Introduction
Peanut is an important legume crop to the southeastern U.S. As a legume, the presence of Bradyrhizobia bacteria in the soil will cause nodulation of the roots and N-fixation to occur. Peanut inoculants place large volumes of viable Bradyrhizobia near the emerging root during germination to improve infection of the root for nodule formation and N-fixation potential. Liquid inoculants, which are sprayed into the planting furrow onto the peanut seed as it is dropped in the ground, have become the most common method of applying Bradyrhizobia to the root zone. However, some planters are not set up to apply liquid inoculants, restricting those growers to use of a sterile peat (sometimes called a powder) formulation that is mixed with the seed prior to loading into the hopper of the planter, or a granular formulation which requires a granular distribution hopper. However, the granular product can be scarce in availability in many areas. There are also growers that do not use any inoculant when peanut has been planted in a standard rotation within a reasonable timeframe, usually within the last three years (Harris, 1997).
Inoculants have a proven record of improving peanut production; especially in fields where peanut is not part of a regular rotation. In a study where peanut was planted in a field for the first time in at least 25 years, Tubbs et al. (2012) reported a 200% increase in nodulation when using a sterile peat inoculant over no inoculant, and a 400 to 500% increase in nodulation when a liquid formulation was applied. There was a subsequent 400 kg/ha yield improvement over non-inoculated peanut with the sterile peat inoculant, and over 800 kg/ha increase where liquid inoculants were used. Liquid inoculant use has resulted in improved nodulation (Bogino et al., 2006) and higher yield (Lanier et al., 2005) over sterile peat inoculants, and over non-inoculated peanut (Jordan et al., 2006; Jordan et al., 2010) in other studies as well.
Another in-furrow product in peanut production is the granular insecticide phorate which primarily controls thrips; with Frankliniella fusca and Frankliniella occidentalis the most prevalent species in peanut. Thrips feeding can injure peanut if it is severe (Todd et al., 1996, 1997). An even greater concern for peanut is that thrips are the vector for transmitting Tospovirus into the plant, leading to TSW which can cause extreme stunting and yield and grade loss (Culbreath et al., 2003). Phorate has been shown to reduce thrips injury and TSW incidence in peanut (Culbreath et al., 2003; Todd et al., 1998). Yet, since phorate is an organophosphate compound, it is highly toxic to many living organisms in addition to thrips. Because the Bradyrhizobia needed for N-fixation to occur is a living organism, there are concerns regarding the placement together and residence of phorate and Bradyrhizobia in the same furrow, for fear of an antagonistic effect reducing the efficacy of the inoculant. Similar research for Bradyrhizobia efficacy with various pesticides used at planting in peanut was conducted by Jordan et al. (2010) on virginia market-type peanuts, but the only insecticide by Bradyrhizobia combination evaluated was with imidacloprid, and only liquid inoculants were used. Another study was also conducted for compatibility of liquid inoculant with acephate insecticide on virginia market-type peanuts (Jordan et al., 2006). However, currently phorate insecticide is commonly used for thrips control in peanut in Georgia, but has not been tested for compatibility with commercial peanut inoculants.
The cultivar ‘Georgia-06G’ (Branch, 2007) (large seed size) is currently the dominant runner cultivar grown in the U.S. However, some peanut product manufacturers are interested in smaller seeded runner peanuts for their products, and smaller peanuts could also save growers money with a lower seed cost if all other inputs and outputs were considered equal. ‘Georgia-12Y’ (Branch, 2013) (medium seed size) is approximately 10% smaller than Georgia-06G (Gassett et al., 2015). Therefore, the objectives of this research were to (a) assess response of peanut to liquid and sterile peat inoculants in the presence or absence of phorate in fields with no history of growing peanut, and (b) evaluate cultivar differences between the widely grown Georgia-06G and the more recently released Georgia-12Y.
Materials and Methods
Field experiments were conducted on the Tifton campus of the University of Georgia (UGA) in Tifton, GA (31.500 N, -83.516 W) during 2013 and 2014. Fields were not irrigated, and planted in a Tifton loamy sand (Fine-loamy, kaolinitic, thermic Plinthic Kandiudults) (USDA-NRCS, 2015) with no prior history of peanut grown on record (dating back over 30 years). Rotation history consisted of primarily corn (Zea mays L.) for silage and pasture grasses, but cotton (Gossypium hirsutum L.) was grown in the area during the summer immediately preceding this trial. A rye (Secale cereale L.) cover crop was grown during the winter prior to both peanut seasons, and was terminated approximately 4 wk prior to planting peanut. Strip-tillage management was used, with rye residue left standing in the field after termination other than the tilled area from the subsoil shank (approximately 18 cm wide and 36 cm deep).
Treatments consisted of two peanut cultivars, three inoculant treatments, and two insecticide treatments. The cultivars were Georgia-06G and Georgia-12Y. The second variable was comprised of Bradyrhizobia inoculant treatments including (a) non-treated, (b) sterile peat formulation (Peanut Power; Verdesian Life Sciences, LLC, Cary, NC) applied directly to seed as 5.0 g/kg of seed and thoroughly mixed prior to loading into seed hopper, and (c) liquid formulation (Optimize liquid inoculant for peanut; Novozymes, Inc., Bagsvaerd, Denmark) applied at 1100 mL/ha using a stream nozzle at total volume of 73 L/ha sprayed over the seed into the furrow prior to closure. The third variable was inclusion or exclusion of phorate insecticide (Thimet 20G; AMVAC Chemical Corp., Commerce, CA) applied at 1.15 kg ai/ha at the base of the planter furrow prior to closure. The experiment was conducted as a factorial arrangement of all possible treatment combinations in a randomized complete block design with four replications each year. Individual plots were two rows wide (0.9 m row spacing) in single row pattern and 14 m long in 2013, and 10.7 m long in 2014 based on size of available area.
Peanut was planted approximately 5 cm deep on 28 May 2013 and 3 June 2014. A standard peanut fungicide program was used based on UGA Extension recommendations and the high risk scenario of the Peanut Rx (Kemerait et al., 2013). Maturity was assessed by the hull-scrape method (Williams and Drexler, 1981), and peanuts were dug on 28 October 2013 and 29 October 2014 followed by mechanical harvest with a peanut picker modified for plots on 5 November 2013 and 4 November 2014.
Peanut yields were adjusted to 7% moisture for uniformity of comparisons and graded according to USDA-AMS grade standards (USDA-AMS, 2014) for total sound mature kernels (TSMK) by the Georgia Federal-State Inspection Service. Nodule ratings were made within 24 hr after digging by counting nodules from five random plants in the plot and averaging the scores, with a score of 0 = no nodules on root, 1 = 1 to 5 total nodules per root, 2 = 6 to 10 total nodules per root, 3 = 11 to 15 total nodules per root, 4 = 16 to 20 total nodules per root, and 5 = 20 and greater total nodules per root. Nodule mass was calculated by removing all nodules from four roots and weighing the dry matter. Active nodules were determined by slicing open 20 nodules randomly subsampled from the four roots used in nodule mass evaluation and visually observing the interior of the nodule for color and moisture. A nodule with a red, pink, purple, or white interior with a glossy appearance of moisture was considered active, while a brown or green interior with a dry texture was considered inactive. All nodule sampling was conducted on 30 July 2013. Days to row closure refers to the number of days after planting until 50% of the row middle is covered with vines touching from adjacent rows (also known as ‘lapping’), and these measurements were observed in both years of the study. In 2014, measurements of foliage color were made on multiple occasions during the crop season with a SPAD chlorophyll meter (SPAD-502; Konica Minolta, Hong Kong) from 10 leaflet samples per plot from recently fully developed leaves and averaged for each plot. SPAD measurements were made on 25 July, 25 August, and 16 September 2014. Also, canopy light wavelength reflectance by normalized difference vegetation index (NDVI) (Rouse et al., 1974) was determined using an active light sensor (Crop Circle ACS-210 [Holland Scientific, Lincoln, NE]) held approximately 0.9 m above the plant canopy and walking at a consistent pace for the length of the plot. These readings were taken on 22 July, 8 and 20 August, and 2 and 17 September 2014. A final plant stand was measured in both years as the average number of taproots per m of row after inversion, counted in 1.5 m increments two times per row in both rows of the plot.
Data Analysis
Data were statistically analyzed using PROC GLIMMIX in SAS 9.2 software (SAS Institute, 2009), with mean separation by pairwise t-tests. Data were combined over years when there was consistency in the data (no interaction of a treatment effect with years when included in the model), and analyzed individually for each year when a variable’s analyses were not consistent across years.
Results and Discussion
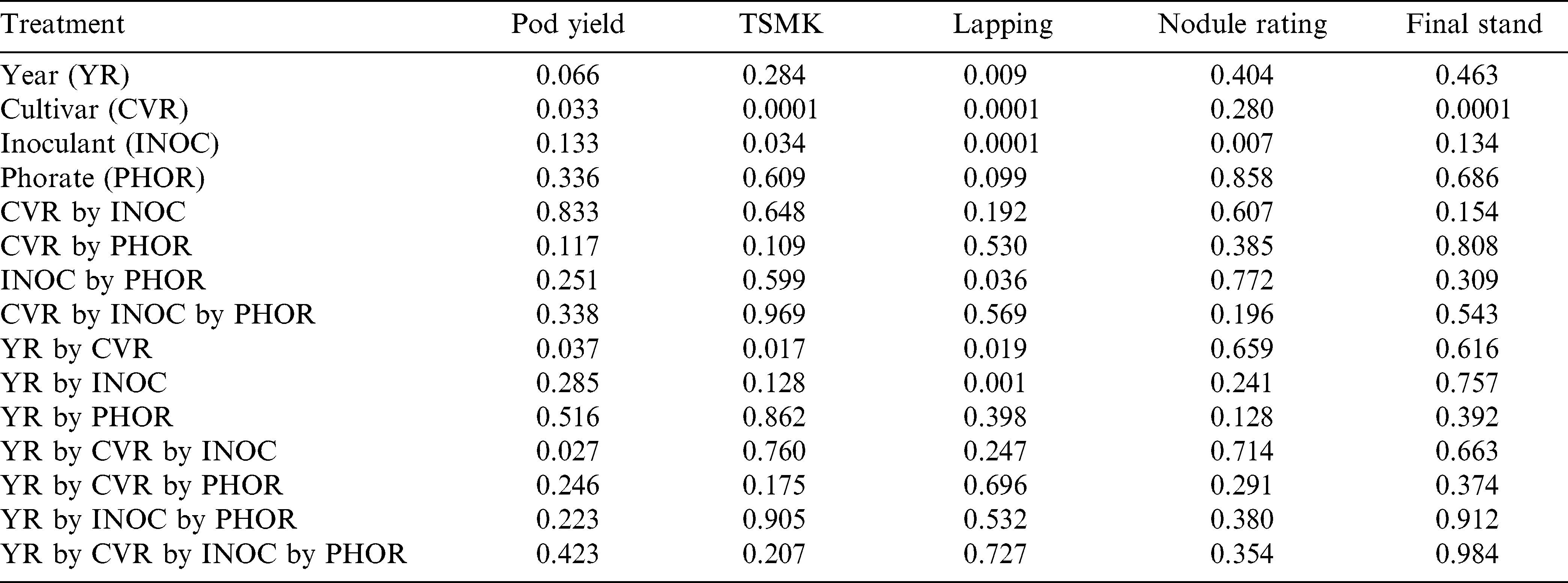
All possible treatment and interaction effects were analyzed for all variables collected. Analysis of variance levels of significance for the variables common to both years (yield, grade, days to row closure, nodule rating, and final plant stand) are shown in Table 1. Since there were interactions for year by cultivar by inoculant for yield, and year by cultivar for grade and days to row closure, analyses for those variables are presented by individual year. An interaction between inoculant and phorate was also observed for days to row closure, but was combined over years. Since year was not a significant effect for grade and nodule rating related to inoculant treatments, those data are combined over years. Final plant stand was only significant for cultivars, so that information is combined over years.
Because there were no interactions between inoculant and phorate treatments for yield or grade, this is an indication that phorate does not reduce the efficacy of Bradyrhizobia peanut inoculants. The only variable with an interaction between inoculant and phorate treatments was days to row closure, where the inclusion of phorate had no effect on non-inoculated (73 d with phorate vs. 75 d without phorate) or liquid inoculant (64 d with phorate vs. 63 d without phorate), but did increase the amount of time until lapping occurred when used together with the sterile peat inoculant (75 d with phorate vs. 66 d without phorate; means are significantly different according to multiple pairwise t-tests at P = 0.05). Phytotoxicity to the foliage of peanut is evident when phorate is used, but it is unclear why vegetative growth might have been impeded when the insecticide was combined with a sterile peanut inoculant in-furrow but not for the other treatment combinations. The rate of lapping/canopy closure in the row middles was quickest using a liquid inoculant regardless of whether phorate was or was not included. Quick canopy closure can assist with controlling weed escapes (Colvin et al., 1985; Hauser and Buchanan, 1981; Wehtje et al., 1984) and soil temperature through shading. Excessive soil temperatures can lead to pollen sterility, reduce flowering and fruit set, and inhibit pegs from entering the soil (Kvien, 1995).
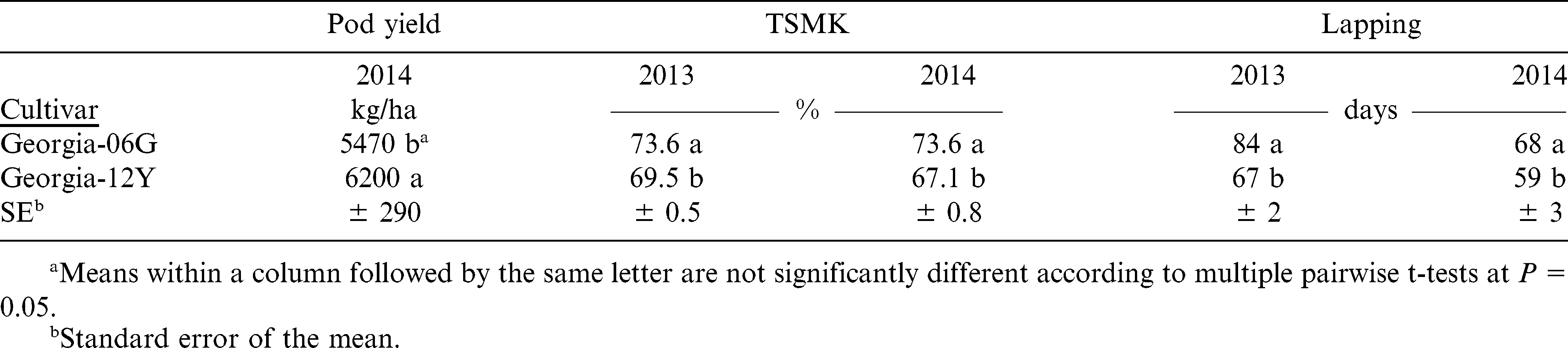
While nodulation was more abundant than expected for a field that had not been planted to peanut within the last 30 years (which will often exhibit no nodulation at all), the data associated with nodulation still followed a predictable pattern in relation to the inoculant treatments applied. The liquid inoculant was superior to both the sterile peat formulation and the non-treated for nodule rating, meaning there were more nodules per plant on average using the liquid formulation (Table 2). It is believed that the liquid inoculant application coats the entirety of the seed and places additional Bradyrhizobia in the soil directly beneath the seed such that the primary root will grow through the bacteria as the root elongates during germination. The sterile peat formulation does not come in contact with 100% of the seed’s surface area, and likewise does not saturate the soil beneath the seed with additional bacteria. Hence, the total nodule mass is improved using the liquid formulation compared to the sterile peat and non-treated plants, although the sterile peat treatment does still improve nodulation over the non-treated as well (Table 3). Both of the inoculant formulations improve the viability of nodule activity compared to the local strains of bacteria already residing in the soil by 20% or more (Table 3). While these positive responses to nodulation were consistently observed, only the liquid inoculant with Georgia-06G peanut resulted in a yield improvement (32% increase) over the non-treated peanuts in 2013. That is consistent with results reported in similar experiments (Jordan et al., 2006; Jordan et al., 2010) where liquid inoculants yielded more than non-treated. However, yields were equal for all inoculant treatments with Georgia-12Y in 2013 (Table 3), and when averaged over both cultivars in 2014 (non-treated = 5790 kg/ha, sterile peat = 5810 kg/ha, liquid = 5910 kg/ha; means are not significantly different according to multiple pairwise t-tests at P = 0.10). But, there was more than a 1% improvement in grade when using the liquid inoculant compared to the sterile peat or non-inoculated treatments (Table 2). The liquid formulation did not provide a significant yield improvement over the sterile peat formulation in this trial unlike those reported by Lanier et al. (2005) which observed as much as a 25% yield improvement and Tubbs et al. (2012) in which a 8% yield increase occurred with liquid compared to the sterile peat formulation. Hence, the liquid formulation is still considered a better option for peanut ahead of both the non-treated and sterile peat formulation because of the nodulation and grade advantage, and other results reported in literature as mentioned above.
For the 2014 iteration of the experiment, there were no interactions between inoculant and phorate treatments for SPAD values on any of the three sample dates. Therefore, respective of the primary objective of this experiment, phorate did not impact efficacy of any inoculant treatment in terms of chlorophyll concentration in the leaves. SPAD values for all non-inoculated peanut (25 July = 41.1, 25 August = 43.1, 16 September = 43.0) were equal to the liquid formulation (25 July = 41.5, 25 August = 43.6, 16 September = 43.1) on all three sample dates. However, SPAD values were lower for the sterile peat inoculant formulation (25 July = 38.7, 25 August = 40.7, 16 September = 40.7) compared to the liquid formulation on all three sample dates.
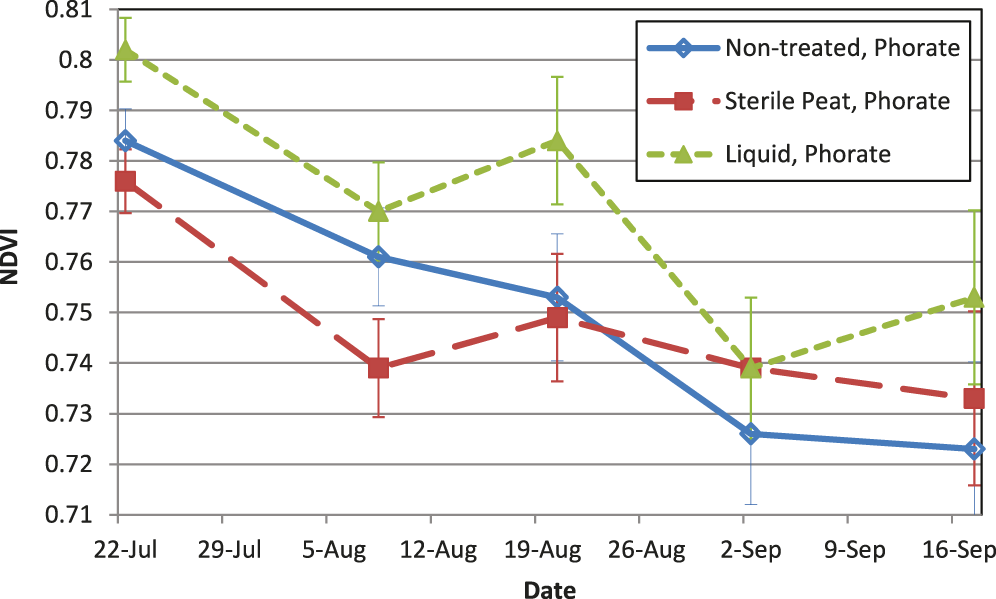
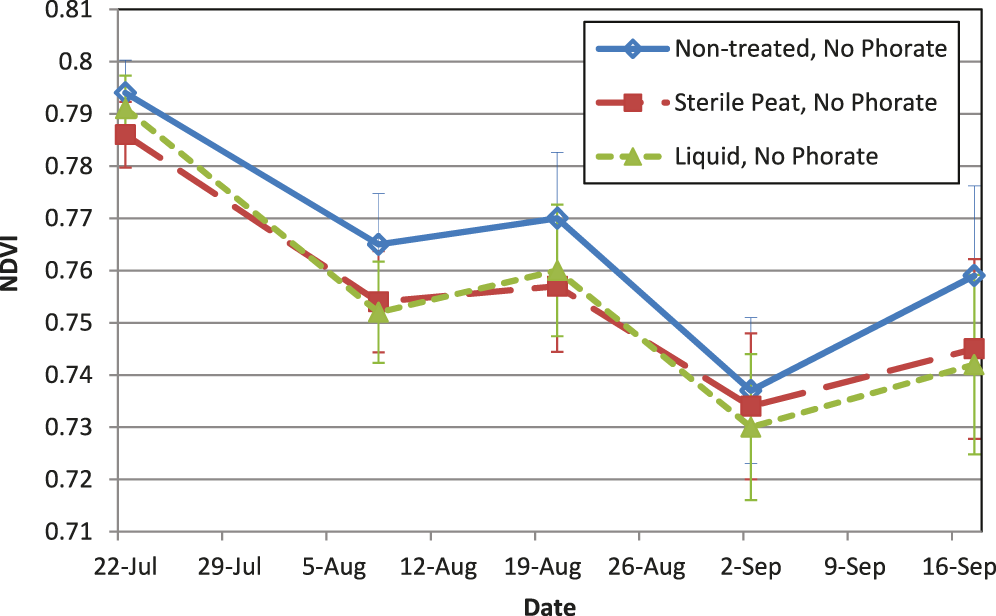
There were interactions between inoculant and phorate related to NDVI on several dates throughout the season. However, the inclusion of phorate with an inoculant did not negatively impact NDVI values compared to the non-treated with phorate (Fig. 1), and actually increased NDVI with the liquid formulation on several dates. Since NDVI is a reflectance value of the entire canopy, it is a function of hue of the canopy and total ground coverage which can be influenced by leaf area index (Carlson and Ripley, 1997). Considering there were no differences in NDVI among treatments when phorate was not included in the furrow (Fig. 2), and the SPAD data demonstrated that there were no differences in chlorophyll content likely ruling out a difference in hue of the foliage, the improvement in NDVI when the liquid inoculant and phorate were both used suggests there was a higher percentage of vegetation covering the soil surface where both the liquid inoculant and phorate were used. Phorate is not considered active in the plant beyond approximately the first month after planting, however improved plant health at the time of NDVI measurements (starting mid-season) may have resulted from the earlier use of phorate by possibly reducing thrips feeding which can cause stunting of the plant. Taking into account both the SPAD and NDVI results from this experiment, the foliage color was not negatively impacted (lighter green in color) with the inclusion of phorate. Jordan et al. (2010) made visual observations of foliage color, and had similar results where the inclusion of the insecticide did not result in a lighter green canopy color (with the exception of one field site). Tubbs et al. (2012) showed that peanuts with a higher NDVI value (at R8 stage) also had higher %N concentration in total vegetative tissue and leaf tissue at the R8-R9 stage of growth, hence NDVI can be an indicator of the amount of N in the plant. This further supports the evidence that in-furrow insecticides are not causing a negative effect on the ability of the inoculant to function properly.
The secondary objective of this project was related to cultivar comparisons of Georgia-06G and Georgia-12Y peanut. The interaction of cultivar and inoculant in 2013 for yield resulted in no difference between cultivars with either the non-treated or sterile peat formulation; however Georgia-06G had a 14% higher yield than Georgia-12Y with the liquid inoculant (Table 3). In 2014, Georgia-12Y yielded 13% better than Georgia-06G when pooled over inoculant and phorate treatments (Table 4). Yet, Georgia-12Y had a significantly lower grade in both years (Table 4). Georgia-12Y had a denser plant stand (18 plants/m) than Georgia-06G (16 plants/m) combined over both years, although both are considered adequate for peanut in single row pattern based on UGA Extension recommendations (13 plants/m or greater), so should not have had an influence on yield or grade in this study. Since growers rely on both yield and grade for the determination of the ultimate payment for their peanuts, additional research is needed to determine if Georgia-12Y has the potential to compete with Georgia-06G as a preferred cultivar with a higher revenue potential. In addition, the different response to inoculants by the two cultivars in 2013 also brings into question whether genetics plays a pivotal role in determining effectiveness of inoculants. Although the cultivar by inoculant interaction was not observed again in 2014, further research should be conducted with more cultivars to determine if genetics are a major factor in peanut response to inoculants.
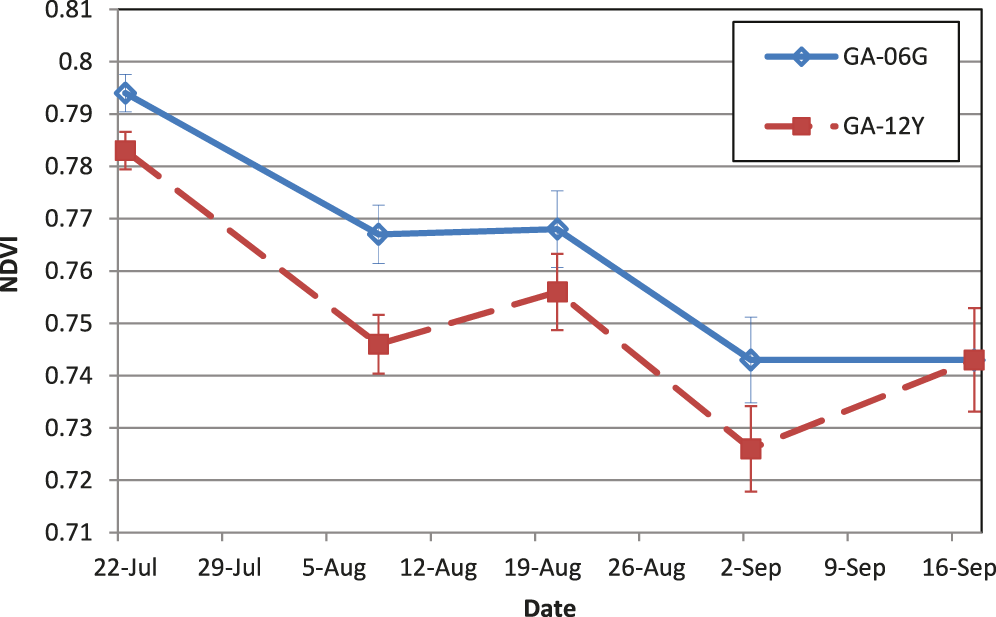
Vegetative growth for Georgia-12Y reached canopy closure 20% faster in 2013 and 13% faster in 2014 compared to Georgia-06G (Table 4). Comparing SPAD values between the two cultivars, they were different at mid-season and just prior to harvest, with Georgia-06G having larger values than Georgia-12Y on both occasions (42.2 vs. 38.6 on 25 July; 43.5 vs. 41.0 on 16 September, respectively; means are significantly different according to multiple pairwise t-tests at P = 0.05). These are similar trends to the NDVI data for those two cultivars over the course of the season (Fig. 3). Hence, Georgia-06G has a darker foliage and appearance than Georgia-12Y.
Summary and Conclusions
Based on these results and supporting literature of similar reports where in-furrow insecticides did not have a negative impact on Bradyrhizobia inoculant performance (Jordan et al., 2006; Jordan et al., 2010), there should be no concern of using phorate insecticide in-furrow in combination with either a liquid or sterile peat inoculant at planting time for peanut. In-season plant health status and growth was not negatively impacted, nor was yield or grade at the end of the season, so growers can proceed with using the combination of materials they prefer without fear of harm to peanut. These data likewise support other research suggesting that the liquid formulation of inoculant provides advantages over the sterile peat formulation, and particularly over non-treated peanut (Lanier et al., 2005; Tubbs et al., 2012). While the liquid formulation does not always provide a superior yield and grade to the sterile peat formulation, it is consistently equal and yield can be up to 25% greater (Lanier et al., 2005), and grade was more than a 1% improvement in this experiment. Compared to non-inoculated peanut in non-rotated fields, yield was as much as 32% greater in this test with a liquid inoculant, and can be up to 200% greater than non-inoculated (Lanier et al., 2005). This can provide an economic advantage in a number of ways, whether directly through yield and/or grade, or indirectly through plant health and growth characteristics that might improve plant nutrition, reduce weed competition, soil temperature, or other similar benefits.
Acknowledgements
The technical efforts of Andy Carter and Michael Plumblee, and the student workers who assisted with data collection, plus the statistical expertise and assistance of Jerry W. Davis were essential to the success of this research and are extremely appreciated. We are also grateful to the Georgia Seed Development Commission for providing seed for these experiments, and the National Peanut Board and Georgia Peanut Commission for providing financial assistance.
Literature Cited
Bogino P Banchio E. Rinaudi L. Cerioni G. Bonfiglio C. and Giodano W. 2006 Peanut (Arachis hypogaea) response to inoculation with Bradyrhizobium sp. in soils of Argentina Ann. Appl. Biol 148 : 207 – 212 .
Branch W.D. 2007 Registration of ‘Georgia-06G’ peanut J. Plant Regis 1 : 120 doi:10.3198/jpr2006.12.0812crc .
Branch W.D. 2013 Registration of ‘Georgia-12Y’ peanut J. Plant Regis 7 : 151 – 153 doi:10.3198/jpr2012.11.0048crc .
Carlson T.N and Ripley D.A. 1997 On the relation between NDVI, fractional vegetation cover, and leaf area index Remote Sens Environ 62 ( 3 ): 241 – 252 .
Colvin D.L Walker R.H. Patterson M.G. Wehtje G. and McGuire J.A. 1985 Row pattern and weed management effects on peanut production Peanut Sci 12 : 22 – 27 .
Culbreath A.K Todd J.W. and Brown S.L. 2003 Epidemiology and management of tomato spotted wilt in peanut Annu. Rev. Phytopathol 41 : 535 – 75 .
Gassett J.D Day J.L. Dunn D.G. and LaHue S.S. ( eds. ). 2015 Georgia 2014 peanut, cotton, and tobacco performance tests Ann. Pub 104 – 6 , Georgia Agric. Experiment Stn., Univ. of Georgia , Athens .
Harris G.H. 1997 Fertilization . pp. 29 – 30 In: Peanut production field guide. Bulletin 1146. Georgia Coop. Ext. Serv ., Univ. of Georgia , Athens .
Hauser E.W and Buchanan G.A. 1981 Influence of row spacing, seeding rates, and herbicide systems on the competitiveness and yield of peanuts Peanut Sci 8 : 74 – 81 .
Jordan D.L Brandenburg R.L. Bailey J.E. Johnson P.D. Royals B.M. and Curtis V.L. 2006 Compatibility of in-furrow application of acephate, inoculant, and tebuconazole in peanut (Arachis hypogaea L.) Peanut Sci 33 : 112 – 117 .
Jordan D.L Johnson P.D. Brandenburg R.L. and Faircloth J. 2010 Peanut (Arachis hypogaea L.) response to Bradyrhizobia inoculant applied in-furrow with agrichemicals Peanut Sci 37 : 32 – 38 .
Kemerait R Culbreath A. Beasley J. Prostko E. Brenneman T. Smith N. Tubbs S. Srinivasan R. Tillman B. Rowland D. Dufault N. Hagan A. Majumdar A. and Monfort S. 2013 Peanut Rx: Minimizing diseases of peanut in the southeastern United States, the 2013 version of the peanut disease risk index . p. 109 – 130 In: Beasley J.P. ( ed. ) 2013 Peanut Update. Spec. Publ. CSS-13-0110 Univ. of Georgia Coop. Ext ., Athens, GA .
Kvien C.S. 1995 Physiological and environmental disorders of peanut . p. 33 – 42 In: Melouk H.A. and Shokes F.M. ( eds. ). Peanut Health Management. Am. Phytopathological Soc St. Paul , MN .
Lanier J.E Jordan D.L. Spears J.F. Wells R. and Johnson P.D. 2005 Peanut response to inoculation and nitrogen fertilizer Agron. J 97 : 79 – 84 .
Rouse J.W Haas R.H. Scheel J.A. and Deering D.W. 1974 Monitoring vegetation systems in the Great Plains with ERTS . p. 309 – 318 In: Freden S.C. and Becker M.A. ( eds. ). Proc. 3rd Earth Res. Tech. Satellite (ERTS) Symp ., vol. 1 , 10-14 Dec. 1974 . National Aeronautics and Space Administration , Washington, D.C .
SAS Institute 2009 The SAS system for windows . v. 9.2. SAS Inst., Cary, NC .
Todd J.W Culbreath A.K. and Brown S.L. 1996 Dynamics of vector populations and progress of spotted wilt disease relative to insecticides use in peanuts Acta Hortic 431 : 483 – 490 .
Todd J.W Culbreath A.K. Brown S.L. Gorbet D.W. Shokes F.M. Pappu H.R. Baldwin J.A. and Beasley J.P. 1998 Development of an integrated management system for spotted wilt disease in peanut. Proc Am. Peanut Res. Ed. Soc 30 : 51 (Abstr.).
Todd J.W Culbreath A.K. Pappu H.R. and Brown S.L. 1997 Thrips as Tospovirus vectors in peanut Proc Am. Peanut Res. Ed. Soc 29 : 17 .
Tubbs R.S Harris G.H. Beasley J.P. Smith A.R. and Smith N.B. 2012 Effect of inoculant and nitrogen application at planting on peanut production in Georgia Online. Crop Management doi:10.1094/CM-2012-0823-01-RS .
USDA-NRCS 2015 Web soil survey http://websoilsurvey.sc.egov.usda.gov/App/WebSoilSurvey.aspx (verified 11 September 2015) USDA Nat. Resour. Conserv. Serv ., Washington, D.C .
USDA-AMS 2014 Farmers’ stock peanuts inspection instructions Available at http://www.ams.usda.gov/sites/default/files/media/Farmers%27_Stock_Peanut_Inspection_Instructions%5B1%5D.pdf (verified 11 September 2015) USDA Agric. Marketing Serv ., Washington, D.C .
Wehtje G Walker R.H. Patterson M.G. and McGuire J.A. 1984 Influence of twin rows on yield and weed control in peanuts Peanut Sci : 11 : 88 – 91 .
Williams E.J and Drexler J.S. 1981 A non-destructive method for determining pod maturity Peanut Sci 8 : 134 – 141 .
Notes
- Dept. of Crop and Soil Sciences, University of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793
- Dept. of Plant Pathology, University of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793
- Dept. of Plant and Soil Sciences, Mississippi State University, Mississippi State, MS 39762 *Corresponding author. E-mail tubbs@uga.edu
Author Affiliations