Introduction
Peanut (Arachis hypogea L.) is an important high-value crop grown annually on over 400,000 ha in southeastern and south-central United States (http://www.nass.usda.gov/). However, good monetary returns associated with high yields and favorable grading factors also depend on maintaining crops free of diseases. One of the most devastating diseases for peanut is Sclerotinia blight, a soilborne disease caused by the necrotrophic fungus Sclerotinia minor (Jagger). The disease has been identified in Virginia, North Carolina, Texas, Oklahoma, and New Mexico (Beute et al., 1975; Porter and Beute, 1974; Sturgeon, 1986; Wadsworth, 1979; Woodard and Simpson, 1993). Favorable conditions for infection by S. minor include relative humidity over 95%, cooler temperatures ranging from 17 to 21 C (Porter and Melouk, 1997), and soil pH around 6.5 (Porter et al., 1982). Good conditions for disease development are met almost each year in Virginia and North Carolina, where yield reductions due to Sclerotinia blight of 10 to 57% have been previously reported (Porter and Melouk, 1997; Smith et al., 1992). Smith et al. (1992) reported that fungicide fluazinam (Omega 500, ISK BioSciences, Cleveland, OH) can provide some protection but multiple applications are required to achieve a 5% or less control. However, the high cost of chemical control may offset the benefits. Significant reduction of price supports due to changes in farm legislation in 1996, coupled with high costs for fuel and pesticides suggest that effective control of this disease will require a genetic approach to disease resistance. The majority of the virginia-type peanut cultivars grown in the VC region are susceptible, and very susceptible, to Sclerotinia blight. Two recently released cultivars including Bailey (Isleib et al., 2011) and Sugg (Copeland et al., 2010) exhibit limited resistance to the disease. No cultivars to date have demonstrated resistance levels that would alleviate the need for fungicide.
The pathogenicity of Sclerotinia minor is related to production of high levels of oxalic acid that damage the cells and induce plant death (Hollowell and Shew, 2001). In cereals, the enzyme oxalate oxidase degrades oxalic acid to carbon dioxide and hydrogen peroxide and prevents infection by oxalic-acid producing fungi (Davidson et al., 2009; Dumas et al., 1995; Dunwell et al., 2008; Hurkman et al., 1994). However, like most other dicots, peanut does not have the appropriate genetic code required for oxalate oxidase production; therefore it does not synthesize the enzyme. Therefore, genetic transformation technologies were recently employed to transfer oxalate oxidase genes from cereals to other crops. For example, soybean (Glycine max L.), tobacco (Nicotiana tabacum L.), and sunflower (Helianthus annuus L.) transformed with a wheat oxalate oxidase gene showed increased resistance to S. sclerotiorum (Donaldson et al., 2001; Hu et al., 2003; Zaghmout et al., 1997. Transformation of three virginia-type cultivars (NC 7, Perry, and Wilson) with a barley oxalate oxidase gene was conducted to induce production of the oxalate oxidase enzyme (Livingstone et al., 2005). Laboratory and field tests indicated that transformed peanut cultivars had improved resistance to Sclerotinia blight over non-transformed parents (Livingstone et al., 2005; Partridge-Telenko et al., 2011). For example, Livingstone et al. (2005) observed 65 to 89% oxalic acid reduction in leaves of transformed vs. non-transformed peanuts, and Partridge-Telenko et al. (2011) indicated in field trials 81% to 86% lower area under the disease progress curve (AUDPC) in 14 transgenic lines in 2005 and 16 transgenic lines in 2006 compared with their non-transgenic parents. These authors also noted that N70, P39, and W73 were resistant to Sclerotinia blight when compared with their parents, NC 7, Perry, and Wilson, respectively. However, before making recommendations for production, field trials are essential in determining that the agronomic and physiological characteristics of the transformed lines were not negatively impacted by the transformation with oxalate oxidase.
The objective of this research was to compare N70, P39, and W73 transgenic lines with their non-transgenic parents, NC 7, Perry, and Wilson for agronomic and physiological characteristics. Because all three cultivars are no longer grown in commercial production, they were further compared for yield potential and grading factors with Bailey, the most popular peanut cultivar grown on over 75% of the peanut acreage in the VC region with partial resistance to Sclerotinia blight, and with CHAMPS, a sensitive cultivar to this disease.
Materials and Methods
In 2009 and 2010, three transgenic lines (N70, P39, and W73), their non-transgenic parents (NC 7, Perry, and Wilson), and control commercial cultivars Bailey and CHAMPS were evaluated at the Hare Farm (37.13 N, 77.28 W) of the Tidewater Agricultural Research and Extension Center near Holland, VA. NC 7 is a high yielding cultivar with high percentage of extra-large kernels (ELK) and fancy pods, and very susceptible to Sclerotinia blight (Wynne et al., 1979). Perry is also a high yielding virginia-type cultivar but ELK and jumbo pod content are lower than NC 7 and Wilson. Perry is less susceptible to Sclerotinia blight than NC 7 and Wilson, and it is the latest maturing virginia-type cultivar (Isleib et al., 2003). Wilson has good yields, but it is susceptible to Sclerotinia blight and other diseases (Mozingo et al., 2004). Bailey is moderately resistant to Sclerotinia blight and CHAMPS is very susceptible to this disease (Mozingo et al., 2006).
Each year, the genotypes were evaluated in two distinct fields for soil type and available water capacity. One field (F3.5) is classified as Eunola (fine-loamy, siliceous, thermic Aquic Hapludults) and has 3.5 cm of available water capacity in the first 25 cm of soil (USDA-Natural Resources Conservation Service, 14 April 2009). The second field (F2.5) is classified as Kenansville (loamy, siliceous, thermic Arenic Hapludults) and has 2.5 cm of available water capacity in the first 25 cm of soil. This field was considered more drought prone than F3.5. In field F2.5, peanut was grown after over 10 years of cotton monoculture. In F3.5, peanut was grown after corn in 2009 and cotton in 2010 in a three year rotation of corn, cotton, and peanut. Each year, the same cultural practices were applied to both fields and they were performed according to the Virginia recommendations for production of high yield and quality (Faircloth and Shokes, 2008) with detailed information described by Balota (2010, 2011). For correct evaluation of yield potential, a maximum input approach was used in both fields and years. Two weeks before planting, 38 L/ha metam sodium 42% was applied. Aldicarb at 35.9 kg ai/ha was applied infurrow at planting for tobacco thrips (Frankliniella fusca (Hinds)) control. Thrips control was maintained during vegetation by foliar sprays with 0.58 kg ai/ha acephate before flower when a 25% threshold leaflet damage was observed (Herbert, 1999). Chlorpyrifos at 2.5 kg ai/ha was applied in late June for corn earworm (Helicoverpa zea (Boddie)) control. Fenpropathrin was used in mid-August for spider mite (Tetranychus spp.) control at a rate of 0.21 kg ai/ha. Biweekly applications of fungicides were started in late June to control foliar and soil-borne diseases using 0.15 kg ai/ha chlorothalonil, 1.26 kg ai/ha prothioconazole, and 0.21 kg ai/ha tebuconazole. Fluazinam at a rate of 0.52 kg ai/ha was used in late August. Supplemental calcium was applied at 636 kg/ha rate as Gypsum 420 (USG Ben Franklin) at flowering. Boron liquid fertilizer (Helena Chemical Company) was applied pre-plant and at the beginning of pod set at a rate of 1.62 kg ai/ha. In late July and August, 1.12 kg ai/ha manganese was also applied as a fertilizer. Pre-emergence (Wilcut et al., 1995) and post-emergence weed control was applied when weeds were observed, and all plots received the same herbicide treatments each year. The genotypes were grown in plots of two-9.8 m rows planted on 0.9 m centers. They were planted on 29 April in 2009 and 4 May in 2010 at a seeding rate of 10.8 seeds m-2. Each year, weather parameters were recorded within less than one mile from the plots. In each field and year, plots were replicated four times in a randomized complete block design.
The total seedling number within each plot was recorded and converted to the number of seedlings/m2 when all germinated seeds were considered fully emerged. Reproductive stages were determined using definitions by Boote (1982) from beginning flower to beginning maturity in all fields and years by visual observations every other day. The growth stage occurrence dates were recorded and transformed to the number of days after planting (DAP) for each growth stage. In 2010, pod maturity was determined by the pod mesocarp color developed by Williams and Drexler (1981). Five plants per plot were collected a week before harvest and approximately 150±5 pods removed. The pod exocarp was then removed through high pressure washing. Pods were placed on a maturity board and grouped by their mesocarp color into white, yellow, orange, brown, and black color groups. Then, the number of pods within each group was counted and percent of white, yellow, orange, brown, and black pods from the total pod pool was calculated. The profile maturity classes by Williams and Drexler (1981) defines white and yellow as immature, and orange, brown and black as mature pods; orange is defined as incipient maturity, and brown and black fully mature pods.
Incidence of Tomato spotted wilt virus (TSWV) (Family Bunyaviridae, Genus Tospovirus) and Sclerotinia blight was evaluated in early September, close to harvest each year. Incidence for TSWV was determined by counting the number of symptomatic plants per plot (plot area was 17.6 m2). Incidence of Sclerotinia blight was determined by counting the number of infection centers in each plot. An infection center was a point of active growth by Sclerotinia minor and included 15.2 cm on either side of that point. Plant stress (e.g., disease, pests and abiotic stress) was alternatively evaluated by the canopy temperature differential (CTD) in 2010, the dry year. Canopy temperature differential was measured as the difference between canopy and air temperature with an AGRI-THERM III™ handheld infrared thermometer (Model 6210L, Everest Interscience Inc., Tucson AZ) with a field of view of 4°. Four measurements per plot (two east and two west) were taken around daily noon and averaged to provide one reading per plot. The measurement procedure included taking one reading of CTD in all plots (genotypes and replications) followed by the second reading, and so on, until four CTD measurements per plot were collected. This way differences in CTD due to temporary cloudiness, wind speed fluctuations, and sun direction and altitude were minimized. In 2010, CTD was measured at beginning bloom and full seed stages and data from both fields were combined.
Plots were harvested in the first week of October in 2009 and the second week of October in 2010 with conventional peanut harvest equipment. Pod yield was determined from the plot weight and adjusted for 7% seed moisture and the foreign material content. A 500 g pod sample was retained from each plot to determine pod brightness and grade characteristics, i.e., % ELK, % sound mature kernels (SMK), % damaged kernels (DK), % sound split kernels (SS), and % total meat (TM). Jumbo and fancy pods are pods large enough to not pass a 13.5 mm spacing on the pre-sizer, and their content was also measured. Jumbo and fancy pod brightness were measured with a colorimeter (model D25LT, Hunter Associates Laboratory, Inc. Reston, VA) and expressed as Hunter L scores (Isleib et al., 1997). Hunter L is a measure of pod albedo with a theoretically black pod scoring 0 and a pure white pod scoring 100. Extra-large kernels are kernels that do not pass a 25.4 by 8.5 mm screen for virginia-type peanut. Sound mature kernels are defined as kernels that do not pass a 25.4 by 6 mm screen. Sound splits are undamaged halves of kernels and DK are inedible peanut kernels due to decay, mold, insect damage, sprouting, freeze damage, or skin discoloration. Total meat includes ELK, SMK, SS, and DK. Gross value was calculated from the federal formula and it is based on a combination of the farmer stock grade factors and pod yield.
The effects of genotype, field, year, and their interactions on the measured traits were assessed with factorial ANOVA from the GLM procedure of SYSTAT® 10.2 (2002, SYSTAT Software Inc, Richmond, CA). Means were separated by Tukey’s Honestly Significant Difference (HSD) test at P = 0.05.
Results
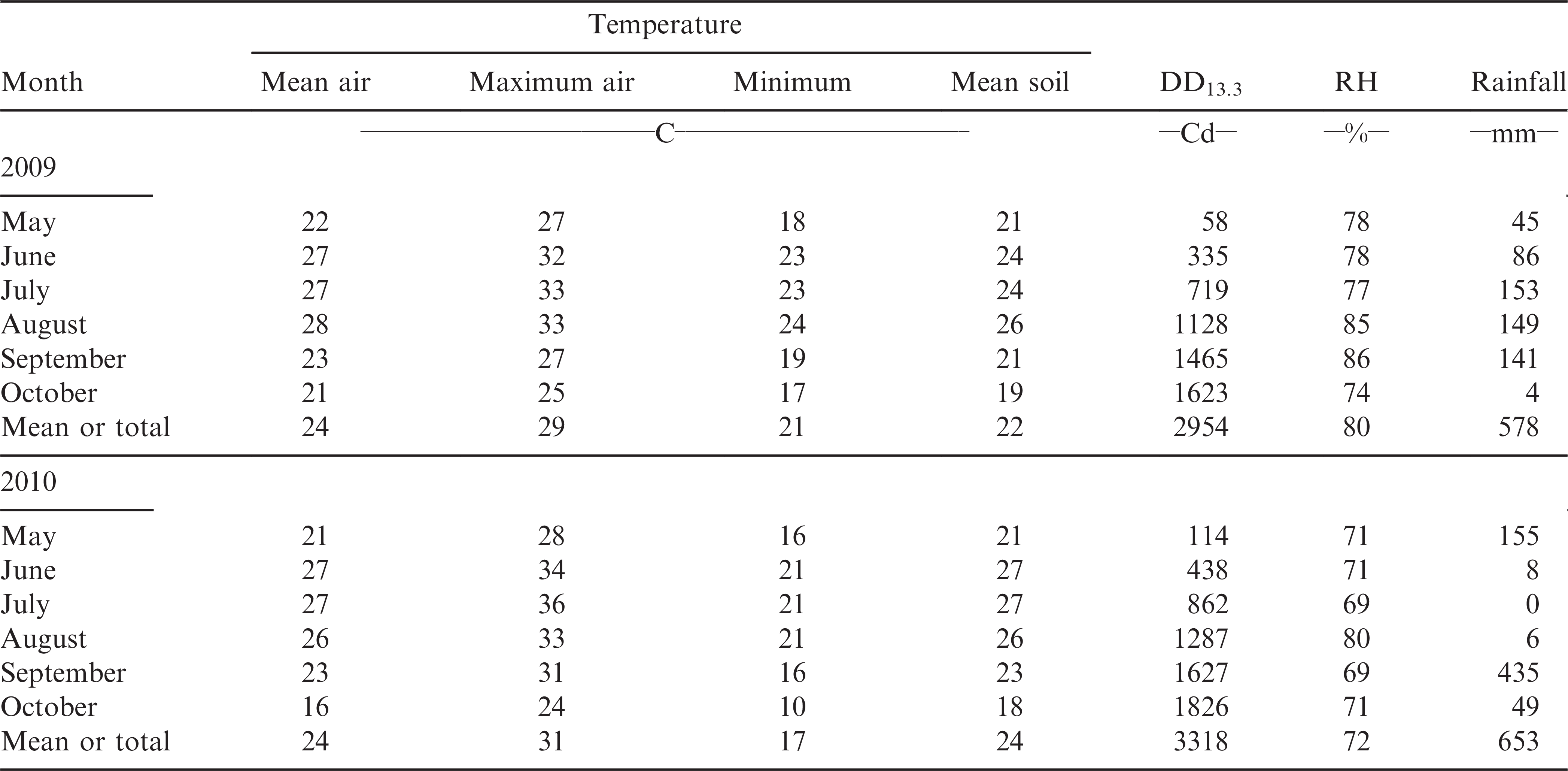
Peanut production was varied over two years due to different weather conditions during the period of most intense peanut growth and development (Table 1). Weather in 2009 was generally cool and wet while in 2010 it was hot and dry. For example, trace rainfall was recorded during June, July, and August of 2010 therefore 24.5 mm of irrigation was applied on 22 June, 7 July, and 21 July to both fields (73.5 mm total). In contrast, during the same time period rainfall totaled 388 mm in 2009. In 2010, a total of 435 mm precipitation were received at the end of September in a few heavy storms that arrived too late to benefit pod production. Both years had sufficient heat units for peanut maturity and optimum light conditions for photosynthesis (Pallas and Samish, 1974) and growth (Table 1).
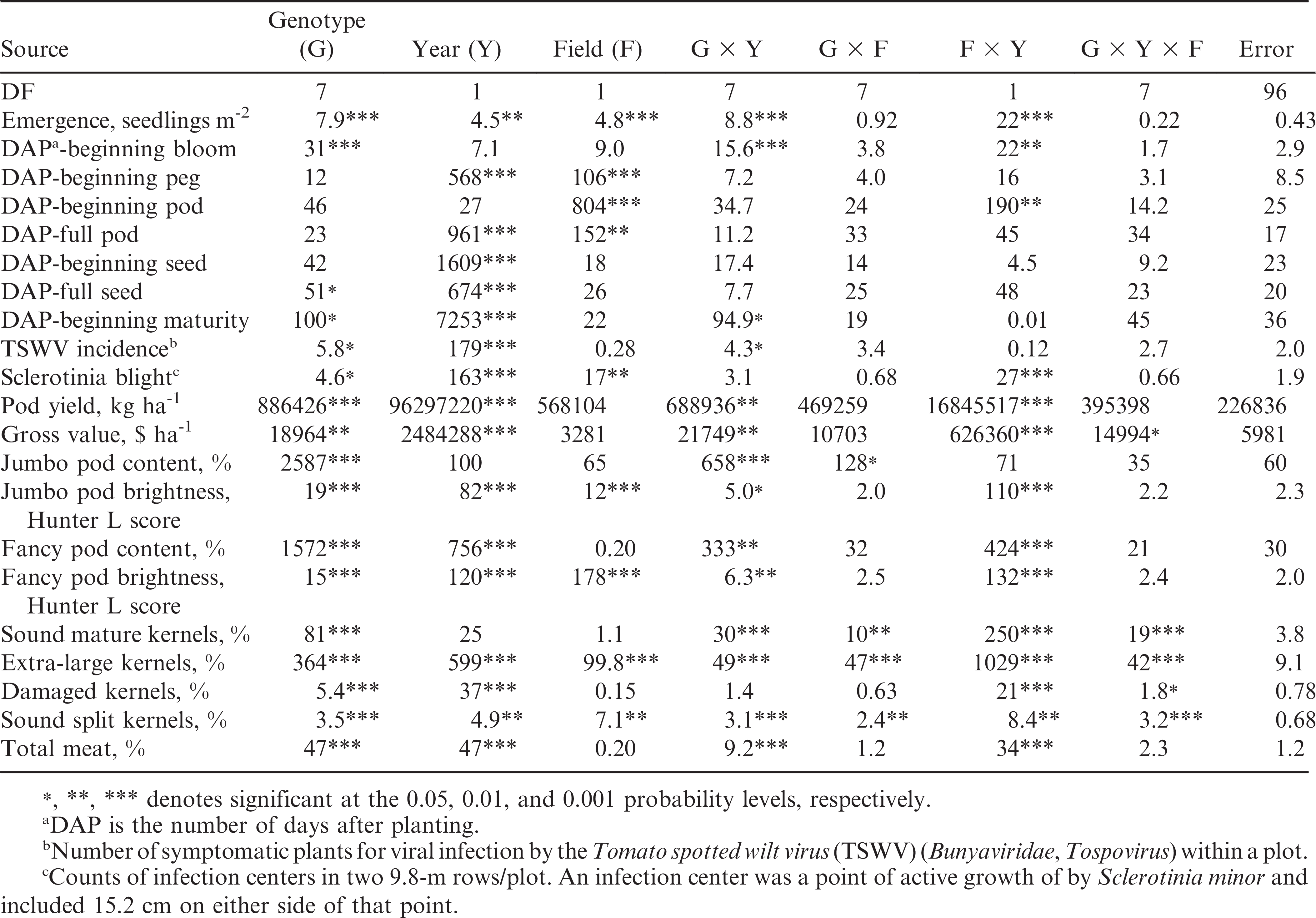
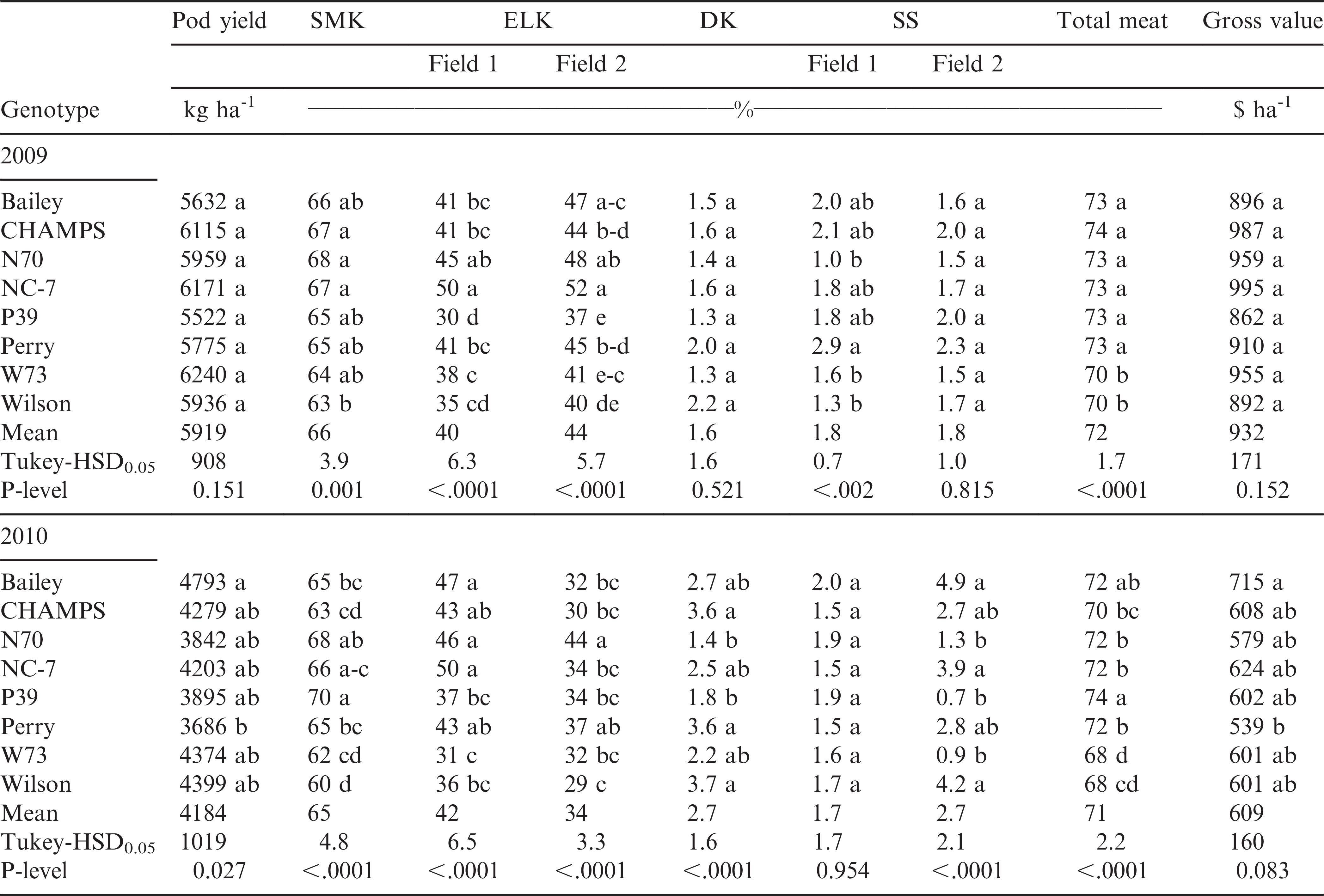
Combined analysis of variance for the effect of genotype, year, field, and their interactions on emergence, growth stages, disease, pod yield, crop gross value, and farmer stock grading factors are shown in Table 2. Genotype had a significant (p ≤ 0.05) effect on all the characteristics analyzed in this experiment excepting the number of DAP to beginning peg, beginning pod, full pod, and beginning seed. Year had a significant effect on all characteristics except for the number of DAP to beginning bloom and pod, and the content of jumbo pods and SMK. Field effect was significant for emergence, the number of DAP to beginning peg, beginning pod, full pod, incidence of Sclerotinia blight, jumbo and fancy pod brightness, and the contents of ELK and SS kernels. The genotype by year and field by year interactions were significant for the majority of the characteristics, but the genotype by field interactions were significant only for jumbo pod content, ELK, SMK, and SS kernels (Table 1). However, the sum of squares of the genotype by field interactions was relatively small for all these characteristics. For the jumbo pod content and SMK there were no main field effects but a strong genotypic effect that could have caused these interactions. For the jumbo pod content and SMK, and all characteristics showing no genotype by field interaction, data from both fields each year were averaged for mean comparisons.
Sum of squares from combined analysis of variance for growth, disease, and agronomic traits including gross value of three virginia-type transgenic lines, their non-transgenic parents, and CHAMPS and Bailey cultivars from two growing seasons and two fields (8 replications) each season at Suffolk, VA, during 2009 and 2010.
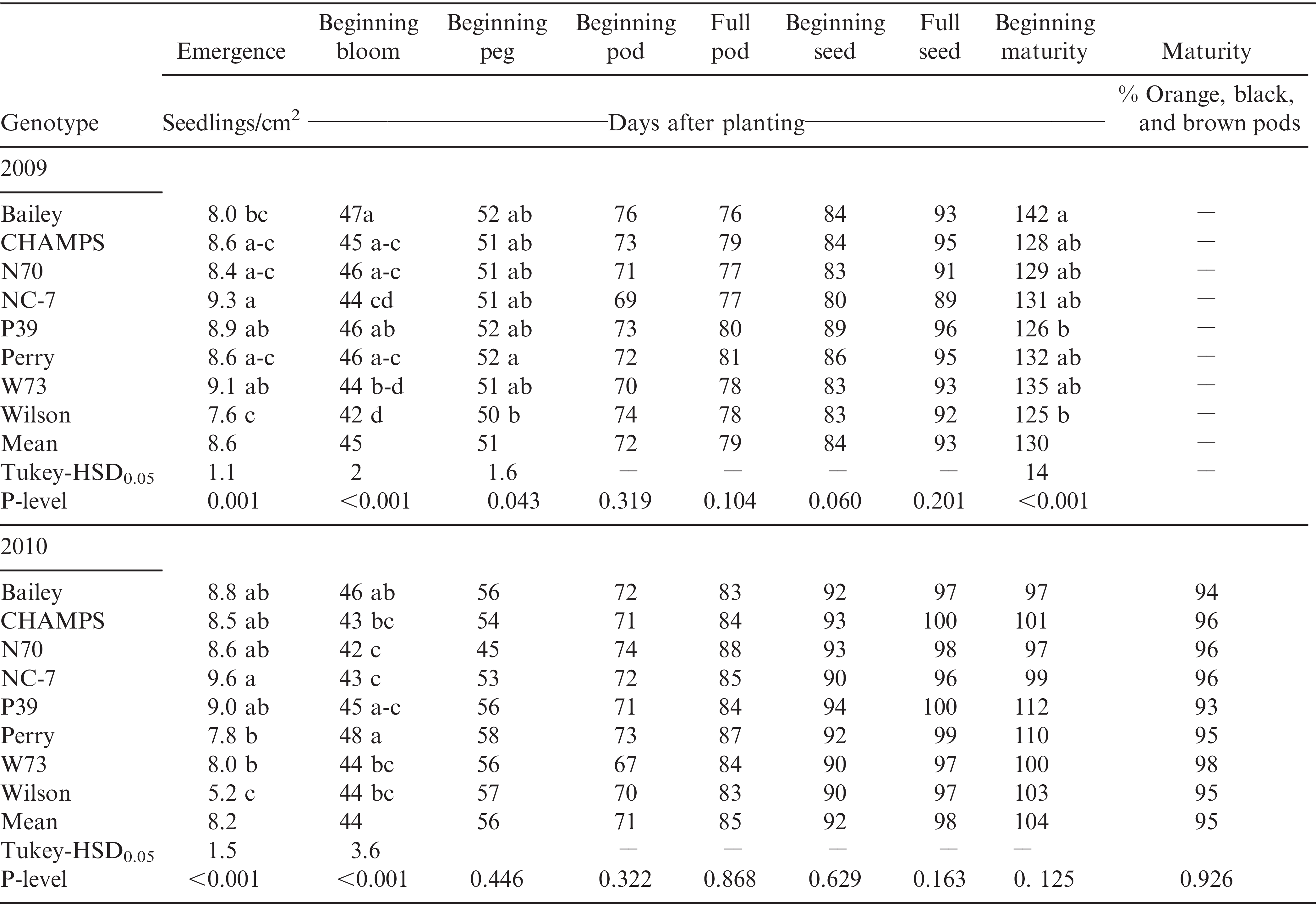
For growth and development, significant (p < 0.001) differences among genotypes were observed for emergence and beginning bloom in both years and beginning maturity in 2009 (Table 3). In both years precipitation was adequate for germination and emergence, resulting in average plant populations of 84,000 plants/ha or 8.4 seedlings/m2 each year. Across years and fields, NC 7 had the highest and Wilson the lowest emergence. With the exception of W73, which had significantly (p < 0.01) higher emergence than its parent, all other cultivars and the transformed lines emerged similarly in both fields each year. Beginning bloom started at 44 DAP in 2009 and 45 DAP in 2010. Overall, Perry, P39, and Bailey required significantly (p < 0.0001) more days to beginning bloom than NC 7, W73, and Wilson in 2009 and CHAMPS, W73, and Wilson in 2010. No significant differences between the transgenic lines, non- transgenic parents, Bailey, and CHAMPS were recorded from beginning peg to full seed in 2009 and from beginning peg to beginning maturity in 2010. In 2009, P39 reached beginning maturity 16 d earlier that Bailey (Table 3).
Emergence, and the number of days after planting to reach beginning bloom, beginning peg, beginning pod, full pod, beginning seed, full seed, and beginning maturity of three virginia-type transgenic lines (N70, P39 and W73), their non-transgenic parents (NC 7, Perry, and Wilson), CHAMPS and Bailey cultivars averaged over two fields (8 replications) each year in 2009 and 2010. Averages for maturity are from two fields (8 replications) in 2010.
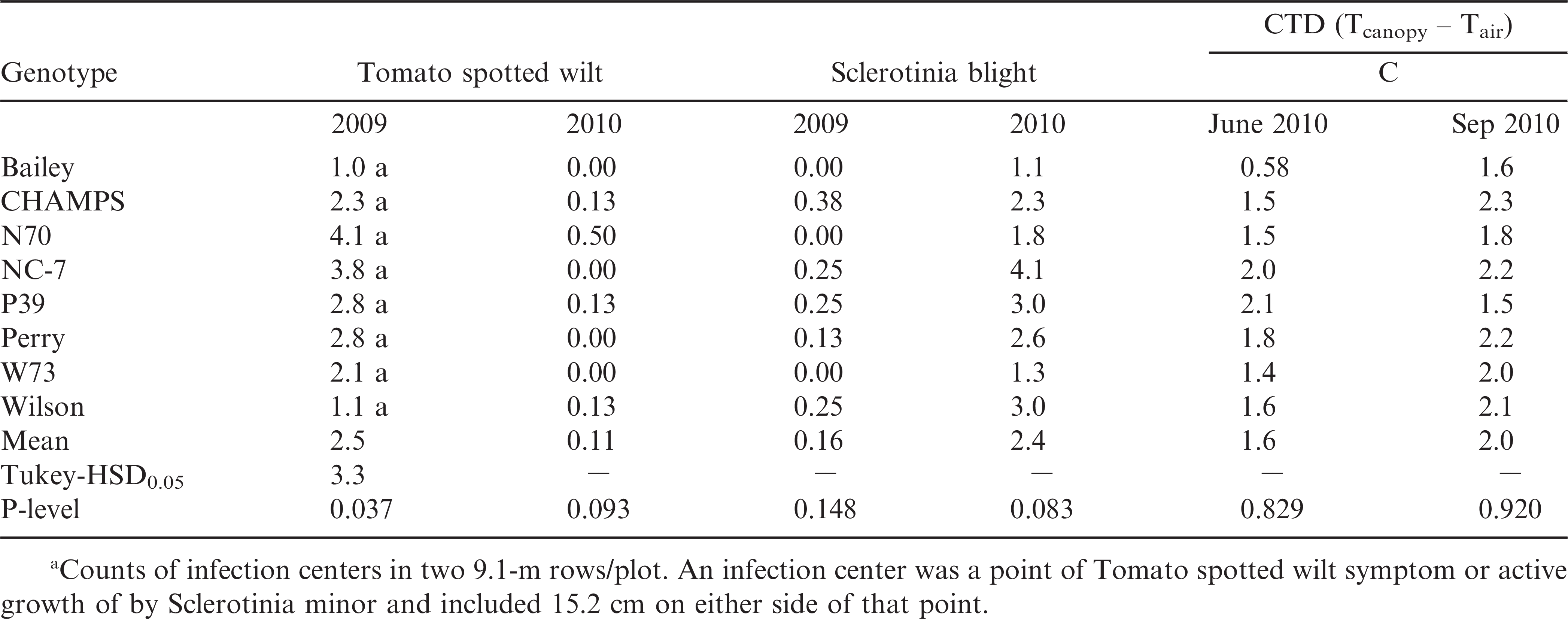
Peanut lines from 2009 were more prone to TSWV and 2010 to Sclerotinia blight disease (Table 4). However, in both years incidence levels were relatively low, not exceeding on average 3.2 infection centers for TSWV in 2009 and 2.4 for Sclerotinia blight in 2010. Each year field F2.5 had less disease, either TSWV or Sclerotinia, than field F3.5. There were no significant differences for disease incidence among genotypes in any year. No significant differences for CTD among genotypes were recorded (Table 4). Canopy temperature values not exceeding 2.5 C above air temperature confirmed that genotypes were not under significant stress in 2010, permitting ideal conditions for yield potential evaluation under the dry environment.
Tomato spotted wilt virus and Sclerotinia blight incidencea of three virginia-type transgenic lines (N70, P39 and W73), their Non-transgenic parents (NC 7, Perry, and Wilson), and CHAMPS and Bailey cultivars averaged over two years and two fields each year. Canopy temperature differential (CTD) averages are from two fields (8 replications) each year in 2009 and 2010.
Yield, ELK, TM, and gross crop value were significantly (p < 0.05) less in 2010 than in 2009 (Table 5). For example, average pod yield was 30% less in 2010 than in 2009 and gross value was 35% less in the dry 2010 than in 2009. Damaged and SS kernels were higher in 2010 than in 2009. Year had no effect on SMK. Overall, Perry and P39 were the lowest, and Wilson and W73 the highest yielding genotypes, comparable to the high yielding cultivars Bailey and CHAMPS (Table 5). Genotypes NC 7 and N70 had intermediate yields. In both years, N70, NC 7, and P39 had overall the highest, and W73 and Wilson the lowest SMK. The lowest ELK was for P39, W73, and Wilson and the highest was for N70 and NC 7. Wilson and W73 had the least TM among the genotypes each year. Genotypic differences existed for the DK and SS content each year and in every field, but the greatest differences were observed in F2.5 in 2010. Interestingly, all transgenic lines had overall significantly (p < 0.05) less damaged and split kernels than the non-transgenic parents, Bailey, and CHAMPS. In general, the gross value of yield was lowest for Perry and P39, and highest for Bailey, CHAMPS and NC 7 (Table 5).
Pod yield, the sound mature kernels (SMK), extra-large kernels (ELK), damaged kernels (DK), sound split kernels (SS), total meat (TM), and gross value of three virginia-type transgenic lines (N70, P39 and W73), their non-transgenic parents (NC 7, Perry, and Wilson), and CHAMPS and Bailey cultivars. When field is not specified, averages over two fields (8 replications) are presented.
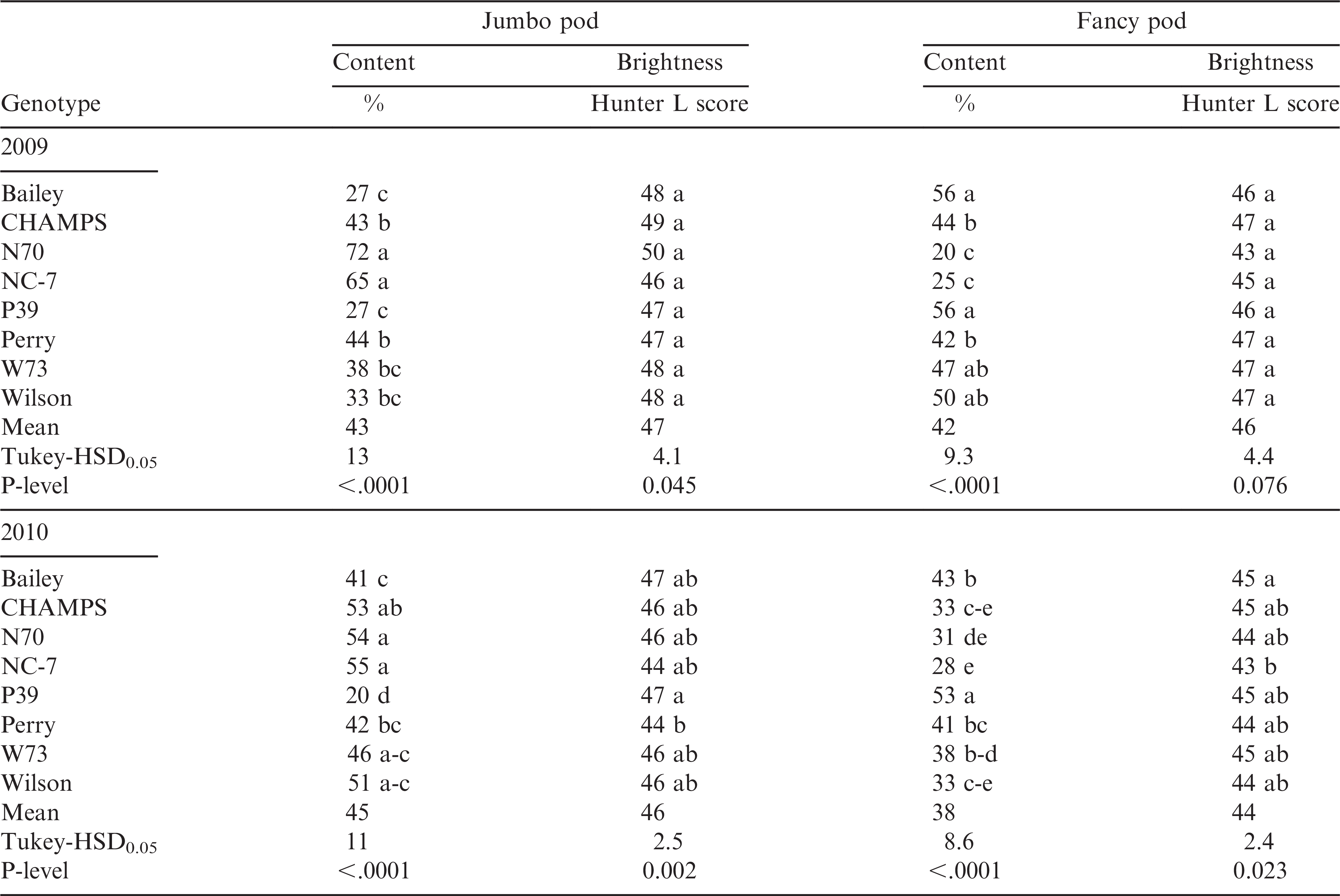
The number of jumbo pods was significantly lower in P39 than its non-transgenic parent, Perry, and the other cultivars except Bailey, W37, and Wilson in 2009 (Table 6). Conversely, fancy pod numbers were greater for P39 than other genotypes with the exception of Bailey, W73, and Wilson in 2009. Jumbo and fancy pod brightness was similar for all genotypes with the exception of P39, which was significantly brighter for Jumbo pods than its non-transgenic parent, Perry, in 2010 (Table 6).
Discussion
The data provide information on the agronomic and physiological characteristics of three transgenic lines expressing oxalate oxidase, their non-transgenic parents, and commercial cultivars Bailey and CHAMPS as check cultivars in absence of severe Sclerotinia blight disease. TSWV and Sclerotinia blight were also nearly absent (e.g. <5%). Environmental stresses that could have been present in the dry year 2010 had minimum impact on the peanut plants based on the CTD data collected at beginning bloom and full seed stages in 2010 and compared with previous literature on peanut (Balota et al., 2012; Sanders et al., 1985). Under these conditions, emergence for W73 was superior to that of its parent, Wilson, which is a favorable characteristic with practical implications for production. In both years of this study, Wilson demonstrated significantly reduced seedling emergence; emergence of 70% of the total seed planted in 2009 and 48% in 2010 was observed. Wilson has been previously identified as deficient in germination, in particular with one year-old seed, and with delayed emergence (Phipps, personal communication). For W73 emergence was 85% in 2009 and 74% in 2010 of the total planted seed. Perry and P39 were the latest maturing genotypes in this experiment. However under the conditions of 2009 and 2010, both only required longer DAP to begin bloom but they were comparable with all other genotypes for occurrence of the other developmental stages and maturity. Interestingly in 2009, P39 matured earlier than Bailey. From this, it can be concluded that all genotypes in this study have relatively early maturity and are suitable for growing in the VC region, including those in the most northern counties of peanut production in Virginia where shorter seasons may be problematic.
In the absence of disease and other stress factors, all transgenic lines were statistically comparable for yield and crop gross value with their non-transgenic parents, Bailey, and CHAMPS, which suggests similar yield potentials and returns. Farmer stock grading factors are very important when considering the gross crop value. For example, premiums are applied for ELK and SMK content. Sound splits are undamaged halves of kernels and a penalty is applied if SS exceeds 4%. When DK exceeds 2.4%, seed is considered segregation 2 or 3 and growers receive only 35% of the gross value for that peanut. P39 showed smaller ELK than its parent and the other genotypes, but the SMK was in general greater than other genotypes. The greatest ELK was for NC 7 and N70. N70 also had high SMK in both years. Reduced ELK and SMK contents were recorded for W73 and Wilson. Overall, all transgenic lines had lower DK and SS than their non-transgenic parents, Bailey, and CHAMPS, which shows that the transgenic lines have less kernel damage and splits in comparison with their parents and currently available commercial cultivars, possibly due to their improved disease resistance. In this study the gross value of the transgenic lines was not statistically different than their non-transgenic parents, Bailey and CHAMPS. However, net value for growing these lines could be higher than for their parents due to improved disease resistance and reduced need for fungicide applications that can offset smaller ELK and SMK kernels of the transgenic lines.
Even though growers do not receive premiums for pod size and brightness, both are very important characteristics for the in-shell peanut market. P39 had significantly fewer jumbo pods than Perry and most of the other genotypes in both years, showing that this line is probably not suitable for the in-shell trading. Even though there is no set criteria for pod size for the in-shell market, cultivars with increased jumbo pod content are preferred by consumers and shellers (Simmons, personal communication). The highest numbers of jumbo pods were recorded for N70 and NC 7, suggesting that these two genotypes would be good choices for the in-shell market. Generally, fancy pods are less bright than jumbo pods. In this experiment pod brightness was good for all genotypes and comparable brightness of the jumbo and fancy pods were observed in both years. Overall, the transgenic lines had similar yield and grading characteristics with their parents, Bailey, and CHAMPS, which indicates their suitability for commercial production in the VC region. These lines are suitable not only because of yield and grading, but also for their disease resistance traits.
Acknowledgements
This work was supported with funding from the National Peanut Board, Virginia Peanut Board, Virginia Carolina Peanut Association and Virginia Crop Improvement Association.
Literature Cited
Balota M Isleib T.G. and Tallury S. 2012 Variability for drought related traits of Virginia type peanut cultivars and advanced breeding lines Crop Sci. 52 ( 6 ): 2702 – 2713 .
Balota M. 2010 Peanut crop physiology related projects at Tidewater Agricultural Research and Extension Center - 2009 Virginia Coop. Ext. Publ. 3009-1460 (Available on-line with updates at http://pubs.ext.vt.edu/3001/3001-1432/3009-1460.html).
Balota M. 2011 Peanut crop physiology related projects at Tidewater Agricultural Research and Extension Center - 2010 Virginia Coop. Ext. Publ. 3101-1532 Available on-line with updates at http://pubs.ext.vt.edu/3101/3101-1523/3101-1523.html.
Beute M.K Porter D.M. and Hadley B.A. 1975 Sclerotinia blight of peanut in North Carolina and Virginia and its chemical control Plant. Dis. Rep. 59 : 697 – 701 .
Boote K.J. 1982 Growth stages of peanut (Arachis hypogaea L.) Peanut Sci. 9 : 35 – 40 .
Copeland S Isleib T. Milla-Lewis S. Shew B. Hollowell J. Pattee H. Sanders T. Dean L. Hendrix K. Balota M. and Chapin J. 2010 Release of ‘Sugg‘ Virginia-type Peanut Cultivar In: 2010 Proceedings of the American Peanut Research and Education Society 42 : 81 – 82 .
Davidson R.M Reeves P.A. Manosalva P.M. and Leach J.E. 2009 Germins: a diverse protein family important for crop improvement Plant Sci. 177 : 499 – 510 .
Dumas B Freyssinet G. and Pallett K.E. 1995 Tissue-specific expression of germin-like oxalate oxidase during development and fungal infection of barley seedlings Plant Physiol. 107 : 1091 – 1096 .
Dunwell J.M Gibbings J.G. Mahmood T. and Saqlan Naqvi S.M. 2008 Germin and germin-like proteins: Evolution, structure and function Crit. Rev. Plant Sci. 27 : 342 – 375 .
Faircloth J.C and Shokes F.M. 2008 Agronomic recommendations and procedures p 10-20. In: Virginia Peanut Guide. J. C. Faircloth (ed.) VPI&SU/Va. Agric. Exp. Stn./TAREC
Herbert D.A. 1999 Insect control in peanut p 38-52. In: 1999 Virginia Peanut Production Guide. Tidewater Agric. Res. and Ext. Center Info. Series 416, Suffolk, VA
Hollowell J.E and Shew B.B. 2001 Oxalic acid production by nine isolates of Sclerotinia minor Proc. Am. Peanut Res. Educ. Soc. 33 : 24 .
Hu X Bidney D.L. Yalpani N. Divick J.P. Crasta O. Folkerts O. and LU G. 2003 Overexpression of a gene encoding hydrogen peroxide-generating oxalate oxidase evokes defense responses in sunflower Plant Physiol. 133 : 170 – 181 .
Hurkman W.J Lane B.G. and Tanaka C.K. 1994 Nucleotide sequence of a transcript encoding a germin-like protein that is present in salt-stressed barley (Hordeum vulgare L.) roots Plant. Physiol. 104 : 803 – 804 .
Donaldson P.A Anderson T. Lane B.G. Davidson A.L. and Simmonds D.H. 2001 Soybean plants expressing an active oligomeric oxalate oxidase from the wheat gf-2.8 (germin) gene are resistant to the oxalate oxidase-secreting pathogen Sclerotinia sclerotiorum. Physiol. Mol. Plant Pathol. 59 : 297 – 307 .
Isleib, T.G., H.E. Pattee, and P.W. Rice A laboratory colorimeter method to measure pod brightness in Virginia-type peanut. Peanut. Sci. 24 : 81 – 84 .
Isleib T.G Rice P.W. Mozingo R.W Mozingo R.W. Bailey J.E. and Pattee H.E. 2003 Registration of ‘Perry’ peanut Crop Sci. 43 : 739 – 740 .
Isleib T.G Milla-Lewis S.R. Pattee H.E. Copeland S.C. Zuleta M.C. Shew B.B. Hollowell J.E. Sanders T.H. Dean L.O. Hendrix K.W. Balota M. and Chapin J.W. 2011 Registration of ‘Bailey’ peanut J. Plant Registrations, 2011, 5 : 27 – 39 .
Livingstone D.M Hampton J.L. Phipps P.M. and Grabau E.A. 2005 Enhancing resistance to Sclerotinia minor in peanut by expressing a barley oxalate oxidase gene Plant Physiol. 137 : 1354 – 1364 .
Mozingo R.W Coffelt T.A. Phipps P.M. Coker D.L. Machado S. and Petrie S.E. 2006 Registration of ‘CHAMPS’ peanut Crop Sci. 46 : 2711 – 2712 .
Mozingo R.W Coffelt T.A. Swann C.W. and Phipps P.M. 2004 Registration of ‘Wilson’ peanut Crop Sci. 44 : 1017 – 1018 .
Pallas J.E. and Samish B.Y. 1974 Photosynthetic response of peanut Crop Sci. 14 : 478 – 482 .
Partridge-Telenko D.E Hu J. Livingstone D.M. Shew B.B. Phipps P.M. and Grabau E.A. 2011 Sclerotinia blight resistance in virginia-type peanut transformed with a barley oxalate oxidase gene Phytopathol.101 786 – 793 .
Porter D.M and Beute M.K. 1974 Sclerotinia blight of peanuts Phytopathol. 64 : 263 – 264 .
Porter D.M and Melouk H.A. 1997 Sclerotinia blight In: Kokkalis-Burelle N. Porter D.M. Rodriguez-Kabana R. Smith D.H. and Subrahmanyam P. ( eds .). Compendium of peanut diseases. APS Press, St. Paul, pp 34-36 .
Sanders T.H Blankenship P.D. Cole R.J. and Hill R.A. 1985 Temperature relationships of peanut leaf canopy, stem, and fruit in soil of varying temperature and moisture Peanut Sci. 12 ( 2 ): 86 – 89 .
Smith F.D Phipps P.M. and Stripes R.J. 1992 Fluazinam: a new fungicide for control of Sclerotinia blight and other soil-borne pathogens of peanut Peanut Sci. 19 : 115 – 120 .
Sturgeon R.V. 1986 Peanut disease loss estimates for major peanut producing states in the United States for 1984 and 1985 Proc. Am. Peanut. Res. Educ. Soc. 18 : 24 – 26 .
Wadsworth D.F. 1979 Sclerotinia blight of peanut in Oklahoma and occurrence of the sexual stage of the pathogen Peanut Sci. 6 : 77 – 79 .
Wilcut J.W York A.C. Grichar W.J. and Wehtje G.R. 1995 The biology and management of weeds in peanut (Arachis hypogaea) p. 207-244. In: H.E. Pattee and H.T. Stalker (eds.) Advances in Peanut Science. Amer. Peanut Res. Educ. Soc., Stillwater, OK
Williams E.J and Drexler J.S. 1981 A non-destructive method for determining peanut maturity Peanut Sci. 8 : 134 – 141 .
Woodard K.E and Simpson C.E. 1993 Characterization of growth and sclerotial production of Sclerotinia minor isolated from peanut in Texas Plant. Dis. 77 : 567 – 579 .
Wynne J.C Mozingo R.W. and Emery D.A. 1979 Registration of ‘NC 7’ peanut (Reg No. 22). Crop Sci. 19 : 563 .
Zaghmount O.F Dang P.D. and Allen R.D. 1997 Expression of oxalate oxidase in transgenic plants provides resistance to oxalic acid and oxalate-producing fungi (abstract no 1152). Plant physiol. (Suppl.) 114 : 227 .
Notes
- First and third authors: Tidewater Agricultural Research and Extension Center, Virginia Tech University, Suffolk, VA 23437; Second author: Cornell Vegetable Program, Cornell University, East Aurora, NY, 14052; Fourth author: Department of Plant Pathology, Physiology, and Weed Science, Virginia Tech University, Blacksburg, VA, 24061 *Corresponding author’s E-mail: mbalota@vt.edu
Author Affiliations