Introduction
Efficient management of pests is important in optimizing peanut yield and economic returns (Mullen et al., 1997; Napit et al., 1988). Making production and pest management decisions relative to peanut can be challenging because many variables affect peanut yield and quality. A wide range of pests can negatively affect peanut yield, including a number of broadleaf and grass weeds (Wilcut et al., 1995), several pathogens causing foliar, stem, and root diseases, several nematode species (Sherwood et al., 1995), and major insect pests (Lynch and Mack, 1995). Costs associated with peanut production can vary considerably, and the additional costs of pest control can influence a grower’s decision to employ management practices. Control costs associated with diseases, insects, and mites are approximately 12% of total annual operating costs in North Carolina (Bullen and Jordan, 2014). Implementation of strategies to manage one pest can increase or decrease the risk of development of other pests, which makes management decisions complex (Herbert et al., 2004; Jordan et al., 1999).
Southern corn rootworm (SCRW) is a primary pest of peanut in fine-textured soils, and direct injury is caused by larvae penetrating the pod to feed on the kernel. Pod scarring occurs when larvae are unsuccessful at penetrating the outer pod wall, and can reduce peanut value. Because larvae are subterranean, scouting is difficult; therefore, SCRW control with insecticides is preventive. The decision to make an insecticide application is based upon a risk index calculation of conditions particular to the site (Brandenburg, 2014).
Disease management in peanut is important to optimize both yield and quality. Uncontrolled diseases result in yield losses of up to 50% (Backman and Crawford, 1984; Besler et al., 2006; Waggoner and Berger, 1987). A number of methods are used to reduce disease incidence in peanut, including rotation, cultivar resistance, avoiding inoculum through field selection, fungicide applications, and fumigation. Tomato spotted wilt (TSW), a systemic disease caused by Tomato spotted wilt virus, can be sporadic, but yield-limiting during times of severe infestation. In 2002, 47% of North Carolina peanut hectares was affected by TSW (Cochran et al., 2003). The virus is transmitted by tobacco thrips, and reduces peanut pod quality as well as overall plant vigor. Tomato spotted wilt cannot be controlled by a single practice, so the incorporation of several methods is necessary in order to reasonably suppress the virus. Those methods include increased plant populations, planting resistant cultivars, planting in twin rows, altering planting dates to avoid peak thrips flight, and applying phorate in-furrow (Culbreath et al., 2003).
Cylindrocladium black rot (CBR), which is caused by the soil-borne fungus Cylindrocladium parasiticum, is a serious disease. Management requires an integrated approach of fumigation with metam sodium, planting partially resistant cultivars and clean seed, rotating crops, and avoiding fields with a history of CBR (Shew, 2014). Sclerotinia blight (SB) caused by (Sclerotinia minor) results in bleached and necrotic lesions on peanut leaves and severely shredded stems. Protectant fungicide applications are often needed, even on partially resistant cultivars, and are applied using weather-based advisories to ensure timely application when environmental conditions are conducive to disease development (Phipps et al., 1997).
Peanut is also susceptible to several plant parasitic nematodes, including northern root knot (Meloidogyne hapla), peanut root knot (Meloidogyne arenaria), and sting (Belonolaimus spp) (Dickson and De Waele, 2005). Two to three year rotations into non-host crops coupled with fumigation with metam sodium, which also controls CBR, can reduce nematode numbers (Phipps and Elliott, 1981; Jordan et al., 2008; Shew, 2014).
Various factors specific to the cropping system must be considered, and weighed by the grower in order to make the most prudent pest management decisions with regard to the environment and the profitability of the farm enterprise. Cultivar selection is important because different cultivars express considerable differences in resistance to individual diseases, especially to TSW, CBR, and SB (Shew, 2014). Planting pattern refers to either single or twin row planting patterns, and the difference in planting pattern and plant population can affect pest reaction. For example, TSW incidence has been shown to be lower in twin rows while incidence of stem rot can increase in twin rows compared with single rows (Baldwin et al., 2001; Lanier et al., 2004). Fumigation with metam sodium can suppress CBR and nematodes (Jordan et al., 2008; Shew et al., 2014). All of these factors and approaches to pest management can interact with each other, and a better understanding of the complexities of pest interactions within a cropping system can aid growers in weighing the costs and benefits of competing control measures.
While the role of twin row planting patterns is well documented in reducing incidence of TSW in peanut, comprehensive research has not been conducted in North Carolina to determine the broader impact of twin row plantings on other pests. Therefore, the objective of this research was to determine how peanut disease management practices interact with planting pattern to affect disease incidence, yield, and market grade characteristics in Virginia market type cultivars.
Materials and Methods
Effect of Planting Pattern on Management of Cylindrocladium Black Rot
Six experiments were completed in North Carolina during 2005, 2006, and 2007; two in farmer-owned fields in 2005 (Bethel) and (Chadbourn), three at the Peanut Belt Research Station located in Lewiston-Woodville in 2006 (Fields A2, B3, and C2) and in one field during 2007 (Field F3). One of the most important management considerations for CBR is the disease history of the field, and each of these locations and fields had a history of CBR. Soil at the Bethel site was an Exum fine sandy loam (fine-silty, siliceous, thermic Typic Paleudult). Soil at the Chadbourn site was a Norfolk loamy fine sand (fine-loamy, siliceous, thermic typic Paleudult). Soil at the Peanut Belt Research Station was a Norfolk loamy sand (fine-loamy, kaolinitic, thermic typic Kandiudults). Plot size was four rows wide (91-cm spacing) and 12.2 m long in Bethel and Chadbourn and 9.1 m long at Lewiston-Woodville.
Treatments included a factorial arrangement of two cultivars (Perry and VA 98R), two levels of fumigation (none or metam sodium at 112 L/ha), and two planting patterns (single or twin rows). The twin row planting pattern consisted of two rows spaced 20 cm apart on 91-cm spacing. Final in-row plant populations ranged from 11 to 14 plants/m and 15-17 plants/m in single and twin row planting patterns, respectively. Metam sodium (Vapam HL, Amvac Chemical Corp., Los Angeles, CA) was delivered 25 cm below projected seed placement in raised seedbeds 2 weeks prior to planting. Aldicarb (Temik 15G, Bayer CropScience, Research Triangle Park, NC) at 1.1 kg ai/ha and Bradyrhizobia inoculant were applied in the seed furrow for each row (single and twin row planting patterns) at planting. The cultivar Perry is considered partially resistant to SB and CBR, but is extremely susceptible to TSW (Isleib et al., 2003). The cultivar VA 98R is partially resistant to SB, moderately susceptible to TSW, and highly susceptible to CBR (Mozingo, 2000; Shew, 2007).
Effect of Planting Pattern on Management ofSclerotinia Blight
Four experiments were completed in North Carolina during 2005, 2006 and 2007; two at farmer-owned fields in 2005 (Corapeak) and 2006 (Roxobel), and one at the Peanut Belt Research Station in Lewiston-Woodville in each of 2006 (Field B3) and 2007 (Field F3). Fields were selected because they had a history of SB in peanut based on the growers’ experience. Soil at the Corapeak site was a Goldsboro fine sandy loam (fine-loamy, siliceous, subactive, thermic Aquic Paleudults). Soil at the Roxobel site was a Norfolk sandy loam (fine-loamy, siliceous, thermic Typic Kandiudults). Soil at the Peanut Belt Research Station was a Norfolk loamy sand (fine-loamy, kaolinitic, thermic typic Kandiudults). Plot size was four rows wide (91-cm spacing) and 12.2 m long at Corapeak, four rows wide (93-cm spacing) and 7.6 m long at Roxobel, and four rows wide (91-cm spacing) and 9.1 m long at Lewiston-Woodville.
Treatments included a factorial arrangement of two cultivars (Perry and VA 98R), two levels of boscalid (Endura fungicide, BASF Corp., Research Triangle Park, NC) (none or boscalid at 0.44 kg/ha), and two planting patterns (single or twin rows). Aldicarb at 1.1 kg/ha and Bradyrhizobia inoculant were applied in the seed furrow at planting.
Methods Common to All Experiments
Weeds were controlled with the same preplant incorporated, preemergence, and postemergence herbicides for all treatments at a given location or year and location combination, but differed across years and locations. Chlorpyrifos (Lorsban 15G, Dow Agroscience, Indianapolis, IN) was applied based upon calculations made for each location using the SCRW index (Herbert et al., 1997, 2004). Fungicides to control leaf spot disease (caused by the fungi Cercospora arachidicola and Cercosporidium personatum (syn. Passalora personata) and stem rot (caused by Sclerotium rolfsii) were applied using recommendations over the entire test area from a weather-based advisory (Brooks et al., 2011; Cu and Phipps, 1993) using a four-row tractor-mounted sprayer calibrated to deliver 122 L/ha.
Foliar damage from tobacco thrips feeding was recorded in 2006 approximately 3 weeks after planting by evaluating 10 of the most recently emerged leaflets for feeding damage using a scale of 1 to 3, where 0 = no foliage damaged and 3 = all foliage damaged. Incidence of CBR was evaluated by counting dead and severely wilted plants in the center two rows of each plot in late September. Plants exhibiting wilting and yellowing were not counted if symptoms of spotted wilt were apparent, whereas wilted plants that yielded to a firm tug on the main stem were assumed to have root and crown rot caused by CBR. Incidence of SB was determined in late September by parting the canopy in the center two rows of each plot and inspecting stems and crowns for symptoms of the disease. Symptomatic plants were marked with surveyor's flags, and all flags were counted when ratings were completed. Leaf spots (both early and late) were evaluated on a 1.2 m section of row in the center two rows of each plot. The section to be rated was determined prior to rating by computer randomization. A rating of plant condition, which is an overall measure of cumulative defoliation and disease, was recorded within 2 weeks prior to digging using a scale of 0 = no disease and 100 = entire canopy expressing disease symptoms.
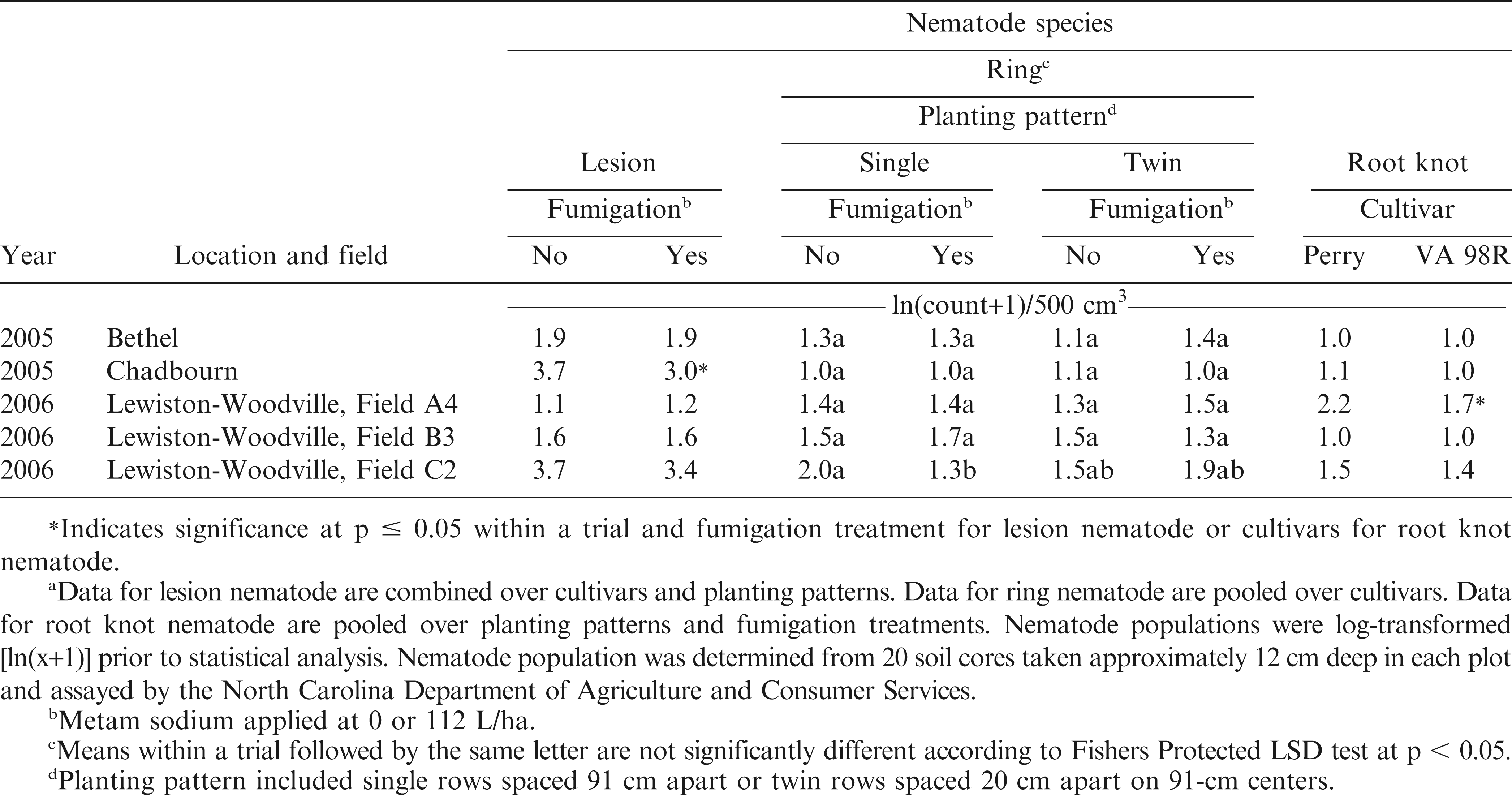
Soil samples were taken from each plot during the first week of June (2005 and 2006) to determine populations of plant parasitic nematodes. Approximately 20 soil cores (10 cm diameter by approximately 12 cm deep) were collected from each plot in a zigzag pattern and combined into one sample per plot. To determine populations of microsclerotia of C. parasiticum, a 500 g subsample of soil was subjected to a modified soil elutriation technique, followed by dilution plating on selective media. Populations were determined from colony counts taken after 5 days of incubation under continuous light (Phipps et al., 1976). To determine nematode populations, a 500 cm3 soil subsample was submitted to the North Carolina Department of Agriculture and Consumer Services, Nematode Assay Section (http://www.ncagr.gov/agronomi/nemhome.htm) for assay. Samples were assayed by a combination of elutriation (Byrd et al., 1976) and centrifugation (Jenkins, 1964).
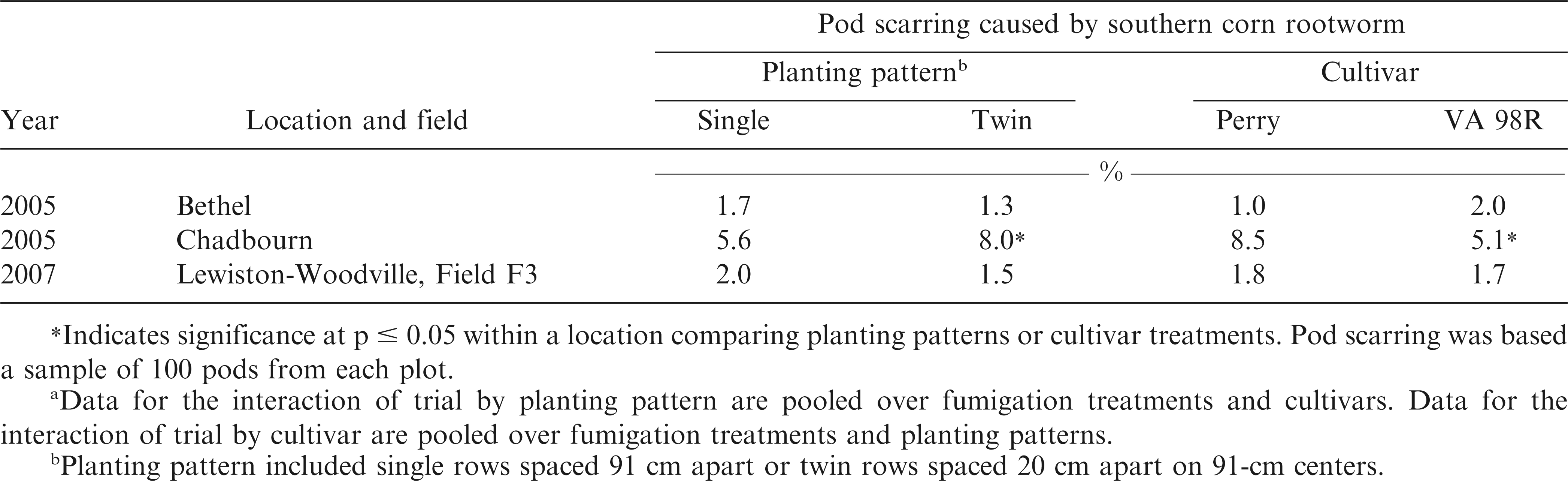
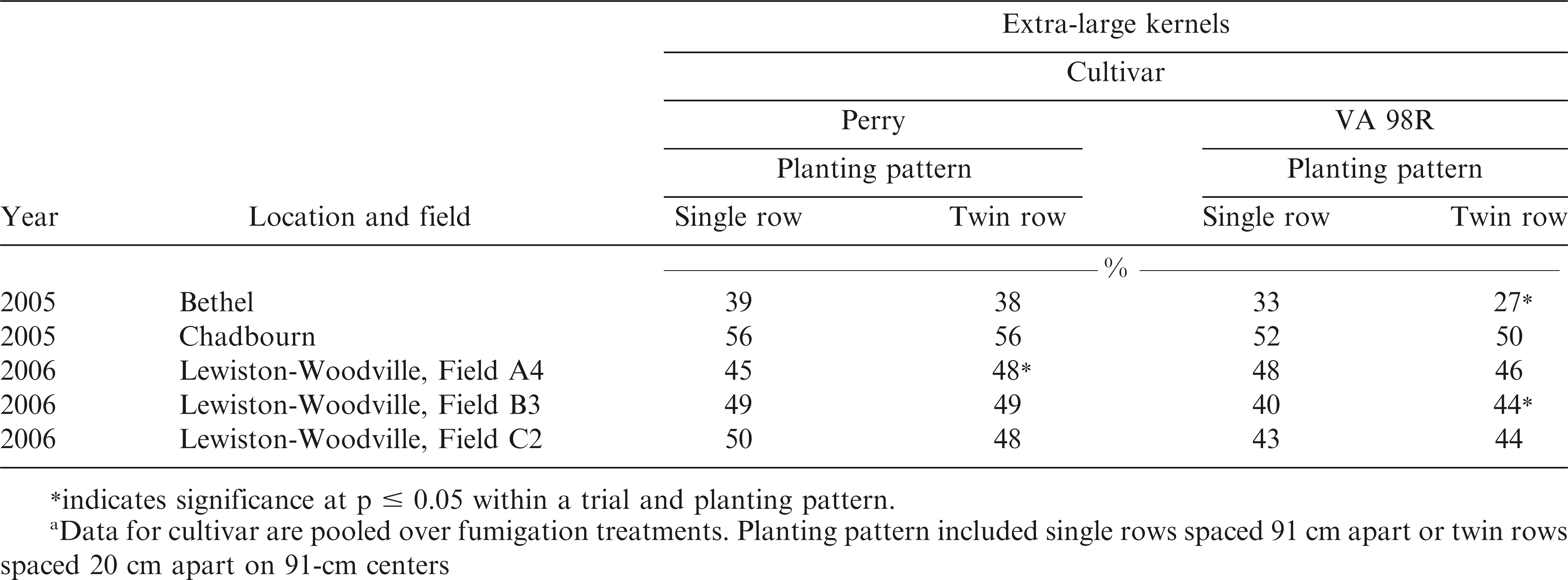
Optimum maturity was determined for each cultivar using the pod mesocarp color technique (Williams and Drexler, 1981). Peanut pods were dug and vines inverted and allowed to air dry for 4 to 7 days prior to harvest. Pods were harvested using small-plot combines and dried to 8% moisture. A 1 kg sample of pods was removed from each plot to determine percentages of fancy pods (%FP), sound mature kernels (%SMK), total sound mature kernels (%TSMK), sound splits (%SS), other kernels (%OK), and extra large kernels (%ELK) using commercial grading standards (USDA, 2005). Pod damage from feeding of SCRW was determined by assessing a 100 pod subsample of the harvested peanuts (Herbert et al., 1997).
The experimental design was a split-plot with cultivars (Perry and VA 98R) serving as the whole plot unit. Depending on the experiment, combinations of either fumigation for CBR suppression or application of boscalid for Sclerotinia blight suppression were combined with planting patterns (single and twin rows) to create sub plots. Treatments were replicated four times. Data for CBR, C. parasiticum, LS, lesion, ring and root knot nematodes, market grade data, pod yield, SCRW, SB, tobacco thrips, and TSW were subjected to ANOVA using SAS (SAS Institute Inc., Cary, NC) appropriate for the factorial treatment arrangement using error terms appropriate for random and fixed effects (Gomez and Gomez, 1984). Means of significant main effects and interactions were separated using Fishers protected LSD test at p ≤ 0.05. Data for plant parasitic nematode populations were log-transformed [ln(x+10)] prior to statistical analysis. Transformed values are presented in the tables.
Results and Discussion
Effect of Planting Pattern on Management of Cylindro-cladium Black Rot
Significant differences were found for inoculum density of C. parasiticum and planting pattern; inoculum levels were higher (p = 0.031, 0.83 vs. 0.58 microsclerotia/cm3 of soil) for single than for twin row planting patterns (data not shown in tables). Planting pattern did not affect CBR incidence (p = 0.7941). However, differences in plant condition rating associated with CBR were noted for cultivars (p = 0.0038), with VA 98R expressing more disease than Perry (12% versus 5%, data not shown in tables). The cultivar VA 98R is more susceptible to CBR than the cultivar Perry (Dong et al., 2008; Isleib et al., 2003; Mozingo, 2000).
Nematode populations were relatively low in all trials. For ring nematode, a trial by planting pattern by fumigation interaction was significant (p = 0.0215), and this interaction most likely was caused by fewer nematodes being present in single rows following fumigation compared with no effect of fumigation in twin rows (Table 1). Placement and concentration of metam sodium in the soil fumigation zone may have contributed to this response. Metam sodium is applied before planting, in one narrow strip in the center of the raised seedbed. In single rows, the seeds are placed down the center of this raised bed, and thus a higher percentage of roots grow within this fumigated soil zone, whereas in twin row planting patterns, the seeds are placed on either side of the center of the raised seedbed. We postulate that the roots in twin row planting patterns may have grown outside of the effective fumigated soil zone, leaving them open to feeding from ring nematode. However, this hypothesis needs to tested by additional research.
The interaction of trial by fumigation was significant for lesion nematodes (p = 0.0145), where fumigation reduced soil populations in the Chadbourn trial but in none of the other trials (Table 1). In one trial in 2006, Lewiston-Woodville Field A4, the population of root knot nematode was lower for VA 98R compared with Perry, resulting in a trial by cultivar interaction (p = 0.0010, Table 1). To our knowledge, the ability of these cultivars to support populations of root knot nematode has not been tested under controlled conditions.
Planting pattern did not affect TSW or interact with other treatments (p ≥ 0.0950). The interaction of cultivar by fumigation was significant for symptoms associated with TSW (p = 0.0480). A higher level of TSW was noted for Perry when fumigated but not VA 98R (data not shown in tables). The cultivar Perry is more susceptible to TSW than VA 98R (Isleib et al., 2003; Lanier et al., 2004; Shew, 2007). Although incidence of tomato spotted wilt was relatively low in these experiments, under higher TSW incidence the value of twin rows would most likely be of higher (Lanier et al., 2004).
The interaction of cultivar by planting pattern was significant (p = 0.0093) for tobacco thrips injury observed 2 weeks after peanut emergence. Interactions of trial (combination of year and location) by all other treatment factors were not significant for thrips injury (p ≥ 0.0557). When pooled over trials and fumigation treatments, there was no difference in tobacco thrips injury when comparing planting patterns for the cultivar Perry (average value of 1.1 on a scale of 1-10, data not shown in tables). In contrast, greater tobacco thrips feeding injury was observed in the cultivar VA 98R when planted in twin rows (value of 1.4) compared with single rows (value of 1.1) (data not shown in tables). These results were surprising given that the same rate of aldicarb was applied in the seed furrow of both single rows and the two individual rows in the twin row planting pattern, so each plant should have been systemically treated with the same amount of active ingredient, regardless of row pattern. Additionally, research shows that twin rows tend to have lower incidence of TSW, higher yields, and improved grades (Baldwin et al., 2001). There have been no reported data suggesting a differential response of the cultivars Perry and VA 98R to thrips feeding; however, differences in cultivar response to tobacco thrips feeding have been demonstrated when the same cultivars are grown in different regions (Culbreath et al., 2000; Smith et al., 1998). Although the difference in thrips injury was relatively minor when comparing row patterns, additional research is needed to determine the reason for this response; especially given that results were significant over three trials and two fumigation treatments.
Interactions of trial by planting pattern (p = 0.0213) and trial by cultivar (p = 0.0009) were significant for pod scarring caused by SCRW. When pooled over fumigation treatments and cultivars, greater damage from SCRW was noted in twin rows at Chadbourn than in single rows even though chlorpyrifos was applied (Table 2). No differences in pod scarring were noted at Bethel in 2005 or Lewiston-Woodville in 2007 when comparing planting patterns. Regardless of planting pattern, the width of the application equipment was 30 cm. The greater damage in the twin rows may have resulted because the diameter of the pegging zone exceeded the width of the insecticide treated soil zone, leaving some pods vulnerable to attack by SCRW. The interaction of trial by cultivar for pod scarring showed less SCRW damage to the cultivar VA 98R than the cultivar Perry at Bethel in 2005, but not at Chadbourn or Lewiston-Woodville (Table 2). This is not surprising given that the cultivar VA 98R reaches optimum maturity during the growing season more quickly than Perry, rendering the pods of VA 98R less susceptible to damage during peak SCRW feeding times (Brandenburg, 2007). Pods that are more fully developed would reduce the likelihood of penetration through SCRW feeding compared with less mature pods (Brandenburg, 2007).
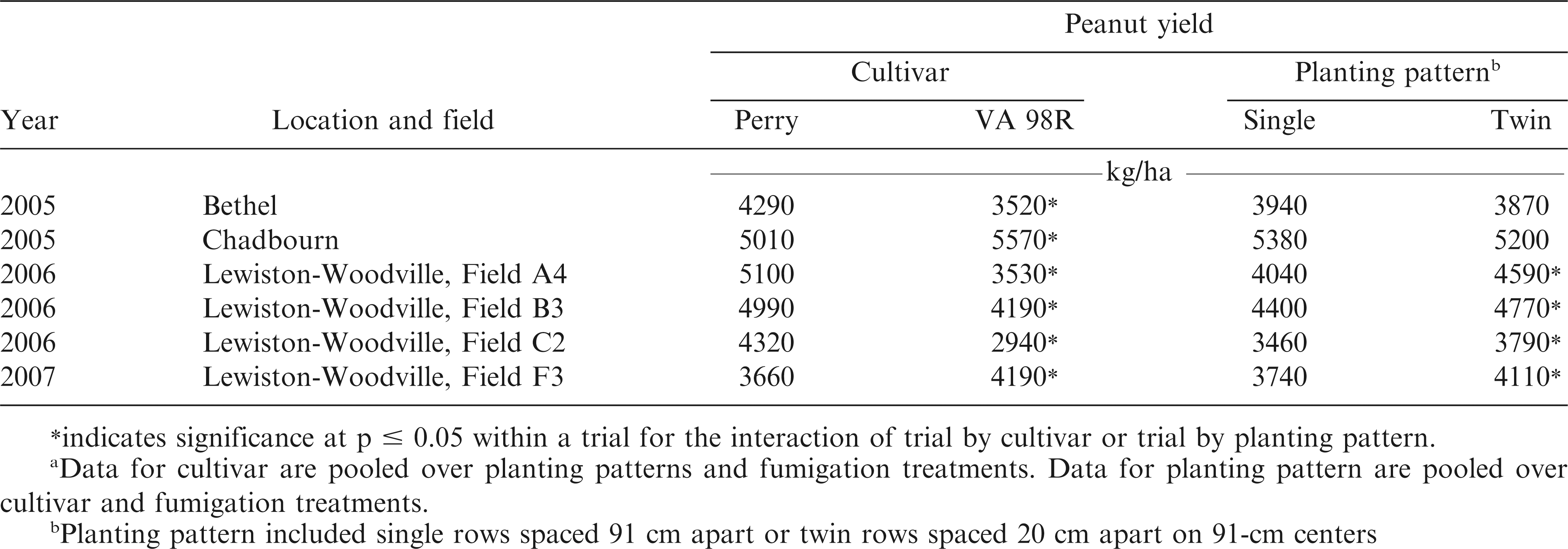
Interactions of trial by cultivar (p = 0.0099) and trial by planting pattern (p = 0.0020) were significant for pod yield. A difference in yield was noted in each trial when comparing cultivars but the differences were not consistent (Table 3). Yield of Perry exceeded that of VA 98R in four trials, while yield of VA 98R exceeded that of Perry in two trials. Perry yielded more at Bethel and Field A4 at Lewiston-Woodville in 2007, which may have been associated with lower CBR incidence for Perry when compared with VA 98R. However, interactions among other factors limit conclusions, especially given the presence of the other pests and diseases as described by the plant condition ratings discussed above. Yield also can be influenced by factors other than disease, including weather and soil type. Average precipitation was lower at Bethel in 2005 and Lewiston-Woodville in 2007 during the growing season (May through September) than in Lewiston-Woodville in 2006.
In 4 of 6 trials, planting peanut in twin rows increased yield by 330 to 550 kg/ha over single rows regardless of cultivar (Table 3). Yield response to planting pattern can be inconsistent (Baldwin et al., 2001; Buchanan and Hauser, 1980; Hurt et al., 2003; Jordan et al., 2001; Tubbs et al., 2011). Increasing the seeding rate or planting peanut in twin row patterns is a key component of risk advisories for TSW management in peanut (Brandenburg, 2014; Hurt et al., 2003). In this experiment TSW incidence was relatively low even though yield was higher in twin rows compared with single rows. Planting peanut in twin rows is more expensive than planting in single rows because the amount of seed, in-furrow insecticide, and inoculant input costs are increased (Tubbs et al., 2011).
In one trial each for Perry and VA 98R, %ELK was highest when planted in twin rows, while in another trial %ELK was highest for VA 98R planted in single rows (Table 4). However, of the 5 trials in the experiment, 2 showed no difference in %ELK when comparing single to twin row planting patterns. Response of Virginia market type peanut to planting pattern can vary with respect to %ELK and %SMK (Table 4). For example, Jordan et al. (2001) reported no difference in both %ELK or %SMK in 7 trials while Lanier et al. (2004) reported that while %ELK did not increase %SMK increased in two years for two cultivars planted in twin rows compared with single rows. Sorensen et al. (2007) reported that %SMK increased by one percentage point for runner market type peanut when planted in twin rows compared with single rows.
Effect of Planting Pattern on Management ofSclerotinia Blight
Planting pattern did not affect SB incidence (p = 0.4031). However, higher seeding rates in twin rows would be expected to increase SB given that the risk of SB increases as the canopy closes (Phipps et al., 1997). Planting pattern did not interact with other treatment factors for yield (p ≥ 0.0769), although the effect of planting pattern needs to be evaluated under higher levels of SB than observed in these experiments.
Conclusions
Collectively, results from these experiments reveal the complexity of selecting IPM strategies in peanut when multiple pests are present across diverse environments and fields. Although interactions among treatment factors were noted, oftentimes responses were independent or interactions could be explained by differences in disease potential among the treatments or locations. However, when making management decisions, it is highly problematic that disease histories frequently are unreliable, as seen in these studies. All fields were selected based on previous history of disease but no disease developed in two of six CBR trials and two of four SB trials. The mistaken CBR history at Chadbourn likely resulted from an earlier misdiagnosed epidemic of TSW but the lack of disease in other trials is harder to explain. Obtaining reliable histories of SB is particularly difficult due to the strong influence of environment and within-field clustering of inoculum in this pathosystem (Phipps et al., 1997).
Research associated with pest management in peanut often focuses on interactions of a specific intervention such as insecticide or fungicide with cultivars or cultural practices such as seeding rates, planting dates, or digging dates with cultivars. The most recent in-depth study in North Carolina with more than two pest management components was conducted in 1997 and 1998 comparing preventative and threshold-based approaches for Virginia market type peanut (Jordan et al., 1999). However, that study did not include fumigation.
Results from this research suggest that pest reaction to twin-row planting pattern in North Carolina with the Virginia market type peanut often will be the same as pest reaction in the traditional single row planting pattern. Yield increases were common for peanut in twin rows compared with single rows irrespective of pest management input. A limitation to results from these and similar large scale experiments is the relatively rapid adoptation of new cultivars and pest management products. For example, the cultivars Perry and VA 98R have been replaced by more recently released cultivars and are no longer grown in North Carolina. However, results from these experiments illustrate the importance of continuing to formulate comprehensive pest management strategies and validating them experimentally as production practices change.
Acknowledgements
Gratitude is expressed to the farmers who provide land for this research; they include Joey Baker (Roxobell site); G. P. Kittrell, (Corapeak site), the Hayes Farm (Edenton site), B. Lennon (Chadbourn site), and K. Mann (Bethel site). We also thank Richard Rhodes, Michael Shaw, Sam Uzzell, and Michael Williams for their assistance as Cooperative Extension agents with the on-farm trials. The authors also thank Dewayne Johnson, Brenda Watson, Joyce Hollowell, and Brian Royals for their technical help and assistance in collecting data. This research was supported by funds obtained from a USDACAR (Crops at Risk) grant and USAID Peanut CRSP project LAG-G-00-96-90013-00.
Literature Cited
Backman P. A. and Crawford M. A. 1984 Relationship between yield loss and severity of early and late leafspot diseases of peanut (Arachis hypogaea) Phytopath. 74 : 1101 – 1103 .
Baldwin J. A. Todd J. W. Weeks J. R. Gorbet D. W. and Culbreath A. K. 2001 A regional study to evaluate tillage, row patterns, in-furrow insecticide, and planting date on the yield, grade, and tomato spotted wilt virus incidence of the Georgia Green peanut cultivar Proc. Annu. Southern Conserv. Tillage Conf. Sustain. Agric. 24 : 26 – 34 .
Besler B. A. Grichar W. J. Starr J. A. Senseman S. A. Lemon R. G. and Jaks A. J. 2006 Effects of peanut row pattern, cultivar, and fungicides on control of southern stem rot, early leaf spot and rust Peanut Sci. 33 : 1 – 6 .
Brandenburg R. 2014 Peanut insect and mite management Pages 88 – 109 In: 2014 Peanut Information. North Carolina Coop. Ext. Serv. Pub. AG-331 .
Brandenburg R. 2007 Peanut insect and mite management Pages 75 – 102 In: 2007 Peanut Information. North Carolina Coop. Ext. Serv. Pub. AG-331 .
Buchanan G. A. and Hauser E. W. 1980 Influence of row spacing on competitiveness and yield of peanuts (Arachis hypogaea) Weed Sci. 28 : 401 – 409 .
Bullen G. and Jordan D. 2014 Peanut production budgets Pages 5 – 10 In: 2014 Peanut Information. North Carolina Coop. Ext. Serv. Pub. AG-331 .
Byrd D. W. Barker K. R. Ferris H. Nusbaum C. J. Griffin W. E. Small R. H. and Stone C. A. 1976 Two semi-automatic elutriators for extracting nematodes and certain fungi from soil J. of Nematol. 8 : 206 – 212 .
Cochran A. Ellison C. Pearce J. Rayburn M. Rhodes R. Shaw M. Simonds B. Smith L. Smith P. and Tyson C. 2003 Results from farmer surveys concerning tomato spotted wilt in North Carolina peanut (Arachis hypogaea) Proc. Am. Peanut Res. Ed. Soc. 35 : 50 .
Culbreath A. K. Todd J. W. and Brown S. L. 2003 Epidemiology and management of tomato spotted wilt in peanut Annu. Rev. Phytopathol. 41 : 53 – 75 .
Culbreath A. K. Todd J. W. Gorbet D. W. Brown S. L. and Baldwin J. A. 2000 Reaction of peanut cultivars to spotted wilt Peanut Sci. 27 : 35 – 39 .
Dickson D. W. and De Waele D. 2005 Nematode parasites of peanut . pp 393 In: Plant Parasitic Nematodes in Subtropical and Tropical Agriculture 2nd Ed Eds Luc M. Sikora R. A. and Bridge J. CAB International .
Dong W. B. Brenneman T. B. Holbrook C. C. and Culbreath A. K. 2008 Evaluation of resistance to Cylindrocadium parasiticum of runner-type peanut in the greenhouse and field Peanut Sci. 35 : 139 – 148 .
Gomez K. A. and Gomez A. A. 1984 Statistical Procedures for Agricultural Research 2nd ed John Wiley and Sons, Inc. , New York .
Herbert D. A. Malone S. Brandenburg R. L. and Royals B. M. 2004 Evaluation of the peanut southern corn rootworm advisory Peanut Sci. 31 : 28 – 32 .
Herbert D. A. Petka W. J. and Brandenburg R. L. 1997 A risk index for determining insecticide treatment for southern corn rootworm in peanut Peanut Sci. 24 : 128 – 134 .
Hurt Brandenburg C. A. R. Jordan D. L. Shew B. B. Isleib T. G. Linker M. Herbert D. A. Phipps P. Swann C. and Mozingo R. W. 2003 Managing tomato spotted wilt virus in peanuts in North Carolina and Virginia North Carolina Coop. Ext. Serv. Pub. AG-638 .
Isleib T. G. Rice P. W. Mozingo R. W. Bailey J. E. Mozingo R. W. and Pattee H. E. 2003 Registration of ‘Perry’ peanut Crop Sci. 43 : 739 – 740 .
Jenkins W. R. 1964 A rapid centrifugal flotation technique for separating nematodes from soil Plant Disease Reporter. 48 : 692 .
Jordan D. L. Beam J. B. Johnson P. D. and Spears J. F. 2001 Peanut response to prohexadione calcium in three seeding rate-row pattern planting systems Agron. J. 93 : 232 – 236 .
Jordan D. L. Brandenburg R. L. Bailey J. E. Johnson P. D. Royals B. M. and Curtis V. L. 1999 Cost effectiveness of pest management strategies in peanut (Arachis hypogaea L.) grown in North Carolina Peanut Sci. 26 : 85 – 94 .
Jordan D. L. Shew B. B. Barnes J. S. Corbett T. Alston J. Johnson P. D. Ye W. and Brandenburg R. L. 2008 Pest reaction, yield, and economic return of peanut cropping systems in the North Carolina Coastal Plain [Online]. Crop Manage . doi: http://doi.org/10.1094/CM-2008-1008-01-RS .
Lanier J. E. Jordan D. L. Spears J. F. Wells R. Johnson P. D. Barnes J. S. Hurt C. A. Brandenburg R. L. and Bailey J. E. 2004 Peanut response to planting pattern, row spacing, and irrigation Agron. J. 96 : 1066 – 1072 .
Lynch R. E. and Mack T. P. 1995 Biological and biotechnical advances for insect management in peanut , pp. 95 – 159 In: Pattee H. E. and Stalker H. T. (eds .) Advances in Peanut Science. Amer. Peanut Res. Educ. Soc , Stillwater, OK .
Mozingo R. W. 2000 Peanut variety and quality evaluation results, 1999 II. Quality data. Virginia Polytech. Inst. and State Univ. Inf. Ser. No. 423 .
Mullen J. D. Norton G. W. and Reaves D. W. 1997 Economic analysis of environmental benefits of integrated pest management J. of Ag.and Appl. Econ. 29 : 243 – 253 .
Napit K. B. Norton G. W. Kazmierczak R. F. and Rajotte E. G. 1988 Economic impacts of extension integrated pest management programs in several states J. Econ Entomol. 81 : 251 – 256 .
Phipps P. M. and Elliott A. P. 1981 Control of northern root-knot nematode on peanut Fungicide and Nematicide Tests. APA. 37 : 196 – 197 .
Phipps P. M. Beute M. K. and Berker K. R. 1976 An elutriation method for quantitative isolation of Cylindrocladium crotalariae microsclerotia from peanut field soil Phytopath. 66 : 1255 – 1259 .
Phipps P. M. Deck S. H. and Walker D. R. 1997 Weather-based crop and disease advisories for peanuts in Virginia Plant Dis. 81 : 236 – 244 .
Sherwood J. L. Beute M. K. Dickson D. W. Elliot V. J. Nelson R. S. Operrman C. H. and Shew B. B. 1995 Biological and biotechnological advances in Arachis diseases, pp 160 – 206 In: Pattee H. E. and Stalker H. T. ( eds .) Advances in Peanut Science. Amer. Peanut Res. Educ. Soc , Stillwater, OK .
Shew B. 2014 Peanut disease management, Pages 110-143 In 2014 Peanut Information North Carolina Coop. Ext. Serv. Pub. AG-331 .
Shew B. 2007 Peanut disease management, Pages 93-138 In 2007 Peanut Information North Carolina Coop. Ext. Serv. Pub. AG-331 .
Smith, O. D. Simpson C. E. Black M. C. and Besler B. A. 1998 Registration of Tamrun96 peanut Crop Sci. 38 : 1403 .
Sorensen R. B. Lamb M. C. and Butts C. L. 2007 Peanut response to row pattern and seed density when irrigated with subsurface drip irrigation Peanut Sci. 34 : 21 – 31 .
Tubbs R. S. Beasley J. P. Culbreath A. K. Kemerait R. C. Smith N. B. and Smith A. R. 2011 Row pattern and seeding rate effects on agronomic, disease and economic factors in large-seeded runner peanut Peanut Sci. 38 : 93 – 100 .
Waggoner P. E. and Berger R. D. 1987 Defoliation, disease and growth Phytopath. 77 : 393 – 398 .
Wilcut J. W. York A. C. Grichar W. J. and Wehtje G. R. 1995 The biology and management of weeds in peanut (Arachis hypogaea), pp 207 – 244 In: Pattee H. E. and Stalker H. T. ( eds .) Advances in Peanut Science. Amer. Peanut Res. Educ. Soc , Stillwater, OK .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity Peanut Sci. 8 : 134 – 141 .
USDA 2005 Peanut inspection program U.S. Gov. Print Office , Washington, DC .
Notes
- Former Graduate Research Assistant, Professor, and Professor, respectively. Department of Crop Science, Box 7620, North Carolina State University, Raleigh, NC 27695.
- Research Assistant Professor, Department of Plant Pathology, Box 7616, North Carolina State University, Raleigh, NC 27695.
- William Neal Reynolds Distinguished Professor, Department of Entomology, Box 7613, North Carolina State University, Raleigh, NC 27695. *Corresponding author Email: david_jordan@ncsu.edu
Author Affiliations