Introduction
Sclerotinia blight, caused by Sclerotinia minor Jagger, is one of the most widespread and damaging peanut diseases worldwide. In the United States, Sclerotinia blight is a problem primarily in Virginia, North Carolina, Oklahoma, and Texas, capable of causing 50% yield loss in heavily infested fields (Porter and Melouk, 1997). For peanut growers, planting resistant cultivars is the most economical approach for managing this disease. Fungicides are frequently used to manage Sclerotinia blight in fields with high levels of disease, but treatments are costly. Sclerotia of S. minor are also viable in the soil for four to five years without a host (Melouk and Backman, 1995); therefore, cultural practices such as multi-year crop rotations may be difficult economically for growers to implement (Damicone and Jackson, 1994). Peanut cultivars with significant resistance to Sclerotinia are available (Chappell et al., 1995; Goldman et al., 1995; Hollowell et al., 2008), although none developed using traditional breeding methods are immune to the disease.
The time required to develop a new peanut cultivar may take more than ten years (Baring et al., 2006; Smith et al., 1991). Multiple years of field testing are needed to identify and verify disease resistance, but it may be possible to accelerate progress by augmenting field trials with greenhouse-based resistance assays. Greenhouse assays may be less limited by seasonality and physical space than field trials, and are thus suitable for screening a larger number of plants. Environmental conditions and pathogen inoculum levels, which frequently vary in the field (Hollowell et al., 2008), may also be controlled more easily in the lab. Lab-based resistance assays for Sclerotinia blight using detached stems (Brenneman et al., 1988; Melouk et al., 1992), whole plants (Cruickshank et al., 2002; Goldman et al., 1995; Hollowell et al., 2008), and leaflets (Hollowell et al., 2003) have been developed. Unfortunately, many of these assays are better at identifying highly resistant and highly susceptible cultivars, and less capable of discriminating among moderately resistant cultivars (Faske et al., 2006; Goldman et al., 1995). In addition, results from greenhouse-based assays may not be completely congruent with results from field trials (Hollowell et al., 2008, 2003). These assays cannot account for plant architecture characteristics that create favorable or unfavorable conditions for disease (Brenneman et al., 1988; Hollowell et al., 2008, 2003).
An alternative, potentially useful method for screening peanuts for resistance to Sclerotinia blight uses oxalic acid. Some fungal plant pathogens, including Sclerotinia spp. (Godoy et al., 1990; Livingstone et al., 2005) and Sclerotium rolfsii (Bateman and Beer, 1965; Kritzman et al., 1977), produce oxalic acid to infect plants. Oxalic acid was used to screen several crops, including common bean (Kolkman and Kelly, 2000), scarlet runner bean (Chipps et al., 2005), sunflower (Noyes and Hancock, 1981), and soybean (Cunha et al., 2010; Wegulo et al., 1995), for resistance to S. sclerotiorum. In these crops, response to oxalic acid exposure, as measured by wilt severity or discoloration in detached stems, discoloration in leaflets, or proportion of lysed cells, correlated with field resistance. However, the oxalic acid assay still shares the same disadvantage as other greenhouse-based assays in that it measures physiological resistance and cannot predict plant avoidance through canopy architecture (Brenneman et al., 1988; Hollowell et al., 2008, 2003). Response to oxalic acid was used to evaluate resistance in peanuts transformed with barley oxalate oxidase (Livingstone et al., 2005); however, this method has not yet been evaluated as a resistance assay for wider selection of conventionally-bred peanuts. The objective of this project was to determine if detached stems of runner and spanish peanut cultivars known to be resistant or susceptible to Sclerotinia blight in the field differed in response to oxalic acid.
Materials and Methods
Six runner and seven spanish peanut genotypes varying in susceptibility to Sclerotinia blight were used to evaluate responses to oxalic acid (Table 1). The following runner genotypes were included: susceptible cultivars Florunner (Norden et al., 1969) and Okrun (Banks et al., 1989); moderately resistant breeding line ARSOK-R143-3 and cultivars Red River Runner (Melouk et al., 2012) and Tamrun 96 (Smith et al., 1998); and the highly resistant breeding line ARSOK-R35. The following spanish peanut genotypes were used: susceptible cultivars Pronto (Banks and Kirby, 1983) and Spanco (Kirby et al., 1989); moderately resistant breeding lines ARSOK-S140-1OL and ARSOK-S1 (PI 670132; Chamberlin et al., 2014), and cultivars Tamnut OL06 (Baring et al., 2006) and Tamspan 90 (Smith et al., 1991); and the highly resistant breeding line ARSOK-S133-3. The relative resistance levels of the breeding lines were assessed on the basis of a minimum of 3 years of field evaluations.
Plants were grown in the greenhouse at 22 to 32 C in 15-cm pots filled with Metro-Mix 350 (Sun Gro Horticulture, Bellevue, WA). Supplemental lighting was supplied by 300-watt LED lights (Sunshine Systems, Wheeling, IL) set at a 14 hr photophase. After six weeks, the two lowermost lateral stems of each plant were cut near the main stem, and main stems were cut immediately below the third or fourth node. Cuts were made perpendicular to each stem using a razor blade, and detached stems were immediately placed in approximately 6 cm of reverse osmosis water. Stems were held in water 60 to 75 min until stems from all genotypes were prepared.
Detached lateral and main stems were rinsed with sterile reverse osmosis water, blotted dry, and placed individually into 5-ml tubes containing 3.5 ml of 0, 20, or 50 mM oxalate solution. Foam plugs were used to secure each stem so that at least 1.5 cm of the stem base was submersed in the solution. Tubes with detached stems were placed in the dark at room temperature (21 to 24 C). Plant response to oxalate exposure was rated after 12, 18, and 24 hr using a modification of the rating scale of Kolkman and Kelly (2000), where 1 = no wilt symptoms, 2 = one leaf wilted, 3 = two leaves wilted, 4 = one petiole collapsed, 5 = two petioles collapsed, and 6 = branch stem collapsed. The length of the bleached, lesion-like stem discoloration in response to oxalic acid exposure was also measured. Tubes were refilled with additional solution at 12 and 18 hr after immersion as needed.
Experiments evaluating the response of runner genotypes to oxalate solutions were conducted separately from the spanish genotypes. A randomized complete block design was used with three replications (three lateral stems and three main stems) per cultivar x oxalate concentration. Blocks were arranged so that the same replication for each cultivar was placed into the oxalic acid solution at approximately the same time. Test tube racks holding individual blocks were placed on separate trays. The experiment was conducted three times between December 2012 and February 2013. Separate analyses were conducted for each market type and stem type. Hypotheses regarding differences in lateral and main stem response (mean wilt scores and length of discoloration) of the differentially resistant genotypes to oxalate were tested using repeated measures ANOVA with an autoregressive covariance structure (TYPE = AR(1)) in SAS (v. 9.2, SAS Institute, Inc., Cary, NC). When a relevant interaction was significant, the SLICE option of the LSMEANS statement was used to examine simple effects.
Results and Discussion
No wilting or stem discoloration was observed at any time period for stems of either peanut market type immersed in water (0 mM oxalic acid). Consequently, these data were removed from the analyses.
Wilt symptoms
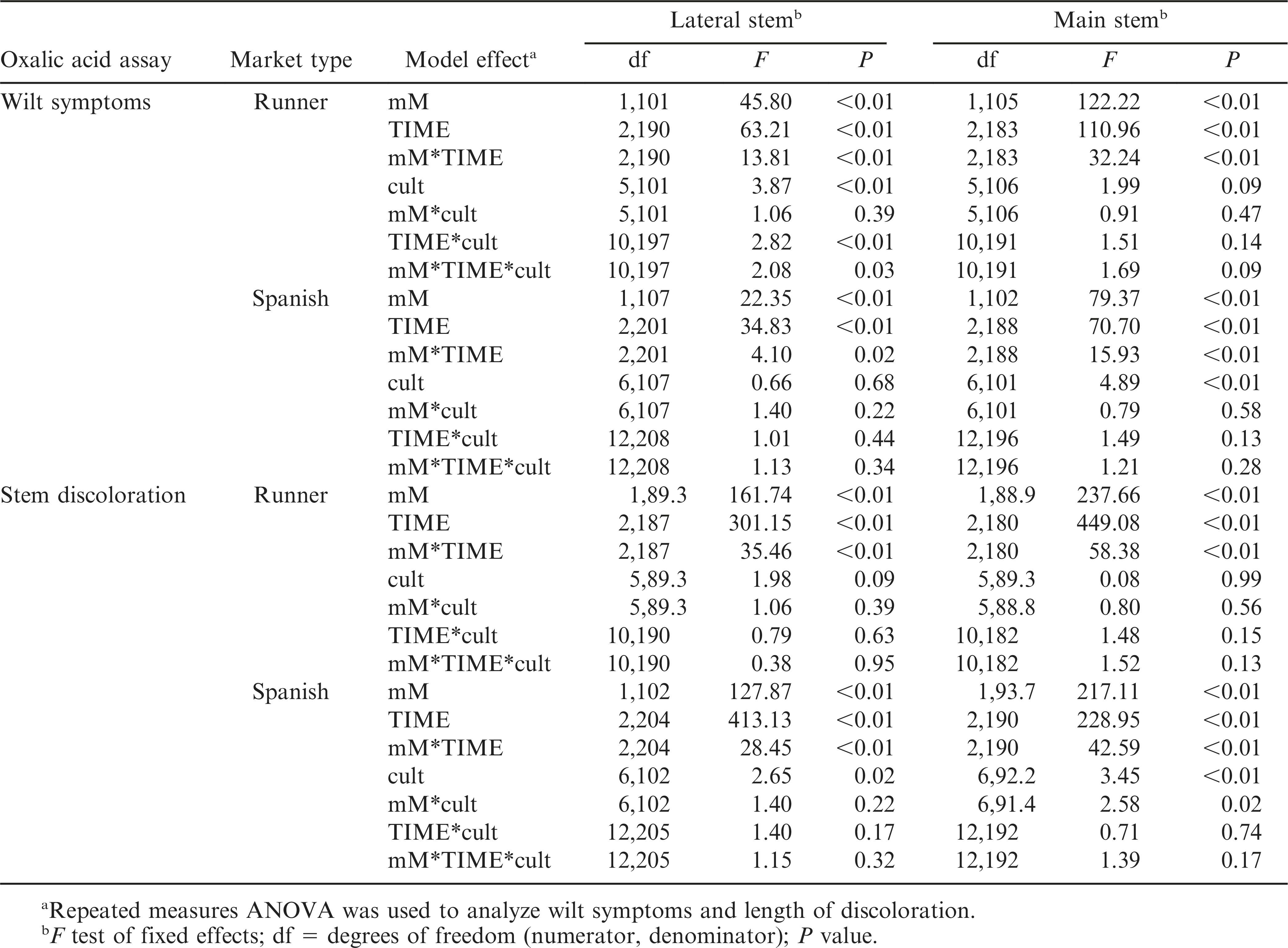
The interaction between oxalic acid concentration and duration of exposure was significant for all stem and peanut market types (Table 2), indicating that the effect of one factor was dependent on the level of the other factor (Fig. 1). In addition, a significant three-way interaction among concentration, time, and cultivar was present for lateral stems of runner peanuts, which made interpretation of the main effects difficult. Cultivar did not have a significant effect on wilt responses of main stems of runners and of lateral stems of spanish genotypes (Table 2). Therefore, differences among cultivars in wilt response were not present or consistent over all concentrations and immersion times (Fig. 1). However, the effect of cultivar on wilt responses was significant in main stems of spanish (Table 2).
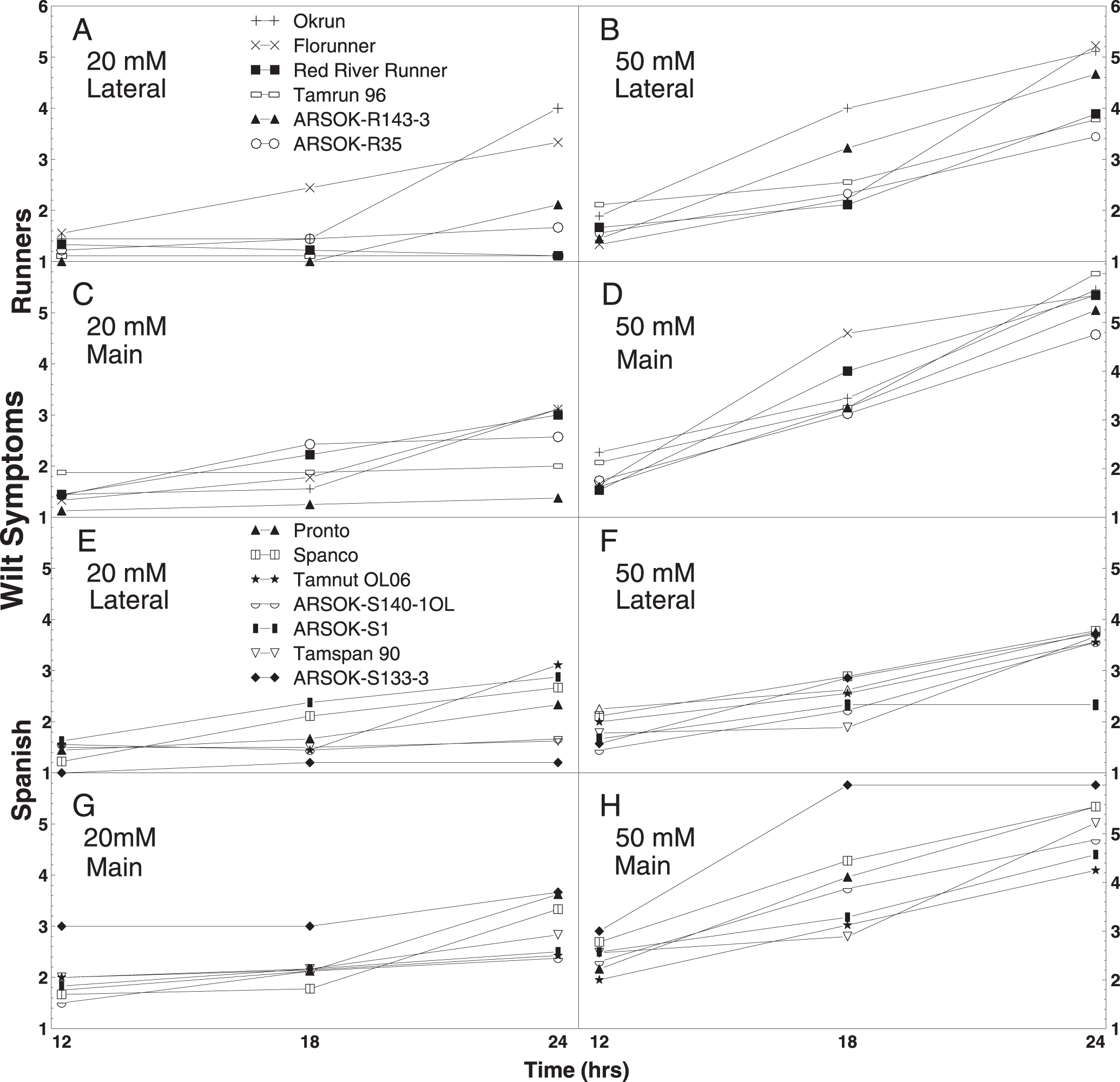
Mean wilting score of runner peanut genotypes Okrun, Florunner, Red River Runner, Tamrun 96, ARSOK-R143-3, and ARSOK-R35 (A-D) and spanish genotypes Pronto, Spanco, Tamnut OL06, ARSOK-S140-1OL, ARSOK-S1, Tamspan 90, ARSOK-S133-3 (E-H). Wilt was rated from lateral and main stems of peanuts immersed in 20 mM or 50 mM oxalic acid for 12, 18, and 24 hr, using a modification of the rating scale of Kolkman and Kelly (2000), where 1 = no wilt symptoms, 2 = one leaf wilted, 3 = two leaves wilted, 4 = one petiole collapsed, 5 = two petioles collapsed, and 6 = branch stem collapsed.
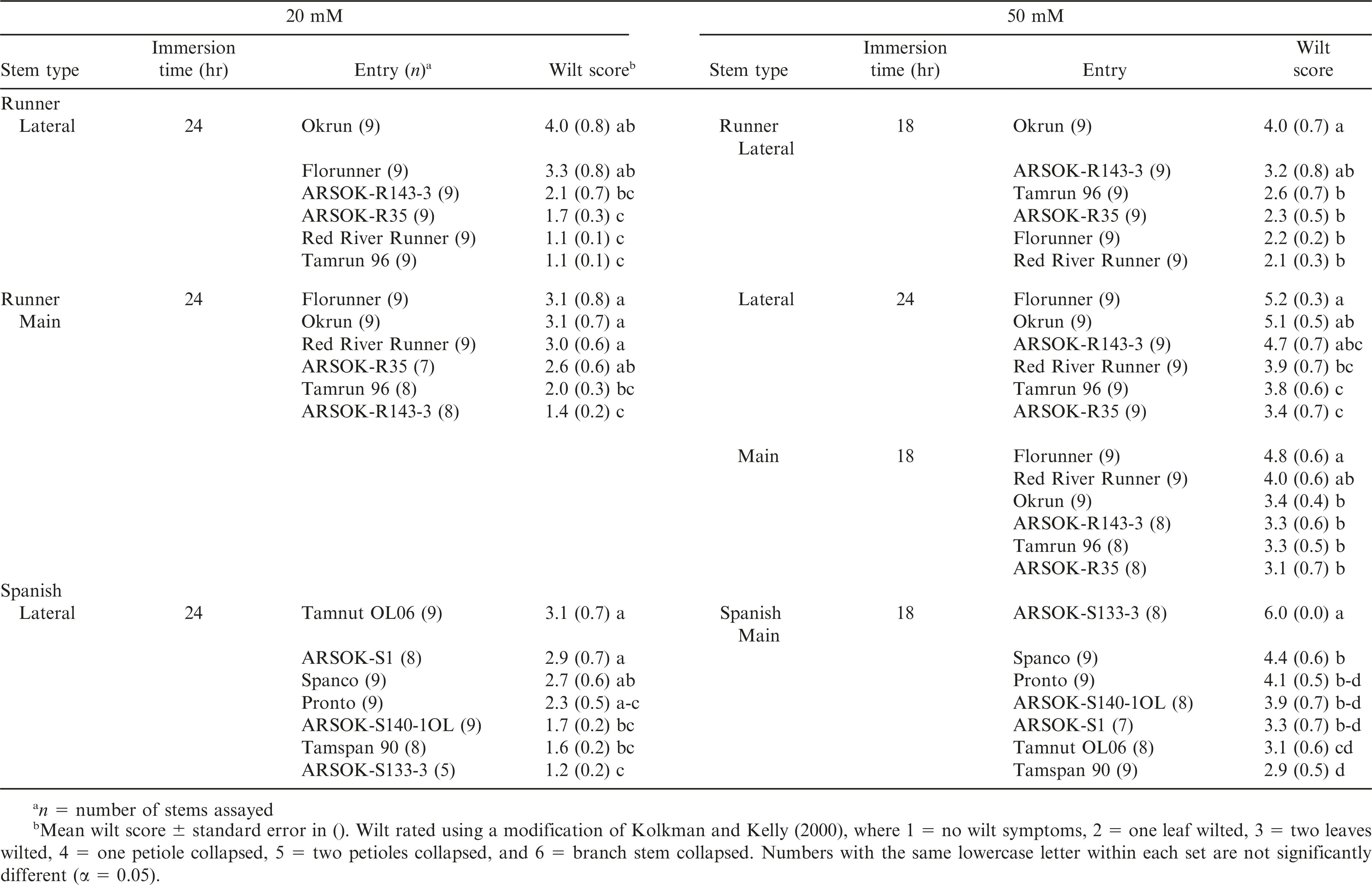
To facilitate interpretation, the wilt responses were further examined so that cultivars could be compared by oxalic acid concentration and duration of exposure. Out of the twelve possible combinations of concentration (20 mM and 50 mM), time (12, 18, and 24 hr), and stem type (lateral and main), five tests among the runners and two tests among the spanish genotypes, resulted in significant differences among cultivars in wilt symptoms (Table 3). No significant differences in wilt severity were observed among the genotypes at 12 hr of exposure. When mean comparisons were adjusted for type-I error, only two tests resulted in significant differences among cultivars: lateral stems of runners immersed in 20 mM for 24 hr, and main stems of spanish in 50 mM for 18 hr (data not shown).
Wilting appeared to be a better indicator of Sclerotinia blight resistance in runners than in spanish peanuts. Okrun and/or Florunner, cultivars known to be highly susceptible to Sclerotinia blight, exhibited the most severe wilt symptoms in all of the statistically significant tests (Table 3). Wilting in response to oxalic acid was less consistent at discriminating the moderately resistant and highly resistant genotypes. In 20 mM oxalic acid, significant differences in wilting were observed at 24 hr after immersion among lateral and main stems of runner entries (Table 3; Fig. 1A). Lateral stems of Okrun and Florunner were significantly more wilted than lateral stems of ARSOK-R35, Red River Runner, and Tamrun 96. Main stems of ARSOK-R143-3 were less wilted than Okrun, Florunner, Red River Runner, and ARSOK-R35, but not significantly different than Tamrun 96 (Table 3; Fig. 1C). In 50 mM oxalic acid, significant differences in wilting among runner cultivars were observed at 18 and 24 hr after immersion for the lateral stems, and 18 hr after immersion for the main stems. Lateral stems of Okrun were more wilted than the lateral stems of other cultivars except ARSOK-R143-3 at 18 hr after immersion. After 24 hr of immersion, lateral stems of Okrun and Florunner exhibited more severe wilt than stems of Tamrun 96 and ARSOK-R35 (Table 3; Fig. 1B). For the main stems, Florunner had more wilt after 18 hr than all cultivars except Red River Runner.
No clear relationship between field resistance or susceptibility and wilting was observed among the spanish entries (Table 3). Significant differences in wilt symptoms among the spanish entries were observed only in lateral stems immersed for 24 hr at 20 mM and in main stems immersed for 18 hr at 50 mM (Table 3; Fig. 1E, 1H). Lateral stems of Tamnut 06 and ARSOK-S1 exhibited more wilt than stems of ARSOK-140-1OL, Tamspan 90, and ARSOK-S133-3 when immersed at 20 mM for 24 hr. Main stems of ARSOK-S133-3 and Spanco immersed in 50 mM for 18 hr were more wilted than Tamnut OL06 and Tamspan 90.
Stem discoloration
A bleached, and occasionally reddish, area of discoloration typically appeared at the base of stems immersed in oxalic acid and lengthened up the stem over time, but only the white lesions were measured. In addition, the white areas on stems often became sunken and somewhat shriveled.
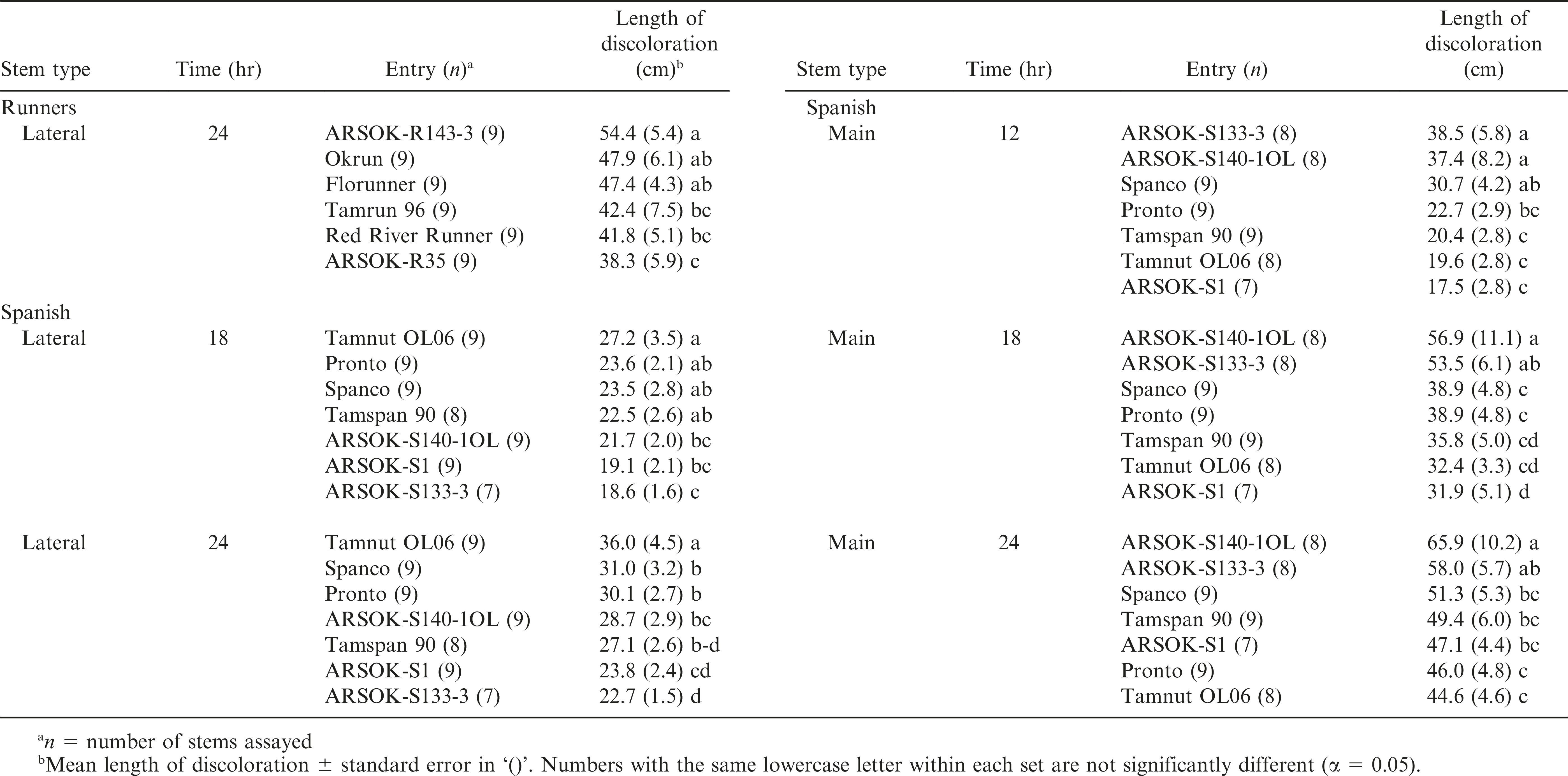
A significant interaction between oxalic acid concentration and duration of immersion was observed for both stem types in runner and spanish genotypes (Table 2). Cultivar did not have a significant effect on length of discoloration in lateral or main stems of runner peanuts. The effect of cultivar was significant for both spanish stem types, but for main stems, this effect was dependent on the level of oxalic acid concentration (Table 2; Fig. 2).
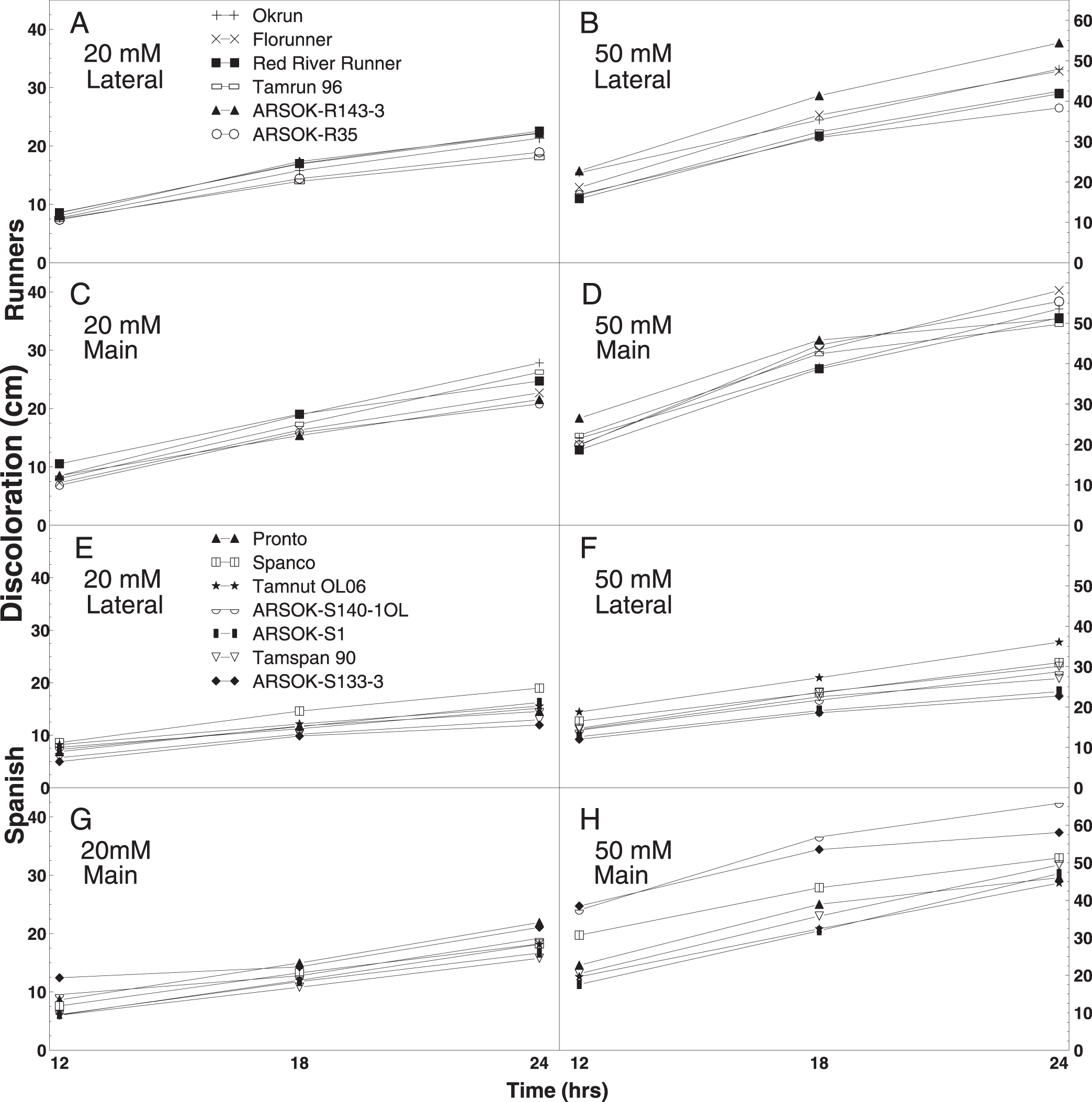
Out of the twelve combinations of concentration, time, and stem type, one test among the runner and five tests among the spanish genotypes resulted in significant differences in stem discoloration (Table 4). All six tests were at the 50 mM concentration. Significant differences in length of discoloration in the runner entries were observed only on lateral stems immersed in 50 mM oxalic acid for 24 hr. ARSOK-R143-3, Okrun, and Florunner had significantly more discoloration than ARSOK-R35 (Table 4; Fig. 2B). Five tests of the spanish genotypes resulted in significant differences among cultivars in stem discoloration; however, severity of stem discoloration was less correlated with field resistance (Table 4). After 18 hr of immersion, lateral stems of Tamnut OL06 were more discolored than ARSOK-S140-1OL, ARSOK-S1, and ARSOK-S133-3. Similar results were obtained after 24 hr: Tamnut 06 had the greatest stem discoloration, and ARSOK-S1 and ARSOK-S133-3 had the least (Table 4; Fig. 2F). Significant differences in discoloration were observed for all times of exposure at 50 mM in the main stems. After 12 hr of immersion, main stems of ARSOK-S133-3, ARSOK-S140-1OL, and Spanco were more discolored than Tamspan 90, Tamnut 06 and ARSOK-S1. After 18 and 24 hr, ARSOK-S140-1OL had more stem discoloration than all other cultivars except ARSOK-S133-3 (Table 4; Fig. 2H).
Mean length of stem discoloration in runner (Okrun, Florunner, Red River Runner, Tamrun 96, ARSOK-R143-3, and ARSOK-R35; A-D) and spanish peanut genotypes (Pronto, Spanco, Tamnut OL06, ARSOK-S140-1OL, ARSOK-S1, Tamspan 90, ARSOK-S133-3; E-H). Stem discoloration in lateral and main stems of peanuts were measured after being immersed in 20 mM or 50 mM oxalic acid for 12, 18, and 24 hr.
Enzymes for breaking down oxalate are present in multiple cereal species, but they are absent from many other crops, including peanuts (Livingstone et al., 2005). Several crop species have been transformed with oxalate-degrading enzymes to create plants resistant to Sclerotinia spp. (Cunha et al., 2010; Dias et al., 2006; Hu et al., 2003; Livingstone et al., 2005). Virginia peanut cultivars genetically transformed with barley oxidase were also highly resistant to Sclerotinia blight in both the lab (Livingstone et al., 2005) and field (Partridge-Telenko et al., 2011). Other, though less-effective, mechanisms for limiting damage from oxalic acid are naturally present in soybean (Wegulo et al., 1995) and common bean (Kolkman and Kelly, 2000; Tu, 1985). Tu (1985) observed differences between susceptible and resistant cultivars of Phaseolus vulgaris in petiole uptake and vein delimitation of oxalic acid. Similar mechanisms may be present in runner peanuts. We found Okrun and Florunner, cultivars known to be highly susceptible to S. minor in the field, consistently exhibited the most severe wilt symptoms when exposed to oxalic acid. The oxalic acid concentrations and immersion times used here, which resulted in significant differences among cultivars, may be useful starting points for identifying physiologically susceptible runner genotypes.
Wilt symptoms in spanish peanuts, and stem discoloration in both runner and spanish peanuts, appeared to be less correlated to known field resistance. A small number of spanish genotypes were used in these experiments, and we may have unknowingly chosen cultivars where canopy characteristics exert a strong influence on field resistance. Improved correlations may be obtained from a different panel of spanish entries, or by using older plants or stems (Brenneman et al., 1988), but these preliminary results indicate that oxalic acid may be of limited use as a resistance assay in spanish peanuts.
Acknowledgements
The authors thank Lisa Myers and Kenneth Jackson for technical assistance. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Literature Cited
Banks D.J and Kirby J.S 1983 Registration of Pronto Peanut (Reg No. 28) Crop Sci 23 : 184 – 184 .
Banks D.J Kirby J.S and Sholar J.R 1989 Registration of ‘Okrun’ peanut Crop Sci 29 : 1574 – 1574 .
Baring M.R Lopez Y Simpson C.E Cason J.M Ayers J and Burow M.D 2006 Registration of ‘Tamnut OL06’ peanut Crop Sci 46 : 2720 – 2721 .
Bateman D.F and Beer S.V 1965 Simultaneous production and synergistic action of oxalic acid and polygalacturonase during pathogenesis by Sclerotium rolfsii Phytopathology 55 : 204 – 211 .
Brenneman T.B Phipps P.M and Stipes R.J 1988 A rapid method for evaluating genotype resistance, fungicide activity, and isolate pathogenicity of Sclerotinia minor in peanut Peanut Sci 15 : 104 – 107 .
Chamberlin, K.D., J.P. Damicone, M.R. Baring, M.D. Burow, C.B. Godsey, R.S. Bennett, H.A. Melouk, and C.E. Simpson Registration of high-oleic peanut germplasm line ARSOK-S1 (TX996784) with enhanced resistance to Sclerotinia blight and pod rot J. Plant Regist ., 9 : 103 – 107 .
Chappell G.F Shew B.B Ferguson J.M and Beute M.K 1995 Mechanisms of resistance to Sclerotinia minor in selected peanut genotypes Crop Sci 35 : 692 – 696 .
Chipps T.J Gilmore B Myers J.R and Stotz H.U 2005 Relationship between oxalate, oxalate oxidase activity, oxalate sensitivity, and white mold susceptibility in Phaseolus coccineus Phytopathology 95 : 292 – 299 .
Cruickshank A.W Cooper M and Ryley M.J 2002 Peanut resistance to Sclerotinia minor and S. sclerotiorum Australian J. Agric. Res 53 : 1105 – 1110 .
Cunha W.G Tinoco M.L.P Pancoti H.L Ribeiro R.E and Aragão F.J.L 2010 High resistance to Sclerotinia sclerotiorum in transgenic soybean plants transformed to express an oxalate decarboxylase gene Plant Pathol 59 : 654 – 660 .
Damicone J.P and Jackson K.E 1994 Factors affecting chemical control of southern blight of peanut in Oklahoma Plant Dis 78 : 482 – 486 .
Dias B.B.A Cunha W.G Morais L.S Vianna G.R Rech E.L de Capdeville G and Aragão F.J.L 2006 Expression of an oxalate decarboxylase gene from Flammulina sp in transgenic lettuce (Lactuca sativa) plants and resistance to Sclerotinia sclerotiorum Plant Pathol 55 : 187 – 193 .
Faske T Melouk H.A and Payton M.E 2006 Comparison of Sclerotinia minor inocula for differentiating the reaction of peanut genotypes to Sclerotinia blight Peanut Sci 32 : 31 – 35.
Godoy G Steadman J.R Dickman M.B and Dam R 1990 Use of mutants to demonstrate the role of oxalic acid in pathogenicity of Sclerotinia sclerotiorum on Phaseolus vulgaris Physiol. Mol. Plant Pathol 37 : 179 – 191 .
Goldman J.J Smith O.D Simpson C.E and Melouk H.A 1995 Progress in breeding Sclerotinia blight-resistant runner-type peanut Peanut Sci 22 : 109 – 113 .
Hollowell J.E Isleib T.G Tallury S.P Copeland S.C and Shew B.B 2008 Screening of Virginia-type peanut breeding lines for resistance to Cylindrocladium black rot and Sclerotinia blight in the greenhouse Peanut Sci 35 : 18 – 24 .
Hollowell J.E Shew B.B and Isleib T.G 2003 Evaluating isolate aggressiveness and host resistance from peanut leaflet inoculations with Sclerotinia minor Plant Dis 87 : 402 – 406 .
Hu X Bidney D.L Yalpani N Duvick J.P Crasta O Folkerts O and Lu G 2003 Overexpression of a gene encoding hydrogen peroxide-generating oxalate oxidase evokes defense responses in sunflower Plant Physiol 133 : 170 – 181 .
Kirby J.S Banks D.J , and Sholar J.R 1989 Registration of ‘Spanco’peanut Crop Sci 29 : 1573 – 1574 .
Kolkman J.M and Kelly J.D 2000 An indirect test using oxalate to determine physiological resistance to white mold in common bean Crop Sci 40 : 281 – 285 .
Kritzman G Chet I and Henis Y 1977 The role of oxalic acid in the pathogenic behavior of Sclerotium rolfsii Sacc Exp. Mycol 1 : 280 – 285 .
Livingstone D.M Hampton J.L Phipps P.M and Grabau E.A 2005 Enhancing resistance to Sclerotinia minor in peanut by expressing a barley oxalate oxidase gene Plant Physiol 137 : 1354 – 1362 .
Melouk H.A Chamberlin K Godsey C.B Damicone J Burow M.D Baring M.R Simpson C.E Dashiell K.E and Payton M 2012 Registration of ‘Red River Runner’ peanut J. Plant Regist 7 : 22 – 25 .
Melouk H.A Akem C.N and Bowen C 1992 A detached shoot technique to evaluate the reaction of peanut genotypes to Sclerotinia minor Peanut Sci 19 : 58 – 62 .
Melouk H.A and Backman P.A 1995 Management of soilborne fungal pathogens , pp. 75 – 85 In H.A. Melouk and F.M. Shokes (eds.) Peanut Health Management, APS Press , St. Paul, MN .
Norden A.J Lipscomb R.W and Carver W.A 1969 Registration of Florunner peanuts Crop Sci 9 : 850 .
Noyes R.D and Hancock J.G 1981 Role of oxalic acid in the Sclerotinia wilt of sunflower Physiol. Plant Pathol 18 : 123 – 132 .
Partridge-Telenko D.E Hu J Livingstone D.M Shew B.B Phipps P.M P.M and Grabau E.A 2011 Sclerotinia blight resistance in Virginia-type peanut transformed with a barley oxalate oxidase gene Phytopathology 101 : 786 – 793 .
Porter D.M and Melouk H.A 1997 Sclerotinia blight , pp. 34 – 36 In N. Kokalis-Burelle et al., (eds.) Compendium of Peanut Diseases, APS Press , St. Paul, MN .
Smith O.D Simpson C.E Black M.C and Besler B.A 1998 Registration of ‘Tamrun 96’ peanut Crop Sci 38 : 1403 .
Smith O.D Simpson C.E Grichar W.J and Melouk H.A 1991 Registration of ‘Tamspan 90’ peanut Crop Sci 31 : 1711 .
Tu J.C 1985 Tolerance of white bean (Phaseolus vulgaris) to white mold (Sclerotinia sclerotiorum) associated with tolerance to oxalic acid Physiol. Plant Pathol 26 : 111 – 117 .
Wegulo S.N Yang X.B and Martinson C.A 1998 Soybean cultivar responses to Sclerotinia sclerotiorum in field and controlled environment studies Plant Dis 82 : 1264 – 1270 .
Woodward J.E Brenneman T.B Kemerait R.C Culbreath A.K and Clark J.R 2006 Sclerotinia blight in Georgia and evidence for resistance to Sclerotinia sclerotiorum in runner peanuts, Online. Plant Health Progress doi: http://doi.org/10.1094/PHP-2006-0531-01-RS .
Notes
- USDA-Agricultural Research Service, Wheat, Peanuts and Other Field Crops Research Unit, Stillwater, OK 74075.
- Department of Statistics, Oklahoma State University, Stillwater, OK 74078. *Corresponding author's email: rebecca.bennett@ars.usda.gov.
Author Affiliations