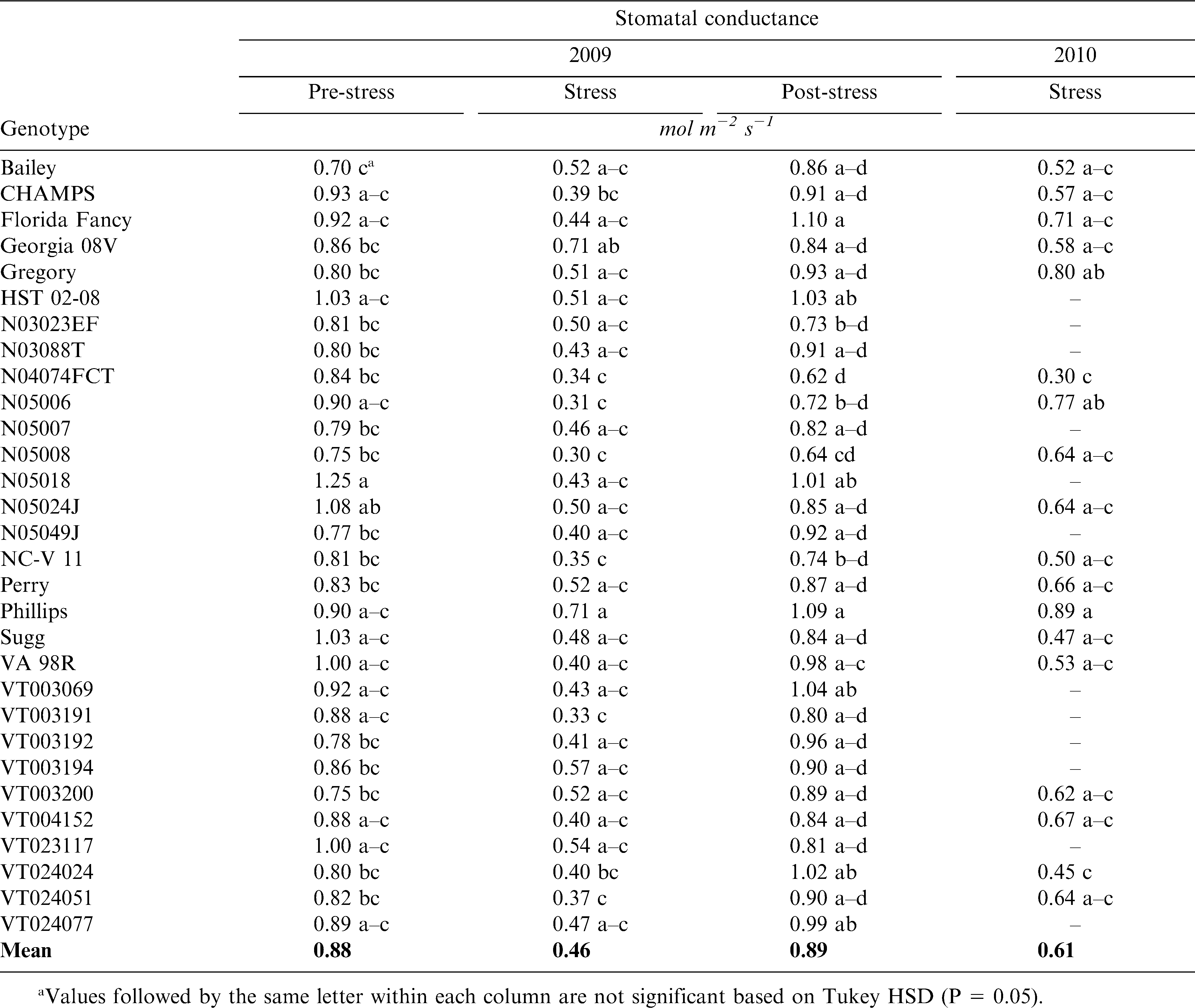
In 2009, thirty and in 2010, eighteen peanut cultivars and advanced breeding lines were evaluated at Tidewater Agricultural Research and Extension Center near Holland, VA (36° 68′ N, 76° 77′ W, 18.9 m elevation) for midday gs and VPD (Table 1). These genotypes represent currently grown virginia-type cultivars and advanced breeding lines with desirable agronomic characteristics tested in the Peanut Variety and Quality Evaluation (PVQE) trials. Genotypes were grown in plots of two 9.1 m rows planted on 0.9 m centers. The soil at this site is classified as Eunola (fine-loamy, siliceous, thermic Aquic Hapludults). Genotypes were planted on 20 April in 2009 and 15 April in 2010. Cultural practices were performed according to Virginia recommendations for production of high yield and quality (Faircloth and Shokes, 2008). Detailed information on the cultural practices is described by Balota (2010, 2011). Plots were replicated two times in 2009 and three times in 2010 in a randomized complete block design with genotype as the only factor. Measurements of gs were taken with a LI-6400 IRGA portable photosynthesis system (LI-COR Biosciences Inc., Lincoln, NE). Equations used to calculate gs were derived from von Caemmerer and Farquhar (1981) and were based on measured flow rates and water concentrations in the sample and reference IRGAs, and a pre-set leaf area of 6 cm2. Equations were provided by the OPEN software v 5 (LI-COR Biosciences Inc., Lincoln, NE). To keep the leaf chamber at ± 0.5 C from air temperature, the block temperature feature of the equipment was used. Relative humidity (RH) inside the chamber was keep at less than 2% variation by using a high flow rate of 500 µmol s−1. During measurements, PPFD was maintained constant 2000 µmol m−2 s−1 with a 6400-02B light source (LI-COR Biosciences Inc., Lincoln, NE). A 6400-01 CO2 mixer (LI-COR Biosciences Inc., Lincoln, NE) was used to inject and maintain a constant concentration of 400 µmol CO2 mol−1 air during measurements. Only fully sunlit leaves were used for the measurements. Leaves were allowed to equilibrate for 60 s before each reading of gs. The middle leaflets of the uppermost fully developed leaf on the main stem were used.
Air VPD was calculated from the saturation vapor pressure from Buck (1981) and mean RH. Air temperature, RH, and precipitation were continuously monitored next to the plots with a Watchdog weather station (Model 2475; Spectrum Technologies, Inc., Plainfield, IL) at 2 m from the ground.
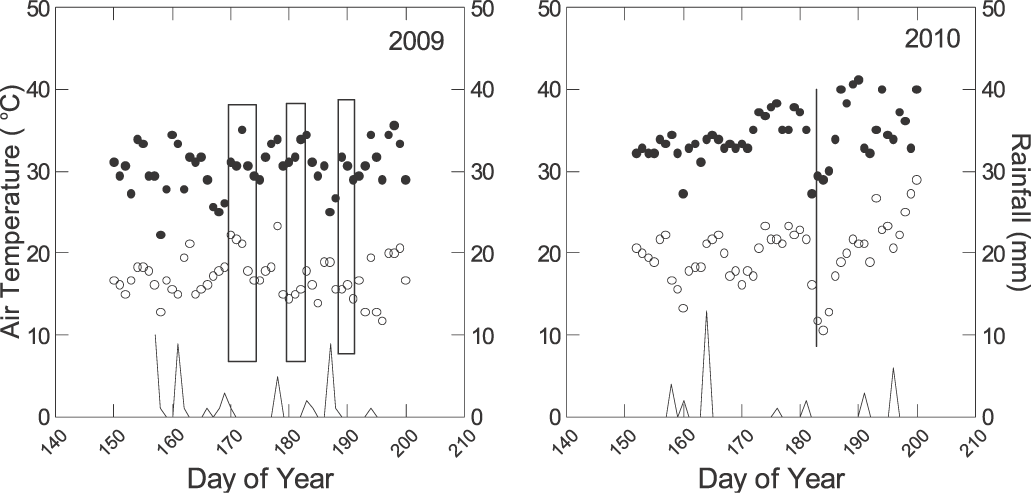
In 2009, measurements were taken on 19, 20, 22, 24, 29 and 30 June, and 1, 7, 8, and 9 July (Fig. 1). On 19, 20, 22, and 24 June plants were at approximately 50% anthesis (R1) (Boote, 1982). No significant rain event was recorded for 10 d prior R1 and this time was considered a pre-stress environment. After approximately 20 d without significant rain, 29 and 30 June, and 1 July, plants were at beginning peg (R2) stage. This time was considered a stress environment as plants in all plots were showing slight leaf rolling later in the afternoon. On 5 July, 25.4 mm irrigation was applied prior to a 10 mm rainfall. Plants were at beginning pod (R3) stage. Time from 7 to 9 July was considered a post-stress environment. This strategy, i.e., sampling on consecutive days, was done in order to obtain a range of temperatures and RH to better characterize the gs response to VPD. Data from consecutive days within each stress level/growth stage were combined for further analyses. Their combination was possible because the genotype × day of measurement interaction was not statistically significant based on the repeated measure analysis. On individual days, all genotypes were measured between 1100 and 1300 h, one leaf per plot. In 2010, measurements were taken on 1 July at R2 stage after approximately 20 d without significant rainfall and irrigation, and when plants in all plots were showing slight leaf rolling later in the afternoon (Fig. 1). The first replication was sampled from 1100 to 1200, the second from 1200 to 1300, and the third from 1300 to 1400. For each replication, one plant from all plots was measured followed by a second plant in the reverse plot order. This sampling strategy was also satisfactory for creating a range of temperatures and RH while minimizing the differences between plots due to the time of measurement.
In 2009, gs was assessed for the interaction of genotype and sampling time (i.e., day of measurement and stress level/growth stage) using the repeated measure design by Cochran and Cox (1957) in SYSTAT. Genotype effect on gs within each stress level/growth stage was further assessed with ANOVA from the GLM procedure of SYSTAT® 10.2 (2002, SYSTAT Software Inc, Richmond, CA). Means were separated by Tukey HSD test at P = 0.05. Simple linear regression equations were fitted to gs and VPD to evaluate the effect of genotype on the transpiration response to VPD (Devi et al., 2010).
Results
In 2009 during pre-stress environment, average temperature during measurements ranged from 28.5 to 33.6 C and RH from 32 to 67%. During the stress environment, temperature ranged from 28.7 to 34.5 C and RH from 30.9 to 47.2 %. During the post-stress environment, temperature ranged from 25.2 to 29.9 C and RH from 41 to 58%. In 2010, the average RH was 39%, air temperature 27.8 C (±0.90 SD), and leaf temperature 27.4 C (±1.86 SD). In 2009, the average air temperature across all environments was 30.2 C (±2.4 SD) and leaf temperature was 32.5 C (±2.6 SD). Across all experiments in 2009, the range of VPD was from 1.29 to 4.22 kPa (±0.56 SD) and in 2010 from 1.78 to 3.57 kPa (±0.23 SD).
Genotype had a significant (P ≤ 0.002) effect on gs in all environments and in both years (Table 1). Phillips was among the highest and N04074FCT the lowest for gs, consistently across environments. In 2009, the repeated measure analysis showed a significant effect of genotype (P = 0.037) and stress level (P < 0.0001) on gs, but the genotype × stress level interaction was not significant (P = 0.290). When common genotypes for 2009 and 2010 were compared, genotype and year showed a significant effect on gs (P = 0.0001), but the genotype × year interaction did not.
The relationship between gs and VPD when all genotypes were combined across individual environments is presented in Table 2. Negative correlations were recorded at pre- and stress environments in both years. Under these environments, average slopes of gs and VPD ranged from −0.46 ± 0.05 to −0.11 ± 0.06 mol H2O m−2 s−1 (Table 2). Under water stress in both years, average slopes were similar, −0.33 ± 0.02 in 2009 and −0.46 ± 0.05 mol H2O m−2 s−1 in 2010. The relationship between gs and VPD was strongest at stress compared with pre- and post-stress, and under stress R2 was 0.57 (n = 180) in 2009 and 0.47 (n = 108) in 2010. When genotypes were combined, no relationship was found between post-stress gs and VPD.
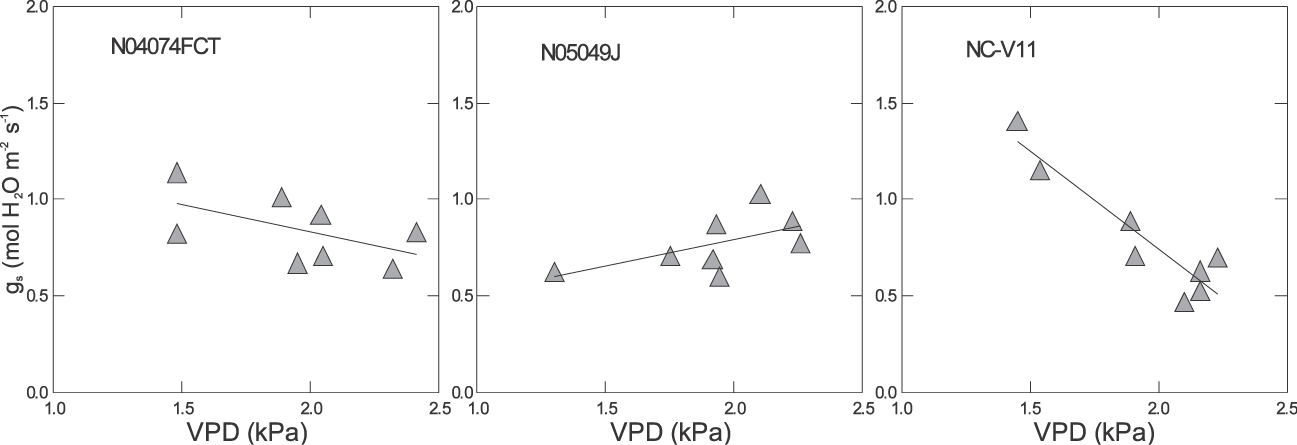
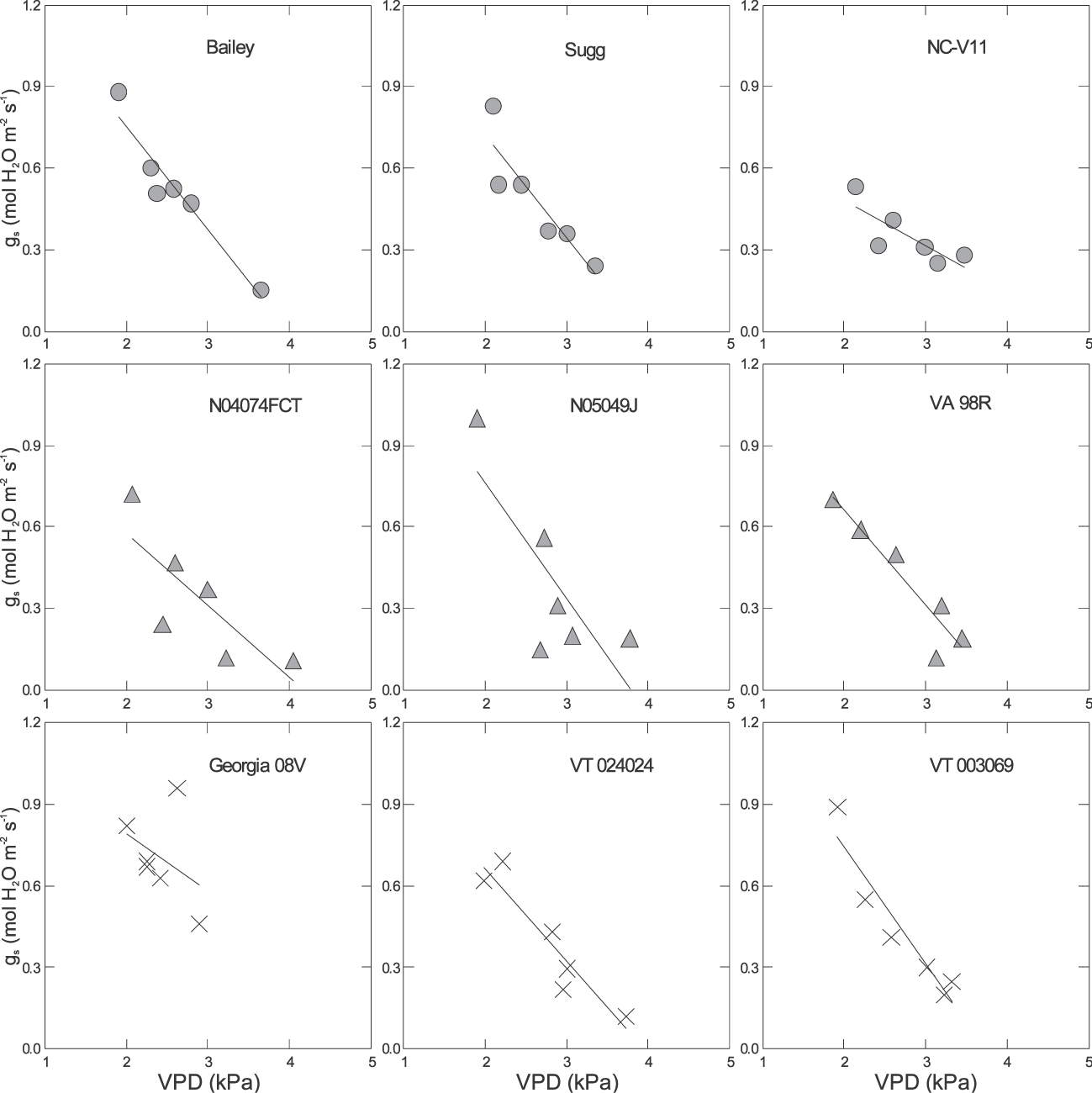
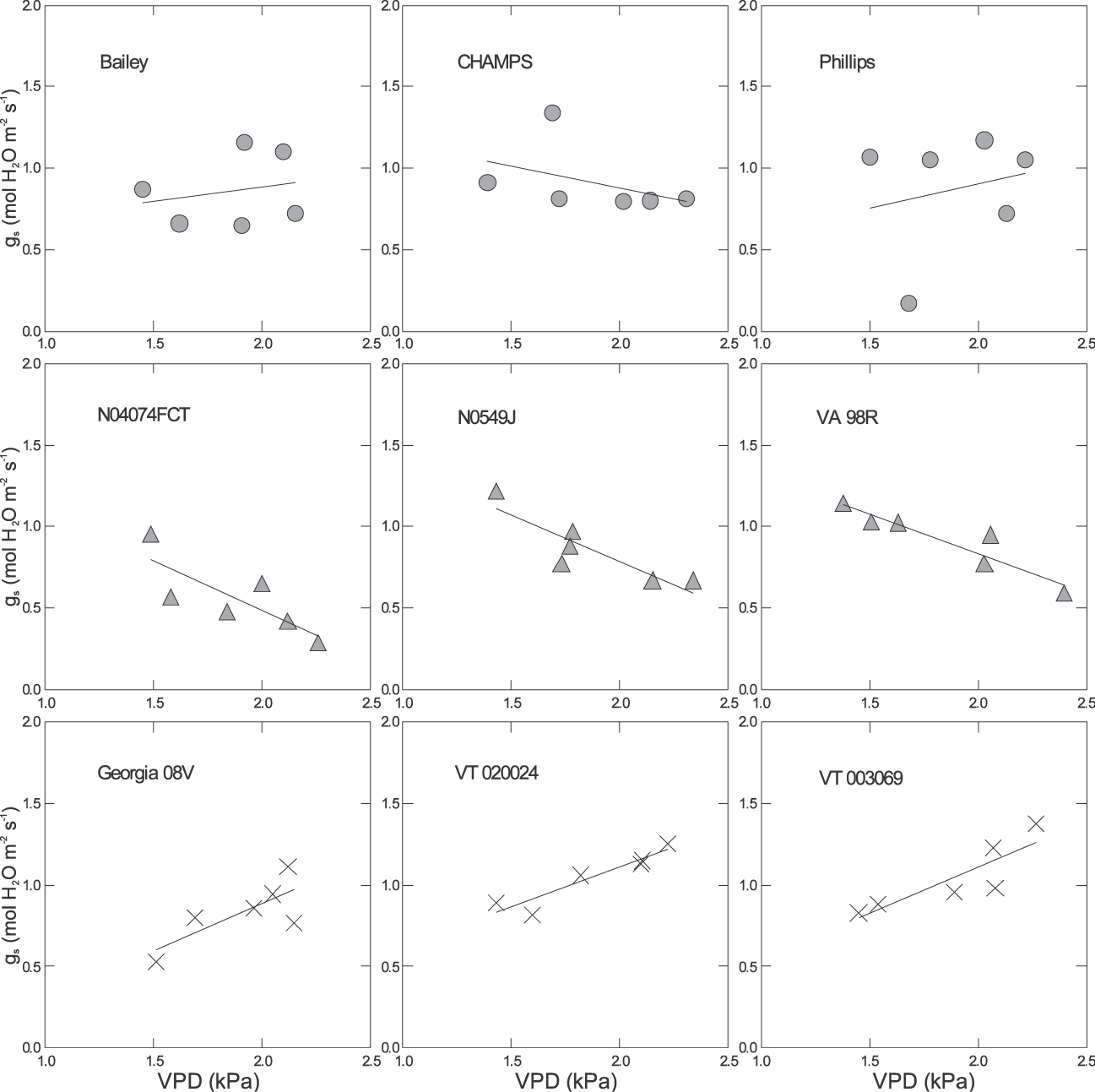
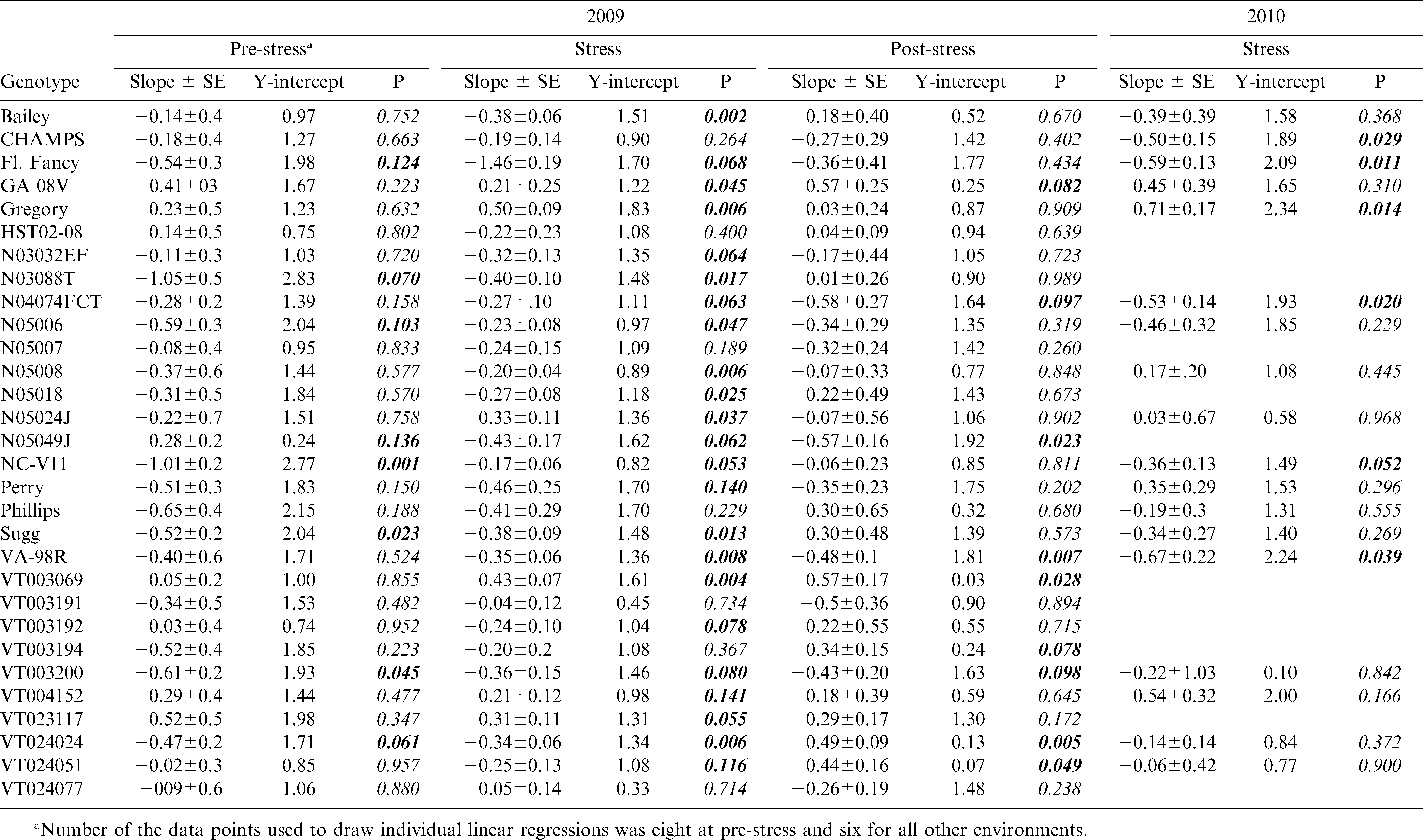
There were significant differences among genotypes for the response of gs to VPD in all environments (Table 3 & Fig. 2– 4). For example under pre-stress environment, when VPD ranged from approximately 1.4 to 2.5 kPa, 11 genotypes including Bailey (Isleib et al., 2011), CHAMPS (Mozingo et al., 2006), Gregory (Isleib et al., 1999), HST02-08, N03032EF, N05007, N05024J, VT003192, VT003069, VT024051, and VT024077 showed no relationship between gs and VPD. For these genotypes, the slopes of the regression ranged from −0.22 to 0.03 mol H2O m−2 s−1 (Table 3). Thirteen genotypes showed a moderate gs decrease with increasing VPD, and those slopes ranged from approximately −0.65 to −0.28 mol H2O m−2 s−1. Florida Fancy, Georgia 08V, Perry, and Phillips were among them (Table 3). For some, the regressions were significant at P = 0.1 or less, but for others the regressions were not statistically significant (Table 3). Five genotypes showed a sharp decrease of gs with increasing VPD at pre-stress. Slopes of gs vs. VPD for these genotypes ranged from −1.05 to −0.47 mol H2O m−2 s−1, and for all the regressions were statistically significant (P ≤ 0.1). They were N03088T, NC-V11, Sugg, VT003200, and VT024024. Finally, genotype N05049J showed an increase of gs with increasing VPD (Table 3). Examples of genotypic response of gs to VPD at pre-stress are presented in Fig. 2. In 2009 under stress, VPD ranged from 2.1 to 3.6 kPa. Stomatal conductance significantly declined with increasing VPD in 25 genotypes (P ≤ 0.1) with slopes ranging from −0.5 to −0.20 mol H2O m−2 s−1. Examples are presented in Fig. 3. In 2010, 6 genotypes showed a significant reduction of gs with increasing VPD; for all others the relationship was weak. These genotypes were CHAMPS (P = 0.029), Florida Fancy (P = 0.011), Gregory (P = 0.014), N04074FCT (P = 0.020), NC-V11 (P = 0.052), and VA 98R (P = 0.039). Under post-stress environment, when VPD ranged from 1.4 to 2.5 kPa, positive relationships between gs and VPD were recorded for Bailey, Georgia 08V, Phillips, VT003069, VT003192, VT003194, VT024024, VT004152, and VT024051. For these genotypes slopes of gs vs. VPD ranged from 0.18 to 0.57 mol H2O m−2 s−1 (Table 3). For some, regression was significant at P = 0.1 or less, but for others regression was not statistically significant (Table 3). Examples of increase of gs with increasing VPD after rain and irrigation events are presented in Fig. 4. N04074FCT, N05006, N05007, N05049J, Perry, VA 98R, VT003200, VT023117, and VT024077 continued to show a significant decline in gs with increasing VPD post-stress and slopes ranged from −0.26 to −0.57 mol H2O m−2 s−1 (Table 3); examples are presented in Fig. 4. For the remaining genotypes no relationship was observed between post-stress gs and VPD.
Examples of response of stomatal conductance (gs) to vapor pressure deficit (VPD) at midday and pre-stress in 2009. For equations of the regressions of gs to VPD, slopes, Y-intercept, and P values are presented in Table 3.
Examples of response of stomatal conductance (gs) to vapor pressure deficit (VPD) at midday and stress in 2009. Equations of the regressions of gs to VPD, slopes, Y-intercept, and P values are presented in Table 3.
Examples of response of stomatal conductance (gs) to vapor pressure deficit (VPD) at midday and post-stress in 2009. For equations of the regressions of gs to VPD, slopes, Y-intercept, and P values are presented in Table 3.
Discussion
Data were collected under rainfed field conditions typical for Eastern Virginia in June and July. During these months plants can experience water stress due to scarce precipitation. Based on relatively high gs values in Table 1, the peanut genotypes were not under severe water stress at any sampling time, even though at certain times they appeared to be stressed. Under severe water deficit, peanut can close stomata and reduce its conductance to nearly zero (Kottapalli et al., 2009), but in our study, a genotype's average gs was no less than 0.46 mol m−2 s−1. Under mild rather than severe water stress, closing stomata in response to increased VPD would seem to be a useful mechanism of conserving water. In our experiment, genotypes, stress level/growth stage, and year showed significant effects on gs, but their interaction did not. This denotes a strong genotypic effect on gs. Genotypes N04074FCT and NC-V11 had consistently lower gs values compared with the other genotypes at all sampling times in 2009 and 2010 (Table 1). Phillips, Gregory, Georgia 08V, and Florida Fancy had higher gs values across sampling times and years, and almost 30% higher than N04074FCT and NC-V11. In part, this can be explained by a different number of stomata per leaf but other factors are involved as well (Balota, unpublished data). When all genotypes were combined under water stress, i.e., average gs was 0.46 in 2009 and 0.61 mol m−2 s−1 in 2010, gs was negatively related to VPD (R2 = 0.57, n = 180 in 2009; R2 = 0.47, n = 108 in 2010), but the relationship was not significant when average gs exceeded 0.88 mol m−2 s−1 (Table 2). However, a wide range of slopes among genotypes were observed in all environments and years (Table 3). In this respect, our field data are in agreement with the green house observations by Devi et al., (2010) that genotypic differences for the response of transpiration to VPD in peanut exist. Genotypes with significant negative relationships of gs and VPD under water stress in both years were Florida Fancy, Gregory, N04074FCT, NC-V11, and VA-98R. While Florida Fancy, Gregory, and NC-V11 are known to be high yielding cultivars, VA-98R and line N04074FCT are not (Wynne et al., 1991; Isleib et al., 1999; Balota, 2010).
Three groups of genotypes were distinct for the response of gs to VPD in our tests. The first group showed decrease or no response at pre-stress, decrease at stress, and no response of gs with increasing VPD at post-stress. Genotypes in this group are high yielding cultivars and breeding lines such as NC-V11, Florida Fancy, Sugg, Gregory, and N05018, but also documented low yielding lines in PVQE tests in VC region, N03023EF, and HST 02-08 (Wynne et al., 1991; Isleib et al., 1999; Balota, 2010). The second group showed slow decreases at pre-stress, decreases under stress, and significant increases of gs with increasing VPD at post-stress. Genotypes included in this group were Georgia 08V, VT003069, VT024051, VT024024, and VT003194. Bailey and Phillips also showed small increases of gs with increasing VPD post-stress, but the relationships were not statistically significant. All these genotypes are high yielding cultivars presently grown for high production in the VC area (Branch, 2009; Isleib at al., 2006, 2011; Mozingo et al., 2006) and advanced breeding lines that were tested for potential release in PVQE tests in Virginia, North Carolina and South Carolina (Balota, 2010, 2011). An important characteristic for these genotypes was their ability to resume transpiration after rain and irrigation, and to fit into the well watered plant model of transpiration increasing linearly with increasing VPD (Sinclair and Bennett, 1998). This suggests that these genotypes recovered well from water stress. Crop production is proportional to its transpiration (de Wit, 1958); therefore, the ability to resume high rates of transpiration after drought episodes and when water becomes available seems to be an important drought tolerance characteristic for the Virginia-type peanut grown in the VC environments. Finally, the third group showed significant decreases of gs with increasing VPD under all environments, with the exception of N05049J, which showed a positive response of gs to VPD at pre-stress and negative at stress and post-stress. None the less all genotypes in this group continued to show declines of gs to VPD increases after rain and irrigation. Based on this experiment, we cannot conclude if the relationship between gs and VPD was typical for moist conditions or somehow stress affected the post-stress relationship for these genotypes. Examples of genotypes in this group are N04074FCT, N05049J, Perry, and VA 98R. Indeed, based on PVQE tests these genotypes appear to have low yields, in particular under sub-optimal growing conditions. For example, N04074FCT was the lowest yielding genotype in PVQE tests at six locations in Virginia, North Carolina, and South Carolina, in 2009 and 2010 (Balota, 2011). In 2010, a record year for sustained drought throughout the VC peanut growing region, Perry and VA 98R had the lowest yields at all PVQE locations among commercial cultivars. Finally, N05049J was the second lowest for pod yield after N04074FCT in PVQE tests in 2008 and 2009 (Balota, 2010). Our data clearly show that genetic differences among tested genotypes existed for gs and the relationship of gs with VPD. Also, based on results from water stress in both years, it appears that not only high but also low yielding genotypes consistently expressed reduced gs with increasing VPD, therefore questioning the importance of this drought tolerance mechanism under our environments. From only 2009 data, there is indication that the relationship of gs and VPD after rather than during water stress could be more important and better associated with yield in dry years for the VC region, but more research is needed to confirm this hypothesis and to elucidate how stomata closure in response to VPD can increase drought tolerance and yield in the VC region.
Acknowledgements
This work was supported with funding from the National Peanut Board, Virginia Peanut Board, Virginia Crop Improvement Association, North Carolina State University, and South Carolina Peanut Board for which authors are very appreciative. Authors are appreciative for Dr. Tom Sinclair's thorough review and suggestions to improve the manuscript.
Literature Cited
Balota M 2010 Peanut variety and quality evaluation results. I. Agronomic and grade data Virginia Coop. Ext. Publ. 3001 – 1432 (Available on-line with updates at http://pubs.ext.vt.edu/3001/3001-1432/3001-1432.html) .
Balota M 2011 Peanut variety and quality evaluation results: agronomic and grade data Virginia Coop. Ext. Publ. 3101 – 1532 (Available on-line with updates at http://pubs.ext.vt.edu/3101/3101-1523/3101-1523.html) .
Boote K.J 1982 Growth stages of peanut (Arachis hypogaea L.) Peanut Sci. 9 : 35 – 40 .
Branch W.D 2009 Registration of ‘Georgia-08V’ peanut J. Plant Registrations 3 : 143 – 145 .
Buck A.L 1981 New equations for computing vapor pressure and enhancement factor J. Appl. Meteor. 20 : 1527 – 1532 .
de Wit C.T 1958 Transpiration and crop Yields. Vol. 64.6 Versl. Landbouwk. Onderz., Institute of Biological and Chemical Research on Field Crops and Herbage , Wageningen, The Netherlands , pp. 24 .
Cochran W.G and Cox G.M 1957 Experimental designs Wiley , New York , 2nd ed.
Devi M.J Sinclair T.R and Vadez V 2010 Genotypic variation in peanut for transpiration response to vapor pressure deficit Crop Sci. 50 : 191 – 196 .
Doorenbos J and Pruitt W.O 1977 Crop water requirements Food and Agriculture Organization Irrigation and Drainage Paper 24. FAO , Rome .
Faircloth J.C and Shokes F.M 2008 Agronomic recommendations and procedures In Virginia Peanut Guide , Faircloth J.C (ed.), VPI&SU/Va. Agric. Exp. Stn./TAREC, 74 p.
Fletcher A.L Sinclair T.R and Allen L.H 2007 Transpiration responses to vapor pressure deficit in well watered ‘slow-wilting’ and commercial soybean Environ. Exp. Bot. 61 : 145 – 151 .
Kottapalli K.R Rakwal R Shibato J Burow G Tissue D Burke J Puppala N Burow M and Payton P 2009 Physiology and proteomics of the water-deficit stress response in three contrasting peanut genotypes Plant Cell Environ. 32 : 380 – 407 .
Isleib T.G Milla-Lewis S.R Pattee H.E Copeland S.C Zuleta M.C Shew B.B Hollowell J.E Sanders T.H Dean L.O Hendrix K.W Balota M and Chapin J.W 2011 Registration of ‘Bailey’ peanut J. Plant Registrations 2011, 5 : 27 – 39 .
Isleib T.G Rice P.W Mozingo R.W Copeland S.C Graeber J.B Pattee H.E Sanders T.H Mozingo R.W and Coker D.L 2006 Registration of ‘Phillips’ peanut Crop Sci. 46 : 2308 – 2309 .
Isleib T.G Rice P.W Mozingo R.W Mozingo R.W Pattee H.E and Pattee H. E 1999 Registration of ‘Gregory’ peanut Crop Sci. 39 : 1526 .
Mozingo R.W Coffelt T.A Phipps P.M Coker D.L Machado S and Petrie S.E 2006 Registration of ‘CHAMPS’ peanut Crop Sci. 46 : 2711 – 2712 .
Sadok W and Sinclair T.R 2009 Genetic variability for transpiration response to vapor pressure deficit among soybean cultivars Crop Sci. 49 : 955 – 960 .
Sinclair T.R and Bennett J.M 1998 Water In Sinclair T.R and Gardner F.P (eds.) Principles of ecology in plant production CAB Int. , Wallingford, UK .
Sinclair T.R Zwieniecki M.A and Holbrook N.M 2008 Low leaf hydraulic conductance associated with drought tolerance in soybean Physiol. Plant. 132 : 446 – 451 .
United Nations Environmental Program 1992 , World atlas of desertification UNEP , Nairobi, Kenya .
von Caemmerer S and Farquhar G.D 1981 Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves Planta 153 : 376 – 387 .
Wynne J.C Coffelt T.A Mozingo R.W and Anderson W.F 1991 Registration of ‘NC-V 11’ peanut Crop Sci. 31 : 484 – 485 .
Notes
- Tidewater Agricultural Research and Extension Center, Virginia Tech
- Department of Crop Science, North Carolina State University *Corresponding author email: mbalota@vt.edu
Author Affiliations