Introduction
The indeterminate characteristic of peanut reproduction and resultant pod maturity can create confusion to identify best digging time for highest peanut yield and grade. In addition, the variability in peanut phenotypic characteristics of commercial cultivars, such as peg strength, may contribute to increased yield or digging loss in some peanut cultivars. Peanut producers and manufacturers recognize that digging too early or too late can reduce yield and grade as well as processing characteristics. Two results of digging early, aside from lower yields, are increased amount of immature peanuts entering storage facilities may increase the risk of mold production (Aspergillus flavus), and immature peanuts can cause off-flavors during roasting. Conversely, digging late may reduce yield by leaving pods on the soil surface (pod loss) caused by mechanical or biological damage to over-mature plants with diseased or weakened pegs due to age. Therefore, harvesting peanut at the optimum time would have greatest peanut yield, least peanut loss, and reduce the risk of mold growth in the warehouse and for the development of off-flavors during roasting. Previous research has shown that peg strength can vary due to agronomic practices, field condition, peg age, moisture content, peanut cultivar, and fungal infections. For instance, Bauman and Norden (1976) showed that cultivars Florunner and Florigiant had the same attachment force but was significantly higher than cultivar Early Runner. Troeger et al., (1976) showed that spanish market type peanut had higher attachment force than either runner or virginia market type peanut. Thomas et al., (1983) using 30 different cultivars (plant introductions) showed that peg strength can vary greatly between cultivars. Chapin and Thomas (2005) showed that fungal infections can reduce peg strength up to 45% compared with healthy pegs. However, pods exhibiting certain disease symptoms (southern stem rot; Sclerotium rolfsii Sacc.) had significantly stronger pegs than those of healthy pods. In addition, Chapin and Thomas (2005) showed no difference in peg strength as pods matured through the mesocarp color classes of yellow 2 to black (Williams and Drexler, 1981). However, peg strength decreased about 32% in over mature black pods compared to mature pods.
The advance of genetic enhancements associated with newer cultivars for increased yield, disease, and insect resistance may also have different peg strengths that could be used to determine pod maturity and possible digging date along with predictive peanut yield. It is unknown how three recently released runner type cultivar peg strengths compare to each other and how variability in peg strength may affect the harvestable yield among cultivars. Williams and Drexler (1981) used mesocarp color to determine pod maturity and predict digging date. This practice has been universally accepted but can be time consuming as well as subjective depending on the person placing the peanut on a color chart and their ability to see color compared to another person. If peg strength values can be used as an indicator of when to harvest instead of the subjective mesocarp (hull) color, growers could possibly increase yield and grade. It is proposed that using peg strength values may be used as a quantitative method to determine digging date thereby reducing the subjective variability of color and pod placement on a color chart. The objective of this research was to compare peg strength of three peanut cultivars with peanut maturity (mesocarp color), peanut yield, and pod loss over various plant dates and multiple harvest dates.
Materials and Methods
This project was conducted for two years (2011 and 2012) in Dawson, GA (31o 47′02″ N 84o 29′ 15″ W) on a Tifton sandy loam soil (Fine-loamy, kaolinitic, thermic Plinthic Kandiudult) with 1 to 2% slope. In 2011, the experimental design was a randomized complete block using a 3 by 3 factorial for peanut cultivar by harvest date with one plant date and four replications. In 2012, the experimental design was a randomized complete block using a 3 by 3 by 4 factorial for plant date, peanut cultivar, and harvest date with four replications. The peanut cultivars used were Georgia-06G (Branch, 2007), Tifguard (Holbrook et al., 2008), and Georgia-09B (Branch, 2010).
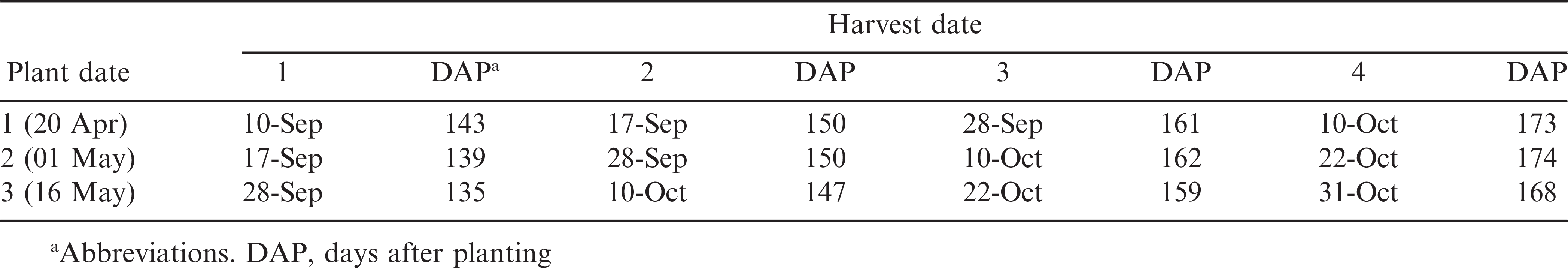
Each site was deep turned, disk harrowed, lime applied (1121 kg ha−1), field cultivated, and bedded on 0.91m row spacing. Individual plots across both years were 1.83 m wide and 12.3 m long. Prior to planting, bed peaks were knocked off so peanut could be planted in moist soil. Peanut was planted at 20 seed m-1 using a six-row vacuum planter. In 2011, peanut was planted 03 May with harvest dates of 12, 26 Sept and 07 Oct or 132, 146, and 157 days after planting (DAP). In 2012, plant dates were staggered at about 14 days (20 April, 01 May, and 16 May) and harvest dates were staggered at 7 to 10 day intervals depending on climatic conditions (Table 1).
Irrigation events were scheduled using IrrigatorPro® and verified using soil water potential sensor measurements for each plant date (2012). All plots were irrigated using overhead irrigation. Soil water potential sensors (MPS-1, Decagon Devices, Inc., Pullman, WA, www.decagon.com) were installed at 25 and 50 cm soil depth and monitored daily. Irrigation events were scheduled following Irrigator Pro’s recommendations or when the average of the two water potential sensors equaled −60 kPa. Herbicide, insecticide, and/or fungicide treatments were applied at manufacturers recommended rates and timing as determined by field scouting.
Prior to each mechanical harvest, four to five peanut plants were randomly hand-sampled from each plot. Plant samples were tagged, returned to the lab, and washed with tap water. Two or three random plants were separated from each sample and excess stems/leaves were removed leaving a 5 to 8 cm stem that held an individual peanut peg and pod. These subsamples were placed in plastic bags and held in a refrigerator until peg strengths were determined. All samples were analyzed within 24 hours after sampling. Peg samples were removed from the plastic bag and individual pods were placed in a “u-shaped” metal bracket that was attached to an electronic force gage (Imada, Inc. Model DS2-11, Northbrook, IL) that would measure peak force. The stem was then pulled manually until the peg broke or detached from the pod. The peak tension for each individual pod was then digitally transmitted to a computer.
In 2012, to measure the relationship between peanut maturity and peg strength, each pod that was pulled from a peg was placed into a consecutively numbered nine by nine cell grid made from 3.8-cm PVC couplings glue-mounted on a screened metal plate (1.3 by 3.2-cm expanded metal mesh) held in place by a wood frame. When all the peanuts from the sampled plants were placed in the PVC grid, another screened plate with a wood frame was placed on top as a lid and held in place with elastic bungee cords. The pods were then blasted using a pressure washer similar to how peanuts are prepared for the hull scrape method (Williams and Drexler, 1981). After blasting, each pod was then removed, in order, from the individual PVC cell and the hull color was determined using the accepted peanut profile board numbered 1 to 25 where each number corresponded to a specific color/maturity level determined by a color chart. The color/number was recorded and then correlated with the tension value recorded when the pod was detached from the peg.
On the same day peanut hand-sampling occurred for peg strength, the respective field rows were dug with a two row inverter for yield analysis. Peanut rows were allowed to dry in the field 3 to 5 days depending on environmental conditions. Each peanut plot was harvested using a two-row peanut combine and collected using a bagging attachment. Bagged peanuts were placed on small sample air dryers and dried to below 10.5% moisture. Each sample bag was weighed and recorded. Each sample was then sub-sampled to retain about a 2000-g sample, stored in paper bags, to be used to determine farmer stock grade. At the end of the harvest season, each subsample had about 1500-g removed, the exact weight was recorded, large sized foreign material (sticks/rocks/etc.) removed, and the farmer stock grade was determined by x-ray analysis (an emerging technology used to determine peanut grade developed at the National Peanut Research Laboratory, Dawson, GA).
Pod loss was determined on each plot following harvest. All excess peanut forage was removed from each plot by hand using pitchforks. A 3-m long by 1.83-m wide area was marked in each individual plot to designate where the digging loss machine would operate. The digging loss machine (called “scavenger”) is a PTO and hydraulic driven experimental implement (designed and manufactured at the National Peanut Research Laboratory, Dawson, GA) with shakers and fans that would separate peanut pods that were lost during the harvest process from soil, rocks, and other debris. The scavenger would sift all material from a 1.83-m wide swath about 5-cm deep. Previous experience with the scavenger implement showed that the 5-cm soil depth was sufficient to collect pods that were lost during the harvesting process. This implement was not designed to collect peanuts missed by the digging process but only those peanuts that were near the soil surface after digging and combining. Separated peanut pods were collected in mesh bags attached to the scavenger. Peanut pods that happened to travel across the scavenger machine and fall to the ground were manually collected and placed in the collection bag. All scavenged sample collection bags were placed on forced-air dryers. After 24 to 48 hrs, each sample was cleaned using a belt cleaner and hand checked to remove any foreign material leaving only peanuts, including pods or loose shelled kernels, etc. Each peanut sample was weighed and recorded to determine pod loss.
Data from each year were analyzed separately due to differences in treatments across years. In 2011, peg strength, pod yield, pod loss, and yield characteristics were analyzed individually by harvest date and cultivar. In 2012, peg strength, pod yield, pod loss, and yield characteristics were analyzed individually by plant date, harvest date, and cultivar. In both years, data were pooled across treatments only when ANOVA F-test showed significance at p≤0.05 level (Statistix10, 2013). Differences between means of peg strength, crop yield, and pod loss were determined using Tukey’s HSD (honest significant difference) pairwise comparison when ANOVA F-test showed significance (p≤0.05). Regression analysis was used to determine if a linear relationship exists between average peg strength versus average peanut yield by harvest date for individual peanut cultivars. Regression analysis was also used to determine if a linear relationship exists between average peg strength versus average pod maturity within cultivar.
Results and Discussion
2011
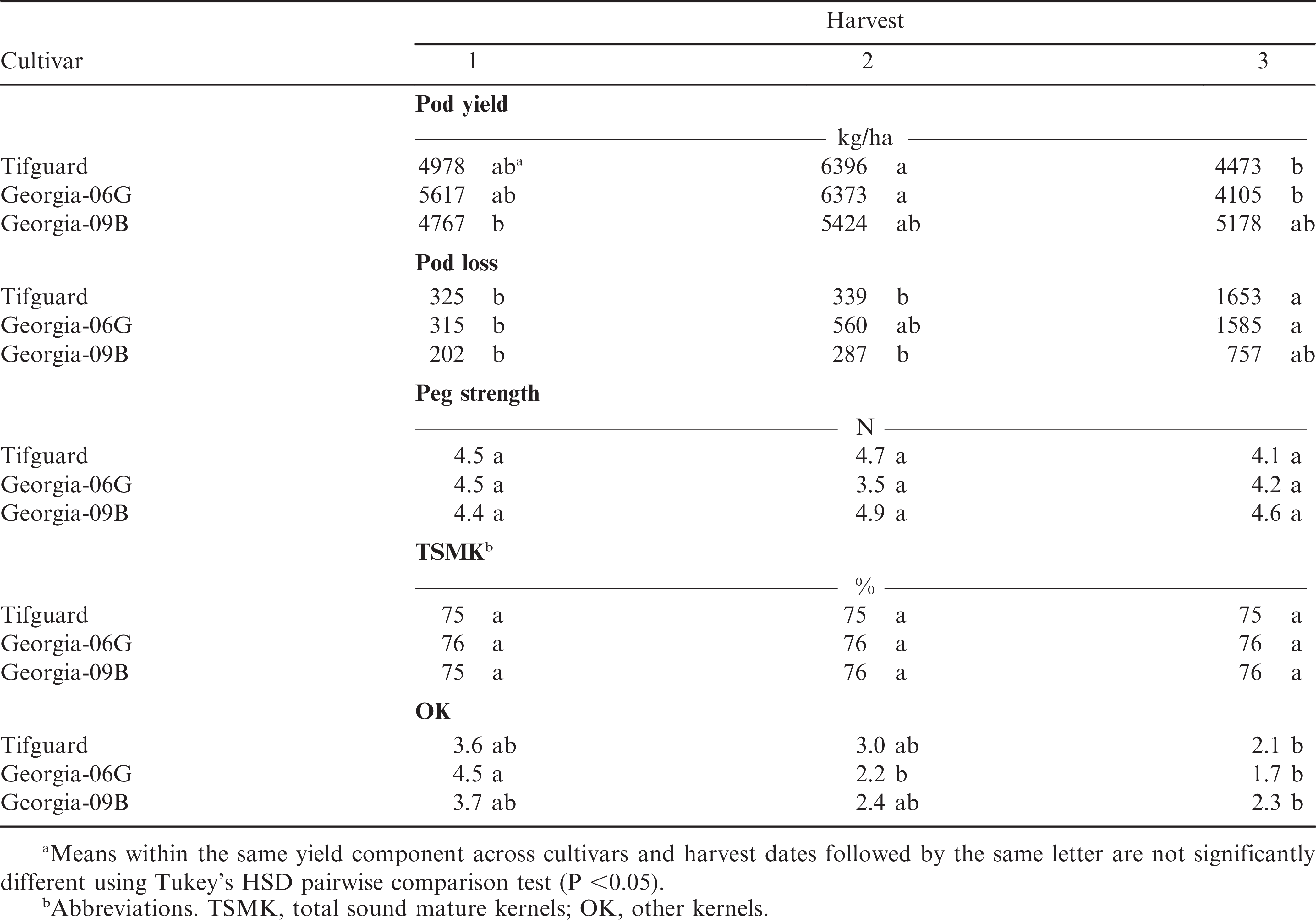
All three cultivars, Tifguard, Georgia-06G, and Georgia-09B, have about the same maturity length compared with Georgia Green (Branch, 1996). It was expected that peg strength would decrease with harvest date resulting in increased pod loss. However, there was no significant difference with peg strength across harvest date or cultivar (Table 2) but there was increased pod loss for Tifguard and Georgia-06G but not with Georgia-09B. The average peg strength was 4.4 N with a range of 3.5 to 4.9 N (Table 2). The average peg strength for these cultivars was lower than those described by Bauman and Norden (1971) at 10 N (Florunner and Florigiant cultivars) and lower than the 5.7 to 8.4 N range described by Johnson George et al. (1988). Thomas et al., (1983) showed peg strengths ranging from 8.3 to 22.1 N for ten commercial cultivars and 12.1 to 37.3 N for 30 plant introductions. Chapin and Thomas (2005) showed peg strengths that ranged from 6.0 to 7.4 N for healthy peanuts and between ∼2.0 to ∼4.0 N for peanuts that were symptomatic for southern stem rot. During this year there was no indication of disease to decrease peg strength, therefore, it is uncertain if these low peg strength values are due to plant cultivar or other environmental factors.
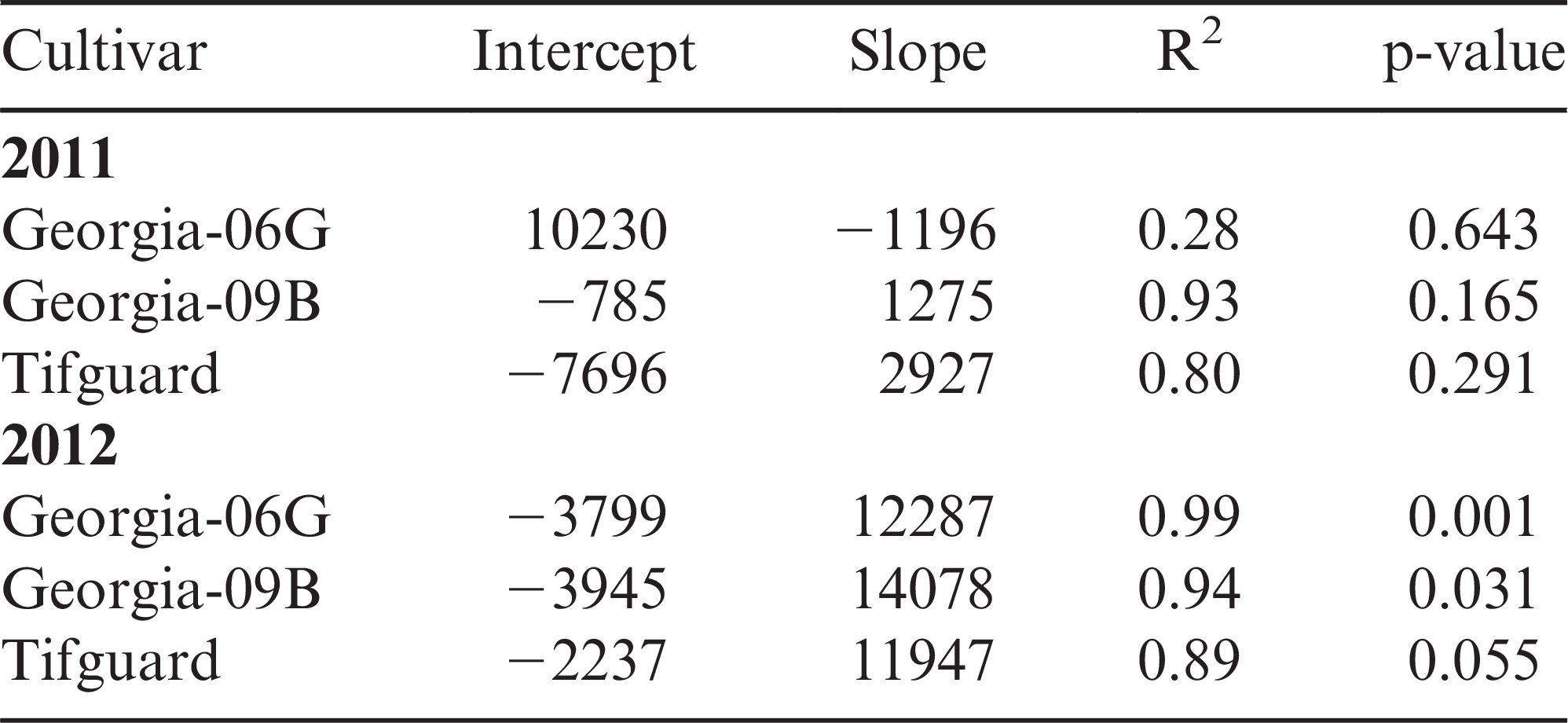
There was no yield difference by cultivar within harvest date (p <0.001) but there were yield differences by cultivar across harvest dates. Both Tifguard and Georgia-06G had lower yield at the last harvest date compared with the second harvest date, while Georgia-09B had consistent yield across all harvest dates (Table 2). There was no difference in peanut grade for total sound mature kernels (TSMK) across cultivars or harvest date. In general, other kernel (OK) values tended to decrease with each harvest date irrespective of peanut cultivar (Table 2).With pod yield tending to decrease and pod loss increasing with plant age, this implies a possible relationship between peg strength and peanut yield. Regression analysis for peg strength versus pod yield showed that both Tifguard and Georgia-09B had strong positive relationship of predicting pod yield with peg strength (r2 = 0.8 and 0.93, respectively; see Table 3). However, Georgia-06G showed a negative relationship where increased peg strength resulted in lower yield with an r2 = 0.28 (Table 4).
2012
Peg strength was statistically different when averaged across plant date (p ≥0.001), harvest date (p≤0.001), and cultivar (p≤0.001). Peg strengths were stronger for plant date one and two compared with plant date three when averaged over harvest date and cultivar. Stronger peg strengths were observed with harvest one and two compared with harvests three and four when averaged over plant date and cultivar. Cultivar Georgia-06G had greater peg strength (6.9 N) than Georgia-09B and Tifguard and Georgia-09B (5.8 N) had greater peg strength than Tifguard (5.5 N), when averaging over plant date and harvest date. The average peg strength for these cultivars during 2012 was lower than those described by both Bauman and Norden (1971) and Thomas et al., (1983) peg strengths described previously. The lower range of peg strengths described by Johnson George et al. (1988) and Chapin and Thomas (2005) at 5.7 and 6.0 N, respectively, are similar to those described in this research. Since the peg strength of these three cultivars were consistently lower for two years compared with those described in previous research, it would seem that these peg strengths are normal for these cultivars.
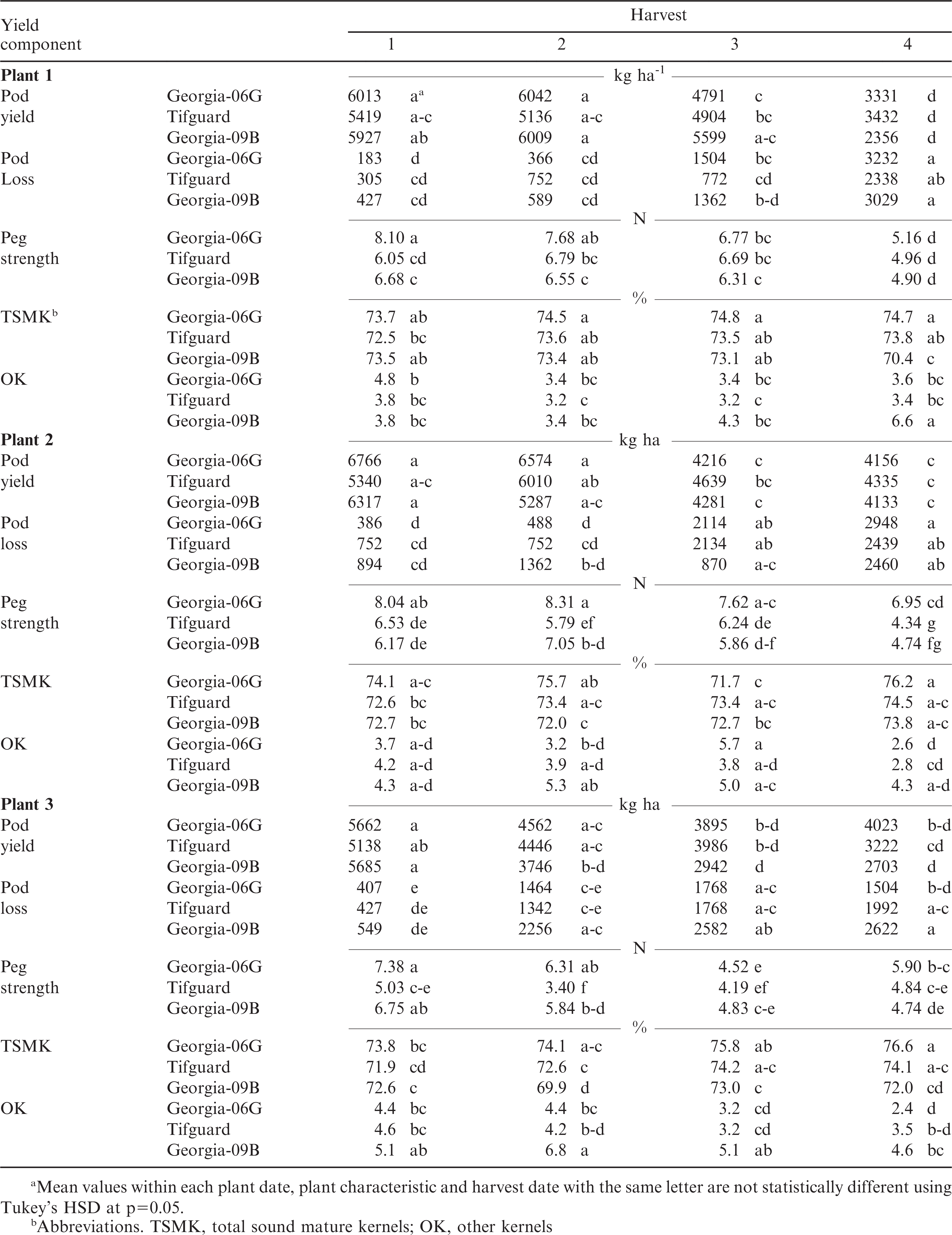
The above averages may show general trends of peg strength by each treatment factor, however, Table 4 shows the interactions of peg strength, pod yield, pod loss and grade characteristics by plant date, harvest date, and peanut cultivar. These data (Table 4) give a better understanding of how each cultivar peg strength changes with plant date and harvest date. Peg strength at plant date 1 ranges from 8.1 to 4.9 N across all harvest dates and cultivars. Plant date two had similar range in peg strength as plant date 1, while plant date three had a lower peg range at 7.38 to 3.4 N. The greatest peg strength was not always at the earliest harvest date and the lowest peg strength was not always at the latest harvest date. These data do show that with each cultivar there were stronger and weaker peg strengths depending on plant date and harvest date. Peg strength data in Table 4 can be used to detail when the peg strength was greatest for a given plant date and harvest date.
Pod yield was statistically different for plant date (p≤0.001), harvest date (p≤0.001), and harvest date by cultivar interaction (p≤0.001) but not for cultivar. Pod yield decreased with the last plant date and the last two harvest dates. These results agree with the general rule that late planted and late harvested crops tend to have decreased yields. Thus, the accepted recommendation to plant on time and harvest on time for best peanut yield should be adhered to by growers. Table 4 shows changes in pod yield across plant date by cultivar and harvest date. Pod yields ranged from a high for Georgia-06G of 6766 kg ha-1 at plant two, harvest one to a low of 2356 kg ha-1 at plant one, harvest four for Georgia-09B. As stated earlier, across all cultivars and plant dates, harvest one and two had similar yields with harvest three and four tending to be lower. Pod yield data in Table 4 can be used to detail when the yield was greatest for a given plant date and harvest date.
There was no difference with pod loss when averaged across plant date or cultivar. There was a difference with pod loss by harvest date when averaged across plant date and cultivar (p≤0.001). As was expected, pod loss increased with each harvest date. Table 4 shows how pod loss increased across plant date and cultivar with harvest date. Pod loss increases over five times from the first (481 kg ha-1) to the last harvest date (2507 kg ha-1) when averaged across plant date and cultivar.
The peanut grade characteristic, TSMK, was not different when averaged by plant date, harvest date, or cultivar. TSMK did show differences within each plant date and harvest date by cultivar interaction. Table 4 show these interactions can be quite dramatic and could change the value of peanuts when sold by the grower. There does not seem to be a pattern of increased or decreased TSMK by plant date or harvest date. TSMK values ranged between 70 and 76% depending on cultivar, plant date, or harvest date.
The peanut grade characteristic of “other kernels” (OK) did show a slight increase with plant date three compared with plant date one and two when averaged across harvest date and cultivar. There was no difference in OK by harvest date when averaged across plant date and cultivar. Georgia-09B did have greater percentage OK compared with Georgia-06G and Tifguard when averaged over both plant date and harvest date. Table 4 shows how values of OK respond to plant date, harvest date, and by cultivar.
Peg Strength Versus Yield and Maturity
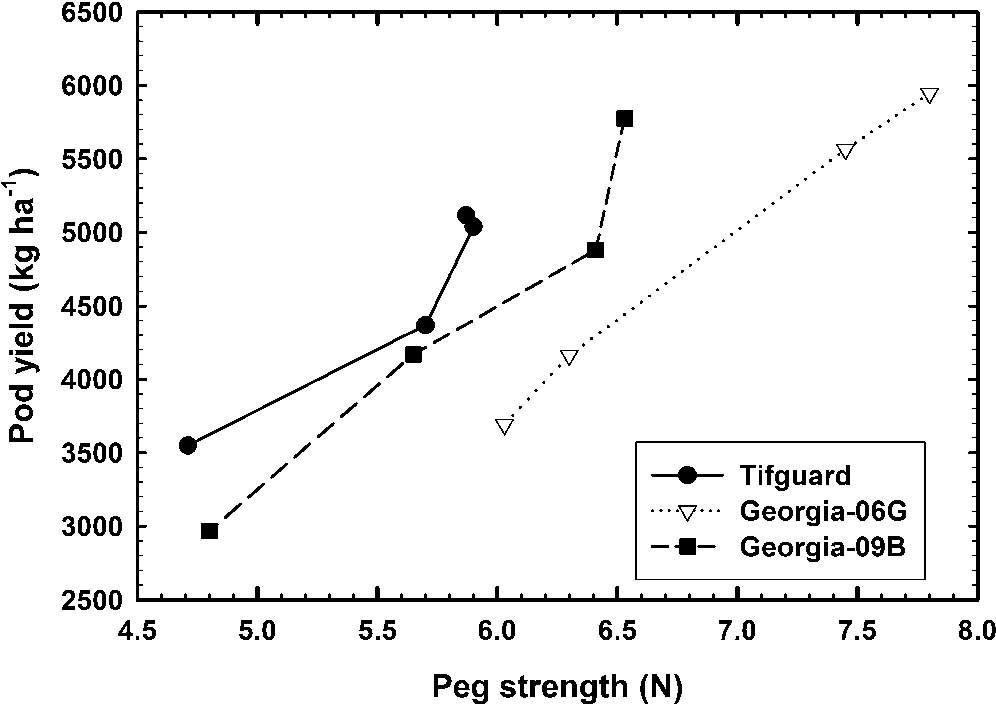
A comparison of peg strength versus peanut yield does show a linear relationship by cultivar (Figure 1). Table 3 shows the linear regression coefficients for each cultivar for pod yield in 2012. Bartlett’s test for equality of variance between cultivars for peg strength versus pod loss indicates no difference in variance (p = 0.4198). Further statistical analysis showed the slope of the line for each cultivar were not statistically different (p = 0.4169) but the intercepts were statistically different (p = 0.0158) confirming peanut yield can vary by peg strength among cultivars.
Peg strength versus peanut yield data indicate a relationship such that peg strength could be used to predict pod yield; however, the difference in the average peg strength from the lowest to the highest peanut yield for Tifguard, Georgia-06G, and Georgia-09B was only 1.15, 1.78, and 1.73 N, respectively. These small differences in peg strengths between high and low yield implies that a very large sample size would be needed to identify the average high and low peg strength. A large sample size would increase collection and preparation time and extend the time required to pull pegs. This large sample size would increase labor requirments compared with the exisiting “hull scrape” method and would therefore probably not be implemented.
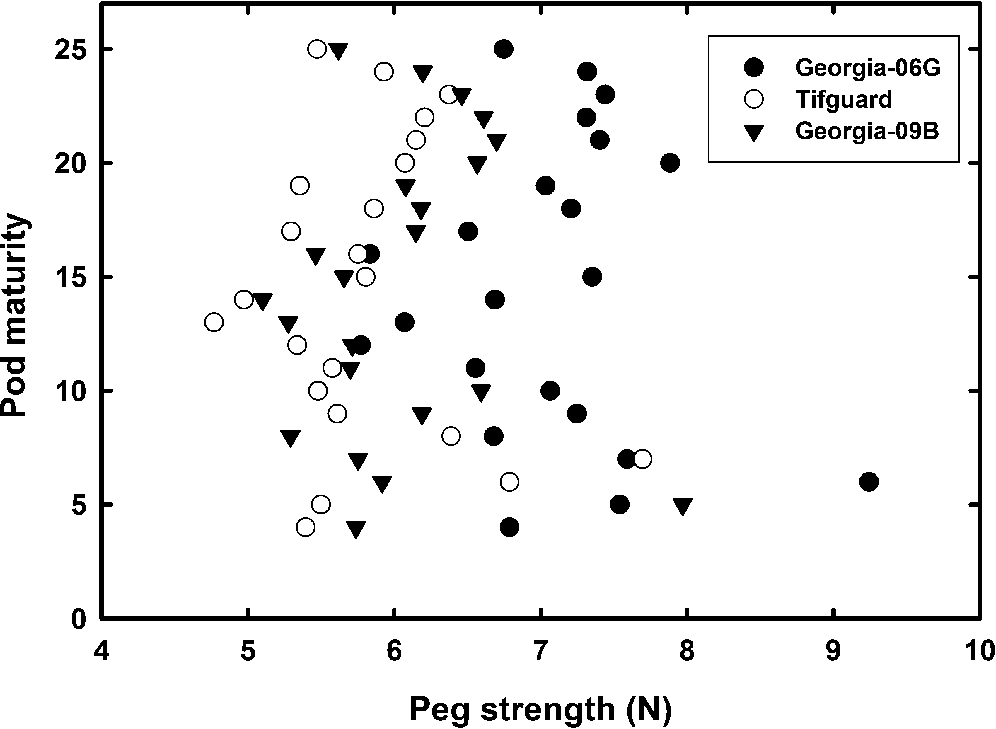
The strong linear relationship between peg strength and peanut yield implies a relationship between peg strength and pod maturity or mesocarp color. Chapin and Thomas (2005) alluded to this peg strength-pod maturity relationship by showing that peg strength was similar for pod maturity through the yellow to black class and decreasing with very mature black mesocarp color. Figure 2 shows the average peg strength for each pod maturity rating (mesocarp color from white to black ranging from 1 to 25) for all cultivars and all harvest dates. Regression analysis showed the slope of the linear regression was near zero with R2 = 0.007 implying the peg strength of an immature peanut could be the same as a mature peanut. Peg strength versus maturity was then averaged within each maturity (color) level and by cultivar. As discussed previously, the cultivar Georgia-06G tended to have stronger peg strengths across the entire maturity profile compared with Tifguard and Georgia-09B, however, there was no relationship between peg strength and pod maturity for these individual cultivars.
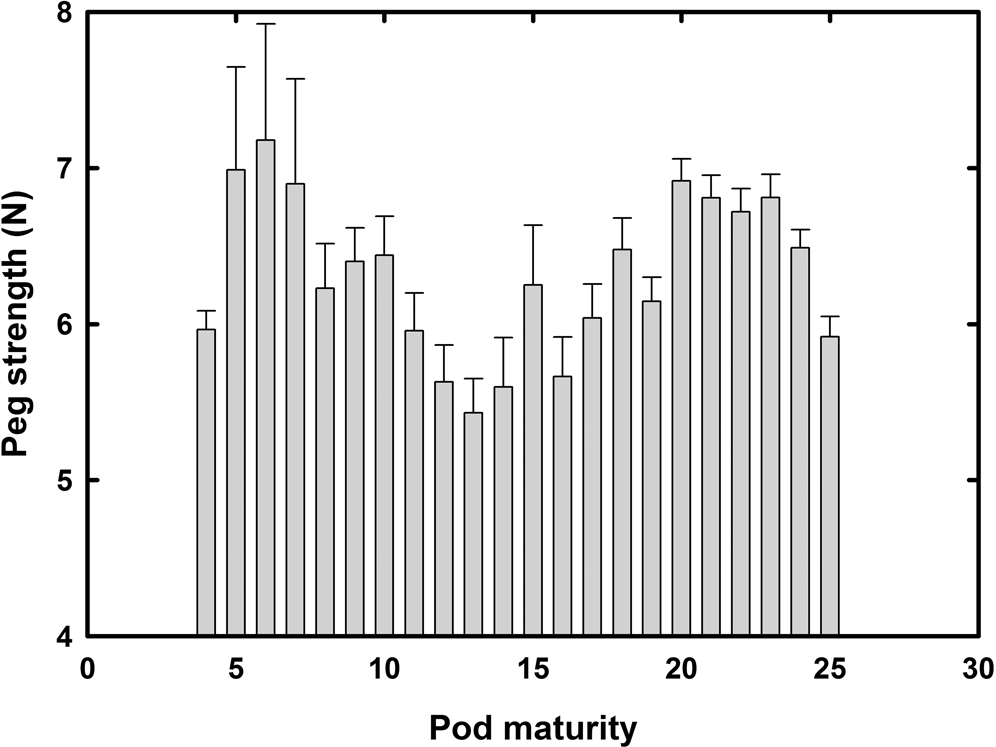
Figure 3 illustrates the mean peg strength across all plant dates, harvest dates, and cultivars and the associated standard error of the mean (error bars) at each mesocarp color level. The standard error was greater with immature peanut and decreased as peanut pods mature (maturity level≥16 to 25). This does not necessarily imply peg strengths of mature peanuts are different than immature peanuts, but does imply that as peanut pods mature they tend to be more stable or have similar peg strength. There was no significant difference in peg strength as peanuts matured. Chapin and Thomas (2005) showed no difference in peg strength as pods matured through the mesocarp color classes of yellow 2 to black. Chapin and Thomas (2005) did show that peg strength did decrease about 32% in over mature black pods compared to mature black pods. This research showed a numerical decrease in peg strength from pod maturity level 20, mature black pods, (6.92 N) to maturity level 25, over mature black pods, (5.92 N) which only about 14% decrease which was about half of what was shown by Chapin and Thomas (2005). Overall, there does not seem to be a relationship between peg strength and pod maturity.
The average peg strength was greater in 2012 (6.7 N) than in 2011 (4.4 N) when averaged across all cultivars and all harvest dates. Different peg strengths across years could be explained by climatic and possible management factors. The average peg strength from first to last harvest date did not change in 2011. In 2012, peg strength was greater in the first two plant dates and harvest dates compared with the first plant date and last two harvest dates. It would seem that peg strength should decrease from the first to the last harvest date implying that older pegs will get weaker. There was no decrease in peg strength in 2011 with harvest date and in 2012 there was a decrease of peg strength versus harvest date but the decrease was variable depending on plant date, harvest date, and cultivar. This could be explained by the indeterminate growth habit of the peanut. As older more mature pegs are weakening, these pods excise, increase pod loss, decrease peanut yield while less mature peanuts become more mature and move from an “orange” to “brown” to “black” maturity class. Thus, as the peanut plant becomes older peanut yield will decrease, pod loss will increase, but average peg strength would remain constant.
Conclusions
Greatest pod yield and lowest pod loss resulted when following accepted best management practices regarding planting and harvesting procedures. A late plant date or a late harvest date tended to reduce yield and increase pod loss. A combination of both a late plant date and a late harvest date will certainly increase the risk of lower peanut yield and increased pod loss. Timely plant dates along with timely harvest dates tended to have greater peg strength. Average peg strengths were different by cultivar and fluctuated with plant and harvest date but tended to decrease as plants passed full maturity. These data show that newer released cultivars can have different peg strengths. Thus, peg strength could be used as a breeding metric for cultivar selection. There was a linear relationship between peg strength and peanut yield, implying peg strength could be used to predict peanut yield. However, the range of peg strength from high to low peanut yield was relatively small. Therefore, to identify this range and to predict peanut yield, a large sample size of peg strengths would need to be collected for each cultivar and range of plant or harvest dates. These large sample sizes would increase the need for manpower, time, and electronic equipment rendering this technique less efficient and probably more costly than the accepted “hull scraping/pod blasting” method currently used. There was no relationship between peg strength and peanut mesocarp color. Therefore, the use of peg strength as a method to determine peanut maturity and eventually digging date was not supported by these data.
Mention of proprietary product or company is included for the reader’s convenience and does not imply any endorsement or preferential treatment by the USDA-ARS.
Literature Cited
Bauman R.W. and Norden A.J. 1971 . Effect of growth regulators and reproductive characteristics of six peanut genotypes . J. Amer. Peanut Res. Educ. Assoc . 3 : 75 – 86 .
Branch W.D. 2007 . Registration of ‘Georgia-06G’ peanut . J. Plant Reg . 1 : 120 .
Branch W.D. 2010 . Registration of ‘Georgia-09B’ Peanut . J. Plant Reg . 4 : 175 – 178 .
Chapin J.W. and Thomas J.S. 2005 . Effect of fungicide treatments, pod maturity, and pod health on peanut peg strength . Peanut Sci . 32 : 119 – 125 .
Holbrook C.C. Timper P. Culbreath A.K. and Kvien C.K. 2008 . Registration of ‘Tifguard’ peanut . J. Plant Reg . 2 : 92 – 94 .
IMADA, Incorporated. 3100 Dundee Rd., Suite 707, Northbrook, IL 60062 . http://imada.com .
Johnson George K Tiwari S.P. and Reddy P.S. 1988 . Genetic variability for peg strength and related characters in groundnut (Arachis Hypogaea) . Indian J. Agric. Sci . 57 : 141 – 143 .
Statistix10 . 2013 . Statistix10: User’s Manual . Analytical Software, P.O. Box 12185. Tallahassee, FL 32317. www.stastitix.com .
Thomas R.J Pettit R.E. Taber R.A. and Jones B.L. 1983 . Peanut peg strength: force required for pod detachment in relation to peg structure . Peanut Sci . 10 : 97 – 101 .
Troeger J.M Williams E.J. and Butler J.L. 1976 . Factors affecting peanut peg attachment force . Peanut Sci . 3 : 37 – 40 .
Williams E.J. and Drexler J.S. 1981 . A non-destructive method for determining peanut pod maturity . Peanut Sci . 8 ( 2 ): 134 – 141 .
Notes
- USDA-ARS-National Peanut Research Laboratory, PO Box 509, 1011 Forrester Dr. SE, Dawson, GA 39842 * Corresponding author email: ron.sorensen@ars.usda.gov
Author Affiliations